Mercuric Naphthalenesulfonate: Looking Beyond the Formula
A Chemical with a Heavy Legacy
Many people glance over a name like mercuric naphthalenesulfonate without a second thought, but chemists who have worked in both analytical and industrial settings recognize that this compound tells its own story. Back in the early twentieth century, researchers explored organomercury chemistry with a spirit of curiosity that would shock today’s more regulation-bound labs. Mercuric salts had already shown usefulness in catalysis, preservation, and even healthcare, and someone realized that tethering mercury to a naphthalenesulfonate backbone let the element mix with aqueous solutions much more reliably than with simple inorganic salts. Industrial chemists found their workhorse. Over time, the unique behavior of this compound kept it relevant — sometimes not for benign reasons.
What Sets It Apart
Chemically, mercuric naphthalenesulfonate combines the heavy metal potency of mercury with the aromatic, polar character of naphthalenesulfonate. In practice, this means a solid, slightly off-white or yellowish powder, stable enough to store away from direct sunlight, yet reactive in the presence of strong reducing agents or complexing ligands. With a molecular weight topping 400 g/mol and solubility profiles that fit plenty of water-based applications, users get a potent mix of persistence and mobility. You find these properties at work in its main uses, from textile processing to specialty intermediates for organic syntheses. Its strength lies in its ability to bond metals as a chelating agent — and anyone who’s ever had to clean up a mercury spill knows just how powerful those bonds can be.
No Shortcuts on Safety
It only takes one story about broken glass pipettes and mysterious neurological symptoms to teach researchers about the risks carried by mercury-based compounds. Mercuric naphthalenesulfonate is no exception. Its toxicology reads like a warning sign: powerful enough to disrupt biological processes, persistent enough to resist breakdown in soil and water. Regulatory agencies around the world call out organomercurials due to their bioaccumulative nature and their risk profile. Long after a drop hits a bench top, you’re dealing with a compound that can enter water tables or expose anyone in the vicinity to danger — and direct skin, eye, or inhalation contact can cause acute and chronic symptoms. Laboratories that still permit its use invest heavily in glove boxes, fume hoods, and dedicated cleanup procedures. Engineers, chemists, and health physicists cooperate on strict protocols that enforce proper storage, labeling, and disposal, often at high cost. These requirements don’t just protect workers; they protect the environment where unnoticed spills could linger for decades.
Research and Rethink: Adaptation under Fire
Chemical companies and research institutions steadily phased out mercury compounds in many roles, shifting instead to less hazardous alternatives. Yet, a handful of applications still lean on mercuric naphthalenesulfonate because today’s chemistry hasn’t fully delivered replacements with the same effectiveness. In certain analytical techniques, as a specific catalyst or reagent, or in niche organic syntheses, this compound pulls off transformations that remain tough for greener chemicals. Such stubborn persistence means research into its toxicity and long-term effects continues. Scientists keep looking for ways to mitigate risk — through encapsulation, limited-scale synthesis, or even digital transformation workflows that replace wet-lab work with computer simulations.
A Look Toward Tomorrow’s Laboratory
I’ve worked with colleagues who felt real nostalgia for the robust chemistry of mercury, but nobody misses the time spent filing paperwork for its use or the late nights making sure a jar didn’t leach vapor into a crowded storeroom. The trend will keep moving away from mercury compounds as new legislative controls arise and newer molecules answer the same challenges with less collateral damage. Funding now goes to green chemistry initiatives, and universities push students to think beyond the solution in a flask — to ask where every molecule goes after it leaves the lab drain or factory outflow. The future for mercuric naphthalenesulfonate isn’t shaped by technological marvels but by a growing awareness that discovery carries responsibility. With every advance in substituting safer compounds or improving industrial processes, users close the chapter on hazardous chemicals that once propped up entire industries. For young researchers reading an old protocol and seeing the word “mercuric,” the question today isn’t “Can I use this?” but “Why did we ever have to?”
What is Mercuric Naphthalenesulfonate used for?
The Chemistry at Play
Mercuric naphthalenesulfonate doesn’t show up in most people’s everyday conversations, but this compound has popped up in specialty chemical supply lists for decades. Chemists and lab staff have used it for one strong reason: its effectiveness as a biocide and catalyst. I learned about these compounds the hard way, spending late nights in an under-equipped lab, mixing and matching catalysts to get slow reactions to run faster and more reliably. Mercuric naphthalenesulfonate stood out for its ability to push reactions forward, especially reactions where other catalysts fell flat.
Where Labs and Factories Turn to It
This chemical plays a role in organic synthesis, specifically as a catalyst. In practice, that means someone trying to bond difficult molecules, especially those involving aromatic rings like naphthalene, often reaches for it. It speeds up sulfonation reactions and delivers consistent results. Pharmaceutical researchers who worked in old-school labs (and even some who still do) have leaned on mercuric naphthalenesulfonate for these kinds of transformations, especially if precision matters. Its biocidal activity also gets used in treating industrial water systems. If you’ve spent time around old factory equipment or water towers, you know that unchecked microbial growth gums up the works. I remember tracing a persistent clog to bacterial slime in a heat exchanger; the answer was a short, controlled dose of an organomercury biocide. It worked—at a cost.
What the Risks Look Like
Effectiveness comes with some real baggage. The toxic nature of mercury compounds gives health and safety teams a nightmare. The facts speak for themselves: exposure to these materials, even in small quantities, carries risks inside the lab and beyond. Mercury compounds disrupt nervous system function. Long-term exposure piles up, hitting the brain and kidneys. I’ve watched colleagues wear full protective gear just to handle a small bottle. Even today, there’s real anxiety when disposing of waste from reactions involving mercuric naphthalenesulfonate, because environmental persistence means mercury doesn’t just vanish. It seeps into soil and water, and eventually ends up in the food chain.
Getting a Handle on the Problem
Everyone I know in chemistry circles who takes this risk seriously has shifted toward alternatives. Real experience in the lab taught me that sometimes a less toxic catalyst just needs an extra hour or a few tweaks in the process. Technology keeps moving forward. Catalysts made of copper, iron, or even enzymes now fill gaps where mercury once ruled. Regulatory bodies have noticed the dangers too. In the United States, using mercury-based compounds triggers hazardous waste protocols. Many countries clamp down on imports, storage, and disposal. This kind of pressure pushes industries and academics toward greener chemistry even faster.
Our Responsibility in Modern Chemistry
Mercuric naphthalenesulfonate marks an uncomfortable chapter in chemical history. The lesson from decades of use and hard-won lab experience: if a chemical delivers strong results but endangers lives or the environment, it’s time to find a better way. Healthier alternatives now offer safer choices for researchers and manufacturers. The push for strict safety rules and greener chemistry isn’t red tape—it’s common sense born from real risk. Taking chemicals like mercuric naphthalenesulfonate off the roster wherever possible keeps people safer and the planet cleaner.
Is Mercuric Naphthalenesulfonate hazardous to health?
What We Know About Its Health Risks
Mercuric naphthalenesulfonate isn’t a name tossed around outside of chemical circles. Dig a little deeper and its dangers seem less distant. This compound mixes heavy metal mercury with naphthalenesulfonic acid, so you get a toxic cocktail right out of the lab. Mercury on its own spells trouble for the nervous system, kidneys, and even the lungs. Compounds like this one don’t lose that edge just because they’re blended with something else.
You’d think strong chemical controls would keep people safe from exposure, but real-life stories paint a messier scene. Everyone hears about big mercury spills or disasters, but small exposures hide in industrial settings: manufacturing, research, maybe even in art or academic labs if someone’s careless. Mercury vapor does not need an open wound to get inside the body. Skin contact or breathing in the dust is enough to cause real harm.
Firsthand Concerns: Workplace Realities
I spent part of my early career working next to a chemical storage room. Over time, I noticed more of my co-workers asking about headaches, nausea, and "how safe the air is." With mercury compounds, those warnings are too easy to ignore. Even when folks knew about mercury poisoning, signs were written off as stress or an old building’s bad ventilation.
Heavy metals stay in the body. It isn’t just about what happens today. Years down the road, early symptoms show up as movement problems or brain fog. The CDC reports that mercury compounds, both organic and inorganic, build up in tissues. The EPA agrees: there’s no "safe" touch or whiff, especially with daily or even monthly exposure.
Gaps in Regulation and Transparency
Rules do exist, but not every country or factory puts worker safety first. In some places, paperwork trumps real risk reduction, so folks who handle mercury compounds wear gloves but get no real respiratory protection. Training works if folks believe there’s a danger: otherwise, risk is brushed off until something major happens.
Disposal brings another headache. Mercury doesn’t disappear. It comes back in water, food, or soil. The World Health Organization tracked persistent mercury in riverbeds and found links with health problems in communities who never handled chemicals themselves. The chain is long, and no step breaks cleanly from the next.
Simple Solutions, Lifesaving Impact
Silence and ignorance make compounds like mercuric naphthalenesulfonate more hazardous. Clear labels, targeted education, and better access to safety equipment all matter. Substitution sits near the top of any responsible list—use something less toxic if it gets the job done. Regular medical exams for workers, real-time air monitoring, and real consequences for companies that cut corners add another layer of protection.
Not everyone gets a choice about what they work with. That’s why honest health information and strong safety culture matter most. Compounds with mercury are risky, and pretending otherwise only makes things worse. Taking the dangers seriously and speaking up is often the difference between long-term health and a lifetime of regret.
What are the storage conditions for Mercuric Naphthalenesulfonate?
Understanding the Risks
Mercuric naphthalenesulfonate doesn’t show up in everyday chemistry classes. This compound falls under those materials that come loaded with real hazards—its mercury content places it firmly on the list of chemicals demanding careful storage. Run-ins with unsafe storage rarely end well. I’ve seen labs forced into deep cleaning or even shut down after a single careless spill of similar mercury compounds. That sticks with you and pushes one to respect what proper handling can prevent.
Temperature and Compatibility
A room with a steady, cool environment makes the safest home for mercuric naphthalenesulfonate. Temperatures up to 25°C work, but heat accelerates chemical breakdown or boosts vapor risks. Mercury contamination brings health consequences—tremors, memory loss, or kidney damage—for anyone who’s exposed over time, so locking down volatility at the storage stage always takes priority. Direct sun and temperature swings make things worse, and in corners with poor airflow vapors can build up fast.
Moisture and Chemical Neighbors
Damp rooms and open containers can create trouble. Water reacts with many mercury compounds, and although naphthalenesulfonate isn’t as reactive as some, a leak transforms a controlled risk into an unpredictable mess. Solid, sealed containers rated for hazardous chemicals shut out moisture. On top of that, keeping mercuric naphthalenesulfonate away from acids, bases, or organics like solvents proves crucial. I’ve watched a single forgotten solvent bottle near mercury salt storage trigger cleanup calls—mixing accidents can spark fires, release toxic fumes, or damage storage rooms down the line.
Ventilation and Security
Good air flow isn’t just a guideline—it saves headaches and hospital visits. A chemical storage cabinet designed for volatile compounds pulls vapors away from people and out of the work space without sending them into the hallway or outdoors unchecked. During an old project, a single sniff of mercury vapor left a lab supervisor feeling ill for hours; after that our department never skimped on ventilation improvements.
Labeling and Access Control
Legible labels and chemical inventories pay off when quick action counts. A labeled container never leads to guessing games or mix-ups. Letting only trained people near mercury compounds blocks a world of trouble before it starts. Lab managers often oversee a secure, keyed-cabinet system. Every year, missing inventory crops up somewhere—a lock and a sign-in log cut down on costly mistakes and keep storage legal.
Disposal and Emergency Preparation
Safe storage ties directly into disposal plans. Collection containers and clear spill kits belong close by. My experience with school labs taught me the value of regular drills and up-to-date mercury spill kits, since panic can turn a small leak into a major contamination. Emergency contacts hung on the wall save precious minutes if something does slip.
Steps Toward Safer Practices
The safest storage results from good training and steady review of procedures. Pairing chemical-specific safety data sheets with clear signage, limiting storage amounts, and running spot inspections all help prevent problems. Serious labs build these habits into routine, cutting down on risks not only for their team but for the neighborhoods around them.
How should I handle spills or exposure to Mercuric Naphthalenesulfonate?
Mercuric Chemicals Bring Real Risks
Mercuric naphthalenesulfonate isn’t something most people want to mess with. This compound often turns up in specialized labs, old industrial products, and chemistry classrooms. Its mercury base makes it dangerous—direct contact or inhalation brings serious risks for nerves, kidneys, and long-term health. Years ago, I helped clean a university lab after a student knocked over a bottle of a similar mercury compound. The clean-up rattled everyone. One thing became clear: staying prepared and taking spills seriously cuts the risk down to size.
Immediate Steps Matter Most
Any direct exposure to mercuric naphthalenesulfonate counts as an emergency. Don’t attempt to “just wipe it up.” Opening the windows or turning on a fan only spreads toxic dust and vapors. Safety at the moment comes down to acting without hesitation. Evacuate anyone who isn’t part of the clean-up. Avoid touching the spill with bare hands. If any gets on skin or clothing, a safety shower—no exceptions. Even a few seconds of delay can mean health problems that last a lifetime.
Personal Protection Is Non-Negotiable
Anyone dealing with the spill or contaminated space needs gear that really protects. Lab coats or Tyvek suits, gloves rated for heavy chemicals, goggles, and—this is key—a quality respirator designed for mercury vapor. The wrong mask gives a false sense of security. In my own experience, gear sometimes gets overlooked during a rush to solve the immediate problem. Better to lose five minutes getting ready than to regret a rushed decision for years.
Containing and Removing the Spill
Keep the area as small as possible. Barriers work—absorbent pads, spill socks, and plastic sheeting help stop any movement of the chemical. Avoid dry sweeping or vacuuming; both spread dangerous dust. Mercury-specific spill kits make the difference. The kit should include powder or paste that binds the mercury for safe collection, plus heavy-duty waste bags approved for hazardous waste sites. Local environmental regulations set requirements for how mercury waste gets disposed of, so stay connected with your building’s environmental health officer or local authority. In my time working with a clean-up crew, I saw firsthand how these folks understand both the letter of the law and the practical steps that protect workers.
Medical and Environmental Follow-Up
People exposed—even by mistake or through indirect contact—must get checked by professionals. Testing isn’t optional. Mercury attacks slowly, but damage grows over weeks and months. Environmental wipe tests help make sure the space becomes safe for work again. Don’t trust smell or appearance. Tools like cold vapor analyzers and surface samples give results that guide the all-clear. Our crew saw some sites shut down for weeks, but health always came first.
Preparation: The Best Solution
Training and spill drills change accidents from disasters into manageable situations. Clear labels, solid protective equipment, and up-to-date spill kits deserve attention every month—not once a year. Workers who handle mercuric compounds should know not just the basics but the details about how these chemicals affect the body and what an emergency looks like. Vigilance and humility—a willingness to ask for help, admit a mistake, or push back on shortcuts—play a big role in real safety. Avoiding the event is always better than cleaning up after it.
Where can I purchase Mercuric Naphthalenesulfonate?
The Search for an Obscure Chemical Raises Bigger Questions
Mercuric naphthalenesulfonate doesn’t show up on shelves at your local hardware store. It’s not the kind of chemical you find by calling a neighborhood supplier. Tracking it down points toward much larger issues around access, safety, and responsibility in today's global marketplace.
Why Is This Chemical Hard to Find?
Mercuric naphthalenesulfonate falls into a class of compounds that combines a mercury salt with a sulfonated aromatic compound. Mercury, as most people know, brings a heavy legacy: neurological toxicity, strict environmental rules, and tough workplace health standards. Mercury compounds raise red flags for regulators worldwide. Many countries have clamped down on sales, with international treaties like the Minamata Convention pressing firms to restrict distribution and find safer alternatives.
Online, a quick search leads to specialty chemical suppliers—often not in your home country—who vet buyers with long forms and requests for end-use declarations. This hardly resembles ordering standard lab chemicals or reagents. I’ve seen chemists wait months for government permissions or get turned down entirely, especially if they can’t prove a clear, regulated purpose.
Responsibility Beyond Regulations
Mercury compounds like this one still see use in specialized research, or sometimes niche industrial catalysts. But the cost and risk can outweigh the benefit. The dangers of mishandling these chemicals are profound, touching not just workers but communities around plants or disposal sites. I’ve worked with research teams who struggle with legacy contamination years after a project wraps. Cleanup never comes cheap or easy.
Supply chains for these restricted chemicals often lack transparency. Illegal trade pops up, driven by demand from labs underfunded or cut off by sanctions: that’s a slippery slope. Governments around the world crack down hard when confiscating mercury sales that don’t follow legal paths, and penalties make the message clear: responsibility takes priority over convenience.
Lessons from Chemical History
History brims with cautionary tales about neglecting chemical risks. From Mad Hatter’s disease due to mercury to forgotten stockpiles leaking toxins, the topic of mercury always brings real consequences. Groups like the EPA, European Chemicals Agency, and WHO have specific lists and controls in place. There’s a reason why chemical suppliers demand so much paperwork and only deal with licensed research groups.
Safer Alternatives and Open Science
Scientists and engineers often succeed by replacing hazardous substances with safer options. Green chemistry isn’t a philosophy; it’s standard best practice in modern industry. In sectors where an old mercury compound once played a role, newer solutions constantly emerge. Collaborating with universities, government labs, and experienced safety officers can help pinpoint better materials or refined procedures.
No question cuts as deep in the laboratory as human health. Chasing mercury-based additives or specialty catalysts—just because “it’s what we’ve always used”—shortchanges everyone involved. Reconsidering that legacy, while keeping up with current regulations and seeking green solutions, represents real progress in science and industry.
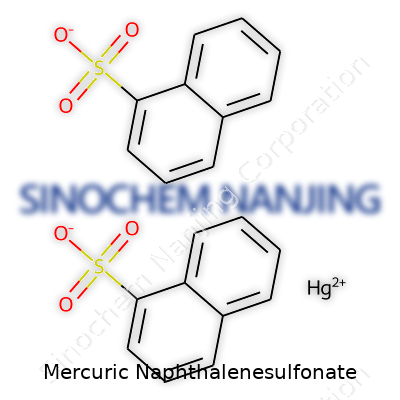
| Names | |
| Preferred IUPAC name | mercury(2+) naphthalene-1-sulfonate |
| Other names |
Naphthalenesulfonic acid, mercuric(2+) salt Mercury(II) naphthalenesulfonate |
| Pronunciation | /ˈmɜːrkjʊrɪk næfˌθæliːnˈsʌl.fə.neɪt/ |
| Identifiers | |
| CAS Number | [3935-95-9] |
| Beilstein Reference | 2041027 |
| ChEBI | CHEBI:131539 |
| ChEMBL | CHEMBL510460 |
| ChemSpider | 17232205 |
| DrugBank | DB06777 |
| ECHA InfoCard | 100.011.086 |
| EC Number | 218-439-6 |
| Gmelin Reference | 67968 |
| KEGG | C18633 |
| MeSH | D008493 |
| PubChem CID | 24861906 |
| RTECS number | PB6125000 |
| UNII | 6XVA6D2595 |
| UN number | UN2996 |
| Properties | |
| Chemical formula | Hg(C10H7SO3)2 |
| Molar mass | 596.89 g/mol |
| Appearance | White powder |
| Odor | odorless |
| Density | 2.35 g/cm3 |
| Solubility in water | Insoluble |
| log P | -0.43 |
| Vapor pressure | Negligible |
| Acidity (pKa) | -3.0 |
| Basicity (pKb) | 6.56 |
| Magnetic susceptibility (χ) | -62.0e-6 cm³/mol |
| Refractive index (nD) | 1.680 |
| Dipole moment | 6.72 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 293.2 J·mol⁻¹·K⁻¹ |
| Pharmacology | |
| ATC code | S01AX01 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled or in contact with skin; causes damage to organs; environmental hazard. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H300 + H330: Fatal if swallowed or if inhaled. |
| Precautionary statements | P260, P262, P264, P270, P273, P280, P301+P310, P302+P352, P304+P340, P305+P351+P338, P308+P313, P314, P330, P391, P501 |
| NFPA 704 (fire diamond) | 3-2-2-MER |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LDLo (oral, dog): 400 mg/kg |
| LD50 (median dose) | LD50 (median dose): 75 mg/kg (oral, rat) |
| NIOSH | WQ9625000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Mercuric Naphthalenesulfonate: "0.1 mg(Hg)/m³ |
| REL (Recommended) | 0.05 mg/m3 |
| IDLH (Immediate danger) | 10 mg/m3 |
| Related compounds | |
| Related compounds |
Mercurous naphthalenesulfonate Mercuric chloride Sodium naphthalenesulfonate Mercuric acetate |

