Mercuric Gluconate: Examining the Journey and the Road Ahead
Historical Development
Mercury-based compounds carved out a permanent spot in the history of both chemistry and medicine well before modern science set safety standards. In the rush to discover new drugs during the nineteenth and early twentieth centuries, the remarkable properties of mercury salts—including mercuric gluconate—raised considerable interest. Early researchers, often driven by the urgent need to treat infectious diseases, turned to these compounds as possible treatments for everything from syphilis to skin infections. This past holds lessons about both the curiosity driving progress and the dangers we uncover in hindsight. As chemical understanding deepened and toxicity concerns gained attention, the use of mercuric salts in medicine waned. Even so, mercuric gluconate’s unique blend of chemical reactivity and solubility continued to attract researchers in chemistry and, later, in analytical applications, demanding a closer look at both the benefits and risks.
Product Overview
Mercuric gluconate, a salt formed by the combination of mercury(II) and gluconic acid, appears as a pale crystalline powder or as a colorless structure that speaks to chemists, the sort you might find behind the lab bench late at night. Its water solubility and reactivity made it stand out from other mercury compounds, offering a ready source of ionic mercury for specialized lab work. The gluconate part, derived from glucose, isn't chosen for nutrition or health benefit here. Instead, it serves as a stable anchor, letting the mercury ion remain available for reaction without flying off into the ether or sticking to every passing molecule. This mix signals why the compound’s presence in chemical inventories hasn’t faded with time, even as people backed away from medical use.
Physical & Chemical Properties
The physical makeup of mercuric gluconate rarely invites fanfare, but it tells an important story about chemical interaction. Its crystalline nature stands in stark contrast to the heavy, metallic feel of elemental mercury. You hold this powder and see none of the liquid metal’s shimmer or dangerous allure; the risk lies within, tied to its structure. Mercuric gluconate dissolves in water, delivering mercury ions into solution. Chemists know that this trait, more than any flashy appearance, decides its use. In solution, mercury ions display high reactivity, forming complexes and kicking off redox reactions. The compound resists decomposition under mild heating, but breaks down at sustained, higher temperatures—an important detail for anyone thinking about transportation or storage. Its stability depends on conditions: certain acids or bases speed up breakdown, and exposure to air and moisture over time creates invisible shifts beneath the surface.
Technical Specifications & Labeling
Handling mercuric gluconate doesn’t fit the routine profile of most lab work. Labels need to shout hazards, not suggestions. User documentation draws attention to high toxicity and guides strict storage: cool, dry, tightly sealed. Container material can’t interact or degrade—glass or certain plastics survive the test. Regulations in many countries demand clear hazard communication, including the mercury icon and warnings about both exposure risks and waste handling. The priority stays on precision—amount, purity, and the presence of any contaminants shape both safety and results, so every vial carries vital batch data. Over years in labs, working with mercury compounds feels like staring at the past and future of safety culture at once: all those gloves, goggles, and ventilation efforts stem from hard-earned lessons about vapor risks and cumulative exposure.
Preparation Method
Making mercuric gluconate in the lab brings together the hallmarks of classic chemistry. Preparation starts with mercuric oxide or basic mercuric salts, which meet gluconic acid or an equivalent gluconate salt in controlled aqueous settings. The desired compound forms as a result of a double displacement, often requiring careful control of pH to prevent the crash of insoluble impurities. Filtration, concentration, and sometimes gentle recrystallization give a final product soft enough to dissolve yet stable in the bottle. Any slip at this stage—wrong temperature, impure reagents, or oxygen exposure—can mean contaminated end product or hazardous fumes. The recipe reflects generations of improvement, but the need for vigilance has never changed, making it a tough test for both technique and patience.
Chemical Reactions & Modifications
Mercuric gluconate acts as a potent source of mercury ions in synthesis and analytical chemistry, often sparking redox and coordination reactions. The mercury(II) ion, sitting in its oxidized state, reacts rapidly with sulfur-containing molecules—especially thiols—forming bonds central to many detection methods for biologically important compounds. Modifying the gluconate ligand, or swapping out ions in the structure, can shift solubility or reactivity enough to fine-tune performance in specific tests. Efforts to develop less hazardous analogues have focused on tweaking these properties to retain chemical usefulness without quite so much risk, but the luster of mercury’s unique chemistry proves hard to replicate. In every reaction involving mercuric gluconate, those involved balance the desire for accuracy and sensitivity with the persistent need for careful control and cleanup.
Synonyms & Product Names
Mercuric gluconate fills the pages of chemical suppliers’ catalogs under several aliases, often listed as mercury(II) gluconate or simply Hg(C6H11O7)2. Older literature describes it as hydrargyri gluconas or mercuric glucose acid salt. If you’ve scoured medical texts from past eras, you might stumble upon legacy names reflecting dated conventions, highlighting how language evolves alongside chemistry. These synonyms hold more than trivia: searching for toxicology data or regulatory restrictions demands knowing them all, because a safety update in one publication won’t always connect the dots with another under a slightly tweaked name.
Safety & Operational Standards
Working with mercuric gluconate presses safety to the foreground. Mercury toxicity refuses to forgive mistakes: inhaled dust, accidental contact, or improper disposal turns lab work into a public health issue. Personal experience has shown that rigid standards—extraction fans at fume hoods, layer after layer of gloves, strict rules for spill management—keep minor incidents from becoming headline news. Nobody strolls into a workspace with mercury compounds expecting a casual experience. Years of case studies and regulatory action mean labs using mercuric gluconate stick closely to national and international guidance—always recorded, logged, and tracked. Disposal routes require dedicated containers and periodic audits, with waste streams destined for specialized hazardous waste treatment, not the regular trash. Those layers of rules and paperwork aren’t red tape—they’re the boundary between routine research and long-term problems for people and the environment.
Application Area
Most use cases for mercuric gluconate have steadily drifted out of human medicine and into the territory of analytical chemistry, industrial testing, and sometimes environmental analysis. Its effectiveness in precipitation reactions, as an oxidizing agent, and in certain electrochemistry approaches keeps it relevant to specialists running specific protocols. In research labs, it can play a part in detecting or quantifying thiol groups, particularly in proteins or industrial samples, though many scientists look for alternatives when possible. Environmental application sometimes includes testing for similar mercury compounds by using mercuric gluconate as a comparison or control. Regulatory limits on mercury in commerce keep the supply strictly managed, slowing any temptation to use it in new, broad consumer or industrial products.
Research & Development
Innovation in this field focuses on finding ways to keep the power of mercury-based reactions without exposing scientists, workers, or communities to their risks. Alternatives like silver compounds, improved organic reagents, or instrumental approaches—chromatography, for example—compete with mercury for analytical roles. Yet many researchers still investigate mercuric gluconate’s specific chemistry, especially where extreme sensitivity or reliability is required. Efforts in molecular engineering and ligand modification aim to blunt toxicity while preserving unique redox and complexation features, but the shadow of mercury’s past toxicity and environmental fallout looms large. In my own time watching R&D teams address these concerns, the move toward greener, safer chemistry inspires hope, but no single answer ends the story.
Toxicity Research
Scientific study on mercury compounds unfailingly circles back to toxicity. Mercuric gluconate presents no exceptions, with animal and cell studies confirming the harms of chronic or acute exposure. Neurological effects top the list—mercury vapor and ions both cross biological barriers and set off a range of health problems. Kidney function, too, feels the impact of long-term low-dose exposures. These insights have shaped tight controls for workplace exposure: air monitoring, surface testing, periodic health checks, and limits on permissible concentrations. The drive for safer labs—more so, a safer world—springs not from regulation alone but from stories of researchers fallen ill and legacy pollution haunting groundwater and fish for decades. Each outcome, each published study, raises the bar for how thoroughly safety must factor into every stage, from synthesis to disposal.
Future Prospects
Debate around the future of mercuric gluconate blends chemistry, risk, and responsibility. Industries searching for sensitive detection methods won’t turn away from mercury’s sharp chemical bite unless alternatives match its performance. Yet no industry, no research field, wants to shoulder the blame for contamination or worker harm that could have been avoided with forethought and investment. Regulatory pressure ratchets higher as scientists raise red flags about low-level mercury exposure and ecological fallout. My experience watching change happen suggests that shifting entire fields away from mercury compounds requires coordinated funding, improved training, technological advances, and a culture shift valuing safety as highly as chemical novelty. The story of mercuric gluconate stands as a challenge for science: how to honor the tools that shaped progress in an era that insists on smarter and safer choices.
What is Mercuric Gluconate used for?
Mercury in Medicine: A Complicated Story
Mercuric gluconate rarely grabs headlines these days, but it still shows up in some scientific and medical conversations. Its name signals its roots: a compound made with mercury and gluconic acid. People with science backgrounds recognize the red flags that come with mercury. For most of the world, mercury means poison, not healing. So, why does mercuric gluconate matter, and should it be used at all?
Where Mercuric Gluconate Shows Up
Years ago, researchers tried building new drugs and disinfectants using various mercury salts. Mercuric gluconate landed on that list. Some labs experimented with it for its antibacterial and antifungal properties. Mercury compounds can disrupt and kill bacteria. In the past, doctors and pharmacists sometimes leaned on these effects to treat skin infections, wounds, and even eye conditions when other options came up short. At one point, some eyedrops and ointments relied on mercuric compounds to prevent or treat infections, especially before antibiotics like penicillin came along.
But history paints a clear picture: toxicity overshadowed the benefits. Mercury lingers in the body, damaging nerves, kidneys, and organs. As drug safety rules grew stricter, the risks of soft tissue damage, neurological effects, and kidney failure proved unacceptable. Modern medical practice dropped mercuric gluconate from its toolkit almost everywhere. Organizations like the World Health Organization and the US Food and Drug Administration now recommend against using any mercury-based therapy for routine care. In many countries, you’d be hard-pressed to find mercuric gluconate on pharmacy shelves.
Current Uses: More Lab, Less Clinic
Small-scale research still sends for mercuric gluconate now and then. Some scientists study mercury poisoning or test new ways to clean mercury out of the environment. These researchers need to understand how mercury compounds interact with cells and tissues. Today, you’ll mostly find mercuric gluconate in the hands of chemists or toxicologists, not pharmacists. Its use in manufacturing or research is tightly controlled by government rules to limit any exposure to workers or the environment.
I had a brush with this in the lab. Graduate school chemistry courses sometimes brought out small vials of dark, powdery substances—always labeled with big, red warnings. Nobody wanted to work with mercury. Handling it meant layers of gloves, goggles, and strict procedures to keep every drop contained. Everyone in the room knew that the story of mercury’s usefulness belonged to a safer past, not the future.
Safer Solutions: What’s Changed
No one wants to see accidental poisonings repeat. Medicine now uses antibiotics, antiseptics like chlorhexidine, and antifungals that offer the same infection control without permanent damage. The science is clear: alternatives outclass mercury compounds in both safety and results.
Looking ahead, regulation remains key. Health workers and patients deserve clear rules that keep dangerous substances off the shelf. For researchers, small, necessary studies of mercury’s behavior only happen under rigorous supervision. If the goal is to clean up contaminated sites or trace mercury’s effects, it’s worth the hassle. But for medicine, the story’s mostly over and better options exist.
In daily life, avoiding old, mercury-based treatments stays simple: check the labels, ask your healthcare provider, and trust that modern drugs do a much better job without the risk.
What are the side effects of Mercuric Gluconate?
Looking Beyond the Label
Most people don’t think of mercury when flipping through a list of medical ingredients, but mercuric gluconate contains exactly that. For years, health professionals have sounded the alarm about the risk tied to using mercury in any compound—its toxicity isn’t some ancient myth. This particular salt, used in some industrial or research settings, brings a heavy load of side effects that should never be downplayed, whether exposure happens through ingestion, inhalation, or skin contact.
Physical and Mental Toll
Getting mercuric gluconate in the body is bad news. Nausea, vomiting, and a burning sensation inside the mouth or throat hit soon after contact. The stomach cramps up. Long-term or large-dose exposure can wreck the digestive tract and kidneys. Many who have worked with mercury compounds have shared how their hands shook and their minds fogged up, not realizing the cause right away. It attacks the nervous system—tremors, mood swings, memory slips, and vision changes show up after enough exposure. In severe cases, people lose coordination and experience hallucinations.
Skin doesn’t get off the hook either. Touching mercuric gluconate can bring rashes or more severe eruptions. It eats away at the tissue, and the body struggles to clear it out. It’s not about a one-off allergic reaction; it’s a full-body threat that lingers.
Kidneys: Bearing the Brunt
Doctors monitor the kidneys closely for mercury exposure for a reason. This compound damages the filters in the kidneys, causing protein to spill into the urine and eventually leading to kidney failure. There are cases where people who handled mercury for years without proper protection ended up needing dialysis before retirement. No job perk is worth that.
Mercury in the Bloodstream
Signs show up in the blood, too. Anemia and low white blood cell counts can leave people vulnerable to illness. Instead of fighting off a cold, the immune system gets stretched thin, sometimes attacking the body’s own tissues. The science backs this up—mercury disrupts cell production and survival.
No Safe Threshold
It doesn’t take much to tip the scales with mercuric gluconate. There’s no real safe dose for humans. Even small exposures, especially in people with other health problems, can cause a chain reaction. As a parent, the thought of mercury exposure near children is chilling—kids absorb toxins faster and react worse than adults.
Poison control centers often struggle to keep up with misinformation about mercury compounds. People sometimes believe a small amount isn’t dangerous, but published toxicology reports and patient stories show the opposite. For example, the Agency for Toxic Substances and Disease Registry has consistently warned about mercury’s chronic and acute effects.
Moving to Safer Alternatives
Most places have already phased out mercuric compounds from routine products and kept them out of consumer hands, though research labs and some countries may still handle them for specialized use. Proper education, strict workplace safety rules, and routine screening for those at risk can help lower numbers of accidental poisoning. Families, schools, and health workers need to stay alert and keep an open line with poison centers.
Safety begins with information. Mercury doesn’t belong in the cabinet—or in people’s bodies. Every step taken to avoid these compounds, whether big or small, adds up to healthier communities.
Is Mercuric Gluconate safe for humans?
The Basics of Mercuric Gluconate
Mercuric gluconate belongs to a group of chemical compounds containing mercury. Some people have come across it in lab settings, where it plays a role in certain experiments. Most folks haven’t seen it in their daily lives, and there's a good reason for that. Mercury in any form brings a lot of risk. Its history stretches back centuries, with stories of its use in old medicines and even as a component in some cosmetic products. Science has moved forward, but questions about safety linger, especially with less common forms like mercuric gluconate.
Mercury and Human Health: A Deep Concern
Over time, researchers have collected a worrying pile of evidence about mercury’s dangers. Exposure to mercury compounds causes serious neurological problems, kidney failure, developmental issues in children, and damage that doesn’t always repair. My own background in chemistry—pulling long hours in university labs—taught me early on about safety protocols for mercury. Instructors never took shortcuts. No one wanted to witness what mercury vapor or accidental spills could do to a colleague or student.
Mercuric gluconate, like other mercuric salts, contains mercury in its +2 oxidation state. The human body absorbs this form quite efficiently if it gets inside through ingestion, inhalation, or skin contact. There’s no practical difference between mercuric gluconate and other mercury salts—elementally, it’s the mercury doing the harm.
Evidence and Regulation
No reputable health authority, including the FDA or the World Health Organization, allows mercuric gluconate in medicines or food. Research still points to the same list of risks: tremors, memory loss, and even death in high enough doses. The EPA and CDC agree that chronic exposure to inorganic mercury compounds is unsafe, regardless of the chemical group they come from.
There’s a famous case where a mercury compound made its way into an unlabeled folk remedy in a community. Investigators found widespread toxic effects, leading to hospitalizations and long-term neurological damage. Reading through these case reports, I felt shock at how quickly the harm can set in. Even with modern medicine, recovery from mercury poisoning is slow and incomplete.
Mercuric Gluconate in Research—A Double-Edged Sword
Some researchers work with mercuric gluconate in tightly controlled environments. Labs use fume hoods, gloves, and thorough disposal plans to protect people and the environment. Yet, accidents still pop up. There is no industrial or medical application where humans benefit from direct contact or exposure.
On rare occasions, experimental studies appear that test trace levels of mercuric gluconate’s effects on cells or animal models. These don’t change the big picture: toxicologists find mercury's risks outweigh any proposed benefit. No evidence shows that this compound belongs in consumer products or health supplements.
How We Move Forward
Education changes everything. Many poisonings occur from a lack of awareness. Keeping mercury-based compounds out of homes—teaching youth about the dangers—lowers those numbers. Policymakers and healthcare providers must take every opportunity to share clear information.
Even as chemistry keeps advancing and new compounds pop up, sticking with clear safety standards protects lives. If someone stumbles across a bottle labeled "mercuric gluconate," the rule stays simple: step away and leave the handling to professionals. Protecting your health usually comes down to what you choose to keep out of your life, not what a label promises.
What is the recommended dosage of Mercuric Gluconate?
Understanding the Risks of Mercuric Gluconate
Mercuric gluconate, a compound containing mercury, doesn't often land in the pharmacy aisle or on a doctor's prescription pad. Most people have heard about mercury and its dangers, mostly in conversations about fish or environmental pollution. This form, bound to gluconate, carries many of the same risks. The human body can't handle mercury well. Even trace amounts can damage nerves, kidneys, and the immune system. Growing up with family working in industrial labs, I remember hearing plenty of cautionary tales about mercury spills and the headaches that followed — literal and otherwise.
Why There's No "Safe" Dosage
There’s no recommended or established medicinal use for mercuric gluconate in modern healthcare. Medical literature doesn’t support its use. Leading health authorities like the FDA and the World Health Organization agree: mercury-based compounds pose health risks that outweigh any possible benefit. EPA literature confirms that mercury poisoning can develop from even low levels of exposure. Although, in the past, some mercury salts made their way into topical treatments or laxatives, time turned up enough horror stories to end those practices. Poisoning symptoms include tremors, personality changes, memory loss, and kidney failure.
The Dangers of Trying to Dose Mercuric Gluconate
Taking any compound containing inorganic mercury, including mercuric gluconate, tends to do more harm than good. No safe threshold exists for human ingestion. Parents, teachers, and public health workers repeat the same warnings: keep mercury away from the home, the classroom, and especially children. If someone accidentally consumes even a tiny amount, medical texts recommend heading straight to the emergency room. Symptoms may not show up right away, so prompt treatment can prevent lasting damage.
Medical Guidance and Regulations
Doctors don’t prescribe mercuric gluconate, and pharmacies won’t stock it for personal use. In the rare instance of laboratory exposure, professionals lean on chelation therapy—treatments that bind to mercury so the body can excrete it. Even then, recovery can be slow and fraught with side effects. Several countries either ban or strictly control the use of inorganic mercury compounds because of the steep public health costs.
Facts from Trusted Sources
Research available from the National Institutes of Health highlights negative outcomes from mercury exposure, including increased risk for developmental issues in children. A CDC report ties persistent mercury use to avoidable hospitalizations and deaths. The United Nations brought forward the Minamata Convention, aiming to reduce mercury emissions and phase out its use in commercial products.
Solutions: Education and Regulation
People turn to dangerous compounds when access to safe treatments is lacking or when misinformation spreads online. Doctors, educators, and regulators must share accurate information, so no one doubts the risks tied to mercury compounds like mercuric gluconate. Better access to proven treatments, clearer labeling on chemical containers, and tighter controls at the industrial level can keep households and workers safe. Constant reminders—and sometimes loud warnings—build a healthier, more informed community.
How should Mercuric Gluconate be stored?
Daily Risks Can Hide In Plain Sight
If you’ve ever taken a look inside a typical laboratory, you’ll know storage rooms rarely look like the photos in the safety posters. Most chemicals arrive with long safety sheets, labels worn off after trips between benches. Mercuric gluconate—less famous than some other mercury compounds—shares the same dangerous reputation. It isn’t a chemical most people handle without a good reason. Yet researchers, educators, and pharmaceutical manufacturers come face-to-face with its risks more often than most realize.
Ongoing Human Health Concerns
Mercuric gluconate contains mercury. That alone should ring alarm bells. Even at low exposures, mercury can damage the nervous system, kidneys, and developing brains. In my own experience working near mercury salts, the lingering awareness of invisible danger lingers over every bottle. You can’t see the vapor. Spills last for years. Safe storage, done right, can mean the difference between a sterile bench and a health crisis that outlasts a career.
Storing Mercuric Gluconate, Step by Step
This isn’t a matter of habit. With toxic salts like these, facts dictate every step. Secure the bottle in a locked, well-ventilated chemical cabinet. Avoid putting it near acids, bases, or organic materials. Heat brings extra risk: even sunlight sneaking in through a window can speed decomposition or change vaporization patterns. At home, temperature swings might only mean spoiled milk. For mercuric gluconate, this becomes a real safety threat. Room temperature storage works, but cooler, steady conditions lower the risk of accidental volatilization.
I remember a supervisor who drilled one rule over and over: treat every mercury compound like a loaded gun. Keep it clearly labeled. Practice double containment: one closed bottle inside a sealed secondary container, all easy to spot and out of reach from casual hands. If there’s a leak, you need to know before it spreads into the air.
Labels, Communication, and Responsibility
Accidents don’t come with warnings. Mislabeled containers led to two close calls I’ve seen up close—both the result of careless handoffs at shift change. Training falls apart when new staff hear a dozen different names for the same powder. Mercuric gluconate goes by obscure labels in some catalogs. Consistent, legible English names and hazard warnings prevent mistakes. Cross-check what’s written against the safety data sheet every time. Don’t assume anything. Take an extra minute at the end of the day. Those little steps are the most human—and most important—preventative measures we have.
Safer Labs Mean Better Science
Regulations exist for a reason. The EPA lists mercury compounds—including mercuric gluconate—as hazardous waste. Proper records help when labs get inspected, but more crucially, they keep facilities honest. I’ve seen too many corners cut for “efficiency.” Every scientist, technician, and student deserves to work in a space where dangerous compounds don’t become legacy problems.
What works in practice is as simple as it is tough to standardize: locked cabinets, clear procedures, and a culture of caution. Ask questions, read the fine print, and keep that bottle off main shelves unless it’s part of an active experiment. It’s not about paranoia. It’s about respect—for the work, and for your colleagues who share that lab today and years from now.
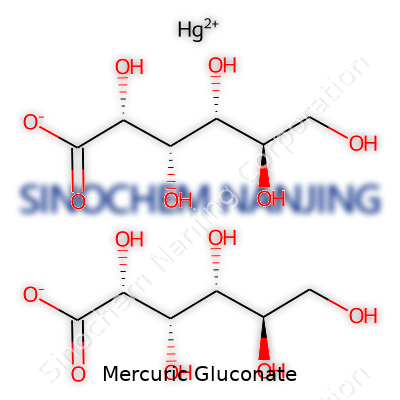
| Names | |
| Preferred IUPAC name | Mercury(II) bis[(2R,3S,4R,5R)-2,3,4,5,6-pentahydroxyhexanoate] |
| Other names |
Mercury digluconate Gluconic acid, mercuric salt |
| Pronunciation | /ˈmɜːrkjʊrɪk ˈɡluːkəneɪt/ |
| Identifiers | |
| CAS Number | 73087-43-9 |
| Beilstein Reference | 75421 |
| ChEBI | CHEBI:75293 |
| ChEMBL | CHEMBL1201607 |
| ChemSpider | 14322058 |
| DrugBank | DB11161 |
| ECHA InfoCard | 100.990.267 |
| EC Number | NA |
| Gmelin Reference | 6767 |
| KEGG | C18797 |
| MeSH | D008567 |
| PubChem CID | 16214105 |
| RTECS number | MW6825000 |
| UNII | 1I88URD85N |
| UN number | UN2025 |
| Properties | |
| Chemical formula | C12H22HgO14 |
| Molar mass | 708.24 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 7.2 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -4.48 |
| Vapor pressure | Negligible |
| Acidity (pKa) | -3.55 |
| Basicity (pKb) | 8.48 |
| Magnetic susceptibility (χ) | -92.0 x 10⁻⁶ cm³/mol |
| Dipole moment | 2.68 D |
| Pharmacology | |
| ATC code | S01AX07 |
| Hazards | |
| Main hazards | Toxic if swallowed, causes severe skin burns and eye damage, may cause respiratory irritation, very toxic to aquatic life. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06, GHS09 |
| Signal word | Danger |
| Hazard statements | Hazard statements: H300 + H310 + H330: Fatal if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | P260, P262, P273, P280, P301+P310, P302+P352, P304+P340, P305+P351+P338, P308+P311, P314, P330, P361, P405, P501 |
| NFPA 704 (fire diamond) | 2-0-2 |
| Lethal dose or concentration | LDLo intravenous rabbit 20 mg/kg |
| LD50 (median dose) | 140 mg/kg (rat, oral) |
| NIOSH | Not Established |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Mercuric Gluconate: "0.1 mg(Hg)/m3 |
| REL (Recommended) | 0.05 mg Hg/m³ |
| IDLH (Immediate danger) | IDHL: 5 mg Hg/m³ |
| Related compounds | |
| Related compounds |
Mercurous gluconate Mercuric acetate Mercuric chloride Mercuric nitrate Gluconic acid |

