Phenylmercuric Acetate: Unpacking the Details and Issues
Historical Development
Chemists started working with phenylmercuric acetate in the early decades of the twentieth century. Researchers and industrialists saw in it a new way to combat bacteria and fungi that threatened everything from crops to medical devices. To many, mercury-based compounds promised both power and control over biological threats, and by the late 1930s, phenylmercuric acetate found widespread use as a preservative and antimicrobial agent. Its role expanded through the postwar boom, showing up in agriculture as a seed treatment and in medicine for topical antiseptics. Demand spiked before people understood the environmental costs and health risks that come with the more reckless use of organomercury compounds. My own early lab work—decades removed from the initial rush—carried lessons from the public health fallout that always follows unchecked chemical enthusiasm.
Product Overview
Phenylmercuric acetate popped up in labs and clinics for one main reason: it stops bacteria and fungi. That made it attractive for creams, eye drops, and seed dressings. The compound forms a white, powdery solid that blends well into solutions or suspensions, holding its shape until deliberately modified by solvents or reagents. These days, the product comes mostly in well-labeled containers due to the tighter restrictions imposed by environmental agencies and health authorities. Safety data sits front-and-center, as the risks are no longer swept under the rug, and requirements for labeling stretch across countries and regulatory regimes.
Physical & Chemical Properties
Phenylmercuric acetate has a molecular formula of C8H8HgO2 and a distinct crystalline shape. Its white appearance led many in the past to underestimate its potential hazards. The compound dissolves well in polar organic solvents but stays insoluble in water, a fact that controlled both its industrial use and its environmental footprint. Its melting point sits near 178°C, and it breaks down slowly under moderate heat, giving off toxic fumes if not handled properly. The molecule reacts with both acids and alkalis, generating hazardous byproducts if mishandled, so it’s no surprise that storage requirements remain strict. Its physical stability made it useful in products like paints and pharmaceuticals, though the same stability complicates efforts to remove it from contaminated sites.
Technical Specifications & Labeling
Manufacturers today provide rigorous breakdowns for every shipment, with purity levels often exceeding 98%. Labels cover not only chemical identity but also warnings, safety instruction, storage conditions, and emergency procedures. My encounters with this compound start with gloves, goggles, and double-checks at every step since ingestion, inhalation, or skin contact all pose serious risks. Regulations force the hand of anyone who handles phenylmercuric acetate, which must be kept away from acids, bases, and flammable solvents. Even small amounts require disposal in controlled facilities. This kind of oversight keeps both workers and the environment safer than earlier decades, when mistakes traveled quietly out of a factory’s back door.
Preparation Method
The route for making phenylmercuric acetate hasn’t changed much since its introduction. Chemists produce it by reacting phenylmercuric nitrate with sodium acetate. The process takes place in an aqueous environment under controlled temperature to prevent unwanted side reactions. Careful washing and filtration remove impurities. I remember early summers in grad school spent purifying organomercury compounds—and I still recall the sharp smell and constant presence of spill kits nearby. Any error or shortcut can create a hazardous situation, so process instructions leave no room for improvisation. Strict adherence stays the rule in every lab I’ve ever worked in.
Chemical Reactions & Modifications
Phenylmercuric acetate owes its biological activity to how it interacts with thiol groups in proteins, breaking down enzymatic function in bacteria and fungi. It can undergo transacetylation or exchange reactions in the presence of various nucleophiles. In the presence of strong acids, it forms phenylmercuric chloride, a compound with its own toxic legacy. Oxidizing agents degrade it further, but not completely, and residual mercury persists, often migrating through ecosystems after disposal. My environmental chemistry colleagues face headaches when tracking these breakdown products in contaminated soils and waters—a burden that underscores just how persistent these compounds can be outside lab settings.
Synonyms & Product Names
If you ever glance through old industrial catalogs, phenylmercuric acetate appears under a stack of synonyms: PMA, acetoxyphenylmercury, and even phenylmercuric ethanoate. I’ve seen suppliers in Europe refer to it as “Mercury, acetate (acetoxy)phenyl-” depending on the regulatory agency or language. These names confuse even experienced researchers, and mismatches still pop up in ordering systems and literature. Careful documentation saves both time and safety, and I’ve learned to double-check chemical synonyms anytime I’m working with mercury-based products.
Safety & Operational Standards
Every decade brings fresh stories of mercury exposure and its aftermath. Phenylmercuric acetate carries risks well understood by anyone who’s spent time reading case studies or safety bulletins. Acute exposure can cause irritation, headaches, and damage to the central nervous system, while chronic contact results in tremors, cognitive problems, and even kidney failure. Strict ventilation, closed systems, and glove boxes dominate any workspace dealing with this compound. Labs enforce real-time air monitoring and mandatory medical checkups for workers. Spills require chemical neutralizers and immediate response—no shortcuts here. My early training drilled these lessons in through repetition and real-world demonstration, not just signs on a wall.
Application Area
Once, phenylmercuric acetate saw huge use in seed-treating, lumber preservation, and as a preservative in cosmetics and medicines. It played a role in eye drops that lasted longer on shelves and paints that resisted mold for years. As toxicity data mounted, these applications narrowed considerably. Health and environmental agencies across North America and Europe banned or severely restricted it in agriculture and consumer goods by the late twentieth century. Some industrial processes still use it for specialized antiseptic and antifungal tasks, but the trend pushes hard toward safer, non-mercury alternatives. My own practice grew more cautious year by year, as new data emerged from regulatory reviews and toxicology studies.
Research & Development
By the 1970s and 80s, toxicologists worked at a furious pace to uncover the full scope of risks tied to phenylmercuric acetate. New detection technologies, from GC-MS to biosensors, helped trace even tiny concentrations in soil, water, or human tissue. Chemists and material scientists tried to tweak the structure, looking for less hazardous analogues or safer breakdown routes. While incremental gains appeared, no mercury-based antimicrobial has shaken off its risk profile completely, and most progress now comes in finding non-mercurial alternatives. Green chemistry teams continue exploring substitutions in both agriculture and medicine—a trend I’ve watched accelerate with each new regulatory crackdown. Still, old contamination remains a tough legacy to manage.
Toxicity Research
Ingesting or inhaling phenylmercuric acetate devastates the body’s nervous system and kidneys. It crosses biological membranes with ease and binds irreversibly to proteins, blocking enzyme action critical to life. Repeated exposure leads to bioaccumulation. Studies from the U.S. EPA and European Union show mercury residues move up food chains, collecting in fish and mammals and eventually back to humans. Efforts to map these pathways rely on years of fieldwork by ecologists and public health professionals. Friends of mine in toxicology can point to regions where careless disposal of mercury compounds left entire communities battling the long-term effects: learning disabilities, cognitive decline, and immune suppression. This research pushed governments and companies to impose ever-tightening controls, with modern scientists forced to justify every use on strictly quantified risk-benefit grounds.
Future Prospects
Most chemical companies and researchers consider phenylmercuric acetate a relic. Safer, greener antimicrobials now take its place. Remediation of contaminated sites remains expensive and time-consuming, eating up grant money and government budgets alike. Ongoing research into bioremediation and chemical breakdown keeps a small scientific community busy, but nobody expects a rebound in phenylmercuric acetate demand. The conversation now focuses on monitoring, clean-up, and alternatives. Education and strict regulation stop new problems from emerging, and old mistakes yield lessons in humility and caution for anyone entering the fields of chemistry, medicine, or agriculture. The future asks for new compounds built on sustainability, not just power, and every generation of scientists must remember the stories behind compounds like phenylmercuric acetate.
What is Phenylmercuric Acetate used for?
How Phenylmercuric Acetate Found Its Place
Up until a few decades ago, the average person might have crossed paths with phenylmercuric acetate without even realizing it. This compound made its way into homes, hospitals, and agriculture fields. In the world of preservatives, nothing worked quite like it. Its job: stop bacteria, fungi, and other pests in their tracks. Products needed longer shelf lives, and farmers wanted stronger crops. For paint manufacturers, a dash of phenylmercuric acetate kept cans from turning funky, even after months on shelves. Pharmaceutical companies trusted it to help ointments last longer and stay safe for use.
Fighting Mold and Bacteria, at a Cost
Anyone who remembers old-school latex paints probably remembers the focus on keeping mold at bay. Mold eats paint, walls get ruined, health risks rise. Phenylmercuric acetate answered the call, offering serious protection. Hospitals and pharmacists leaned on it too, especially where sterility mattered. Many eye drops and topical creams relied on its presence to fend off contamination.
Agriculture followed a similar storyline. Farmers needed answers for fungal diseases that threatened food security. Seeds treated with phenylmercuric acetate saw improved chances of sprouting healthy and strong, delivering larger harvests. This chemical delivered solutions that, for a while, looked like a win-win.
Hard Truths: Risks Nobody Can Ignore
Stories about mercury poisoning come up for good reason. Phenylmercuric acetate contains a form of mercury that builds up in the environment and inside living things. This creates a troubling legacy. Chronic exposure to mercury compounds leads to kidney damage, nervous system issues, and even birth defects. Published health studies link this kind of exposure to real, serious symptoms, not just hypotheticals. Fish in polluted lakes show high mercury levels, and families depending on that fish suffer the most.
Governments didn’t look away. By the late twentieth century, health agencies in the United States and other countries started restricting phenylmercuric acetate. The EPA’s Toxic Substances Control Act lists it among chemicals demanding close watch or outright bans. Paints and personal care products featuring it fell out of favor, with safer options rising up to replace them. European guidelines go even further, keeping strict limits on all forms of mercury in consumer products.
Exploring Better Alternatives
Building safer lives means retiring old habits. Chemists now focus on preservatives based on less toxic ingredients. Paint and pharmaceutical industries have started turning toward compounds such as parabens and isothiazolinones. These bring fewer health risks but still offer protection against contamination. Farmers have also pivoted, with universities and research institutes sharing new solutions to fend off crop disease.
For anybody working with vintage products, or cleaning up old farm buildings, awareness matters. Treat mystery powders and liquids with suspicion. Regulatory agencies provide advice and disposal services to keep people safe. Educating workers and families about legacy chemicals keeps accidental poisonings off the table.
Accountability and Ongoing Vigilance
Transparency about what goes into paints, crops, and medicines has made a difference. Companies share ingredient lists openly. Science keeps evolving, and what seemed acceptable in past generations comes under fresh scrutiny. Pulling unsafe substances off the market is not about limiting progress—it's about learning from the past and refusing to compromise on health for short-term profits. At the end of the day, the story of phenylmercuric acetate echoes across chemistry, medicine, and agriculture: real safety means constant improvement informed by real evidence.
Is Phenylmercuric Acetate hazardous to health?
Understanding the Problem
Phenylmercuric acetate comes from a group of chemicals containing mercury. Many people come across it in older paint, certain pharmaceutical products, and old lab storerooms. The stuff was used to stop mold and bacteria growth. On a daily basis, I see folks throw out old paint cans or ask about vintage cosmetics without thinking twice about the label. Mercury doesn’t stay in one place – it tends to spread, especially through touch or inhalation.
Health Concerns and What Studies Reveal
Doctors have warned for decades: mercury hurts the brain, the nervous system, and kidneys. Even short-term exposure to phenylmercuric acetate can trigger skin rashes, breathing problems, or eye irritation. Some research, like a 2012 review from the World Health Organization, links even low-dose exposure over time to memory trouble, tremors, and mood swings. I’ve met a few hobbyists who work with old thermostats or electronics and later develop strange symptoms, but rarely connect it to old chemicals.
What makes phenylmercuric acetate risky is how easily it gets absorbed through the skin and lungs. Once inside, mercury builds up. Even at work, many people never realize they’re handling a toxic compound because small amounts rarely carry an odor or warning sign. The US Environmental Protection Agency says there’s no safe level for mercury in any form.
Regulations and Hidden Hazards
Strict laws now control most uses of mercury compounds. The US Food and Drug Administration banned mercury-based preservatives in over-the-counter drugs long ago. The European Union, Canada, and Japan put heavy restrictions on its use in paints, fungicides, and eye drops. Still, older homes and medical collections sometimes keep these products tucked away. Dangerous labeling in forgotten supply closets or flea market items remains a real risk, especially for children and pregnant women who are more vulnerable to mercury’s toxic effects.
Protecting Yourself and the Community
Common sense counts a lot here. I always recommend reading old product labels carefully and never guessing about “mystery” bottles or powders. At our local hazardous waste drop-off, staff wear gloves and masks just in case someone turns in mercury compounds. If you spot a label with phenylmercuric acetate or any mention of mercury, keep it sealed and never pour it down the drain or in the trash. Most cities run special collection events to keep these toxins out of landfills and water supplies.
Doctors need more awareness of chemical risks, too. Cases of mercury poisoning sometimes get misdiagnosed or go unnoticed. A real solution involves better education for everyone. Safe disposal programs work, but only if people know about the dangers lurking in forgotten cabinets or basement shelves.
Trust between scientists, health workers, and regular folks matters a lot. It helps to talk openly about the health effects of chemicals like phenylmercuric acetate, not just rely on warning labels and rules. Old science doesn’t always cover what we know now—mercury-based chemicals affect people in subtle but lasting ways. Listening to the stories of those exposed can push decision-makers to promote safer alternatives for everyone.
What safety precautions should be taken when handling Phenylmercuric Acetate?
Understanding the Hazard
Phenylmercuric acetate shows up in labs and some older industries, but it’s far from benign. I remember the first time I pulled a dusty bottle of it from a storage cabinet during undergraduate research, and the warning labels were hard to ignore. This stuff can hurt your skin, lungs, and nervous system. That’s not fearmongering—check the scientific literature or the CDC’s recommendations, and mercury compounds pop up repeatedly as persistent workplace hazards. I’ve seen cases where poor handling led to compromised health for workers and grim contamination for the environment.
Personal Protective Equipment: The Real MVP
Every time I handled phenylmercuric acetate, rubber gloves never felt like overkill; they felt like insurance. Nitrile gloves fight off penetration, unlike cheaper latex. I bundled up in a lab coat, with every snap button closed up. Splash-proof goggles wrapped tightly around my eyes, since a single drop in the eye could mean a trip to the emergency room. Some colleagues used full face shields in places without good ventilation.
Chemical fume hoods can make a huge difference. Pulling air and vapors away from your face keeps inhalation risks down. A lab director once showed me how a few careless moments at an open bench could send traces of mercury straight up your nose. One sniff isn’t harmless, as long-term exposure can trigger tremors, memory loss, and kidney problems.
Storage—and Respect
Even storage turns risky without attention to detail. Dark, cool cabinets, away from acids and oxidizers, prevent unwanted reactions. Strong, tight-sealing containers, with mercury-specific hazard labels, warn others before they reach for a potentially deadly powder. I’ve witnessed old containers leak, seeping toxic dust onto shelves, so secondary containment—plastic bins or trays to catch spills—makes sense for everyone’s peace of mind.
Spill Management and Waste
Spills need smooth execution, not panic. I practiced mock drills, using mercury spill kits stocked with sulfur powder, special wipes, and a mercury vacuum. Standard vacuums just push the hazard around; they don’t trap it. After cleanup, double-bagged waste goes in a labeled container, then heads to a certified hazardous waste facility. Regular trash won’t cut it.
Training: The Real Safety Net
What stuck with me most from my years in research was how much training kept mistakes in check. Reading protocols isn’t enough. Hands-on supervision with an experienced handler drives home the seriousness mercury demands. Newcomers often think they don’t need supervision, but the risks hide in small lapses—rushing, skipping PPE, or guessing instead of checking.
Bigger Picture: Reducing the Need
Fact is, the best safety comes from not using phenylmercuric acetate whenever possible. Many labs have switched to less toxic preservatives because of environmental and health regulations. Alternatives usually cost more, but no price matches the toll of mercury exposure on employees and their families. I've supported those shifts in my own workspaces—sometimes begrudging at first, until I met a retired chemist who lived with nerve damage from exposure years ago.
Looking back, every safety step around this compound paid off. Gloves, hoods, training—they aren’t just bureaucracy or window dressing. They keep poison from ending up where it shouldn’t. Safety culture starts with respecting how dangerous phenylmercuric acetate can be. Skip that lesson, and the risks become real in a hurry.
How should Phenylmercuric Acetate be stored?
The Risks Demand Serious Respect
Phenylmercuric acetate isn’t your everyday chemical. It’s a mercury compound, and anyone who’s handled mercury—whether breaking an old thermometer or reading toxicology reports on industrial spills—knows the damage it brings. Short-term exposure can hurt lungs, kidneys, and the brain. Over time, small mistakes add up, and workplace carelessness can mean big health costs. Stories from labs and older industrial sites stick with people. Anyone working with or near phenylmercuric acetate has a responsibility not just to themselves, but also to the people they might unintentionally expose.
Real-World Storage: Keeping Problems Contained
Storing phenylmercuric acetate never qualifies as a casual decision. I remember a chemist sharing a harrowing tale about a mislabeled jar that ended up sitting on a dusty shelf until someone finally realized the risk. The key is a system that doesn’t trust luck. Put this chemical in a sealed, airtight container—glass with a secure lid or a chemical-resistant bottle works best. Labels matter: they need to be clear, bold, and durable, listing the full name and warning about both toxicity and mercury content. Even small drips or accidental exposures can linger in a space longer than anyone expects.
Genuine safety shows up where you store it. Phenylmercuric acetate ought to be kept in a cool, dry spot out of direct sunlight. People skip this step too often, and high temperatures just build pressure in containers, or speed up dangerous breakdowns of the compound. I’ve seen storage rooms neglected, with years of heat and humidity buckling old shelves—don't let that be your situation. Avoid contact with acids or oxidizing agents (such as bleach), since dangerous reactions have left more than a few workers in urgent care.
Often, the only thing standing between a safe lab and a disaster comes down to who last checked the storage log, or whether someone insists on routine checks. Frequent audits—actual, eyes-on inspections, not just pencil-whipping checklists—make a difference. Double-check the integrity of containers and monitor for any signs of leaks. I’ve seen small procedures, like secondary containment bins or spill trays, prevent big messes. Handling spills quickly with the right mercury-absorbent materials saves headaches and the environment.
Human Factor: Training Goes Beyond Posters
The culture in a workspace influences how chemicals get treated. In places where workers actually feel responsible for each other, safety starts looking like teamwork. That comes from hands-on training and updates when things change—not just handing out a safety sheet filled with jargon. It helps to have older employees share actual stories—those close calls stick with people better than any rulebook. Health monitoring offers another important layer, because early signs of mercury poisoning don’t always show up on day one.
Looking Forward: Solutions That Stick
Many workplaces still keep procedures on autopilot, assuming “the old way” works. Digital inventory systems and routine training updates help everyone stay on the same page. Sharing incident reports openly prevents repeat mistakes. If your workplace allows it, swap out hazardous compounds where safer alternatives exist. Unsafe shortcuts never pay off. Proper storage, honest communication, and a careful eye save lives and reputations. Chemical safety is personal: every missed step endangers someone.
What is the chemical structure and formula of Phenylmercuric Acetate?
Why Chemical Details Matter
Chemistry shapes daily life and public health in more ways than most notice. Looking at phenylmercuric acetate, you are looking at a compound that played a big role as a fungicide and antimicrobial agent for decades. Its reputation—effective, but flagged for toxicity—comes down to exactly how its chemical structure interacts with living things.
The Formula Up Close
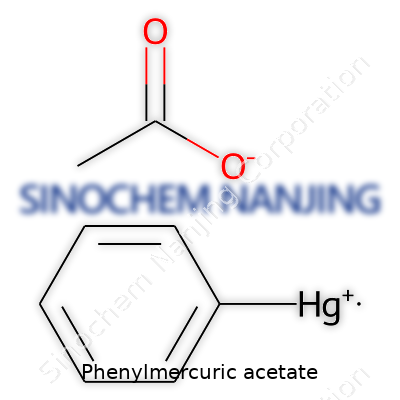
Phenylmercuric acetate carries the formula C8H8HgO2. Dissecting the structure provides a clearer idea of why this compound behaves as it does. The basic anatomy pairs a phenyl group (that’s a benzene ring, C6H5–) directly connected to a mercury atom (Hg). This molecule meets an acetate group (–OCOCH3), which completes the picture.
Lay it out: the mercury atom sits in the middle, bonded to both the phenyl group and the oxygen of the acetate. If you imagine the molecule visually, the phenyl ring anchors at one end, mercury forms the link, and the acetate group branches out, wrapping up the connection. The full IUPAC name reads as (acetato-O)phenylmercury.
Real-World Implications
Reading about molecular formulas starts to mean more after seeing where those details lead us. In college labs, I remember running tests on substances like phenylmercuric acetate, following strict protocols because of the risks tied to each atom on the table. Mercury compounds penetrate cells easily. The phenyl group ramps up the molecule’s lipophilicity, giving it a way into living tissues and helping it persist there. Paired with the acetate, the compound dissolves well enough in water to find its way into many environments—especially old hospital walls, paints, and agricultural products.
It hit home that every chemical shape can shape the world beyond the lab. The risks here come from the mercury atom sandwiched into this structure. When phenylmercuric acetate breaks down, it can leave behind inorganic and organic mercury. Scientists have tied these breakdown products to toxic effects on the nervous system, kidneys, and environment. Even trace contamination leads to calls for strict storage, disposal, and handling. Countries started phasing out mercury preservatives once the health risks couldn’t be ignored.
Pursuing Safer Alternatives
Discussing phenylmercuric acetate pushes you to ask bigger questions: How do we balance federal regulation, historical use, and daily exposure? I’ve seen universities swap out mercury-based fungicides for sodium benzoate, silver ions, or plant-based extracts. Not all replacements tick every box for cost or efficacy, but advancing green chemistry standards offers hope. Biocompatible alternatives and careful waste management help tackle the bigger problem of environmental mercury. Raising awareness in classrooms, workplaces, and policy circles could prevent old mistakes from repeating.
Final Thoughts
Phenylmercuric acetate’s formula—C8H8HgO2—is more than textbook trivia. Understanding its chemical makeup links right to health, industry, and regulation. Real safety starts with knowing exactly what’s in a compound and why structure matters in the first place.
| Names | |
| Preferred IUPAC name | acetoxy(phenyl)mercury |
| Other names |
Acetic acid, phenylmercury(II) salt PMA Phenylmercuric ethanoate Phenylmercury acetate |
| Pronunciation | /ˌfiː.nɪl.mɜːˌkjʊr.ɪk əˈsiː.teɪt/ |
| Identifiers | |
| CAS Number | 62-38-4 |
| Beilstein Reference | 1718733 |
| ChEBI | CHEBI:44949 |
| ChEMBL | CHEMBL9419 |
| ChemSpider | ChemSpider ID: 22939 |
| DrugBank | DB00764 |
| ECHA InfoCard | 100.005.445 |
| EC Number | 200-532-5 |
| Gmelin Reference | 3975 |
| KEGG | C18814 |
| MeSH | D010624 |
| PubChem CID | 6766 |
| RTECS number | OV3325000 |
| UNII | J15K7A69RO |
| UN number | UN1674 |
| Properties | |
| Chemical formula | C9H10HgO2 |
| Molar mass | 339.73 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 2.62 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 1.39 |
| Vapor pressure | 1 mmHg (20°C) |
| Acidity (pKa) | 7.1 |
| Basicity (pKb) | 6.34 |
| Magnetic susceptibility (χ) | '-183.5×10⁻⁶ cm³/mol' |
| Refractive index (nD) | 1.570 |
| Dipole moment | 2.07 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 359.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -261.0 kJ/mol |
| Pharmacology | |
| ATC code | S01AX07 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or absorbed through skin; causes burns; may cause genetic defects; highly toxic to aquatic life. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. H373: May cause damage to organs through prolonged or repeated exposure. H410: Very toxic to aquatic life with long lasting effects. |
| Precautionary statements | H260, H300, H310, H330, H373, H410, P210, P222, P223, P231, P232, P260, P262, P271, P273, P280, P284, P301+P310, P302+P352, P304+P340, P308+P313, P311, P320, P330, P361, P391, P403, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-2-Acidity |
| Lethal dose or concentration | LD₅₀ oral (rat): 33 mg/kg |
| LD50 (median dose) | LD50 (median dose): 14 mg/kg (oral, rat) |
| NIOSH | TP9625000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Phenylmercuric Acetate: "0.01 mg/m³ (as Hg), skin |
| REL (Recommended) | 0.01 mg/m³ |
| IDLH (Immediate danger) | 10 mg/m3 |
| Related compounds | |
| Related compounds |
Phenylmercuric nitrate Mercuric acetate Phenylmercuric borate |

