Manganese Nitrate: A Deep Dive into an Essential Inorganic Compound
Historical Development
Exploring the progress of manganese nitrate takes us into the early advances in industrial chemistry. Before modern technology arrived, chemists depended on trial and error in dusty workshops, mixing nitric acid with manganese oxides and learning through smoky reactions and stained glassware. As the nineteenth century rolled in, advances in analytical chemistry gave people the chance to measure purity, reactants, and temperature with more precision. I recall studying vintage laboratory guides and realizing how critical manganese nitrate became to early pigment production and metallurgy. It helped drive innovation in the glass industry, and over time, facilitated better performance in batteries and electronics. The route from accidental discovery to essential reagent mirrors the broader journey of inorganic chemistry, moving from curiosity to industry staple.
Product Overview
Manganese nitrate remains a keystone inorganic chemical, commonly encountered as its tetrahydrate form appearing as pink crystals. Used both in laboratories and industries, this compound plays a hands-on role in creating catalysts, fertilizers, ceramics, and batteries. A quick scan around research supply catalogs highlights how vital this compound has become—not just in its pure crystalline form, but as a foundation for mixtures and precursor solutions in advanced manufacturing. I have seen tech startups ordering manganese nitrate by the barrel as they attempt to ramp up battery pilot lines and energy storage devices. This product stands out not just for traditional processes, but increasingly as an ingredient in emerging clean energy research.
Physical & Chemical Properties
At room temperature, manganese nitrate typically comes as a transparent, pinkish crystal with a tendency to liquefy as it pulls in moisture. This tendency—deliquescence—means it can turn into a puddle if left in humid air. The nitrate salt dissolves easily in water, forming an acidic solution. Manganese nitrate's oxidizing nature means it reacts readily with fuels and reducing agents—chemists take care not to store it with organic or combustible materials. I've seen storage cabinets in university labs fitted with humidity controls and warnings for both corrosion and combustibility, underlining the compound’s dual character as both a helpful reagent and a potential hazard if used carelessly. Chemically, the presence of both manganese and nitrate ions yields an array of reactivities, allowing it to serve as an oxidant as well as a source for manganese cations in various syntheses.
Technical Specifications & Labeling
Labeling for manganese nitrate today stresses clarity: product grade, concentration, hydration state, and trace impurity levels must be visible. Technical buyers look for CAS numbers, ECCN codes, lot tracking, and details about storage requirements—including the specific maximum humidity safe for long-term storage. Suppliers provide guidance on safe handling and proper disposal, echoing regulatory focus on waste minimization. From my own procurement experience, the best vendors share detailed certificates of analysis with measured levels of trace metals, ensuring each batch meets application-specific purity needs. Good labeling fosters safe use, traceability, and regulatory compliance across borders.
Preparation Method
The production of manganese nitrate in industry rests on controlled reactions between manganese(IV) oxide and dilute nitric acid, usually under temperature control to avoid unwanted byproducts. In small-scale laboratory settings, I’ve repeated this synthesis by slowly dripping nitric acid onto powdered manganese dioxide, tracking temperature, and letting the gas evolve safely in a vented hood. Filtration and crystallization follow, with a slow evaporation method yielding the hydrated crystals. Large-scale manufacturers automate these steps, purify the final product with vacuum filtration, and stabilize it in moisture-tight packaging. Control over factors like acid strength and temperature sets apart technical-grade from reagent-grade output.
Chemical Reactions & Modifications
Manganese nitrate often acts as an oxidizer in synthesis, kicking off redox reactions for preparing other manganese compounds—manganese oxides, for instance, which appear in batteries and ceramics. Heating manganese nitrate leads to decomposition and evolution of nitrogen oxides, leaving behind ever-more-oxidized manganese residues. I’ve seen it used as a source of manganese ions in solution for ion-exchange reactions, as well as in sol-gel methods where its solubility and reactivity fine-tune the properties of resulting nanomaterials. Tinkering with stoichiometry, pH, and other reagents allows chemists to tailor the manganese oxidation state, pushing research forward on new electrode materials and catalysts.
Synonyms & Product Names
In scientific and technical literatures, manganese nitrate surfaces under names such as manganous nitrate, manganese(II) nitrate, or simply Mn(NO3)2. Some older reference texts use Latinized forms or trade names, while commercial catalogs specify hydration states—manganese(II) nitrate tetrahydrate being the usual. From experience with global suppliers, I’ve noticed the importance of clarifying product identity in specifications, since confusion between manganese(II) and manganese(IV) compounds can ruin months of experimental planning.
Safety & Operational Standards
Handling manganese nitrate demands respect for both its chemical and health risks. In the lab, technical staff wear gloves, goggles, and lab coats, since skin and eye contact produces irritation and ingestion or inhalation can have toxic effects. Regulatory authorities, such as OSHA or the European Chemicals Agency, set exposure limits and require proper labeling under GHS rules. Facilities store this compound away from organic, combustible materials and ammonia-based reagents to control fire and explosion risk. I’ve walked into facilities where polypropylene containers sit inside sturdy vented cabinets, all with humidity sensors, and every operator knows not to eat, drink, or smoke where manganese nitrate is present. Training, good labeling, and frequent risk reviews are the norm—not simply to check a box, but to protect real people from long-term hazards.
Application Area
Manganese nitrate’s uses reach deep into materials science, battery development, pigment production, and even agricultural micronutrients. Manganese forms part of essential redox reactions in plant metabolism, and chemists use the nitrate for trace manganese in foliar sprays and soil supplements. In the ceramics industry, this compound acts as a source for glazes and pigment colorants, creating violet and brown hues that persist in high-temperature firings. Electronics manufacturers prize high-purity manganese nitrate for synthesizing cathode materials in lithium-ion and other advanced batteries. During my research days, I watched electroplating firms rely on manganese nitrate to control the manganese layer’s thickness and integrity. Every major application area builds on the compound’s blend of reactivity, solubility, and compatibility with large-scale, reproducible processes.
Research & Development
The current wave of research focuses on expanding manganese nitrate’s use for clean-energy devices and catalysts. Innovative laboratories harness its properties to create novel manganese oxides, spinel structures, and other mixed-metal compounds crucial for battery and fuel-cell technology. Recent journals show syntheses using manganese nitrate that achieve better-controlled particle sizes, porosity, or crystallinity, all supporting next-generation storage or environmental remediation solutions. I have collaborated with teams who pushed for greener synthesis pathways, using less acid, less waste, or more efficient recycling strategies in order to meet both environmental and economic challenges tied to scale-up. The compound’s widespread availability and relatively low cost open possibilities for researchers in both nimble startups and major institutions.
Toxicity Research
Ongoing studies track manganese nitrate’s health impacts over both short and long timelines. Acute exposure brings skin, eye, and respiratory tract irritation; chronic or repeated exposure raises deeper questions around neurological effects. Animal studies track systemic effects at different exposure levels, and public health research investigates manganese’s role in air or water pollution where industrial use lapses occur. From field experience, managing dust and aerosol generation tops the list in safe manufacturing and research settings. Reliable air handling equipment, chemical-resistant gear, and routine health screens for staff keep exposure well below regulatory thresholds. Attention to proper disposal, containment, and spill management further reduces the risk of environmental contamination or public health impacts.
Future Prospects
Looking ahead, manganese nitrate stands poised for greater demand as clean energy demand surges. Batteries, fuel cells, and green catalysts all pull for higher volumes, stricter purity, and new forms that fit advanced processing routes. Research into recycling spent manganese materials picks up speed, seeking closed-loop solutions that reduce mining and waste while feeding quality feedstocks back into the loop. The versatility of manganese nitrate—serving as a source, a reagent, and an intermediate—keeps it central in both legacy and emerging technologies. Long-term sustainability, safe operational practices, and high-grade product control will shape its evolution in academic, industrial, and environmental applications worldwide.
What are the main uses of Manganese Nitrate?
The Unsung Helper in Agriculture
Manganese nitrate often ends up in fertilizer mixtures. Crops strain without enough manganese, showing poor growth and yellowed leaves. Farmers see firsthand how even a little gap in key nutrients hurts their harvest. Some crops, like soybeans and oats, respond quickly to a dose of manganese. Applying manganese nitrate helps them bounce back. There’s no need for a complicated chemical mystery here: the goal is strong roots, steady yields, and healthy food on tables. Unlike more common nutrients, manganese works deep in plant cells, powering vital reactions. Fertilizer makers count on manganese nitrate because it dissolves well in water, so it spreads evenly across fields. Crops get a fair shot, and families down the line eat better.
Better Batteries and Electronics
Industrial companies working on batteries have learned how manganese nitrate adds value. In the past few years, as more people use electric vehicles and portable devices, battery makers look for chemicals that deliver reliable results. Manganese nitrate brings a source of pure manganese for making cathode materials in lithium-ion batteries. These batteries power phones and cars, and rely on manganese for longer life and greater energy. If you’re tired of a phone that quits out early, thank the researchers who zeroed in on materials like this. People chasing breakthroughs in green tech depend on manganese nitrate to keep the momentum going. By supporting the shift away from fossil fuels, it quietly shapes daily life.
Ceramics, Dyes, and Pigments
Artists and manufacturers know the difference a strong pigment makes. Ceramics and tile makers value manganese nitrate as a coloring ingredient. Fired glazes come out with an earthy brown or violet hue. In the world of dyes, textile producers apply manganese nitrate, giving fabric lasting color and a distinctive look. My own family worked in local ceramics for years, and the way a batch of glaze changed with an extra pinch of manganese amazed everyone in the workshop. Little adjustments in chemical mixes could mean a masterpiece or a reject. That hands-on experience explains why this chemical holds a steady spot on supply shelves.
Laboratory Reactions and Research
In research spaces, manganese nitrate acts as a starting point for more complex manganese compounds. Chemists mix it into experiments, exploring new catalysts for water treatment or material science. Lab teams use manganese nitrate because it’s simple to measure and reacts predictably. Safer handling means less risk to people in the lab, compared with some tougher alternatives. This accessibility means colleges, as well as big research companies, can make progress without breaking the bank. Projects in nanotechnology or environmental cleanup often begin with straightforward ingredients, and manganese nitrate helps lay the groundwork.
Addressing Risks and Moving Forward
No chemical stands without trade-offs. Manganese nitrate requires careful storage. Contact with moisture or heat causes it to break down and release nitrogen oxides, which harm lungs. Production facilities use well-ventilated spaces and sealed containers to cut down on exposure. Both safety teams and regulators shape the rules on how workers interact with it. For those who worry about environmental effects, closed-loop processes and neutralization steps can keep runoff out of rivers and lakes. Investing in safer packaging and worker training makes sense; the companies that lead in those fields build trust with neighbors and customers.
Is Manganese Nitrate hazardous or toxic?
Digging Into Manganese Nitrate
Folks working in labs, the metal industry, or crop science might run into manganese nitrate more than most people ever will. You’ll see it as a pink or pale violet powder, easy to overlook—until you realize what it actually can do. This compound pops up in fertilizer production, chemical processing, and even battery manufacturing. The less-familiar name shouldn't signal less risk. It needs the same respect people give to more notorious chemicals.
Health Risks – More Than Just a Dust Problem
Think about handling bags of fertilizer or runoff from industrial waste. Breathing in manganese nitrate dust or getting it on your skin is not the same as harmless chalk. When inhaled, manganese compounds can tinker with the nervous system. Repeated exposure can lead to a condition called manganism, featuring muscle weakness, tremors, and trouble with memory. The U.S. EPA lists manganese as an air pollutant, and for a good reason: chronic exposure links directly to neurological effects, usually worse among welders or factory staff.
Skin and eye contact also demand attention. The compound can cause burning pain or persistent irritation. Swallowing even a small amount can lead to stomach trouble, and it only gets more dangerous with higher doses. Once in the bloodstream, the substance disrupts how your body manages enzymes and nerve signals.
Environmental Hazards on the Table
Manganese nitrate doesn't stay put. Left on bare ground, it dissolves into water and drifts downstream. Aquatic life pays the price. In rivers and lakes, too much manganese disrupts the health of fish and plants, throwing natural cycles off balance. Over time, farm runoff with manganese nitrate can taint drinking water supplies, which spells trouble for both people and animals nearby.
Fact-Based Solutions That Actually Work
Ignoring safety doesn’t cut it in workplaces that deal with manganese nitrate. The Occupational Safety and Health Administration (OSHA) sets strict exposure limits. Good ventilation, dust collectors, and protective equipment like gloves and goggles protect workers who don’t want the substance soaking into their skin or lungs. Everyday folks won’t end up in trouble handling store-bought batteries or walking by farms, but the people on the front lines—factory techs, farmers, and waste handlers—need to know what this stuff does.
Disposal calls for just as much care. Dumping manganese nitrate down a drain or tossing it out with regular trash isn’t an option. Facilities follow hazardous waste guidelines, and for a reason: even small leaks can leach into soil or water. There’s nothing glamorous about double-checking containers and using chemical spill kits, but skipping those steps invites long-term messes that cost real money and real health.
Solid Information Matters
I’ve seen the cost of complacency in work as a science communicator: folks who didn’t learn the real risks, who got sick after years of exposure. Honest, readable safety info saves jobs and lives. Reliable instructions on storage, clean-up, and handling let people keep their livelihoods and avoid trading health for a paycheck. Clear rules at work and strong community right-to-know laws mean people don’t have to work in the dark.
Behind every chemical label, there’s a line between safety and disaster. Manganese nitrate doesn't grab headlines, but it deserves upfront facts and consistent respect. Solutions don’t have to be fancy—just practical, enforceable, and based on what we already know.
What is the chemical formula of Manganese Nitrate?
Getting the Formula Right
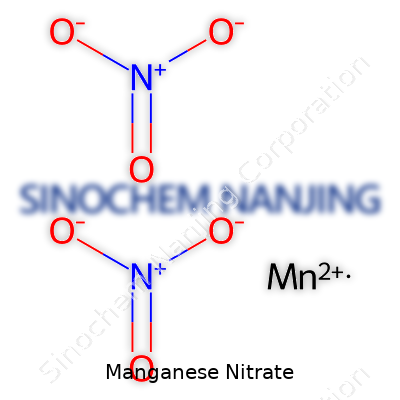
Manganese nitrate has the chemical formula Mn(NO3)2. In science class, we all memorized formulas, but it’s easy to forget how they play out in the real world. This simple arrangement of symbols influences everything from fertilizer blends to oxidizing reactions in labs across the globe. Manganese sits with a +2 charge in this salt, joining up with two nitrate ions, each weighing in with a -1 charge. It keeps everything balanced, but out of the classroom, things get more interesting.
What Purpose Does Mn(NO3)2 Serve?
Every job site and every lab bench has its unsung workhorses. Manganese nitrate pops up in fertilizers, helping crops get the trace minerals they crave. I watched growers in the Midwest tweak their soil chemistry to push yields higher, and smaller details like trace element selection made a real impact. In industrial chemistry, this compound keeps the wheels turning for oxidation processes. It doesn’t take center stage, but it’s there, behind better batteries and purer catalysts.
Quality and Safety Take Priority
Anyone who’s handled nitrates knows safety isn’t just a suggestion. Manganese nitrate, like other strong oxidizers, demands respect. The formula itself signals this—nitrates release oxygen, so the risk of fire or explosion hangs in the air when conditions go wrong. Companies face regulatory oversight for storage and shipping, and everyone from warehouse workers to high school teachers follows tight procedures. Overexposure also brings health risks. Breathing in manganese over long stretches can affect the nervous system, a lesson learned in factories long ago. Proper gear and steady monitoring make a difference.
Problems and Practical Solutions
In the field, contamination and runoff remain a challenge. Farmers see quick growth, but careless use lets manganese leach into waterways. Downstream, aquatic life takes the hit. The solution isn’t a mystery: modern farmers deploy soil tests and only add what’s needed. Local regulations already exist for nitrate fertilizers, creating a baseline, but stewardship means going further. Recycling and controlled-release products cut down on waste.
On the industrial side, manufacturers work to replace hazardous substances with greener options where possible. Research teams check if catalysts with lower toxicity can handle the same jobs, all while tracking production waste. Education and training play a part too, since anybody who handles the compound should understand both the risks and the safest handling methods. Well-trained hands reduce accidents.
Facts Put Knowledge to Work
Mn(NO3)2 isn’t just numbers and letters on a label; it’s a building block that crisscrosses agriculture, industry, and research. Getting the formula right matters, not just for passing tests but for making smart, safe, and responsible choices at every stage. Accuracy supports safety, productivity, and environmental stewardship. From the lab to the field, these details help shape outcomes in real lives.
How should Manganese Nitrate be stored safely?
Understanding What You're Dealing With
Manganese nitrate doesn’t get the spotlight like some household chemicals, but it deserves careful respect. Common in laboratories and industries—where it plays a role in fertilizers, ceramics, and metal treatments—it comes with baggage: it’s an oxidizer and can support fire, especially if it’s near combustibles like paper or oil.
Getting Storage Right from the Start
There’s no shortcut here. Put manganese nitrate anywhere near heat sources or sunlight, and you’re adding risk. The best spot is a cool, dry, ventilated storage room, away from direct sunlight or any HVAC vent that might blow dust around. Standard metal shelving works, but don’t crowd it. I learned the hard way in my university lab—stuffing too many bottles on one shelf invites accidents.
If moisture sneaks in, manganese nitrate absorbs water and may cake or clump, increasing handling trouble. Humidity control can make or break good storage. Running a small dehumidifier or adding silica gel packets nearby helps. Always check the area for any pools of water or pipe leaks overhead. Water plus chemical equals a cleanup nobody wants.
Staying Clear of the Wrong Companions
Manganese nitrate doesn’t play nice with everything. Mixing with flammable chemicals or organic materials boosts the chance of something going wrong. A basic rule: keep oxidizers away from fuels, even in storage. Store it away from acids and bases, too, since strange reactions can surprise you. Some workplaces use color-coded shelving—red for flammables, yellow for oxidizers, blue for health hazards—which takes the guesswork out, especially during busy shifts.
Pays to Go Beyond Labels
Don’t trust a faded label or half-peeled tape. Every container should have a clear identification label, date received, and, if possible, an “opened on” date. New staff can slip up fast if containers aren’t marked. One time in a small plating shop, I found two almost identical jars—one contained manganese nitrate, another a benign salt. One misplaced scoop would have spelled trouble.
Choose the Right Containers
Glass is popular for storing reactive salts, but plastic works if it’s high quality and doesn’t degrade. Never transfer this chemical to a container with a missing or broken lid. Air exposure can lead to degradation, and spills invite corrosion around the storage room. Check seals every few months; replace any cracked jars or loose lids without delay.
Watch the Number of People with Access
Fewer hands, fewer headaches. Not every staff member needs access to high-risk substances. Locking cabinets or a dedicated storeroom can keep misplacement or theft in check. Years of managing supply closets have taught me this: an open-door policy increases the odds of mistakes. Assign one or two people as “key holders” who make sure safety protocols aren’t just empty words.
Prepare for Accidents Before They Happen
Don’t wait for a near-miss to review safety gear. Every manganese nitrate storage area should have chemical-resistant gloves, face shields, and a chemical spill kit close by. Keep a clear list of emergency contacts posted on the door. Practice regular spill response drills—complacency causes more damage than equipment ever does.
Knowledge Sticks Around Longer Than Luck
Some old-school safety habits matter—like logging every container’s movement in and out of storage, or making sure the ventilation fan kicks in every morning. These routines turn into muscle memory. Good storage habits can sound boring, but they keep workplaces and people safe. It’s not about paranoia; it’s about respect for chemistry’s power. That lesson lasts longer than luck every time.
What is the solubility of Manganese Nitrate in water?
The Chemistry Behind the Dissolve
Anyone who has dropped manganese nitrate crystals into a glass of water has noticed how quickly they vanish. This reaction isn’t just a trick. Manganese nitrate’s solubility in water comes from the nature of its ions. It usually lands on shelves as a pale-pink solid—specifically, Mn(NO₃)₂·4H₂O. Add it to water, and this salt gives up those nitrate groups and manganese ions without much fuss, forming a transparent solution. At room temperature, one hundred grams of water can hold up to about 65 grams of manganese nitrate. That’s hefty, compared to many other common salts.
Why This Solubility Matters Beyond the Lab
In my days working with chemistry sets and later in university labs, I learned quickly that not all salts play nicely with water. Some, like manganese nitrate, jump right in. Its ease in making solutions means industries and researchers like dealing with it. You’ll often see this property put to work in fields from fertilizer production to the development of specialty ceramics and catalysts.
Water treatment operators, for example, sometimes rely on manganese nitrate as a source for manganese, which helps oxidize and clean certain contaminants. Agriculture industries have also spotted the value. Trace manganese finds its way into crop soils, and this compound’s water-friendliness makes dosing straightforward. No messy residue, no tricks to coax it into liquid form. It’s so soluble you’ll need to keep solutions in well-sealed containers, or the crystal will pull in water straight from the air.
Risks and Environmental Thoughts
It helps to keep an eye on exactly how much manganese you’re putting into a system. While manganese is part of healthy soils and some water supplies need it, too much can throw things off balance fast. Too much manganese nitrate dissolving uncontrolled into waterways or soil can lead to toxicity—in plants, animals, and sometimes people.
I’ve seen operators overdo nutrient dosing, thinking more equals better. The result? Manganese accumulates. Some groundwater becomes bitter or even unfit for nearby homes. In humans, excess manganese can harm the nervous system, sometimes creating symptoms that look like Parkinson’s disease. These aren’t far-off tales. Recent years brought stricter limits in countries like the United States and Canada, after decades of assuming manganese held little risk.
The solution starts with education and monitoring. Processes using manganese nitrate need real checks on how much gets released and strong spill-handling practices. I remember a good friend working in industrial safety who often reminded his team: “There’s no magic cleanup for water contamination. Keep it contained first.” For science classrooms and small labs, using the right eye protection and minimizing direct contact just makes sense. Public guides for safe storage and disposal—beyond dusty manuals—give both staff and newcomers real tools to keep things in check.
Reliable Sources and Research Backing
All this information comes from trusted sources in chemistry reference books, government water quality reports, and hands-on experience. Reliable solubility numbers for manganese nitrate show up both in Merck indexes and published chemical handbooks. Current environmental research on manganese’s impact is widely available through health organizations and academic reviews, reflecting an up-to-date understanding.
Understanding the way manganese nitrate dissolves opens a window, not only on a neat chemical reaction but also on decisions we make in farming, manufacturing, and public safety. Facts guide safer use. A little research and smart practice go a long way toward keeping benefits front and center and risks under control.
| Names | |
| Preferred IUPAC name | Manganese(II) nitrate |
| Other names |
Nitric acid, manganese(2+) salt Manganese(II) nitrate Manganese dinitrate Manganese(II) nitrate tetrahydrate Manganese bis(nitrate) |
| Pronunciation | /ˈmæŋɡəniːz ˈnaɪtreɪt/ |
| Identifiers | |
| CAS Number | 10377-66-9 |
| Beilstein Reference | 358661 |
| ChEBI | CHEBI:63056 |
| ChEMBL | CHEMBL4291638 |
| ChemSpider | 157422 |
| DrugBank | DB11357 |
| ECHA InfoCard | 03e9b9e2-4177-4e72-acf8-3c8f3eaf539d |
| EC Number | 233-828-8 |
| Gmelin Reference | 13519 |
| KEGG | C02348 |
| MeSH | D008360 |
| PubChem CID | 24584 |
| RTECS number | OO9625000 |
| UNII | 55D14O5989 |
| UN number | UN1477 |
| Properties | |
| Chemical formula | Mn(NO3)2 |
| Molar mass | 164.946 g/mol |
| Appearance | Pink crystalline solid |
| Odor | Odorless |
| Density | 2.29 g/cm³ |
| Solubility in water | Soluble |
| log P | -3.14 |
| Acidity (pKa) | 3.47 |
| Magnetic susceptibility (χ) | +1300e-6 |
| Refractive index (nD) | 1.401 |
| Viscosity | 12 cP (25 °C) |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 247.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −451.6 kJ/mol |
| Pharmacology | |
| ATC code | V07AB32 |
| Hazards | |
| Main hazards | Oxidizer, harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS05, GHS07, GHS09 |
| Pictograms | GHS07,GHS05 |
| Signal word | Danger |
| Hazard statements | Hazard statements: H272, H301, H314, H332 |
| Precautionary statements | P210, P220, P221, P264, P280, P301+P330+P331, P305+P351+P338, P306+P360, P370+P378, P403+P233, P501 |
| NFPA 704 (fire diamond) | 2-0-2-OX |
| Flash point | 130 °C (266 °F) |
| Autoignition temperature | 180°C |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD₅₀ (oral, rat): 410 mg/kg |
| LD50 (median dose) | > 4,320 mg/kg (rat, oral) |
| NIOSH | SN26350 |
| PEL (Permissible) | PEL: 1 mg/m³ |
| REL (Recommended) | REL (Recommended Exposure Limit) of Manganese Nitrate: "1 mg/m³ (as Mn), 8-hour TWA (NIOSH) |
| Related compounds | |
| Related compounds |
Manganese(II) oxide Manganese(II) carbonate Manganese(II) sulfate |

