Mercuric Oxalate: Looking Beyond the Lab Bench
Tracing the Arc from Discovery to Modern Use
Mercuric oxalate might not spark excitement outside chemistry circles, but stepping back into history, it’s clear the compound’s discovery wasn’t just a result of idle curiosity. Centuries before digital databases and glove boxes, researchers chased new reactions, guided by hand-written notes and a real risk to themselves. Chemists noticed long ago that mixing mercury salts with oxalate solutions formed a white, sometimes yellow-tinged substance. Early work often tangled with guesswork, since modern analytical techniques hadn't been developed. Benches lined with beakers and the smell of chemicals remind me of research stories from departmental archives, where the smallest observations opened gaps in conventional wisdom. Later advances allowed scientists to pin down the formula—HgC2O4—a moment that shifted it from a mysterious residue to a compound with a defined identity.
Understanding Mercuric Oxalate’s Place in Chemistry
Simple at first glance, mercuric oxalate is anything but basic. Once isolated, the white, faintly yellow crystalline powder turns heads in more niche labs. Mercury’s notorious toxicity shadows its chemistry, demanding respect, experience, and concrete safety measures. Touching or breathing in its dust can cause real harm, a constant reminder that the science has consequences. Its chemistry starts with its formula: one mercury atom, paired with the oxalate ion, gives plenty of chance for reactions with acids, heat, or sunlight. Stability doesn’t rank high on its profile—in time, exposure to light can break it down, releasing mercury vapors, which no one wants to take lightly in the workplace.
Getting Down to the Essentials: Properties and Handling
You find physical and chemical properties shaping every decision, from the tools you choose to how you clean up afterward. Mercuric oxalate crystals don’t dissolve well in water, a detail that helps in separating them out from solution. Heat is a different story; even slight warmth can prompt the release of mercury, pushing labs to monitor temperatures closely. Handling viable samples means using proper labeling, ensuring hazard warnings are visible, and storing them where light or accidental bumping can’t spark trouble. Every bottle calls for a trained eye, a steady hand, and a healthy respect for its dangers. No matter how controlled the environment, a moment’s oversight can cause more problems than a full week’s research can fix.
How Chemists Make It: Preparation Insights
Preparation of mercuric oxalate can act as a rite of passage for those willing to dance with danger for the sake of science. Most methods echo variations on mixing a soluble mercury salt—like mercuric chloride or nitrate—with oxalic acid. My time watching these processes taught me the importance of patience: slow addition, steady stirring, and the use of protective barriers. Once the yellowish precipitate forms, careful washing and drying follow. Trying to rush will lead to contaminated, impure crystals. Reagents must be pure, glassware clean, and waste managed under the strict eye of hazardous materials protocols. Letting down your guard, even for a second, betrays both the compound’s hazards and your colleagues’ safety.
Chemical Reactions and Tweaks
Mercuric oxalate isn’t a dead end in chemical synthesis. Under the right conditions, it undergoes various chemical transformations—thermal decomposition yields mercury, carbon dioxide, and carbon monoxide. Throw in acids, and you see the liberation of oxalic acid and other mercury salts. Organic chemists, always seeking better routes to tough molecules, sometimes use it as a catalyst or as a reagent to introduce mercury into more complex structures. Each pathway holds its own risks, but the transformations offer glimpses into new reactivity and reaction control. Watching these reactions unfold under the right fume hood reminds you how fine the line is between curiosity and caution.
Names in the Literature
Science deals with synonyms as much as with structure, and mercuric oxalate bears several. Some call it mercury(II) oxalate, others note its Latin flavor as mercurii oxalas. Each label points back to the same challenging compound, adding to the confusion when diving into older scientific literature. What matters is remembering that no matter its name, the risks and research carried out with it deserve consistency and clarity, not shortcutting due diligence in handling or reporting.
Navigating Safety Without Shortcuts
Mercury’s hazards have shaped entire chapters of laboratory training. Every encounter with mercuric oxalate demands not just gloves and goggles, but a culture that refuses to cut corners. Eating, drinking, or even pipetting by mouth—once common in older labs—don’t belong near this substance. Spills force instant response, with powder scattered by air currents threatening both people and equipment. Safe disposal, air circulation, and detailed logs aren’t just paperwork—they’re built on hard lessons from earlier mishaps. Those entering the field hear stories of forgotten mercury drop traps and the real dangers of chronic exposure that linger long after the lab has gone dark for the night. These protocols weren’t born out of bureaucracy but out of experience, injuries, and long-term studies linking mercury to neurological problems and kidney damage.
Applications: Where Theory Meets Reality
Despite its hazards, mercuric oxalate found uses that made risks feel more like calculated decisions. Analysts reaching for more sensitive detection methods in early years often used mercury compounds to test for trace metals or organic compounds in water or soil. Inorganic synthesis projects, especially those needing controlled sources of mercury, relied on this compound to kick off chain reactions. Its reliability in certain catalytic settings—where small amounts were key—once made it an attractive option, even as safer alternatives started chipping away at its dominance. None of these applications survives without vigilant oversight; regulators and in-house safety teams set limits that reflect a hard-earned balance between insight and health risks.
Pushing for Progress: Research and Development
Watching the direction of research today, I see fewer students handling mercuric oxalate, but investigators still look for what its unique chemistry can reveal. Current studies try to harness its properties without exposing workers or the environment. Green chemistry initiatives nudge labs toward safer, more sustainable reagents. The challenge involves finding alternatives that deliver similar results without legacy hazards. Researchers develop encapsulation techniques, digital simulations, and in-line detection to minimize direct handling. Projects funded under environmental and occupational safety initiatives take these stories to heart, using past experience not as a reason for nostalgia but as fuel for smarter protocols.
Toxicology: Learning the Hard Way
Mercury’s reputation as a heavy metal toxin overshadows every discussion of mercuric oxalate. Chronic exposure stories, tracked over lifetimes, tell of tremors, memory problems, and kidney damage. Regulatory agencies stepped in with blood level monitoring, restricted use, enforced fume hoods, and called for the development of less hazardous substitutes. Data from long-term health studies forced industry and academia to adjust, rethinking how compounds like this fit into broader standards for workplace health. Modern safety checks trace their roots back to chilling reports of mishandling and slow poisoning—not abstract statistics, but real stories of loss and missed warning signs. Changes come slowly in science, but the cumulative effect of those safety bulletins, lawsuits, and evolving regulations built the world where every beaker matters as much as every person holding it.
Looking Ahead: Future Directions and Choices
As regulatory schemes grow tighter and innovation drives new directions, mercuric oxalate’s future hangs in the balance. Demand falls anywhere people can use safer compounds or automated processes. Some branches of physical chemistry and materials science still draw on its reactivity, so legacy stocks get used with greater care and even more redundancies in safety. Education now leans on virtual demonstrations, videos, and strict oversight when actual samples feature in the teaching lab. I see a future where its direct handling fades, replaced by molecules designed for both curiosity and peace of mind. Old habits may linger in places struggling to afford newer options, but change always starts with awareness and shared responsibility across generations of chemists. This compound isn’t just a line in a catalog; it’s a reminder that science grows not just by finding new answers but by respecting the lessons that come with each risk taken and each problem solved.
What are the main uses of Mercuric Oxalate?
Laboratory Applications
Most people outside of chemistry forget how many substances have a history far messier than any high school science class would admit. Mercuric oxalate fits that bill. It’s best known among researchers as a reagent in chemical experiments—researchers value it for specific redox reactions, particularly because it can act as an oxidizing agent. Its ability to bump electrons around unlocks routes for studying reaction pathways. Many older protocols in inorganic chemistry lean on it for making other mercury compounds, where it acts as a handy intermediate.
I’ve seen its use mostly in specialized studies—nobody turns to mercuric oxalate for routine tasks. Unlike sodium chloride or even potassium permanganate, it doesn’t show up in every lab drawer. Its toxicity demands care. Many chemists I know reach instead for safer items, but for experiments investigating mercury’s properties or decompositions, mercuric oxalate has stuck around. It decomposes when heated, producing carbon monoxide, carbon dioxide, and mercury vapor, making it useful in demonstrations of thermally driven breakdowns or in advanced coursework.
Historical Presence in Analytical Chemistry
Through much of the twentieth century, analytical chemists used mercuric oxalate for detecting and quantifying certain organic compounds. Its reaction with specific functional groups lets researchers identify and separate substances, offering a sharp tool for root-cause investigations. In my own academic training, I’ve spotted old manuals recommending it in protocols for confirming the composition of complex mixtures, capitalizing on its reactivity. Times have changed, and alternative reagents that present less risk to people and the environment have replaced it in most labs.
Bridge to Photographic Technology
Not many folks know mercuric oxalate once played a niche role in photography. Before smartphones, the chemistry behind photographic images saw constant tinkering. Historical records show it formed part of processes aimed at producing images with high contrast or experimenting with alternative photographic prints. These methods used its sensitivity to light and its ability to produce metallic mercury on exposure. Few modern printers rely on it any longer, but it’s fascinating to see the evolution of image technology and which chemicals have faded into obscurity.
Safety and Environmental Concerns
Mercuric oxalate comes with big red flags. Mercury compounds carry health risks—chronic exposure or improper handling can cause nerve damage and other serious problems. Laws in America and across Europe now restrict use to trained professionals with proper containment, and disposal follows tight rules. I always stress to students and colleagues that old school chemistry sometimes left a trail of environmental headaches, and part of responsible science means learning from those mistakes. Innovations today steer research away from toxic substances like mercuric oxalate toward greener, safer options.
Finding Alternatives
Modern labs look for replacements for historical reagents like mercuric oxalate whenever possible. Green chemistry pushes for safer and more sustainable methods, both to protect workers and to limit impact on the planet. I’ve watched research culture shift—chemists scrutinize old protocols, rewrite experiments, and train new generations to think about the whole lifecycle of a substance. While the story of mercuric oxalate includes scientific advances, it serves as a reminder that innovation needs to value health and safety at every step.
What safety precautions should be taken when handling Mercuric Oxalate?
Respecting the Risk
Mercuric oxalate rarely comes up in everyday conversation, but in labs and chemical plants, its dangers get real attention. Older chemists and safety trainers always share stories of accidents. Just a whiff of mercury vapor from spillage or a mix-up can cause lasting harm. Smarter safety starts by accepting this chemical doesn’t forgive mistakes—exposure leads to serious kidney, lung, and nervous system damage. Oxalate compounds have their own risks, like burning skin and messing with body chemistry. Treating this stuff lightly is like jumping a fence marked “biohazard”—the consequences stick with you.
Ventilation and Containment
The first job is controlling where those particles or vapors might travel. If the work area lacks good ventilation, you’re asking for trouble. Mercury salts don’t just poison through touching the skin—the dust can coat anything, and the vapors escape whenever you heat, grind, or even move the container wrong. Fume hoods with tested airflow ratings aren’t just for show. Anyone in the room needs to know how airflow works and recognize if the hood alarm sounds off. Bagging the material at every transfer limits spills. Label spills drills help everyone stay sharp for the real emergency.
Personal Protection Isn't Optional
Compliance means zero shortcuts in the lab. Nitrile gloves, disposable lab coats, and face shields all play specific roles. Cotton and standard latex tend to soak up mercury compounds, creating hidden threats. Splash goggles protect eyes from oxalate burns. Working in teams, watching each other’s technique, and always wearing respiratory protection when powders move should become muscle memory. Practiced hands always check gloves and ties before opening bottles, and switching out contaminated gear on the spot keeps mercury out of lunch and car handles.
Safe Storage and Preparation
Storing mercuric oxalate away from other chemicals—especially organics, metals, and acids—prevents surprise reactions. Locked safety cabinets marked with hazard signs mean everyone knows where to expect trouble. Pre-weighing outside the main workspace, possibly in secondary containment, limits the odds of contamination. Regular inventory checks also catch any old bottles slowly degrading. Anyone who prepares the material should keep detailed process logs to catch patterns and changes in reaction outcomes.
Waste Doesn't Disappear
Every scrap, powder, or contaminated glove should go directly into mercury-safe containers. Waste streams get logged and moved with disciplined regularity. Sending anything down the sink, even trace amounts, taints plumbing and the broader water supply. Most communities require chemical waste contracts, and real responsibility means personal follow-up until the material has been fully treated or encased. I’ve seen whole labs shut down over a forgotten mercury spill that nobody cleaned up quickly.
Training Pays Off
A manual on the shelf won’t protect anyone. Group training breaks down each hazard and lets folks practice emergency techniques under supervision. Practicing spills, reviewing case studies, and quizzing on reactions grow real awareness. Rushed jobs put everyone at risk, so supervisors and senior staff need to build in time for careful work. Fresh faces learn best by watching deliberate, cautious routines—if someone shrugs off goggles or skips the hood, it sets back the whole team.
Treat Every Step as Essential
Safety with mercuric oxalate isn’t a checklist to rush through. It comes from a culture of looking out for each other, treating the chemical with the seriousness it demands, and keeping every step in the open. Consider it a group promise—everyone leaves healthy, every day.
How should Mercuric Oxalate be stored?
Risk Factors That Demand Caution
Mercuric oxalate brings its own set of safety concerns. It reacts violently with heat, friction, and shock. You might find it in research labs, niche chemical manufacturing, or educational settings. Every story I’ve heard from seasoned chemists points to the same lesson: don’t underestimate it. Any accident involving mercuric oxalate leaves a mark, whether through toxic mercury exposure or property damage. A close friend once handled similar compounds too casually; his carelessness resulted in a small but costly fire. That experience made me rethink every routine in the lab.
Why Environment and Security Matter
Mercuric oxalate breaks down under hot or bright conditions, releasing toxic mercury vapor, oxides of carbon, and corrosive fumes. Even a brief temperature spike in storage can set off a chain reaction. Keeping it cool means choosing a dry, well-ventilated space, out of sunlight. Specialized refrigerators with strict temperature controls help keep it stable. Good ventilation isn't just a bonus; it removes any fumes that leak from aging bottles or faulty seals. I’ve seen labs that neglect airflow, only to get stuck dealing with headaches, unexplained odors, or sudden alarms.
Containers Built to Last
Ordinary plastic never cuts it. Only airtight glass or chemically stable containers stop moisture and air from speeding up decomposition. A clear, bold label is essential—no smudged handwriting or cryptic abbreviations. Mixing up bottles could mean disaster. In my early days, a mislabeled reagent led to hours of cleanup and a lingering chemical stench. I learned the hard way: every cap needs checking, every label needs clarity.
Location Is Not Just a Detail
A secure chemical cabinet with a lock keeps both unauthorized hands and accidental jostling at bay. Heavy shelves, mounted firmly to the wall, hold the weight without tipping over. Positioning counts for a lot. I avoid placing sensitive chemicals above eye level, since reaching for them can jar containers or create spills in a split second. Segregating reactive chemicals solves half the problem before it starts—never store mercuric oxalate near acids, bases, or flammable solvents. A little extra space between containers cuts cross-contamination risks.
Staff Training and Emergency Readiness
Classic lab training covers safe handling, but drills and refreshers make a bigger impact. New team members need hands-on guidance for every routine. Up-to-date material safety data sheets should sit close at hand, not buried in a digital folder or dusty binder. I’ve seen teams freeze up during a spill; clear, practiced protocols make all the difference. Spill kits stocked with mercury-absorbing compounds, gloves, face shields, and disposable aprons belong close to storage areas—not across the hall, not on another floor. Quick response stops most accidents from getting worse.
Real-World Solutions
A thoughtful storage plan pays off. Chemical inventory management software tracks shelf life, runs audits, and reminds staff when supplies reach expiration. Real-time monitoring alerts you if fridge temperatures creep outside the safe range. Investing in high-quality PPE and keeping detailed incident logs keeps everyone accountable. Open conversations about near-misses build a culture of safety, not blame.
Mercuric oxalate demands respect, not fear. Careful storage, training, and teamwork reduce risks, letting research run smoothly without putting health on the line.
What are the potential health hazards of Mercuric Oxalate?
Understanding What You're Dealing With
Mercuric oxalate doesn't sound friendly, and it isn’t. This chemical combines mercury, a powerful neurotoxin, with oxalic acid. Most people never come across this substance outside of labs or industrial work, but those who do can't afford ignorance. Its risks aren’t just theoretical—stories from experienced chemists and old safety data sheets hold enough warnings to make anyone pause before handling it.
Mercury Exposure: More Than Just a Warning Label
Mercury poisoning sits behind a lot of tough stories in industrial history. Coughing, tremors, memory problems, and kidney issues have plagued those working with quicksilver—an older term for mercury compounds. Researchers focus on methylmercury and elemental mercury most often because people eat fish or spill liquid mercury in homes. Mercuric oxalate, though, operates in a similar, harmful way: the body absorbs mercury compounds through the skin, lungs, or gut. Once inside, it damages the nervous system, targeting movement, speech, mood, and even hearing.
Health agencies like the CDC and NIOSH rank all forms of mercury as acutely toxic. There’s no harmless level of exposure, especially for kids or pregnant women, whose brains and nervous systems are still developing. The oxalate part adds its own dangers—oxalic acid, in high enough doses, damages kidneys and can kill cells by binding up calcium. That turns possible spills or ingestion events into medical emergencies.
Everyday Risks and Real-Life Consequences
The trouble with mercuric oxalate runs past its chemical makeup. Dust, vapors, and spills don’t kindly stay put. Poor ventilation in an older lab? Accidental drop on a warm hotplate? These events lead to airborne particles that workers can breathe in before anyone notices there’s a problem. Some chemists share stories of skin rashes or nosebleeds from lab accidents; not everyone walks away unharmed.
Long-term risks grow every time someone brushes off the need for gloves or a fume hood. Heavy metals like mercury build up in the body. Over years, exposure turns into chronic illness—shaky hands, mental fog, sleep issues, irreparable kidney damage. For lab workers or factory staff who aren’t told the risks, it might take years before a sharp doctor puts the symptoms together.
Change Comes From Awareness and Better Practices
No one grows up hoping to deal with mercury exposure. The best defense starts with honest training. Good chemical handling habits come from mentors in the lab—the people who double-check the MSDS sheets, insist on real gloves, and teach younger staff to work under hoods every time. Medical checkups for those handling mercury compounds aren’t overkill; they save lives by catching health problems before they get out of hand.
Labs and factories lower risk with proper ventilation, strict storage, and real accountability. Up-to-date spill kits, proper hazardous waste disposal, and clear emergency instructions help the most during accidents. For years, swapping mercury-based reagents for safer alternatives has proven possible for many industrial processes—the switch takes commitment, not magic.
Accountability Goes Both Ways
Nobody should work with dangerous chemicals alone. Safety officers, managers, and employees each play a part. When people speak up about hazards and push for better gear, the risk to everyone drops. Respect for mercuric oxalate isn’t about paranoia—it’s about basic decency, common sense, and knowing that everyone deserves to go home healthy.
What is the proper disposal method for Mercuric Oxalate?
Mishandling Chemicals Has Real Consequences
Experience in the chemistry lab teaches plenty about the risks of certain substances. Mercuric oxalate throws a double punch: mercury and oxalate both threaten health and the environment if ignored. Mercury in any form, especially inorganic compounds like mercuric oxalate, brings out the worst of hazardous waste—plaguing nervous systems, waterways, and even the air. Mistakes happen, but a chemical like this rarely gives second chances. Stories of improper chemical disposal stick in memory because the cost shows up in contaminated water or wildlife suffering. So, treating mercuric oxalate as just another jar in storage invites trouble.
Throwing It Out Is Never an Option
No responsible scientist dumps mercuric oxalate down the drain or tosses it in the trash. Doing so risks local drinking water and can damage municipal wastewater treatment systems. Many community water supplies already struggle to keep mercury out; extra load from careless disposal makes the problem worse. New research shows mercury buildup sneaks up in riverbeds and migrates through the food chain, so it doesn’t vanish with a quick flush.
Laws Set Clear Boundaries
Environmental regulations come down especially hard on mercury compounds. In the US, the Resource Conservation and Recovery Act (RCRA) classifies mercuric oxalate as hazardous waste. Fines for illegal disposal can reach tens of thousands of dollars—universities and companies have faced these costs for skipping the disposal process. Similar strict rules apply in Europe and Asia, emphasizing collection, containment, and proof of responsible disposal.
The Right Way Goes Beyond the Lab
Emptying vials or cleaning glassware used for mercuric oxalate starts with labeling everything. Unlabeled waste gets lost, and lost containers threaten everyone nearby. Facilities with hazardous waste programs gather chemical waste in sealed, compatible containers—usually high-density polyethylene or glass, because other plastics break down when exposed to mercury salts.
Trusted waste companies pick up and transport the containers for treatment and disposal. Staff at those companies don full protective gear and treat the waste in ways that capture mercury, often through chemical precipitation, specialized filtration, or even distillation techniques that recover elemental mercury. Collected mercury goes to secure storage or is converted into forms less likely to enter the eco-system again.
Reducing Risk Starts With Smart Procurement
Working with mercuric oxalate makes anyone think twice about how much to order for experiments or projects. Keeping only what’s needed, and switching to safer reagents when possible, slices future disposal headaches and reduces the risk of accidental exposure. Some companies and universities have adopted zero-mercury policies—encouraging alternatives and offering training in safe storage and disposal practices.
Hazard Awareness Saves Lives—and Land
Years spent in both teaching and research labs taught that no policy replaces basic caution and education. Meeting with Environmental Health and Safety officers turns out to be just as important as the science itself. Disposal may seem like a chore, but mishandling a compound like mercuric oxalate means taking a gamble no lab should risk. Best practice is always clear: treat every container with respect, document every gram, and let the professionals handle what’s left.
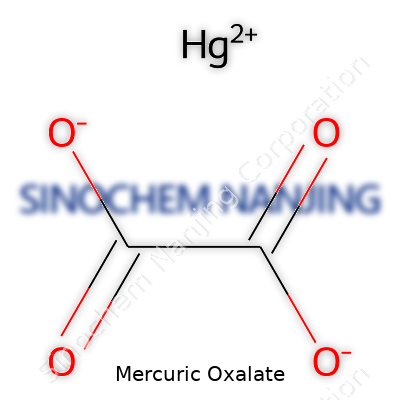
| Names | |
| Preferred IUPAC name | mercury(II) ethanedioate |
| Other names |
Oxalic acid, mercury(2+) salt (1:1) Mercury(II) oxalate Dioxalatomerury Mercury oxalate Mercury(2+) oxalate |
| Pronunciation | /ˈmɜː.kjʊ.rɪk ˈɒk.sə.leɪt/ |
| Identifiers | |
| CAS Number | 593-86-2 |
| Beilstein Reference | 1711464 |
| ChEBI | CHEBI:31780 |
| ChEMBL | CHEMBL1201607 |
| ChemSpider | 20205621 |
| DrugBank | DB01444 |
| ECHA InfoCard | 100.998.595 |
| EC Number | '231-871-7' |
| Gmelin Reference | 79065 |
| KEGG | C18738 |
| MeSH | D008629 |
| PubChem CID | 24406 |
| RTECS number | MA8050000 |
| UNII | T3W5XF3EXW |
| UN number | 1629 |
| CompTox Dashboard (EPA) | DTXSID8044424 |
| Properties | |
| Chemical formula | HgC2O4 |
| Molar mass | 320.40 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 6.28 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | -4.37 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 3.45 |
| Magnetic susceptibility (χ) | -41.7·10⁻⁶ cm³/mol |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 291.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -567.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -576 kJ/mol |
| Pharmacology | |
| ATC code | V09AA11 |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled; causes damage to organs; very toxic to aquatic life with long lasting effects. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | Hazard statements: H301, H330, H373, H410 |
| Precautionary statements | P260, P262, P273, P280, P301+P310, P302+P352, P304+P340, P305+P351+P338, P308+P310, P314, P320, P330, P501 |
| NFPA 704 (fire diamond) | 3-2-2-POISON |
| Autoignition temperature | 130 °C (266 °F; 403 K) |
| Lethal dose or concentration | LD50 oral rat 35 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 12 mg/kg |
| NIOSH | MU8225000 |
| PEL (Permissible) | PEL: 0.1 mg/m³ |
| REL (Recommended) | 0.05 mg/m³ |
| IDLH (Immediate danger) | 5 mg/m3 |
| Related compounds | |
| Related compounds |
Mercury(I) nitrate Mercury(II) nitrate Mercurous chloride Mercuric chloride |

