Exploring Mandelonitrile: Challenges, Uses, and the Road Ahead
Historical Development
In tracing the long march of synthetic chemistry, mandelonitrile pops up early. The compound sneaks its way into the textbooks as a classic example of a cyanohydrin—one made most famously by mixing benzaldehyde with hydrogen cyanide. Throughout nineteenth-century Europe, scientists hunched over their glassware figured out routes to combine natural aromatic compounds with noxious cyanide, all in pursuit of understanding natural flavors, fragrances, and the chemical fingerprints of things we eat and breathe. Mandelonitrile originally interested researchers for its connection to bitter almonds. That it could be coaxed from a nut and then turned back into almond flavor gave it a whiff of both danger and culinary delight. Over time, mandelonitrile’s relevance grew in lockstep with interest in pharmaceuticals and organic synthesis—serving as both tool and puzzle for generations of chemists.
Product Overview
Nowadays, mandelonitrile appears as a specialty item in the toolkit for scientists and industry. It sports a chemical formula of C8H7NO and often serves as a precursor for dyes, agents used in laboratories, and as a stepping-stone for other specialty chemicals. As anyone who has handled almond oil in a lab can attest, there’s a sharp, distinctive aroma that comes with handling this compound—one both inviting and a little unnerving, given its ties to cyanide.
Physical & Chemical Properties
Mandelonitrile sits as a colorless to pale yellow liquid or sometimes a crystalline solid, depending on storage and temperature. It sports a melting point a little above room temperature, leaving it halfway between solid and liquid in most laboratories. It dissolves easily in organic solvents. One recurring challenge comes from its instability in water, where it can break down and release hydrogen cyanide, reminding everyone this isn’t a compound to take lightly. This property gives mandelonitrile a distinct edge: beneficial for controlled reactions, dangerous if ignored.
Technical Specifications & Labeling
Any chemist eyeing mandelonitrile will notice bottles stamped with strict hazard warnings. Labels reference its toxic nature, especially warning about inhalation, ingestion, and skin contact. Containers typically mention the presence of hydrogen cyanide risks and feature color-coded stickers. In practical terms, anyone storing or using mandelonitrile needs to keep it away from acids and water, which can spark the formation of cyanide gas. In my own training, I never forgot the shudder of respect we developed in handling this kind of bottle—gloves, goggles, and well-ventilated hoods as the default kit for even a routine transfer.
Preparation Method
Synthesizing mandelonitrile in a lab usually means reacting benzaldehyde with hydrogen cyanide. This reaction proceeds under controlled, cold conditions, since cyanohydrins tend to fall apart if you rush or heat things up. Old-school chemists performed the synthesis using wet chemistry and delicate temperature control, always alert to the dangers posed by free cyanide. Today, safer technology and better ventilation have replaced some hazards, but the fundamental chemical risks remain. Alternative synthetic routes aim to bypass the need for free hydrogen cyanide using other reagents or in situ generation—small steps, but ones driven by hard-won experience and serious pilot studies intended to cut down on accidents and environmental releases.
Chemical Reactions & Modifications
Mandelonitrile’s reactive cyanohydrin structure opens up many chemical pathways. The compound dehydrates to benzonitrile under acidic or basic conditions. Through controlled hydrolysis or enzymatic reactions, chemists convert it to mandelic acid—a compound that’s put to use in some cosmetic and medical applications. My own use of mandelonitrile as a building block in organic syntheses brought home just how sensitive and interesting this molecule proves. Under the wrong conditions, cyanide can split off, so keeping an eye on temperature, pH, and reagent sequence spells the difference between innovation and accident.
Synonyms & Product Names
Mandelonitrile has picked up a fair share of alternate names in popular literature and in laboratories: phenylglycolonitrile, α-hydroxybenzyl cyanide, and even amygdalonitrile nod to its origins in almond oils. These names mostly describe the same compound, though packaging and commercial sales sometimes swap in trade names. In any regulatory or export paperwork, mandelonitrile always comes with its unique chemical identifiers, since authorities rightfully track its movement for safety and compliance reasons.
Safety & Operational Standards
Handling mandelonitrile demands full attention and rigor. Besides the cyanide risk, the compound can irritate the skin, eyes, and lungs. Laboratory protocols steer all manipulations into chemical hoods. Spills get handled by trained staff armed with respirators and cyanide antidotes close at hand. Facilities using mandelonitrile train every worker in emergency procedures, often including simulated spill drills and first-aid practice for cyanide poisoning. From my experience, you never grow casual with something this unforgiving, so robust institutional safety culture forms the backbone of every operation touching cyanohydrins.
Application Area
Mandelonitrile’s main claim to fame grows from its role as a precursor in organic synthesis. Chemical manufacturers build off its backbone to create many products, such as pharmaceuticals and fine chemicals. In medicine’s early days, some researchers even investigated its breakdown product—hydrogen cyanide—as a medicinal agent, though modern science quickly moved away from such dangerous experiments. Today, the compound’s main value lies in its chemistry, fueling research into new drugs, dyes, and biochemical probes. It also provides a useful standard in analytical chemistry for those measuring cyanohydrin content in foods and plant materials.
Research & Development
Much current research on mandelonitrile explores ways to mitigate its hazards while maximizing its synthetic potential. Scientists try to tack on chemical groups or use enzyme-catalyzed approaches to get desired products without releasing free cyanide. Some work focuses on “green chemistry” approaches that reduce or recycle hazardous wastes and improve process yields. Others look at safer derivatives for pharmaceutical or cosmetic uses. In my time in a chemical research group, colleagues showed how tweaking reaction conditions or swapping solvents trimmed down risk without giving up on product quality. Incremental improvements in process safety, containment, and detection keep pushing the boundaries of what’s possible without sacrificing the well-being of workers.
Toxicity Research
Mandelonitrile’s toxicity casts a long shadow and receives continual attention from toxicologists and regulatory authorities. Animal studies document acute toxicity tied to its capacity to release hydrogen cyanide, resulting in symptoms typical for cyanide exposure—headaches, rapid breathing, convulsions, and even death at high doses. Chronic exposure can also cause nerve and organ damage. The compound never appears in consumer products as an active ingredient for this reason. Ongoing toxicity studies try to pin down safer handling limits, test new antidotes, and set appropriate exposure thresholds for workers. As a chemical researcher, I saw firsthand the emphasis on personal protective equipment, real-time air monitoring, and rigorous training—nothing left to chance, knowing how unforgiving a small slip with cyanohydrin chemistry can be.
Future Prospects
Looking out on the future, the hope is to keep digging for safer, more sustainable ways to use mandelonitrile and its derivatives. Green processing stands out, aiming to reduce cyanide emissions, cut down on hazardous waste, and reuse solvents. Advances in continuous-flow chemistry hold promise—containing the reaction and improving control over temperature and exposure. Ongoing development of new catalysts, including biological enzymes, could allow industry to bypass the most hazardous sequences entirely. The search for practical substitutes keeps rolling, especially in pharmaceutical chemistry, where safety and environmental impact face increasing scrutiny. Through better oversight, new technology, and hard-earned lessons from decades of experience, the road ahead for mandelonitrile looks a little less treacherous than the one already traveled.
What is Mandelonitrile used for?
What Mandelonitrile Brings To The Table
Mandelonitrile may sound like something best left to the lab, but its roots reach deep into both history and practical chemistry. This compound, with its bitter flavor and ties to organic synthesis, shows up in some surprising places. Experts frequently refer to it as an “aromatic nitrile” because of the cyanide group attached to a benzene ring. The attention often gravitates to its relationship with cyanide—rightly so, as chemistry class scares make lasting impressions. The reality behind its uses stretches far beyond simple cautionary tales.
Role in Pharmaceuticals
Years ago, I stumbled across a research paper while searching for the history of heart medications. The name Mandelonitrile popped up. Turns out, this chemical forms a base structure for producing pharmaceuticals such as laetrile (sometimes called amygdalin), once touted as a cancer therapy. Doctors don’t recommend laetrile for cancer anymore due to lack of effectiveness and safety concerns, but the way chemists engineer these transformations remains fascinating. Using Mandelonitrile as a stepping stone, professionals craft diverse compounds—some helpful, others only experimental. Its structure allows easy modification, unlocking pathways to synthetic drugs targeted for both research and potential treatment down the line.
Chemical Synthesis and Industry
In my early lab experience, Mandelonitrile acted as a crucial ingredient for preparing chemicals nobody could buy off the shelf. Manufacturers draw on its structure to make benzaldehyde, another building block with its own set of jobs in flavors and fragrances. Not limited to perfumes and sweets, these compounds matter in the design of agrochemicals and specialty products. Efficiency and purity become top priorities, as mishandling Mandelonitrile leads to the creation of hydrogen cyanide—a toxic danger. Safety isn’t just a slogan. The dangers push everyone to respect protocol, double-check each procedure, and stay engaged when someone reaches for that pre-weighed bottle.
Natural Connections and Food
Look at an apricot kernel or a bitter almond and you’ll find Mandelonitrile nestled among the natural compounds. Nature uses it as a defensive molecule, built into plants as a way to keep pests at bay. Decades ago, stories popped up warning about eating too many apple seeds or bitter almonds, connecting those warnings straight back to the chemical transformations involving Mandelonitrile. Though you rarely hear about accidental poisoning from everyday fruit these days, food safety law keeps a close watch, tracing the journey of cyanogenic glycosides from orchard to lunchbox.
Challenges and Ways Forward
Working with hazardous chemicals leaves little room for error. Chemists, industry leaders, and safety professionals continue refining protocols to limit exposure to cyanide. Regulatory agencies step up to formalize safe use, offering extensive training and clear rules about storage and disposal. Emerging green chemistry trends bring in alternative pathways for both synthesis and waste management, reducing the need to handle high-risk intermediates like Mandelonitrile directly.
Closing Thoughts on Relevance
Every time industry or research relies on Mandelonitrile, it sparks a mix of innovation and concern. Bridging natural products, pharmaceuticals, and manufacturing, this compound serves as a touchstone for chemistry’s progress as well as its perils. Following the best science, sharing honest information, and investing in safer technologies strengthens trust and steers chemistry forward.
Is Mandelonitrile toxic or hazardous?
Poking Into the Facts
Mandelonitrile might not come up in dinner conversations, but it is worth knowing about, especially for folks who work in labs, chemical manufacturing, or even advanced farming. It’s an organic compound—comes from almonds, apricots, and a few other pits. Simple story, but curiosity grows because it has a cyanide group attached, and that group definitely rings alarm bells for toxicity and hazards.
What Happens in the Body?
My hands have shaken a bit when I’ve handled risky chemicals in the lab, and mandelonitrile earned my respect early on. When this compound ends up in the body, enzymes can turn it into benzaldehyde and hydrogen cyanide. Now, hydrogen cyanide is big trouble. It keeps cells from using oxygen, so even a small dose can lead to poisoning. Symptoms fly in: dizziness, confusion, trouble breathing, even seizures. That makes the label “toxic” pretty hard to argue with.
People grill health professionals about risk, and the facts are up front. Mandelonitrile’s threat depends on how much you take in, how you get exposed, and what other health problems you might already have. A single whiff may not topple a healthy person, but eating or inhaling enough can flatline major organs fast. Poison control centers have case files on accidental exposure—it’s rare but plenty real.
Handling and Real-World Experience
Folks sometimes ask about safe handling. A good pair of gloves only goes so far if someone ends up breathing powder or spills it on their skin. Laboratories use mandelonitrile as an intermediate for making pharmaceuticals or as a starting point for synthesis, but proper ventilation and secure containers become non-negotiable. Personally, watching colleagues rush when handling cyanogenic materials always drummed home one rule: never get careless. One spill or sniff during an experiment could put someone in the ER.
Accidental poisonings connect most often to improper storage or using mandelonitrile outside controlled settings. Shop teachers, backyard chemists, and even small business owners can miss the red flags unless training is constant and clear. Stories circulate about forgotten vials causing headaches—and worse—when curiosity got ahead of caution. Every spill brings risk, and cleanup needs more than just soap and water. Poison control numbers belong on speed dial.
Risks and Solutions
Strict regulations surround this chemical in countries that value safety. Workers need training, and companies install fume hoods, emergency showers, and make sure antidotes are on hand. Sodium thiosulfate and hydroxocobalamin treat cyanide poisoning, but hospitals warn that response time decides between life and death.
Awareness solves a big piece of the puzzle. Chemistry students and hobbyists track safety data sheets and wear protective equipment. Manufacturers train their teams, and labeling rules cover every shipment. Inspections do their job, but the real difference comes from treating the chemical with respect at every step.
Mandelonitrile has a place in industry and research, but everyone from warehouse managers to grad students should know its risks. It’s not a household chemical, and nobody should treat it like just another bottle on the shelf. Education and careful handling mean fewer accidents, and that saves lives, plain and simple.
How is Mandelonitrile stored and handled safely?
Why Mandelonitrile Matters
Working with mandelonitrile doesn’t grab headlines, but its use in labs and industry carries a set of real and pressing safety demands. Derived as a cyanohydrin, mandelonitrile includes a cyanide group—something that immediately raises red flags for anyone who’s spent time around chemicals. Cyanide compounds are notorious for their risk profile, so mishandling even a small amount of mandelonitrile can quickly turn into a serious incident.
Straight Talk: Health Risks and Exposure
Mandelonitrile doesn’t mess around. Hitting the bloodstream, it turns into cyanide. Symptoms don’t stop at headache and dizziness; exposure can push respiratory function to its limit, even leading to loss of consciousness if you’re unlucky or careless. That’s not theoretical—emergency rooms across the world know what happens when compounds like this aren’t given thoughtful treatment. Even skin contact with a liquid sample or a single, unlucky inhalation in a closed workshop sparks trouble.
What Safe Storage Really Looks Like
Decades of chemical work have hammered home that safe storage starts long before the actual shelving. Sound storage doesn’t mean hiding a bottle away in the back room. The best practice is a double containment—the main vessel sits inside a secondary chemical-resistant tray. Only chemical cabinets built for toxics should hold mandelonitrile. Forget about wooden cupboards or mixed-use lab shelves. Strong steel or polymer cabinets, labeled brightly and with physical locks, give peace of mind that no one stumbles across it by accident.
Temperature and humidity add another layer. Material Safety Data Sheets aren’t just paperwork; they’re distilled lessons learned from hard experience. For mandelonitrile, storing cool and dry wins every time. Any spike in heat can cause pressure to build, so ventilation in your chemical room isn’t optional. In shared spaces, storing it away from acids and oxidizing agents stops any cross-reaction possibility in its tracks.
How to Handle Without Drama
Years in the lab teach that personal protection isn’t just protocol—it’s self-preservation. Gloves rated for chemical use, full-length lab coats, and eye protection form the backbone. Respirators only come out for high-risk tasks, but having one on hand makes a difference when vapors threaten. Spills and cleanups don’t get “make it up as you go.” A spill kit for cyanides, kept close to storage, should include absorptive materials, neutralizing agents, and a step-by-step guide. Workers need training, not just for handling but for what happens when something escapes containment—the crew that knows what to do shaves vital seconds off emergency response.
What Works for Real-world Safe Practice
Effective safety means drilling everybody—students, technicians, even seasoned chemists—on strict procedures. Written protocols, checklists, and peer reviews catch slip-ups before they turn into accidents. Security measures limit access. Only trained, authorized people open those cabinets. Eyes washes and showers stay unobstructed, with regular checks to guarantee they’re ready if a mishap happens. Local exhaust, like fume hoods, always gets used during dispensing and weighing.
Waste doesn’t get poured down the sink—dedicated hazardous waste containers, labeled and treated as if the worst could happen, keep people and the environment out of danger.
Turning Knowledge Into Action
A cautious approach, shaped by lessons from history and a deep respect for the risks, spells out the safest path. Any lab using mandelonitrile keeps strict control, demands real preparation, and never takes shortcuts. That way, its usefulness—in synthesis, research, or teaching—fits hand in hand with responsible stewardship.
What is the chemical formula of Mandelonitrile?
Digging Beneath the Surface of Mandelonitrile
Mandelonitrile carries a simple, exact formula: C8H7NO. On paper, it looks pretty standard—eight carbons, seven hydrogens, one nitrogen, one oxygen. But there’s a more compelling story behind those numbers. The molecule sits at the crossroads between chemistry and biology, linked to a family of compounds called cyanogenic glycosides.
How Experience Meets Fact
For years, chemists have studied mandelonitrile because it pops up in both labs and nature. This molecule occurs in the seeds of certain fruits like apricots and bitter almonds. Anyone who’s looked into plant toxins knows its reputation. Through the natural breakdown of mandelonitrile, hydrogen cyanide gets released—a fact heavily studied in food safety and toxicology. The atom-count tells only half the story; it’s the arrangement that packs the punch. This connection between an everyday fruit and a potentially deadly poison pushes researchers and regulators to learn more about its pathways.
Why Chemical Formula Knowledge Goes Beyond Textbooks
With large amounts of information circulating online, basic chemical data often gets overlooked. Still, knowing the formula C8H7NO can help filter out misinformation, especially when people encounter health fear-mongering or overblown news. Reliable resources like PubChem, the FDA, and the Merck Index validate this core information. Professionals need this knowledge to spot errors early—errors that can sway public opinion or even guide policy the wrong way.
Implications in Real Life and Science
When I first came across mandelonitrile in a college chemistry course, we obsessed over laboratory synthesis and its behavior as an intermediate in organic synthesis. The risk of hydrogen cyanide release meant everyone wore extra protective gear in lab. The lesson stuck. Even in small quantities, incorrect handling could cause acute toxicity. Many regional poison control centers keep data on cyanogenic compounds for this very reason. Meanwhile, pharmaceutical chemistry uses mandelonitrile to develop drugs for research into heart medication and cancer therapy, capitalizing on its reactivity and selectivity.
The Human Side of Scientific Transparency
Food science looks at mandelonitrile as part of a larger safety puzzle. Apricot kernels, for instance, pack a real punch because they break down into cyanide upon ingestion. Direction from health agencies focuses on limiting consumption for children. The European Food Safety Authority and U.S. Food and Drug Administration have released public advisories on these seeds, drawing from studies where quantifying the breakdown products started with understanding the chemical formula.
Pushing for Practical Solutions
Fixing misinformation and improving safety needs strong outreach from both educators and industry voices. Clear, fact-driven education around molecules like mandelonitrile helps families make better choices—avoiding unnecessary panic, reducing poisoning risks, and guiding safe product design. Companies handling large volumes rely on standardized chemical identifiers, robust labeling, and training to protect workers. Even gardeners benefit from understanding what lies inside fruit pits or enhancing compost safety.
Reliable Knowledge Builds Trust
The formula C8H7NO grounds any discussion about mandelonitrile in verifiable science. By focusing on accurate, accessible chemistry, specialists help balance innovation with safety and keep communities informed. That’s the kind of knowledge that delivers real value, day after day.
What are the applications of Mandelonitrile in industry or research?
Opening the Door to Specialty Chemicals
Some compounds leave quiet but lasting fingerprints across many fields. Mandelonitrile stands out this way. Chemists value it for what it helps to build. Used mainly as a key intermediate, mandelonitrile opens the door to specialty chemicals—especially those with benzaldehyde or cyanide groups. Forget blockbuster pharmaceuticals for a minute. Big chemical companies rely on mandelonitrile to ramp up production for fragrances, pesticides, and even medicines. Its main job: acting as a bridge, helping build larger, more complex chemicals.
Pharmaceuticals: Where Small Changes Matter
Medicinal chemists often want to modify a drug’s shape to test what works best for the patient. Mandelonitrile works like a Lego piece in this space. It can transform into amygdalin—once famous for its links with "Laetrile" in alternative cancer treatments. While science has rightfully cast doubt on those therapies, mandelonitrile remains a foundation for new research. Recent studies look at shaping anti-inflammatory, antiviral, or anticancer compounds using parts derived from mandelonitrile. Academic research often tests new synthesis routes because mandelonitrile's simple skeleton can be counted on to produce a wide range of useful building blocks.
Agrochemicals: Feeding Farmers’ Demands
Pesticides and herbicides don’t show up out of thin air. Large-scale farms rely on precisely engineered chemicals. Manufacturers use mandelonitrile to produce benzonitrile derivatives—common ingredients in crop protection. Brands tweaking molecular structure for better weed or pest control usually experiment with mandelonitrile first. Its stable backbone supports multiple modifications, helping deliver compounds that last longer on crops or break down faster in the environment, depending on what’s needed. While not famous on labels, mandelonitrile's role as an industrial workhorse keeps many plates spinning in the agricultural sector.
Fragrances and Flavors: The Scent Behind the Scenes
Fragrance creators need molecules that mimic or support natural aromas. Benzaldehyde and almond-type scents often begin with mandelonitrile. Getting the subtle bitter-almond aroma for perfumes and flavorings connects back to careful chemical steps involving this compound. Synthetic chemists pick mandelonitrile because its structure handles the process well. The high purity possible during its conversion means safer, cleaner end-products—a must in any flavoring destined for food.
Lab Workhorses: Teaching and Innovation
Synthetic chemistry professors still hand out mandelonitrile for advanced lab exercises. It’s central to teaching nucleophilic addition and synthesis techniques. Students get hands-on with real-world reactions, watching colors change or crystals form. Those experiments are about more than theory—they shape tomorrow’s scientists. My own early days practicing reaction monitoring involved mandelonitrile: simple setup, reliable outcome, satisfying results. This routine lab experience gives confidence before heading into more complex industrial syntheses.
Sticking to Safety and Scaling Up
Using mandelonitrile at an industrial scale doesn’t go without risk. Strict safety measures stay in force—excessive exposure can release dangerous hydrogen cyanide. Factories invest in containment and air quality monitoring. Europe, the United States, and major Asian chemical markets routinely review safety guidelines. Responsible sourcing has improved, thanks to regulatory bodies stepping in and newer green chemistry methods cutting waste. Companies on the cutting edge work to minimize byproducts, improving not just bottom lines but environmental footprints as well.
Future Paths and Sustainable Chemistry
Mandelonitrile continues to anchor big and small discoveries. More chemists try greener synthesis, turning to enzymes and milder methods to produce benzaldehyde derivatives. Tracking the impact on food, soil, and chemical safety, researchers keep this molecule in rotation—not because it’s flashy, but because it works. Industry relies on such tools; as better technology appears, mandelonitrile will remain a fixture behind the developments that keep modern life moving.
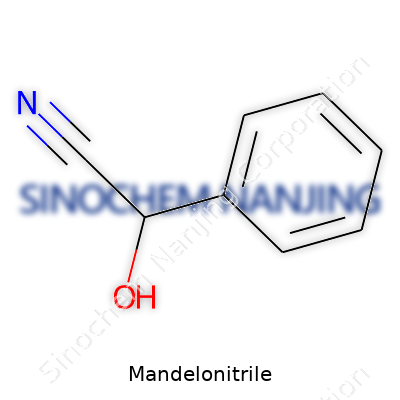
| Names | |
| Preferred IUPAC name | 2-hydroxy-2-phenylacetonitrile |
| Other names |
Benzaldehyde cyanohydrin α-Hydroxybenzyl cyanide |
| Pronunciation | /ˌmænˌdɛl.oʊˈnaɪ.trɪl/ |
| Identifiers | |
| CAS Number | 532-28-5 |
| Beilstein Reference | 1208738 |
| ChEBI | CHEBI:2868 |
| ChEMBL | CHEMBL3217346 |
| ChemSpider | 66385 |
| DrugBank | DB02925 |
| ECHA InfoCard | 100.003.159 |
| EC Number | 201-978-6 |
| Gmelin Reference | 7874 |
| KEGG | C01761 |
| MeSH | D008334 |
| PubChem CID | 6989 |
| RTECS number | NN1575000 |
| UNII | 5D9130048H |
| UN number | UN2811 |
| Properties | |
| Chemical formula | C8H7NO |
| Molar mass | 133.16 g/mol |
| Appearance | Colorless to pale yellow oily liquid |
| Odor | Bitter almond |
| Density | 1.104 g/mL at 25 °C |
| Solubility in water | Slightly soluble |
| log P | 0.53 |
| Vapor pressure | 0.01 mmHg (25°C) |
| Acidity (pKa) | 12.0 |
| Basicity (pKb) | 8.86 |
| Magnetic susceptibility (χ) | -61.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.528 |
| Viscosity | 1.247 mPa·s (20 °C) |
| Dipole moment | 2.98 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 217.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -87.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1266.8 kJ/mol |
| Pharmacology | |
| ATC code | N02BB05 |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled. |
| GHS labelling | GHS02, GHS06 |
| Pictograms | GHS06, GHS08 |
| Signal word | Danger |
| Hazard statements | H302: Harmful if swallowed. |
| Precautionary statements | P261, P264, P270, P271, P301+P310, P330, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-0 |
| Flash point | Flash point: 113°C |
| Autoignition temperature | > 300 °C |
| Lethal dose or concentration | LD50 oral rat 120 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral, rat: 15 mg/kg |
| NIOSH | MW6125000 |
| PEL (Permissible) | PEL: 5 ppm |
| REL (Recommended) | 12 h |
| IDLH (Immediate danger) | 25 mg/m3 |
| Related compounds | |
| Related compounds |
Benzaldehyde Mandelic acid Amygdalin Prunasin Phenylacetonitrile |

