Phenylmercuric Pentachlorophenoxide: A Closer Look at Its History, Properties, and Future
Historical Development
Watching the history of Phenylmercuric Pentachlorophenoxide unfold tells a story of changing scientific priorities and shifting regulations. Chemists first synthesized it in the early 20th century, chasing better antimicrobial agents for paints, textiles, and paper. In the decades after World War II, demand soared as people focused on industrial growth and public health, not always pausing to weigh long-term harm from toxic compounds. By the 1970s, as researchers rang alarm bells over organomercurials in the environment, public agencies and scientific journals started to press for more restriction and oversight. Through all this, industry experts and toxicologists have argued about the right balance—between genuine protective benefits and mounting evidence about mercury’s dangers.
Product Overview
Phenylmercuric Pentachlorophenoxide comes from the blend of two potent chemical groups: an organomercury structure paired with a pentachlorophenol group. Manufacturers turn to this compound as a fungicide and antibacterial agent, counting on its strong ability to kill unwanted microbes in a range of products. You see it pop up in preserved paints, wood treatments, and even some medical disinfectants. Markets and approvals have changed fast, but the substance once maintained a key spot for stopping mildew and spoilage, especially in places where humid weather pushes other options to the sidelines.
Physical & Chemical Properties
The material usually takes the form of a fine white to off-white powder, carrying a characteristic chemical odor, and sometimes forming slightly yellowish crystals with prolonged storage or environmental exposure. It resists easy breakdown by air and water, showing low volatility under ordinary conditions, and tends to dissolve better in organic solvents like chloroform or ethanol than in water. The molecule combines mercury and chlorine atoms, so it reacts differently from more common organic substances. At room temperature, it stays stable unless exposed to ultraviolet light or excessive heat. All these features add up to a chemical that acts with staying power in whatever product holds it.
Technical Specifications & Labeling
Labeling for Phenylmercuric Pentachlorophenoxide usually lists active ingredient percentages, batch numbers, and clear warnings about toxicity, targeting both professional handlers and regulatory compliance. Formulators keep the mercury content tightly controlled, often keeping below 0.1% to 0.3% in finished goods. Product labels draw attention to personal protective equipment, possible inhalation risks, and disposal guidelines. Modern regulations push for QR codes or tracking numbers as added assurance, since supply chain transparency now factors so heavily in chemical safety across many countries.
Preparation Method
People in the chemical industry rely on a reaction between phenylmercuric acetate and sodium pentachlorophenoxide, carrying the process out in controlled reactor vessels, under careful temperature and pH management. Operators add phenylmercuric acetate in solvent, stir with sodium pentachlorophenoxide, then precipitate and filter the crude product. The product gets washed and sometimes recrystallized for higher purity. Each batch takes close monitoring to avoid excess mercury waste and to keep product purity in the range suitable for final industrial or commercial use.
Chemical Reactions & Modifications
Phenylmercuric Pentachlorophenoxide stays stable with most common materials but breaks down with strong acids, bases, or reducing agents, releasing mercury ions and chlorinated phenols. Chemists have studied ways to shift its properties—sometimes they modify the molecule for slower breakdown or for tailored action against specific fungal strains. These tweaks sometimes lower the volatility or tweak the binding to surfaces, but every modification needs fresh toxicity and leaching tests, since mercury and chlorophenolic breakdown products both count among persistent environmental challenges.
Synonyms & Product Names
Over the years, people in the industry have called Phenylmercuric Pentachlorophenoxide by several names, which can muddy the regulatory waters and confuse workers. Trade names like PMPP, Phenylmercury penta, or, less formally, "phenyl mercuric salt of pentachlorophenol" pop up in registrar lists and supply trade logs. Local suppliers sometimes switch names to bypass regulatory hurdles. Standardization of naming in official inventories and harmonized lab documentation really matters to keep global oversight effective and to prevent mistaken substitutions, which can prove costly or dangerous.
Safety & Operational Standards
Staying safe around this compound demands genuine care and respect for longstanding health warnings. Direct contact or prolonged inhalation triggers mercury poisoning symptoms—from mild skin irritation to serious neurotoxicity and kidney stress in exposed workers. Most industrial settings call for air-tight mixing vats, fume hoods, and rigorous worker health checks. I’ve seen entire sites shut down during audits for stretching safety guidelines around organomercurials, and once mercury hits a spill control plan, it's a tough and expensive clean-up. Regulatory agencies like OSHA and EPA set permissible exposure limits far below what’s allowed for other antimicrobial agents. A slip-up doesn’t simply mean a fine—sometimes workers end up in the hospital and companies lose their licenses to operate.
Application Area
Paint makers and wood preservation companies used to rely on Phenylmercuric Pentachlorophenoxide for its strong fungicidal action, especially where weather or moisture made mold growth an ongoing fight. Textile mills and paper factories leaned on it to keep raw materials from rotting or discoloring. Hospitals applied versions of the product for surface disinfection. Shifts in regulatory stances moved many of these industries to less hazardous options, but in regions with limited market access or older infrastructure, the compound hasn’t vanished—sometimes showing up in private inventories or legacy stocks.
Research & Development
Scientists keep probing alternatives—or less hazardous analogs—that offer similar microbial control without the environmental fallout. Some focus on encapsulated delivery systems to trap mercury in place, stretching the useful lifespan of treated goods while shrinking leaching into soil and waterways. Others measure the breakdown products, watching for safe routes to degrade or bind leftover mercury. Every promising result leads to complex field studies, and in research labs, there’s always pushback from legacy operators who cite cost and performance as barriers to full replacement. Funding for greener chemistry, especially in the antimicrobials field, grows each year as concerns stack up in academic journals and community groundwater surveys.
Toxicity Research
Every time toxicologists sit down with samples containing phenylmercuric compounds, headaches mount. The data connect mercury from this source with neurological problems, impaired fetal development, and long-term impacts on wildlife populations. Fish, birds, and humans all absorb organomercurials, often suffering the consequences years after exposure. Chronic low-level exposure tends to fly under the radar, especially in communities near industrial discharge or using untreated water near legacy sites. As new lab techniques grow more sensitive, links between exposure and real health problems sharpen. Global health bodies now recommend all possible users find safer alternatives. The push for routine testing of food and water, especially in heavily industrialized regions, comes in direct response to these risks.
Future Prospects
Looking ahead, pressures from government crackdowns and industry health standards seem set to keep shrinking demand for Phenylmercuric Pentachlorophenoxide. Sectors with heavy use, like paint and paper, keep migrating toward non-mercurial substitutes, even if the alternatives bring new sets of challenges. Research into green chemistry continues to pick up pace, with projects aiming to safely dismantle old stocks and cap leaching from contaminated sites. My experience working on remediation teams suggests the cleanup of this chemical lags far behind best intentions, often hitting roadblocks on cost and technical limits. International agreements such as the Minamata Convention on Mercury push companies and countries to rethink old habits and stamp out loopholes. For people working in affected industries, pivoting means investing in worker retraining and tougher environmental auditing, but the benefit shows up in cleaner water, healthier workers, and a smaller toxic legacy for the next generation.
What is Phenylmercuric Pentachlorophenoxide used for?
The Chemical at a Glance
Phenylmercuric pentachlorophenoxide rarely crops up in household conversations, but anyone digging into the history of preservatives or agricultural chemicals will bump into it. Born from a time when mercury got tossed into products from paints to medicines, this compound packs a punch — not just as a long-lasting preservative but as a strong fungicide and bactericide.
Why It Showed Up Everywhere
Landscapers, textile manufacturers, and paint makers once counted on this chemical to fight mold and mildew. I grew up visiting greenhouses where the staff grabbed anything they could to stop fungus in its tracks. If wood needed to avoid rot, companies might have chosen phenylmercuric pentachlorophenoxide. Same story for paints designed to survive mildew in damp conditions. In some cases, pharmaceutical companies included similar mercury compounds in topical antiseptics. At the time, the health risks of mercury didn’t get the same attention they do now.
The Risks That Changed Minds
The facts about mercury and its compounds have changed how scientists, regulators, and the public view chemicals like phenylmercuric pentachlorophenoxide. Exposure to even small amounts of organic mercury can cause serious damage to the nervous system. Multiple cases, especially those involving children, showed that what stopped mold could harm people for years.
Environmental health experts raised alarms as mercury compounds started piling up in soil and water. Fisheries near areas with regular use of mercury-based fungicides wound up with contaminated catches. Pregnant women and young kids faced the greatest danger due to the way mercury builds up in the body and can cause developmental delays.
Times Have Changed: Alternatives Replacing Mercury Compounds
After seeing these hazards spelled out in the news, governments around the world put strict rules in place. The United States Environmental Protection Agency, Canada, the European Union, and others either banned or sharply restricted mercury compounds in the 1970s and 1980s.
The industries that once relied on phenylmercuric pentachlorophenoxide turned to safer alternatives. Modern paint companies use compounds like isothiazolinones, formaldehyde-releasers (with safety precautions), and other advanced chemicals to fight mold and mildew. Hospitals use alcohol-based disinfectants instead of mercury-based antiseptics. Agriculture shifted toward organic and lower-impact fungicides, reducing risks to farm workers, food, and waterways.
Looking Forward: Lessons from Phenylmercuric Pentachlorophenoxide
Chemical history like this matters because people keep discovering new uses for old chemicals — and new harms too. My own skepticism about miracle ingredients comes directly from cases like this: promising shelf life, until the real costs show up later.
Stronger science, transparency in labeling, and tough regulation have helped, but people need to stay alert. Reading safety sheets, asking questions about ingredient lists, and supporting laws that protect against toxic chemicals ensure that the next generation faces fewer health risks.
Just because something stops mold or keeps paint fresh doesn’t mean it should land in homes, playgrounds, or rivers. The story of phenylmercuric pentachlorophenoxide stands as a reminder: being thorough with safety likely prevents much bigger problems.
Is Phenylmercuric Pentachlorophenoxide safe to use?
Understanding Phenylmercuric Pentachlorophenoxide’s Risks
Phenylmercuric pentachlorophenoxide rarely shows up in everyday conversation, but it has a past in industry as a preservative and fungicide. Having worked in a lab years back, the name still sticks with me, mostly because mercury-based chemicals demand extra caution. The first thing anyone learns working with mercury compounds is to check twice and triple check disposal methods—because once they seep into the environment or get inside the body, it’s hard to undo the damage.
What the Research Tells Us
Researchers have known for decades that mercury compounds cause both acute and chronic toxicity. Phenylmercuric pentachlorophenoxide contains organic mercury, a class linked to neurological problems, kidney damage, and birth defects. In the 1970s and 1980s, cases surfaced where workers exposed to this stuff developed tremors, skin rashes, and, at high enough levels, serious nervous system damage. The numbers do not lie: one study in Clinical Toxicology tracked organomercury exposure and found even low-level contact accelerates risk. It’s not just about getting a big dose all at once; repeated, small exposures over years build up in organs.
The Regulatory Landscape
Regulators in most countries chose to restrict or ban phenylmercuric compounds in consumer goods back in the 1990s. The U.S. Environmental Protection Agency and the European Chemicals Agency both flagged it as too risky for regular use outside strictly controlled industrial processes. It's not just about law, but about community health. Even in the rare cases where phenylmercuric pentachlorophenoxide pops up—such as legacy agricultural sites or old paints—special cleanup crews handle it with full protection.
Many people ask: if regulations are so strict, why does this chemical surface in safety debates at all? The answer boils down to legacy use and poor disposal practices. Runoff from old materials I remember working with sometimes turned up in soil samples—far away from where it was used—because mercury refuses to just disappear. It stays put, and in the right conditions, can even turn into more toxic forms.
Respecting the Health Impacts
Personal stories help shape understanding here. A friend’s family lived near a plant dumping mercury waste, and he struggled with learning disabilities his whole life. Doctors connected it to mercury found in their water. Hard to ignore facts like these. Every major public health group agrees the safest amount of mercury exposure is as close to zero as possible.
Safe Alternatives and Prevention
Modern science has handed us better choices. Non-mercury fungicides, biocides, and preservatives do the job without the health baggage. Simple changes in industry and agriculture—swapping out toxic chemicals for safer ones—slash the risk for everyone. As someone who spent years in the chemical field, embracing new, less hazardous substances takes work, but it’s worth it. People sleep at night knowing the backyard isn’t hiding a dangerous secret.
Abstract science only goes so far. Real safety sometimes means saying no to a product with a toxic past and choosing an alternative. Knowledge, solid research, and public transparency move us toward a world where chemicals like phenylmercuric pentachlorophenoxide fade quietly out of use.
What are the storage requirements for Phenylmercuric Pentachlorophenoxide?
Understanding the Risks
Phenylmercuric pentachlorophenoxide is not the kind of chemical anyone wants sitting forgotten on a shelf. It’s not only toxic, but also hazardous to water, air, and anyone who comes into contact with it. For folks who handle chemicals in the lab or warehouse, this isn’t just another name on a list—it’s a substance that calls for careful handling.
Choosing the Right Space
This compound reacts with plenty of things—especially acids, some metals, and heat. I remember walking into storage rooms and noticing that the best-run labs keep their most reactive chemicals separate, away from foot traffic and moisture. For Phenylmercuric pentachlorophenoxide, a cool, dry, and well-ventilated area makes all the difference. Heat and sunlight can trigger decomposition, and that’s never good.
Container Safety
Glass or sturdy polyethylene containers—with tight, non-metallic lids—stand up to corrosion better than anything made of metal. No one wants to see a leaky container after a weekend, and with this compound, spills can get dangerous fast. In real lab settings, secondary containment trays catch any drips before they spread. It's a small step that spares a lot of headaches and cleanup.
Isolation and Segregation
Chemicals like this deserve their own shelf or cabinet, far from acids, reducing agents, food, or anything for human use. Storing these near incompatible materials turns a simple storage mistake into a recipe for disaster. Anyone working in a lab or warehouse should check the labels twice and re-read the segregation chart. A little diligence prevents mixing substances that just shouldn’t get close.
Access Control and Safety Resources
Only folks with training should get near this stuff. Locked cabinets and clear warning signs keep visitors, students, or new hires from wandering into trouble. One of my earliest mentors repeated, “If you treat hazardous chemicals with casual care, you’ll only learn the hard way.” Proper storage isn’t just about following rules; it’s about respecting the risk and protecting everyone who might walk in—intentionally or not.
Environmental Considerations
Mercury-based compounds harm aquatic life and build up in the food chain. These risks make it even more important that containers are leak-proof and checked regularly for any sign of deterioration. Waste goes straight to hazardous waste collection, never down the drain or in the trash. In places I’ve worked, supervisors walk through monthly just to double-check on aging bottles and any mislabeling.
Planning for Emergencies
Anyone storing Phenylmercuric pentachlorophenoxide should make sure spill kits, gloves, and goggles are nearby—not tucked away in another room. Emergency numbers and procedures need to stay visible. Fire extinguishers rated for chemical hazards hang within reach. These aren’t just formalities—they’ve saved lives and minimized harm more than once.
Better Practices, Safer Spaces
Real safety comes down to culture and consistency. Regular audits, updated inventory lists, and quick response to damaged or outdated material help keep things under control. Investing in proper training and safety gear beats cutting corners. When it comes to hazardous chemicals, a bit of caution and respect keeps people, property, and the environment safer now and in the future.
How should Phenylmercuric Pentachlorophenoxide be handled and disposed of?
Why Phenylmercuric Pentachlorophenoxide Matters
Working in labs and talking with folks in maintenance over the years, I’ve seen nerves shot at the mention of mercury chemicals like phenylmercuric pentachlorophenoxide. The name alone can clear a room. There’s reason for this. This compound isn’t just a mouthful — it packs real health risks. Exposure can impact the kidneys, the central nervous system, and pile on the environmental damage if ignored.
Handling Doesn’t Mean Cutting Corners
Some shops keep this stuff on shelves for fungus control in paints or as a preservative in some older formulas. I’ve noticed sloppy habits around older stockpiles, especially when workers figured “a mask and gloves” might do the trick. Not true. Skin contact, fumes, dust — every route in is a hazard. Wearing heavy-duty gloves, chemical splash goggles, and a lab coat becomes smart practice, not overkill. I even tell people, double up that protection with a face shield and mercury-rated respiratory cartridge, since breaches with a compound like this don’t always announce themselves.
Keep this out of regular work spaces. Dedicated, locked chemical storage with secondary containment keeps it far from accidental spills. In cramped janitorial closets or supply rooms, it’s just an accident waiting to happen. Ventilation helps, too. I remind coworkers to open windows, use local exhaust hoods — fresh air moves vapors out, which means fewer headaches and fewer mistakes.
Spill Response: No Room for Improvisation
Once I helped clean up after a small mercury spill, the lesson stuck — skimping on cleanup supplies or expertise has ripple effects. Granular mercury spill kits help, but most regular janitors haven’t trained for mercury compounds. That’s why it's better to alert certified hazmat teams who know the drill and use special mercury absorbents and HEPA vacuums rather than brooms or vacuums from the closet.
I’ve seen well-intentioned staff use paper towels and trash bins, sending the problem down the line. That’s how mercury enters drains, then rivers — and communities pay the price. Seal off the area, prevent vapor spread, call in trained professionals sooner rather than later.
The Garbage Can Isn't an Option
Disposing of phenylmercuric pentachlorophenoxide by tossing it in the dumpster or pouring it down the sink? Never a safe choice. Laws in most places treat this as “hazardous waste,” which makes sense. Local waste authorities or environmental firms offer collection and disposal programs specifically for mercury-contaminated items. Label everything — jars, rags, your gloves — and put them in a chemical waste container with a tight lid, not a plastic grocery bag. Storing it safely until collection stops curious hands and accidental leaks.
Better Policies, Stronger Training
Over the years, I’ve watched companies drift from required safety reviews, even as chemicals age on stockroom shelves. Mandating annual hazard training and record audits keeps everyone alert. Supervisors and staff need real drills on spill response, not just signatures on a form. Manufacturers and supply catalogs can do their part, too, by flagging hazards more clearly and supporting trades with up-to-date disposal contacts.
Community take-back events have worked well for old paint and batteries, so why not for high-risk chemicals? Cash-strapped schools and small businesses especially need access to safe disposal so dangerous shortcuts aren’t their fallback.
Keeping Safety Real
Ignoring chemicals like phenylmercuric pentachlorophenoxide just won’t fly. Following solid handling habits, calling in trained teams for cleanup, and sticking to legal, professional disposal means looking out for coworkers, neighbors, and the environment. That approach feels right because the costs of taking shortcuts show up for years to come.
What are the potential health hazards of Phenylmercuric Pentachlorophenoxide?
Understanding The Chemical
Phenylmercuric Pentachlorophenoxide doesn’t roll off the tongue, but its strong-sounding name matches the risks it carries. This chemical was once a go-to for industrial uses, especially in paint, fabric, and wood preservatives. Chemists designed it to keep mold, bacteria, and other pests away—a job it did well. Over time, though, workers and scientists began noticing risks that far outpace any benefits.
Mercury: Playing With Fire
Mercury lurks in the name. That’s the red flag. This heavy metal has caused countless problems for people working in factories or living near contaminated sites. With every touch or breath, mercury slips through the skin or lungs. In my own experience talking to factory maintenance teams, I heard stories of headaches, tremors, and persistent fatigue—classic symptoms of mercury exposure.
Chronic mercury poisoning attacks the nervous system. Focus gets lost easily, hands start shaking, and memory fades. Some workers shared that trouble sleeping and muscle weakness followed them home. Medical journals back them up: mercury exposure can hurt kids’ brain development, slow growth, and damage kidneys.
A Toxic Tag Team: Phenylmercuric Compounds and Pentachlorophenol
The story doesn’t end at the word “mercury.” This chemical also contains pentachlorophenol, another product with a bad reputation. Pentachlorophenol can cause skin rashes, sore throats, and coughing. At high levels, it wrecks the liver and can trigger a toxic reaction throughout the whole body. Animal studies even connect long-term exposure to cancer, which lines up with findings from workers exposed for years on end.
The worst risk comes when both toxic agents combine. Imagine workers sanding or scraping paint, releasing fine chemical dust that they inhale over hours in a tight workspace. Each breath brings both mercury and pentachlorophenol—two heavy hitters with a track record for harming people.
Why Regulation and Replacement Matter
Government agencies recognized the pattern of harm decades ago. The U.S. bans the use of phenylmercuric compounds in interior paint for this very reason. The European Chemicals Agency warns users about the danger. The Consumer Product Safety Commission lists it among restricted substances. Still, I’ve heard from some small shops and importers who stumble across old stock or mislabeled batches. These end up forgotten on back shelves, where risks sit unchecked.
No easy fix exists for workers who already breathed in months—or years—of fumes. The most effective solution: replace risky chemicals with safer alternatives. Paint and preservative industries have found substitutes that break down easily and lack the toxic punch of mercury or chlorinated compounds. In my time working on workplace safety programs, I’ve seen a clear drop in health complaints wherever safer chemicals replaced older, toxic products.
Education brings the first line of defense. Workers and users need to know which labels signal danger and how to store or dispose of hazardous products. Health monitoring for those who have handled these substances should be standard. Regular blood and urine screening catches mercury buildup before symptoms spiral.
Health Above All
Working with chemicals means taking their effects seriously. The stories coming out of the 1970s paint rooms should remain in industry memory. Phenylmercuric Pentachlorophenoxide brings real health hazards that reach far beyond the factory gate. Replacing it and similar toxic compounds with less harmful agents protects not just those holding the brush or mixing tanks, but entire communities downstream and downwind.
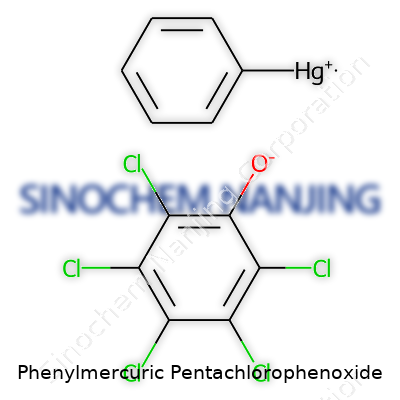
| Names | |
| Preferred IUPAC name | Phenylmercury 2,3,4,5,6-pentachlorophenoxide |
| Other names |
Merfen PMPCP |
| Pronunciation | /ˌfiːnaɪlˌmɜːrˈkjʊrɪk ˌpɛnkloʊrəˈfɛnɒksaɪd/ |
| Identifiers | |
| CAS Number | 13477-40-6 |
| Beilstein Reference | 1858733 |
| ChEBI | CHEBI:53156 |
| ChEMBL | CHEMBL2107629 |
| ChemSpider | 151172 |
| DrugBank | DB13520 |
| ECHA InfoCard | 100.017.159 |
| EC Number | 230-320-2 |
| Gmelin Reference | 8720 |
| KEGG | C18796 |
| MeSH | D010662 |
| PubChem CID | 25835 |
| RTECS number | OV9625000 |
| UNII | 68W76G00K1 |
| UN number | UN2020 |
| CompTox Dashboard (EPA) | DTXSID6020148 |
| Properties | |
| Chemical formula | C12H5Cl5HgO |
| Molar mass | 500.16 g/mol |
| Appearance | White or yellowish powder |
| Odor | Odorless |
| Density | 1.98 g/cm³ |
| Solubility in water | Insoluble |
| log P | 0.5 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 14.0 |
| Basicity (pKb) | 8.94 |
| Magnetic susceptibility (χ) | -84.0e-6 cm³/mol |
| Refractive index (nD) | 1.632 |
| Viscosity | Viscous liquid |
| Dipole moment | 2.96 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 509.9 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -174.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | −561.3 kJ mol⁻¹ |
| Pharmacology | |
| ATC code | S02AA07 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or absorbed through skin; causes burns; may cause mercury poisoning; very toxic to aquatic life. |
| GHS labelling | GHS02, GHS06, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H300 + H310 + H330: Fatal if swallowed, in contact with skin or if inhaled. |
| NFPA 704 (fire diamond) | 3-2-3-🌐 |
| Lethal dose or concentration | LD50 (rat, oral): 25 mg/kg |
| LD50 (median dose) | LD50 (oral, rats): 41 mg/kg |
| NIOSH | 'SB6125000' |
| PEL (Permissible) | 0.1 mg/m3 |
| REL (Recommended) | 0.1 mg/m3 |
| IDLH (Immediate danger) | IDLH: 2 mg/m³ |
| Related compounds | |
| Related compounds |
Phenylmercuric acetate Phenylmercuric borate Phenylmercuric chloride Phenylmercuric nitrate Phenylmercuric oleate |

