Mercuric Pyrosulfate: A Substance with Layers of History, Science, and Scrutiny
Historical Backdrop
People rarely consider how chemicals like mercuric pyrosulfate found their way from the early laboratories into modern books and research journals. Long before environmental standards and regulations, chemists explored the behavior of mercury compounds with keen curiosity, sometimes with little concern for hazards that seem obvious today. Mercuric pyrosulfate emerged as part of this wave of experimentation, tucked into the expanding catalog of inorganic salts that opened doors to sulfur chemistry. Early mentions of mercuric pyrosulfate link back to studies attempting to map the mercuric ion’s activities as it interacts with sulfuric acid, an essential reagent that powered industry revolution and chemistry curriculums. As uses for mercury grew, so did the documentation of these compounds, though often shadowed by crude safety practices that harmed more than they helped. Even libraries and chemical supply rooms in the twentieth century stored this substance with a nonchalance rarely seen today, reflecting how awareness around toxicity builds over time and experience.
Outlining the Product
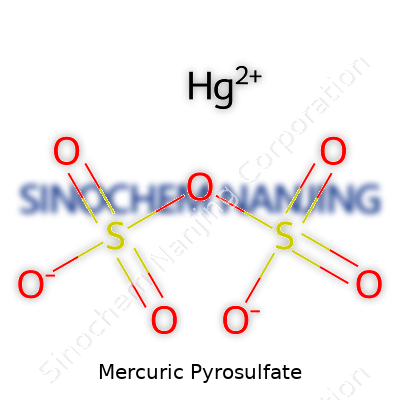
Mercuric pyrosulfate sits on the shelf as a white, sometimes off-white, crystalline solid. Its formula, Hg2S2O7, encapsulates two mercury atoms locked between sulfate groups—a pairing that points to its strong oxidizing potential and high reactivity with reduced sulfur or organic compounds. Among laboratory chemists, the compound developed a reputation both for its aggressive chemical behavior and its notorious mercury content. Its commercial chaperoning through the decades often landed it in the hands of academic and industrial researchers, but less so within general educational labs due to rising safety concerns. I remember the first time encountering a bottle marked as such; the unassuming powder, sealed under warning labels, prompted a long safety briefing. Even today, those unaccustomed to handling highly toxic substances hesitate before removing the cap. Even in chemical storage, the double warnings—mercury and strong oxidizer—stand out in a crowded inventory.
Physical and Chemical Properties
Ask most chemists about mercuric pyrosulfate, and the first thought likely jumps to its stability and toxicity. The compound appears as a crystalline powder, typically odorless, and strongly attracts moisture from air. It dissolves slowly in water, forming a solution that hovers at an acidic pH due to sulfate release. In practice, even the physical handling of this substance requires careful manipulation, never leaving a grain behind, because mercury compounds like this one contaminate with ease and do not simply wash away. Chemically, mercuric pyrosulfate stands as a potent oxidizer. In the lab, it oxidizes organic materials and reacts violently above certain temperatures, releasing sulfur dioxide, oxygen, and elemental mercury vapor—an especially concerning trio in unventilated spaces. Those who spend time around mercury salts learn quickly to judge conditions, double check containment, and appreciate the unique twinning of sulfur chemistry and heavy metal risks at play.
Specifying Technical Details and Labeling
Looking at the technical side, labels for mercuric pyrosulfate often feature a flurry of pictograms and hazard statements—most prominently, the skull and crossbones, environmental hazard tree, and oxidizer symbols. The detailed warnings aren’t just for show. Every gram demands tracking due to international regulations targeting heavy metal movement, especially mercury. In regions with strict health codes, even the smallest sample comes wrapped in secondary containment and supported by transport licenses. Lab managers and custodians treat expired stock almost like radioactive waste. Packing, labeling, and storage become a logistical exercise where clear identification, secure sealing, and up-to-date records separate safe chemistry from accidental exposure. Sometimes, older labels betray their age by omitting crucial risk phrases, a tell-tale sign that the bottle dates to less rigorous regulatory times.
Preparation Method
Mercuric pyrosulfate doesn’t materialize by accident—it comes from the careful reaction between dry mercury(II) sulfate and concentrated sulfuric acid at elevated temperatures. A careful balance keeps the reaction mixture from running away or decomposing prematurely. Having practiced similar syntheses in the past, it becomes apparent how much precision, patience, and cleanliness matter, especially around reactive mercury compounds. The process often requires glassware resistant to attack from strong acids, with condensation and venting systems that keep harmful fumes from accumulating. The intermediate stages generate both heat and acid vapor, so experienced practitioners pay close attention to color changes, air quality, and temperature ramps. Safety always takes top billing, and those who forget this typically don’t get a second chance.
Exploring Chemical Behavior and Modification
Once on hand, mercuric pyrosulfate opens new doors for reaction chemistry. It acts as a strong oxidizer because of the high reactivity between mercury and sulfur-oxygen frameworks. In organic syntheses, it sometimes serves to strip electrons from heat-stable substrates, triggering ring transformations or the breakage of stubborn carbon-sulfur bonds. Under other conditions, the compound reacts with water and dilute acid to revert back to mercury(II) sulfate and sulfuric acid—a practical demonstration of equilibrium, but hardly good news for environmental safety. Research in the field pushes boundaries in modifying this compound, but always shadows progress with a hard look at by-product management, recycling, and safe disposal. These days, many labs prefer alternative, less hazardous oxidants when the classic chemistry once reserved for mercuric pyrosulfate can be replicated by cleaner, greener means.
Alternative Names and Identities
Anyone searching for mercuric pyrosulfate might stumble across a handful of synonyms in older textbooks or regulatory documents: mercury disulfate, mercury(II) pyrosulfate, and even mercurous sulfate in rare cases, though strictly speaking that last one describes a different oxidation state. The key is the chemical formula; unless it reads Hg2S2O7, the bottle likely contains something else. Names change as regulatory language and nomenclature rules evolve, and the compound’s presence in literature can often reflect the era of the publication. Some researchers use the systematic name or slip back into the comfort of older terms, reminding us that chemical language rarely stands still.
Health and Safety Concerns
Mercuric pyrosulfate brings health concerns that demand respect. Occupational handling guidelines arrive with good reason. Mercury compounds are neurotoxic, even at low exposure levels, and this particular salt adds in strong oxidizing properties, amplifying risk. Splash, spill, or inhalation incidents quickly escalate, requiring immediate evacuation and medical attention. Working in labs that used mercury salts regularly, the lessons came hard and fast: avoid skin contact, double-glove, work in a fume hood, and never treat waste casually. Some regions adopted legal limits for airborne mercury and strictly controlled waste streams. Personal stories abound of hasty accidents leading to chronic effects. Modern practice, shaped by these stories, now leans hard into ventilation upgrades, spill kits, and exposure monitoring. If a lab suffers a near-miss, the event often reverberates through the institution, sharpening protocols and ramping up training for all who enter.
Application and Research Directions
Historically, mercuric pyrosulfate found its value in analytical chemistry, especially in oxidation reactions where stubborn substances resisted milder treatment. A handful of industrial syntheses relied on its sharp reactivity, particularly in sulfur and nitrogen chemistry. These days, the search for greener, less toxic alternatives puts pressure on even long-standing applications. Research continues, though, to find cleaner recycling methods for mercury, better ways to capture emissions, and improved alternatives that maintain the desired chemical reactivity without the toxic baggage. Laboratories that still use mercuric pyrosulfate tend to deploy it sparingly and with a careful audit trail, often as a comparator against newer oxidants rather than a staple reagent. Training the next generation, researchers now fold environmental and safety considerations into every experimental design, sharing both the chemistry and the consequences that come with it.
Toxicity Research
No discussion about mercuric pyrosulfate finds completeness without delving into toxicity. Researchers tracked effects from low-dose, chronic exposure to dramatic incidents involving spills and fires. Studies on laboratory and environmental exposure demonstrate symptoms ranging from headaches and tremors to irreversible neurological impairment. Animal studies informed strict occupational limits, and high-profile cases prompted industry-wide changes in use and disposal practices. Devastating as mercury exposure can be, progress in medical and toxicological research now includes better detection methods for mercury in air, water, and biological samples. The modern lab never expects to operate without material safety sheets, exposure response plans, and a ready supply of proper containment solutions. As case reports surfaced over the decades, the most significant lesson reinforced the cost of ignoring hazard warnings on a bottle as unassuming as mercuric pyrosulfate.
Future Prospects
Looking ahead, the fate of mercuric pyrosulfate balances on a knife edge between continued scientific interest and growing environmental concerns. Regulatory crackdowns on mercury force manufacturers to minimize production or phase out use altogether. The chemistry community faces mounting pressure to substitute less toxic, more sustainable options wherever the reaction specifics allow. Research efforts now pivot on safer oxidizing agents and mercury-free methodologies, especially in fields where end-users prioritize both performance and planet. That doesn’t mean the old chemistry vanishes overnight. Archives and historical projects still cite mercuric pyrosulfate, while its story continues to instruct chemists on the price of scientific progress that comes untethered from environmental and health responsibility. Younger scientists see lessons not only in the possibilities the compound once unlocked, but also in the demanding stewardship needed for hazardous reagents in an era that values not just discovery, but also the long-term health of workers and ecosystems alike.
What is Mercuric Pyrosulfate used for?
Tough Chemistry, Tough Questions
Mercuric pyrosulfate doesn’t sound like something most folks talk about over coffee, yet this chemical finds itself tucked away in certain labs and research environments. As someone who spent years working with analytical chemistry teams, I remember how tightly chemicals like this got locked up. Not just because of their toxicity, but due to their specialized uses.
Where Mercuric Pyrosulfate Steps In
Mercuric pyrosulfate plays a niche, yet important, role as a reagent in some analytical chemistry methods. Most people working in conventional labs probably haven’t used it, but those tracking down trace elements or organic substances in samples might have crossed its path. It helps break down organic materials, digesting complex molecules to leave behind components scientists then identify and measure.
One major use involves testing food or water samples for specific contaminants. The chemical reacts strongly, allowing labs to tease out pieces of information that would be tough to get through other reactions. With the rise in food safety concerns and water quality scandals, demand for highly accurate testing has become part of daily life all over the world.
Risk Runs Deep
I remember the first time our team handled a bottle marked with mercury salts. We listened close to every word in the safety briefing. Mercuric pyrosulfate contains mercury—an element known for its toxicity and environmental impact. Getting careless with this stuff can spell real danger for people and for the environment. Many mercury compounds stick around in soil and water, and living things absorb and accumulate them up the food chain.
Industry regulations landed for a reason. Years of exposure and case studies revealed mercury’s ability to damage nerves, kidneys, and even child development. Labs now use containment procedures and protective gear because the risks go beyond spills. Mercury fumes can cause trouble before anyone realizes what’s going on.
Pushing Toward Safer Solutions
Chemists stand at a crossroads. They need reliable testing methods, yet the old standbys can come with serious costs. Several countries started offering grants and incentives for the development of less hazardous reagents. Some universities research green chemistry approaches, where safer compounds substitute for legacy reagents like mercuric pyrosulfate. Progress comes in slow steps; old methods die hard in the scientific world, mainly because trust matters more than novelty when stakes involve public health or clean water.
Real change hinges on investment and on how seriously research labs, companies, and regulators respond to both environmental threats and worker safety issues. Every time a lab swaps in a safer alternative, it inches science toward smarter and greener solutions. Regulatory agencies can help by approving new methods more quickly once they prove reliable, and by supporting projects focused on green innovation.
Looking Forward
Mercuric pyrosulfate continues to serve a specialist role, but its challenges keep scientists searching for better answers. People inside and outside the lab deserve transparency and safety. By learning from decades of experience and supporting fresh research directions, society can limit risks without sacrificing crucial advances in testing and quality control.
What are the safety precautions for handling Mercuric Pyrosulfate?
Understanding the Risks
Mercuric pyrosulfate sits in the family of chemicals that most folks don’t want anywhere near their skin, lungs, or workspace. It brings the hazards you’d expect from something with both mercury and sulfur in its structure. Breathing in its dust or fumes can mess up your kidneys and nervous system. Skin contact may lead to rashes or more severe reactions down the line. Swallowing even a bit would put someone in serious danger. Protecting health isn’t just about following the rules; it keeps you and your coworkers safe for the long haul.
Personal Experience in the Lab
Back in my student lab, bottles with a skull-and-crossbones label always got special attention. Mercuric compounds topped the list. I wore gloves that actually blocked mercury, not just thin latex from a box store. Nitrile or neoprene gloves, along with sturdy lab coats and face shields, proved worth the trouble. Eye protection with side guards beats out flimsy prescription frames every time. Once, a colleague learned this the hard way—a single spill on her wrist, even through a tear in the glove, meant a visit to occupational health and weeks of checks. Nobody wanted to repeat that.
Ventilation and Equipment Control
Fume hoods aren’t for show. They pull toxic vapors out of your breathing space—a must when handling mercuric pyrosulfate, which doesn’t always sit still in a beaker. The chemical can release dangerous gases, especially if someone accidentally heats it up too far or mixes it with the wrong thing. Closed containers, wiped clean before opening, avoid spreading invisible dust. Regular checks of the airflow and airflow direction matter more than most think; some labs skip this, only to find out too late when the monitor lights up red.
Spill Management
Dropping mercury-based powder calls for calm, not a scramble to grab paper towels. Absorbent materials for mercury cleanup work, but even better is training. In my lab, we drilled for small spills—a sprinkle, not a pile—but everyone knew when to call in the professional hazmat crew. After even a minor incident, the area stayed off-limits until testing said it was clear.
Storage Practices
Dry, tightly sealed containers in a locked chemical cabinet keep mercuric pyrosulfate from accidental mix-ups. Storing it away from acids, bases, and organic material proved wise; reactive spills love unplanned chemistry. Labels showed the full chemical name and the date, not just shorthand. This way, nobody grabbed a forgotten bottle that had deteriorated or started to leak fumes.
Waste Disposal: No Shortcuts
You can’t throw leftovers in the trash or pour them down the drain. Trained staff packaged any scrap or spill residue in lined drums, marked for hazardous waste pickup by certified handlers. Local regulations dictate the details, but the key lesson: out of sight does not mean out of trouble. Tracking what leaves the workspace counts for future audits and in case of community scrutiny.
Learning and Teaching
Too many folks skip refreshers on chemical safety after passing an introductory course. Explaining what happens after a glove fails or a seal breaks makes it real for newcomers. Bringing in stories from OSHA’s case files, or a near-miss from your own history, keeps attention sharp. Ultimately, a healthy lab is everyone’s responsibility, and clear communication stands as the best protection.
What is the chemical formula and molecular weight of Mercuric Pyrosulfate?
Chemical Identity: Formula and Weight
Mercuric pyrosulfate rolls off the tongue for seasoned chemists but gets eyebrows raised in folks less knee-deep in flasks and beakers. The chemical formula for mercuric pyrosulfate is Hg2S2O7. Its molecular weight clocks in at 562.72 g/mol. That’s a big number—mercury is dense, and this compound doubles down on elemental heft by having two mercury atoms in the mix for every molecule.
Stepping Beyond Formulas: Real-World Relevance
There’s a reason specialists don’t just toss around “mercuric pyrosulfate” like it’s table salt. This compound plays a role in chemical analysis, especially where measuring or oxidizing certain substances gets tricky. Chemists use it to break down organic materials and get accurate readings. No surprise—two heavy metals linked with strong oxidizing groups pack a serious punch.
Working with materials like this requires both respect and solid knowledge. Mercury as an element doesn’t have a glowing reputation. Documented cases of mercury poisoning have followed accidents and negligence for over a century. Handling a compound with two atoms of mercury means personal protective equipment isn’t just a nice idea; it’s essential. Lax safety rules can turn curiosity and exploration into long-term health troubles.
Practical Applications and Cautions
Lab technicians lean on mercuric pyrosulfate for analytical chemistry, especially where less aggressive chemicals come up short. Oxidizing stubborn organic samples sometimes calls for muscle like this. In my own university lab days, we learned early on about the trade-off between analytical power and safety risks. The compound gives researchers the tools to analyze tough samples but shoots up red flags for safety and waste disposal.
Natural curiosity comes with the risk of mishandling, especially where mercury is involved. Past spills or mishaps have shown how easily vapors or residues can create hazards. Even tiny amounts left behind can lead to lasting environmental trouble, so proper training shouldn’t just be a box to check. I remember a story from a former coworker: a minor spill set off a facility-wide cleanup, revealing just how unforgiving mercury compounds can be.
Supporting Safe and Responsible Use
Following up-to-date safety standards helps keep people and their surroundings protected. Chemicals with mercury require storage in specialized containers, away from where children, pets, or food are nearby. Research groups and industries that use mercuric pyrosulfate should plan for responsible disposal, including collection by trained hazardous waste handlers. Reviewing material safety data sheets before starting work makes a difference. Regular audits of storage and procedures catch problems before they become accidents.
Many research institutions now look for alternatives to mercury-based chemicals. Some new oxidizers offer similar performance, with fewer health and environmental risks. Switching from legacy reagents like mercuric pyrosulfate often means retraining staff. Adopting new protocols takes time, but safer labs and cleaner environments justify the investment.
So, understanding both the technical side—Hg2S2O7, 562.72 g/mol—and the practical risks creates a culture where exploring complex chemistry can be both effective and safe. Knowing what’s at hand, and using it well, pays off for everyone around the table.
How should Mercuric Pyrosulfate be stored?
Why Proper Storage Matters
Storing chemicals like mercuric pyrosulfate goes far beyond just tossing a bottle on a shelf. I’ve worked with a variety of toxic and sensitive compounds, and I’ve seen what happens when people cut corners. Fumes creep, glass corrodes, labels fade—mistakes build up until real risks start showing up. Mercuric pyrosulfate isn’t the sort of chemical you just leave sitting out. Its toxicity sits at levels few would feel comfortable with. Even trace mercury can haunt your workspace for years. This stuff affects people’s health, the next batch of students in your lab, and the groundwater if a jar breaks near a drain. That kind of risk should push anyone to rethink storage from the ground up.
Understanding the Risks
This compound contains mercury, and that ring of red hazard diamonds on the label isn’t just for show. Inhaling its dust, vapors, or letting it touch your skin carries a real chance of poisoning. Stories from old chemists often focus on how simple lapses in storage led to lifelong neurological problems. It’s not dramatic, it’s just a slow accumulation of mistakes—until someone gets hurt. Many health agencies around the world, including the CDC and OSHA, put tight restrictions on mercury exposure. That means you should look at every storage step as a chance to block leaks, spills, or mishandling.
Storage Tips from Real-World Experience
Mercuric pyrosulfate prefers dry, stable conditions. I always pick a well-ventilated chemical cabinet—ideally a sturdy one lined with spill-resistant shelves. Cabinets for corrosive or toxic substances usually offer secondary containment trays, which catch any drips. In my time working with mercury compounds, I’ve seen rogue crystals turn up on bottlenecks or lids, especially in humid environments. Silica gel packets in the cabinet soak up stray moisture and slow down deterioration; this isn’t overkill, it’s just common sense.
Direct sunlight and heat quickly break down many compounds, and this one is no exception. Cool, shaded storage keeps the bottle contents stable, minimizing both vapor leaks and decomposition. Some folks think refrigeration is always better—it's not. Mercuric pyrosulfate can absorb moisture and start creating puddles or corrosive residue if chilled in a way that lets frost build up. Room temperature works better for mercury salts, except in the hottest climates where you should use an air-conditioned storage room.
Labeling, Security, and Training
Every time I walk into a lab, faded or missing labels always jump out to me. Chemicals last longer than tape and ink, so updating labels is part of the safety routine. Labels with large text, hazard symbols, and last opened dates really do save lives. A chemical with a missing label goes straight to disposal, no exceptions.
Mercury compounds attract strict controls, so secure storage is a must. Doors should have locks, and only trained staff handle the keys. In places where regulations demand it, keep a logbook to show exactly who accessed these bottles and when. Anyone handling mercuric pyrosulfate deserves thorough instruction, with direct, practical training—not just a sign-off sheet. Mistakes drop fast once people understand what happens if things go sideways.
Planning for Spills and Disposal
No storage method completely removes risk. If a bottle breaks, you need spill kits with materials specifically rated for mercury compounds—not just generic pads or baking soda. At the sites where I’ve worked, proper cleanup supplies and disposal drums always sat close to storage. Coordination with local hazardous waste handlers and emergency contacts comes next. Don’t leave people guessing which number to call if the bottle tips over.
Final Thoughts on Safe Storage
Safe storage isn’t about fear, it’s about respect. Respect the chemical, respect everyone sharing the workspace, and surround these bottles with habits that protect both health and reputation. Only through solid routines and a little extra caution do you avoid hard lessons later on.
Is Mercuric Pyrosulfate hazardous to health or the environment?
Understanding the Risks
Mercuric pyrosulfate catches the attention of chemists mainly for its role in laboratories. On paper, it sounds like just another compound, but the story takes a serious turn once you look at health and environmental risks. Mercury-based substances rarely pass unnoticed—too many headlines have hammered home mercury’s impact across generations.
Human Health Hazards
Handling mercuric pyrosulfate, even in small quantities, sets off a cascade of hazards. This compound enters the body mostly through inhalation of dust or direct skin contact. Based on reported cases and workplace experience, exposure can lead to symptoms such as skin rashes, eye irritation, and damage to digestive organs. The real trouble starts because the body doesn’t flush out mercury easily—once it gets in, it settles in tissues including the brain, kidney, and liver.
Long-term or repeated exposures make the list of problems grow: tremors, memory loss, kidney damage, mood swings—the list feels endless. Mercury compounds sometimes cause irreversible changes in the nervous system, a fact well documented over the decades. One only needs to look at incidents in industrial sites that failed to control vapor or dust. Increased regulations came after communities saw soaring rates of neurological illness.
Chemistry labs take these risks seriously. Safety protocols require gloves, fume hoods, goggles, and careful monitoring, not because of an abstract worry, but because accidents with mercury compounds can ruin careers and devastate lives. My own experience as a lab tech taught me to never cut corners: just a tiny droplet on bare skin means a visit to the nurse and a lengthy incident report.
Environmental Impact
Environmental authorities keep a tight eye on mercury releases, with good reason. Once mercuric pyrosulfate enters water or soil, it doesn’t just sit harmlessly—bacteria can convert it into methylmercury, a far more toxic form taken up by fish and moved up the food web. Families dependent on fishing end up with contaminated food, and public health departments scramble to issue advisories.
Every tiny spill or poorly managed laboratory discharge adds up, contributing to the kind of contamination that plagued Minamata Bay in Japan or rivers near old manufacturing sites. Mercury pollution links directly to birth defects, cognitive delays, and wildlife losses. Cleanup efforts demand millions and still leave scars on ecosystems for decades.
Looking Toward Solutions
No silver bullet exists. Reducing risk means strict storage rules, professional waste handling, and persistent training in safe handling. Laboratories replace mercury reagents with safer alternatives whenever possible. Regulators push for better reporting and audits, and environmental agencies press industries to track emissions.
Communities near research facilities or industries often push for tougher oversight. The movement for “green chemistry” means researchers develop new methods that side-step hazards entirely. These changes demand time and budget, but the payoff is clear: fewer health emergencies, cleaner rivers, and peace of mind for those working on the front lines.
Trust comes from informed action. Those who work with or live near places that use mercuric pyrosulfate rely on leaders to keep hearing their concerns, back up safety measures with funding, and keep pushing for low-risk science.
| Names | |
| Preferred IUPAC name | Mercury(II) pyrosulfate |
| Other names |
Mercury(II) pyrosulfate Mercury disulfate Pyrosulfuric acid, mercury(2+) salt |
| Pronunciation | /ˈmɜː.kjʊr.ɪk paɪˈrɒl.sfɛɪt/ |
| Identifiers | |
| CAS Number | 7487-66-9 |
| Beilstein Reference | 1206984 |
| ChEBI | CHEBI:131374 |
| ChEMBL | CHEMBL1201673 |
| ChemSpider | 21569842 |
| DrugBank | DB06772 |
| ECHA InfoCard | 04d00eaf-316f-42aa-8e13-dcc8e3e0110a |
| EC Number | 231-998-3 |
| Gmelin Reference | 16244 |
| KEGG | C18683 |
| MeSH | D010529 |
| PubChem CID | 25136 |
| RTECS number | OW6825000 |
| UNII | WL40K6D9T7 |
| UN number | UN1673 |
| CompTox Dashboard (EPA) | `DTXSID2030295` |
| Properties | |
| Chemical formula | Hg₂S₂O₇ |
| Molar mass | 464.64 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 3.98 g/cm3 |
| Solubility in water | Slightly soluble |
| log P | -2.1 |
| Acidity (pKa) | -4.0 |
| Basicity (pKb) | 7.4 |
| Magnetic susceptibility (χ) | -47.7·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.64 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 239.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -880 kJ/mol |
| Pharmacology | |
| ATC code | V09AA18 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled or in contact with skin; causes severe skin burns and eye damage; may cause damage to organs through prolonged or repeated exposure. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS05 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. |
| Precautionary statements | P260, P262, P273, P280, P302+P352, P304+P340, P305+P351+P338, P310, P314, P330, P361, P391, P501 |
| NFPA 704 (fire diamond) | 3-0-2 |
| Lethal dose or concentration | LD50 oral rat 57 mg/kg |
| LD50 (median dose) | LD50 (median dose): 21 mg/kg (oral, rat) |
| NIOSH | WX3850000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Mercuric Pyrosulfate: 0.1 mg/m³ (as mercury) |
| REL (Recommended) | 0.05 |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Mercury(II) sulfate Mercury(II) oxide Mercury(II) chloride |

