Mercuric Arsenate: Examining Its Role Through History, Science, and Safety
Historical Development
Mercuric arsenate has roots that go back over a century, tracing back to an era where chemistry labs often brimmed with curiosity and a risky lack of caution. Early researchers looked to combine the unique features of mercury and arsenic compounds, both notorious for their toxicity yet valued for their peculiar chemical traits. Demand in analytical chemistry shaped initial interest, and for a long time, nobody flinched at glass vials filled with ominous crystals. Textbooks from the late 19th century documented uses and preparations as routine, glossing over long-term health risks. Industrial interests swelled in the mid-20th century as accuracy in chemical detection required more selectivity than standard reagents provided. Looking back, that drive for precision often eclipsed any heed for personal safety.
Product Overview
Mercuric arsenate, known among chemists by its formula Hg3(AsO4)2, sticks out because it combines two heavy-element ions with a strong oxidizing arsenate group. In practice, it shows up largely as a fine powder or salt, forming pale yellow or off-white crystals. Reliable suppliers today emphasize purity, sourcing it mostly for tightly controlled research use. Names like mercury(II) arsenate, mercury arsenate, and even trimeric mercury arsenate pop up in scientific catalogs, though seasoned experts know all these refer to the same risky substance. Nobody outside of specialist work handles it casually, since its hazards overshadow any lingering nostalgia for old-school chemical libraries.
Physical & Chemical Properties
The compound tips the scale as a heavy, nearly insoluble solid at room temperature. Under strong heating, it decomposes, releasing toxic vapors—a grim reminder of its mercury-arsenic backbone. Mercuric arsenate doesn’t dissolve easily in water, but acid brings it into solution fast, which influences how it reacts in analytical and industrial contexts. The ionic bonds, paired with the stubborn stability of the arsenate group, keep the crystals from breaking down under weak solvents or mild heat. Beyond hazards, this unique resilience explains why it became a tool for precise chemical analysis. It keeps its color and structure unless exposed to extremes or strong reducing agents.
Technical Specifications & Labeling
Technical documentation for mercuric arsenate always stresses a minimum assay, usually upwards of 98% Hg3(AsO4)2, with trace impurities tightly controlled. Labels carry a stark warning: toxic if inhaled, swallowed, or contacted. Labeling as per GHS guidelines flags hazards like H300 (fatal if swallowed) and H410 (very toxic to aquatic life with long lasting effects), none of which leave room for misinterpretation. Suppliers punch lot numbers and safety verbiage in bold, meant to jolt even the groggiest lab worker into attention. Storage instructions don’t mince words either: dry, airtight, shielded from any incompatible reagents, and out of reach from untrained hands.
Preparation Method
Laboratory preparation mixes aqueous solutions of mercuric nitrate and sodium arsenate under cold, well-vented conditions. The reaction happens almost instantly, with yellow-white mercuric arsenate precipitating out. Chemists filter the solid, rinse it to limit unreacted materials, and dry it under reduced pressure. Every step invites scrutiny, especially with fumes and contamination risks. Even a minor spill pushes lab personnel to deploy spill kits, evacuate, and don full PPE. Direct handling happens behind shielding, under hoods, and with mercury- and arsenic-rated gloves. Old protocols from pre-war textbooks seem naive, bordering on reckless, compared to today’s rigors.
Chemical Reactions & Modifications
Mercuric arsenate doesn’t play nicely with reducing agents. Exposure to sulfides or hydrogen reduces the mercury component to metallic form, which drives some researchers to explore its redox properties further. Acidic conditions break down the solid to soluble mercuric and arsenate species; alkaline treatment, though slower, also prompts decomposition. Attempts to synthesize organometallic derivatives usually fail, as the parent structure resists modifications common with either mercury or arsenic on their own. In some analytical protocols, chemists rely on its insolubility to separate interfering ions in solution, but these techniques demand extreme vigilance.
Synonyms & Product Names
Known in scientific circles as mercury(II) arsenate or trimeric mercury arsenate, suppliers sometimes list it under archaic indexes as white mercuric arsenate. The overlap between nomenclature can confuse newer chemists or safety personnel. Catalog numbers and CAS registry entries aim to clarify, yet it’s easy to miss distinctions with similar-sounding mercury or arsenic compounds. This makes careful documentation and labelling indispensable to prevent dangerous mixups. Risk-management protocols call for training on synonyms before anyone pulls a container from inventory.
Safety & Operational Standards
Every safety guideline written today for mercuric arsenate draws from hard-learned lessons, often documented with somber case studies. Inhalation or skin contact brings rapid symptoms—neurological effects, kidney damage, and severe gastrointestinal distress. Chronic exposure strips health silently before obvious damage shows up, so nobody familiar with the compound skips personal monitoring or prompt decontamination if exposed. Spills trigger full containment, sometimes with entire lab spaces ruled off-limits until proper cleanup happens. Handling protocols set firm thresholds on allowable quantities, typically measured in grams at most, and mandate double containment within working areas. Emergency procedures require specialized mercury and arsenate neutralization kits, along with dedicated waste collection and offsite hazardous disposal. Trained personnel test workspace surfaces regularly, because any lax attitude courts disaster.
Application Area
Mercuric arsenate found early adoption in gravimetric and colorimetric analytics, offering chemists a reliable standard for detecting certain cations. Its performance made it a staple in classic qualitative analysis, though tighter safety regulations in recent decades slashed its use dramatically. Environmental labs and some niche analytical services still request the compound for testing of interfering ions in complex matrices where few options rival its selectivity. Academic settings now use it mostly as a reference standard under sealed conditions, never as a routine reagent in open labs. Some legacy industrial applications linger, especially where alternatives prove less reliable or where legacy instrumentation ties calibration to mercury-arsenic responses.
Research & Development
Recent research accents detection and remediation instead of field expansion. Most current studies delve into ways to analyze, contain, or neutralize mercuric arsenate before it enters the environment. Advances in analytical chemistry push for replacement reagents or develop sensor arrays to pick up trace amounts in waste streams. Material scientists pursue encapsulation strategies to immobilize residual stocks or investigate chemical pathways to break it down efficiently. I’ve watched a rising focus on portable detection equipment and biosensor technology, which reflects a shift from broad laboratory use to strict stewardship and rapid response to spills or leaks.
Toxicity Research
Toxicity studies around mercuric arsenate have taken grim discoveries and forced the scientific world to approach the compound with heavy respect. Both mercury and arsenic bear a stained legacy of occupational poisonings and environmental disasters—their combination compounds those hazards. Chronic exposure studies show accumulation in vital organs, neurological dysfunction, and a troubling legacy of carcinogenicity and teratogenicity. Testing with environmental models reveals rapid damage to aquatic organisms and slow, but inescapable, buildup in food webs. Researchers now work with only the strictest ethical approvals and limit animal studies in favor of computational toxicology and advanced in vitro cell modeling.
Future Prospects
Looking forward, mercuric arsenate faces near-extinction as a working chemical outside forensic analysis and legacy system management. Regulatory bodies tighten controls every year, and substitutes keep catching up in reliability and selectivity. Work also leans into solutions—rapid detoxification techniques, in-situ destruction technologies, and robust trace monitoring. Green chemistry now dominates strategic planning, focusing laboratory innovation on reagents that match or exceed performance without risk or environmental regret. Education around proper hazardous materials handling has become as important as the chemistry itself. I've seen more institutions treat every gram of mercuric arsenate as a transient relic, prioritized for safe destruction or long-term secure containment over further experimentation.
What is Mercuric Arsenate used for?
Why People Care About Mercuric Arsenate
Mercuric arsenate sits on a short and infamous list of compounds laced with both mercury and arsenic—elements that have left a complicated mark on human health and the environment. In labs and industrial circles, those names bring up two words nobody wants to hear: toxic exposure. People want straightforward answers about what purpose this dual-heavy-metal compound could serve, and why it crops up in scientific records more often than in commercial catalogs. From my own time working with chemical safety databases, certain substances draw more concern than curiosity, and mercuric arsenate fits that bill. It doesn't end up in daily use products or on the shelves of most supply rooms, for good reasons tied to safety and evolving scientific priorities.
The Narrow Paths of Use
Practical uses for mercuric arsenate have mostly come out of old-school laboratory procedures. Chemists have used it as a chemical reagent, especially in early analytical techniques aimed at measuring trace amounts of chloride ions. Years back, detection kits and environmental monitoring sometimes relied on mercuric arsenate as a part of their systems because of its ability to react in predictable ways. This practice tapered off as the risks became more obvious, and as better, safer materials hit the market.
Another historical point: Some researchers explored mercuric arsenate for its potential as a pesticide or wood preservative in the early 1900s, during an era less governed by environmental rules and long before most countries acknowledged the hazards tied to persistent heavy metals. Looking back now, it's hard to justify the risk, knowing what science has uncovered about mercury and arsenic leaching into soils and water—and the slow, painful process of cleaning up after them.
The Weight of Health and Environmental Risks
Mercuric arsenate compounds carry a high profile in health and environmental safety training. Both building blocks—mercury and arsenic—rank among the most notorious poisons found in nature. Nobody needs reminding about mercury poisoning scandals tied to fishing towns or the long epidemics of arsenic-tainted water. When mixed into one molecule, those risks don’t disappear, they often amplify.
Mercury attacks the nervous system and kidneys, especially when fumes or compounds slip into the body unnoticed. Arsenic leaves its own trail of illness: skin lesions, cancer, cardiovascular trouble, and more. Even short spills or workplace exposure can turn routine handling into disaster. Facilities working with mercuric arsenate follow some of the strictest safety guidelines out there—think glove boxes, vacuum systems, air filters, and regular medical check-ups for lab employees. From my old lab experience, nobody enjoys opening a bottle labeled with either element, never mind both at once.
Moving Toward Safer Chemistry
Despite old literature describing mercuric arsenate’s roles, today’s labs and industries rely less on heavy metals and more on alternative compounds. Advanced sensors, green chemistry breakthroughs, and improved detection methods now deliver results without unconscionable risk. Many scientific journals no longer recommend using mercuric arsenate at all. Regulators in the US, EU, and many Asian countries keep a sharp eye on mercury and arsenic. Disposal rules leave no room for shortcuts, with legal requirements for certified hazardous waste handling, long-term monitoring of disposal sites, and ongoing research into decontamination methods.
Modern chemistry aims to serve both innovation and health, not just the bottom line. Substances like mercuric arsenate remind us that progress in the lab also means responsibility—choosing chemicals that not only get the job done, but also keep people, and the planet, safe.
Is Mercuric Arsenate hazardous to health?
Understanding the Chemistry
Mercuric arsenate carries two heavy hitters—mercury and arsenic—inside a single chemical formula. Both of these elements have been making headlines for years, linked to a wide range of health problems. The dangers don't just spring from the laboratory. These compounds show up in industrial settings, sometimes in agriculture, and occasionally in older scientific collections. Whenever they turn up, people start asking questions about risks.
Direct Health Effects
The science is pretty clear. Exposure to either mercury or arsenic can cause real trouble for the body. Mercury focuses its damage on the nervous system, creating problems with memory, movement, and sometimes even mood. Arsenic targets multiple organs. Over time, it damages the liver, lungs, skin, and bladder. Inhaling, touching, or accidentally swallowing mercuric arsenate creates a double threat that health experts treat with serious concern.
Stories from workplaces remind us how dangerous these substances can get when safety steps get skipped. Even brief contact—think accidental skin exposure or inhaling a bit of dust—can start health problems in a way that’s tough to reverse. In my experience covering industrial safety, I have seen how a few overlooked precautions can change lives forever. Prevention always costs less than dealing with the fallout of heavy-metal poisoning.
Community and Environmental Exposure
Mercuric arsenate doesn’t stay where it’s put. It can settle into soil, mix with water, and spread through air. People living near industrial sites face a higher chance of coming into contact with dangerous chemicals. Children play in yards without thinking about what might be under the surface. Families drink water without testing for hidden hazards. In rural areas, old pesticides sometimes leave behind traces of compounds like mercuric arsenate long after the original use has faded from memory.
Research published by the World Health Organization links heavy metals with everything from acute poisoning to long-term cancers. Arsenic, in particular, connects with higher rates of skin and lung cancer. Mercury builds up in the body over time, so people often don’t notice until symptoms take hold. The evidence stacks up: exposure at any age brings more harm than good.
Alternatives and Solutions
Some countries choose a strict approach—outright bans, tough limits on disposal, clear warnings for anyone who might use or store mercury- and arsenic-based products. Even so, older stocks and legacy pollution sometimes slip through the cracks.
Safer substitutes give people practical options. Modern labs and farms can rely on chemicals posing much lower health risks. Responsible companies spend extra up front to avoid the lifelong medical bills tied to mercury and arsenic. I have spoken with small manufacturers who stopped using dangerous compounds once they discovered environmentally friendly replacements. The change rarely breaks the bank and often brings peace of mind.
Regulations need backup. Preventing accidents calls for honest training, clear labeling, and safe disposal procedures. Community groups play a powerful role, too, fighting for testing and cleanup in forgotten corners. Sharing information and facing risks head-on matters more than ever, especially in a time when environmental health is top of mind for families and workers alike.
What are the safety precautions for handling Mercuric Arsenate?
Recognizing the Real Hazards
Mercuric arsenate combines some of the most dangerous elements we can encounter in a lab: mercury and arsenic. Both rank near the top of lists meant to keep folks safe from chemical exposure. Many people overlook how just a small lapse in attention can lead to real harm. On one hand, we’ve got mercury, known for its ability to enter the body through inhalation or even simple skin contact. On the other hand, arsenic attacks organs quietly, leaving lasting damage. The mix of the two multiplies risks in ways that don’t leave much room for error.
Layers of Personal Protection Matter
There’s no shortcut to safety gear. Durable gloves, chemical splash goggles, and solid lab coats keep exposure at bay. I’ve watched someone lose weeks to a careless splash during a cleanup that could have been avoided with the right gloves. Air filters and face shields build another barrier, especially in rooms with no strong airflow. Respiratory protection isn’t a luxury in any lab using mercuric arsenate. The stuff vaporizes fast enough to sneak into the lungs before you realize anything's wrong. Relying on a fume hood isn’t just smart—it's non-negotiable.
Work Habits That Save Lives
Good habits carry just as much weight as gear. Always label containers clearly and check them before opening. No multitasking around this powder. Phones stay far from the bench, food and drinks as well. Washing hands becomes more than a casual hygiene measure. It breaks a chain that could otherwise carry invisible traces to door handles, pens, and eventually, the mouth or eyes. Practicing clean workspaces at all stages saves trouble.
Controlled Storage and Careful Handling
Storing mercuric arsenate in locked, ventilated cabinets keeps it out of unwary hands. I remember one time in school when someone left a bottle on the wrong shelf; it ended up in a room that saw student foot traffic all week. Only group supervisors or those trained on chemical hazards should access these bottles. Even then, double-sealing bottles in shatterproof containers gives a bit more peace of mind. Spilled powder shouldn’t ever get swept up like dust. Instead, using wet methods reduces airborne danger, and observed protocols for hazardous waste turnout make disposal safer for people down the chain.
Emergencies: Response Without Panic
If exposure happens, speed matters. Skin contact? Strip off affected clothes, hit the nearest eyewash or shower, and scrub for at least 15 minutes. Inhalation accidents demand immediate fresh air and medical help. My own run-in with a broken thermometer, much lower risk by comparison, still taught me how quickly mercury can be inhaled. Quick thinking—opening windows, calling for help—makes a tough situation manageable. Emergency numbers should be taped to walls in plain sight. It’s also worth running practice drills from time to time; saving precious minutes in a real crisis holds more value than anyone realizes until it's too late.
Training and Oversight Form the Backbone
Training sessions help everyone remember what's at stake handling materials like mercuric arsenate. Refresher courses and on-the-spot reminders drill the habits into daily routine. Experience doesn’t guarantee immunity from mistakes, but it does make folks less likely to forget important steps. Supervisors who communicate clearly and check on safety rules often build a culture that thinks about protection first, not last.
What is the chemical formula of Mercuric Arsenate?
Chemical Formula and Structure
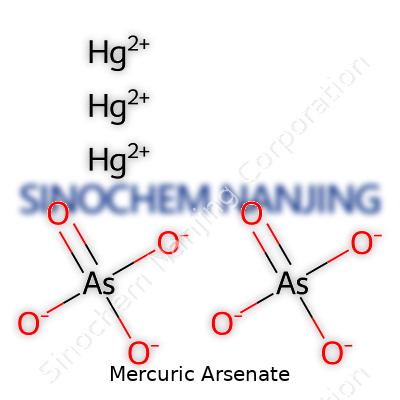
Mercuric arsenate goes by the formula Hg3(AsO4)2. That formula captures three mercury(II) ions bonding with two arsenate ions. With a background in both academic and industrial labs, I've seen how the arrangement of atoms tells you a lot about the reactivity and behavior of a compound. Here, mercury sits in the +2 oxidation state, and arsenic comes in at +5 inside the arsenate anion. In practical terms, understanding this structure matters for anyone working in chemistry, environmental science, or toxicology.
Real-World Applications and Concerns
Mercuric arsenate never shows up on a shelf in a hardware store. You see it mainly in research labs, with careful controls, because both mercury and arsenic pose huge health risks. From personal experience in toxicology coursework and fieldwork, even tiny amounts of mercury compounds can wreak havoc on biological systems — not just humans, but also the water and soil ecosystems around us. Arsenic brings its own baggage, with a long history of poisoning and contamination.
Researchers and environmental scientists track compounds like mercuric arsenate to get a handle on heavy metal pollution. Industries once used mercury and arsenic in pesticides and pigments, and legacy contamination still sits in soils near old manufacturing sites. Knowing the precise formula lets experts model how the compound might break down, how it moves through water or soil, and which safety protocols keep people out of harm’s way.
Why Formula Knowledge Matters
Some chemists might overlook a formula unless they're working directly with a substance. In my experience, the details really count, especially with hazardous materials. Labels and safety sheets demand total accuracy so workers and emergency crews can respond fast if a spill occurs. Hospitals and poison centers rely on this info, too, to figure out the best treatment for exposure. There’s a real-world urgency to getting the details right. I remember one training session with emergency responders, where the formula dictated what gear and neutralizing agents they’d carry to a scene.
Current Challenges and Solutions
Legacy pollution remains a top issue. In some regions, arsenic and mercury compounds still leak into water supplies, causing chronic health problems like nerve damage and cancer. Fighting back takes a mix of good science, clear communication, and political will. Remediation crews dig up and isolate toxic soil. Water treatment systems get upgraded with better filtration, often based on a deep understanding of chemistry. Teaching the next generation of chemists and engineers to respect these formulas can change outcomes for entire communities.
Lab professionals rely on strict guidelines for managing chemicals like mercuric arsenate. Routine audits, up-to-date safety protocols, and transparent reporting help lower the risk of accidental releases. Field testing and advanced analytics have come a long way over the past decade, making it easier to monitor groundwater and prevent long-term harm. Connecting the dots between a simple formula and the health of people and environments shows the stakes involved.
Moving Forward
Accurate chemical knowledge turns into meaningful action in the real world. Keeping mercuric arsenate contained and monitored depends on strict compliance with regulations and ongoing investments in safer alternatives. As a chemist and citizen, I’ve learned that science serves society best when we treat small details like chemical formulas as foundations for bigger solutions, not just trivia.
How should Mercuric Arsenate be stored and disposed of?
A Substance That Demands Respect
Mercuric arsenate doesn’t leave much room for error. It brings both mercury and arsenic into the room—two elements that carry serious health risks. A single careless action can trigger lasting harm for workers, local water sources, and those living nearby. Sitting in labs or storerooms across research facilities and industry, this chemical needs careful hands and a healthy dose of respect at every step.
Choosing the Right Storage Approach
Glass bottles work well for holding mercuric arsenate because the material stands up to strong, toxic compounds. Tightly fitted lids keep fumes from escaping and limit spills. Metal shelves might seem strong enough, but corrosion creeps in when chemicals leak or even when vapors linger. That’s why I always stick with lining cabinets with chemical-resistant coating. Separate the bottle from acids and organic materials—a spill or fume blend causes reactions faster than most people imagine.
Any storeroom housing mercuric arsenate deserves locked access. Only trained staff handle this chemical. This isn’t about building barriers for the sake of it; this is about protecting the janitor, the researcher, and the delivery driver who might not know the risk. Clear labeling matters. Labels don’t get buried under tape or marker. I see people slack off as time passes, so scheduled audits help catch when something slips through the cracks.
Disposal Puts Community at Stake
Rarely does one person face the fallout from mishandling mercuric arsenate. Mercury and arsenic pollute soil and water quickly, especially in high rainfall or porous ground areas. Dumping even a small amount down a drain stirs a chain of contamination that’s tough to reverse, moving from plants to fish to kids who play outside. Most chemical labs partner with licensed hazardous waste service companies, making sure the compound lands in a secure container for transport. These companies use incinerators and chemical treatment sites designed for heavy metals—local landfills won’t cut it, not even for trace amounts.
Gloves, goggles, and heavy aprons sound basic, but too many ignore them, thinking one small session won’t matter. Direct exposure builds up in the kidneys and nervous system, with symptoms showing up months or years later. Cleanup crews use HEPA vacuums and chemical neutralizers, not the everyday mop and bucket. The equipment then goes out with chemical waste, never back to the janitor’s closet.
Accountability Matters More Than Ever
Leaving this task to “the pros” doesn’t let labs or businesses off the hook. State and federal environmental agencies demand strict records for each step—from storage through to the final disposal certificate. This level of oversight can frustrate, but I’ve seen it save organizations huge fines and massive PR disasters after someone takes shortcuts. OSHA, the EPA, and state health departments publish clear guidelines because the consequences stretch far beyond paperwork. Checklists, logs, and outside audits catch mistakes before they spiral.
People who’ve studied incidents with mercury or arsenic poisoning agree: casual or rushed disposal turns a local problem into a public health crisis. Safe storage and disposal practices depend on training, discipline, and a willingness to think beyond the next budget year. A little extra care pays off for everyone who breathes the air or pours a glass of water nearby.
| Names | |
| Preferred IUPAC name | Trimercury arsenate |
| Other names |
Mercury arsenate Mercury(II) arsenate Dimerco Mercuric diarsenate |
| Pronunciation | /ˈmɜː.kjʊ.rɪk ˈɑː.sə.neɪt/ |
| Identifiers | |
| CAS Number | 7784-45-4 |
| Beilstein Reference | 1205742 |
| ChEBI | CHEBI:86341 |
| ChEMBL | CHEMBL1231201 |
| ChemSpider | 22587 |
| DrugBank | DB13765 |
| ECHA InfoCard | 100.029.152 |
| EC Number | 232-064-2 |
| Gmelin Reference | 14820 |
| KEGG | C18797 |
| MeSH | D008618 |
| PubChem CID | 24849 |
| RTECS number | OV9625000 |
| UNII | PJ7M727897 |
| UN number | UN1679 |
| Properties | |
| Chemical formula | Hg3(AsO4)2 |
| Molar mass | 398.41 g/mol |
| Appearance | White powder |
| Odor | Odorless |
| Density | 6.47 g/cm³ |
| Solubility in water | Insoluble |
| log P | -0.26 |
| Vapor pressure | 0 mm Hg (20 °C) |
| Basicity (pKb) | 2.96 |
| Magnetic susceptibility (χ) | −73.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 2.4 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 333.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -910.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -675.4 kJ/mol |
| Pharmacology | |
| ATC code | V09AA11 |
| Hazards | |
| Main hazards | Toxic by inhalation, ingestion, and skin absorption; causes damage to kidneys, liver, and central nervous system; carcinogenic; environmental hazard. |
| GHS labelling | GHS06, GHS08 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H300 + H330: Fatal if swallowed or if inhaled. H350: May cause cancer. H372: Causes damage to organs through prolonged or repeated exposure. |
| Precautionary statements | P260, P262, P264, P270, P271, P301+P310, P302+P352, P304+P340, P308+P313, P314, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-0-☠ |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LDLo oral rat 25 mg/kg |
| NIOSH | WA4500000 |
| PEL (Permissible) | 0.01 mg/m3 |
| REL (Recommended) | 0.01 mg/m³ |
| IDLH (Immediate danger) | 10 mg/m3 |
| Related compounds | |
| Related compounds |
Mercurous arsenate Mercury(II) oxide Mercury(II) chloride Lead(II) arsenate |

