Magnesium Tert-Butoxide: A Deep Dive into Its Chemistry and Role in Industry
Historical Development
The curiosity about magnesium compounds began long before laboratories bristled with glassware and digital controls. Seeking alternatives to harsher alkoxides and driven by organic synthesis demands, chemists in the mid-20th century turned their attention to magnesium tert-butoxide. Publications surfaced hinting at its utility in making complex molecules, while earlier forms in the 1920s centered around simple inorganic salts. With the rise of organometallic chemistry in the 1960s, researchers refined preparation methods, observing how subtle adjustments in synthesis delivered cleaner products. I recall my own early Organic Chemistry professor showing faded notebooks from the 1970s, scribbled with magnesium tert-butoxide experiments—sometimes messy, always pushing boundaries. This gradual evolution shaped its broad acceptance for laboratory innovation.
Product Overview
Magnesium tert-butoxide stands out as a white, often amorphous, powder. Chemists value it for its strong basicity and steric bulk, which lets it influence reactions with selectivity. This compound doesn’t simply function as another strong base; its magnesium center brings special reactivity to condensation and deprotonation reactions. The tert-butyl groups make it less prone to moisture sensitivity compared to lighter alkoxides, yet it still demands careful handling. In my own work, we favored it for making beta-keto esters with greater yields, sidestepping problems that plagued sodium and potassium bases.
Physical & Chemical Properties
Presented as a volatile white powder with a faint alcohol-like odor, magnesium tert-butoxide draws attention with a melting point near 130°C. It shows poor solubility in water but dissolves in ethers and other nonpolar solvents. Its tendency to absorb water suggests the need for sealed containers in storage. The magnesium-oxygen bonds bring a unique profile, making it both reactive and selective in transformations. The compound's crystalline structure resists air-stable handling, which means working under dry and inert conditions becomes second nature to those experienced with this reagent.
Technical Specifications & Labeling
Magnesium tert-butoxide comes labeled with purity specifications, typically above 98% for laboratory-grade bottles. Vendors include batch numbers, recommended storage temperatures, and cautionary pictograms for flammability and health hazards. You spot terms like anhydrous or stabilized, which refer to moisture content and freshness. I’ve seen many a mishap from unclear batch labeling, where traces of hydrolysis altered expected outcomes, underscoring how crucial clear and detailed technical data can be for experimental reproducibility.
Preparation Method
Synthesizing magnesium tert-butoxide usually means reacting magnesium turnings with excess tert-butanol, sometimes with a catalytic iodine to spark reactivity. Heating this mixture in suitable solvents—diethyl ether or tetrahydrofuran—eases conversion. The reaction releases hydrogen gas, so anybody at the bench pays close attention to venting. Once complete, oils and byproducts get washed away, and the compound crystallizes as a fine powder. Handling large batches takes skill, as impurities such as secondary alkoxides can form if temperatures swing too high or if atmospheric moisture sneaks in.
Chemical Reactions & Modifications
As a base, magnesium tert-butoxide enables key organic reactions, such as Claisen condensations and enolate formation. Its bulky tert-butyl groups steer selectivity toward less hindered sites, often producing cleaner product profiles. In my postgraduate work, using it in Michael additions trimmed byproduct formation, a benefit that potassium or sodium alkoxides just couldn’t match. Also, it enters exchange reactions with other alcohols, forming mixed alkoxides, and in cross-couplings, can facilitate transmetalation steps. Some inventive labs report modifying the magnesium alkoxide backbone with ligands, tuning electronic effects for asymmetric synthesis purposes.
Synonyms & Product Names
In chemical catalogs, magnesium tert-butoxide may go by names like magnesium 2-methyl-2-propoxide, tert-butoxymagnesium, or t-butylmagnesium oxide. Brand names rarely pop up, but you do spot proprietary blends for more complex magnesium alkoxides in advanced research settings. Having worked with a half-dozen suppliers, I've noticed minor differences in naming conventions, requiring careful attention to specification sheets. These discrepancies reinforce the need for clear communication in laboratory protocols.
Safety & Operational Standards
Working with magnesium tert-butoxide means respecting its reactivity. Fine powders easily ignite if exposed to open flame, and inhalation causes respiratory irritation. Laboratories store it in desiccators with ample warning stickers, and personal protective equipment—gloves, goggles, lab coats—is nonnegotiable. Local exhaust ventilation helps handle airborne particles. We mandated pre-lab safety walkthroughs, having learned that a splash or spill can escalate quickly. In my time training undergraduates, I stressed immediate cleanup of spills and maintaining strict dry conditions, since even a bit of ambient water transforms the powder into caustic slush.
Application Area
Magnesium tert-butoxide plays a major role in making pharmaceuticals, agrochemicals, flavors, and advanced polymers. Its ability to form magnesium enolates brings innovation to novel drug scaffolds and catalyst development. Startups focusing on greener chemistry gravitate to it to avoid the sodium and potassium waste streams. In my experience, flavor chemists rely on it for selective formation of key esters. Materials scientists push its limits for new polyether and polycarbonate structures, seeking the right combination of strength and flexibility. The breadth of its use keeps expanding, especially as more synthetic chemists explore non-traditional bases in challenging bond constructions.
Research & Development
Academic and industry labs run steady streams of research on magnesium tert-butoxide. Projects address tailored ligands for stereoselectivity or green chemistry tweaks, like solvent-free syntheses. In grant panels, I’ve seen funding go toward exploring its use in tandem with transition-metal catalysts for more sustainable cross-coupling. Publications in the last decade highlight new applications, such as forming carbon-carbon bonds under milder conditions or minimizing catalyst loading. These advances spur even more tests to identify side reactions, optimize scale-ups, and reduce purification steps, all aiming for efficiency and minimal waste.
Toxicity Research
Toxicological studies show that magnesium tert-butoxide poses both acute and chronic risks. Direct skin contact causes burns, and inhaling dust damages mucous membranes. The tert-butyl component carries a risk of central nervous system effects at high exposure, so workplace monitoring and medical surveillance matter. Environmental exposure brings concern over aquatic toxicity, pushing regulators to monitor waste disposal closely. In risk assessments I’ve reviewed, proper ventilation and disposal protocols stand out as the simplest ways to avoid incidents. Accidental ingestion, though rare, produces symptoms such as abdominal pain and vomiting, so rapid medical attention remains critical. Researchers continue to compare its toxicity profile with similar alkoxides, emphasizing a balance between performance and user safety.
Future Prospects
Step by step, magnesium tert-butoxide finds new applications as synthetic needs evolve. Chemists look to it for more selective, energy-efficient routes to complex molecules. The growth of green manufacturing, combined with stricter environmental regulation, pushes researchers to optimize its production, recycling, and waste control. Ongoing work aims to lower cost barriers for industrial-scale preparation while minimizing health hazards with improved formulation and safer packaging. With the boom in advanced materials and pharmaceuticals, scientists explore even wider applications, like flow chemistry and microreactors where rapid, high-yielding transformations demand precision. As labs worldwide share their findings, the compound’s profile rises steadily as a cornerstone in modern synthetic toolkits.
What is Magnesium Tert-Butoxide used for?
What Sets This Compound Apart
Magnesium tert-butoxide doesn’t pop up in dinner conversations, but for anyone who steps into a lab, its place on the shelf matters. This white powder acts as a strong base, and it’s not something you find at the local pharmacy. Chemically, it’s known for its bulkier structure compared to other alkoxides. That’s not just trivia; that structure makes a difference in how reactions turn out. In the world of organic chemistry, a small change in a molecule’s shape changes everything—kind of like how a single piece out of place ruins a puzzle.
Applications That Impact Science
Lab textbooks mention magnesium tert-butoxide in the same breath as big-name reactions. For instance, it steps in during the formation of carbon–carbon bonds, a fundamental move in building anything from pharmaceuticals to plastics. Magnesium tert-butoxide plays a big role in Claisen condensations. Anyone who has tried these reactions learns that the wrong base leads to unwanted byproducts. One study published by researchers at the University of California described how using this specific base improved yields and kept the reactions on track. That sort of impact saves time, money, and raw chemicals, reducing waste in the process.
Another application involves selective deprotonation. In non-technical terms, it helps chemists remove just the right hydrogen atoms from molecules. That matters in fine chemical production—think of painkillers, advanced plastics, or flavorings for food. Its bulky nature means it less often grabs the “wrong” atom, improving the chance of making only the desired product. Life is full of enough mistakes; having reagents that cut down on lab errors matters.
Why Attention to Safety and Training Matters
Despite its usefulness, magnesium tert-butoxide reacts vigorously with water and air. I remember a young chemist once left a bottle slightly open, and the result was a smelly, smoky mess that sent us clearing out the lab for ventilation. This experience left a lasting impression: respect what you don’t fully understand. Safety data from Sigma-Aldrich highlights the risks, urging gloves, goggles, and dry conditions, especially since it can ignite spontaneously.
Labs often invest in dry boxes—special sealed containers that keep air and moisture away—just to handle chemicals like this. Some labs save on these expensive containers at first, risking costly incidents later. The upfront investment pays off by preventing damage to property and risk to staff. This trade-off is familiar in many workplaces, but in chemistry, the consequences hit a bit harder.
The Push for Greener Choices
Magnesium tert-butoxide gets its fair share of criticism from environmental consultants. Its flammability and the challenge of disposing leftovers means waste management becomes a real issue. Some companies have started exploring alternatives, searching for compounds that deliver solid results with fewer safety risks. At a conference last year, one researcher presented a new catalyst that performed similar reactions but produced less flammable byproduct.
Chemists aren’t sentimental about tradition. If an option appears that works faster, safer, or greener, labs will move toward it. As regulations keep tightening, it won’t surprise me to see more universities and manufacturers looking for replacements or improving safety systems to keep magnesium tert-butoxide from causing trouble.
What is the chemical formula of Magnesium Tert-Butoxide?
Behind the Letters: What is Magnesium Tert-Butoxide?
Chemists sometimes joke that the hardest part of organic synthesis is just finding the right bottle on the shelf. Magnesium tert-butoxide, with its cryptic name, falls into that category. The actual chemical formula looks like this: Mg(OC(CH3)3)2. It means a magnesium atom holds onto two bulky tert-butoxide groups. In straightforward terms, each terb-butoxide is a carbon atom bound to three methyl groups and an oxygen, which connects to the magnesium. The formula looks intimidating, but each part describes something any lab chemist has likely wrestled with in a reaction flask.
Why Magnesium Tert-Butoxide Draws Attention
Labs turn to magnesium tert-butoxide for its strong base qualities. Sometimes, you need a base that stands up to water, doesn’t get knocked around by side reactions, and brings the bulk to keep things selective. In college, sleepless students usually learned about sodium or potassium tert-butoxide. Magnesium’s bigger cousin is a different beast. It gives you more control over certain organic syntheses—especially when you need that extra push but don’t want things to spiral out of control through unwanted side products. That’s value for any researcher keeping budgets tight and timelines even tighter.
What Formula Reveals About Handling and Safety
Anyone handling this compound understands the need for care. The formula, Mg(OC(CH3)3)2, tells us about sterics—the real crowding that influences how this molecule behaves. Those tert-butoxide groups surrounding the magnesium limit what can come close. For practical chemistry, it reduces unwanted reactivity, but also presents real-world headaches for storage. This compound fights water. Left out in humid air, it breaks down, sometimes rapidly. Proper storage involves air-tight bottles, low humidity, perhaps even an inert atmosphere. These steps come directly from understanding the formula. The more methyl groups, the bigger the target for water in the air. It isn’t just theoretical—it’s the difference between solid science and wasted supplies.
Magnesium Tert-Butoxide on the Larger Stage
On an industrial scale, this compound’s formula signals why it fits in certain syntheses. Pharmaceutical makers rely on strong, non-nucleophilic bases during key steps. Let's say you're creating a new drug and you need to remove a proton from a sensitive molecule. A base like magnesium tert-butoxide is almost surgical—it’s big, clunky, and less likely to grab onto something it shouldn’t. Using it instead of sodium or potassium bases sometimes means fewer byproducts, which cuts down on waste. The formula tells production chemists exactly how much they need, how pure the stuff must be, and what precautions to take so there’s no dangerous buildup of flammable tert-butyl alcohol during disposal.
Balancing Opportunity and Challenges
The formula also reminds us that advances often rely on basics—clear communication about what’s inside the bottle. Scientists make better decisions and safer workplaces when they trust what the formula tells them. Disaster strikes when a simple mislabeling causes someone to use the wrong base, possibly invalidating a whole sequence of expensive reactions.
Magnesium tert-butoxide, through its formula, stands as one more example of the way fundamental chemistry supports innovation, safety, and efficiency. Knowing those letters and numbers is never just about passing a quiz. It’s about making the next project run smoother—and about avoiding one more cleanup on Friday afternoon.
How should Magnesium Tert-Butoxide be stored?
Understanding Real Risks
Magnesium tert-butoxide doesn’t get much press compared to common household chemicals, but those handling it in research labs or industry know the stakes. This white, powdery solid can catch fire in air and reacts fiercely with water or humidity. Just a whiff of air can start trouble, so proper storage isn’t up for debate—it’s a must for anyone who cares about safety, health, and keeping a clean record.
My Lab Days: Lessons in Discipline
During my early years at a university lab, I saw a bottle of magnesium tert-butoxide left next to a sink. Within hours, its cap showed signs of warping. Every old hand in the lab shared a version of the same warning: never trust a poorly sealed container, and never treat the stuff like table salt. It drove home the lesson that routine and discipline keep people from making costly mistakes.
The Importance of Dry, Inert Conditions
Solid facts back up these anecdotes. Magnesium tert-butoxide reacts with moisture to form isobutylene and magnesium hydroxide. This means even air with regular humidity spells risk. Most sources agree: an airtight container always works best, with storage in a glovebox purged with nitrogen or argon. This strips away the biggest threat—accidental contact with air and water—and helps sidestep accidents like fire or toxic fumes.
Location, Location, Location
Anyone who has shared a lab knows, storage space isn’t just about shelf space—it’s about traffic, temperature, and buffer zones. Never place reactive substances like magnesium tert-butoxide near acids, flammable solvents, or oxidizers. An isolated, clearly labeled spot on a dedicated shelf inside a low-humidity desiccator lowers chances of error. The few minutes it takes upfront pay off with every trouble-free month.
Labeling: No Shortcuts
It sounds obvious, but always label the container—date received, user initials, and hazard warnings beat a bare jar every time. This isn’t just bureaucracy. When new staff join or an emergency hits, clear labeling can save time and prevent guesswork under pressure. From what I’ve seen, a small investment in labeling outperforms the best memory or spreadsheet on a bad day.
Training Makes the Difference
No one is born knowing how to handle chemicals like magnesium tert-butoxide. Effective training includes regular drills and open conversations about close calls. People need to know why moisture is the enemy, and what backup steps to use if the unexpected happens. Share stories, not just bullet points. The best advice often comes from people who’ve handled the material themselves and know where the process can break down.
Waste and Accident Response
Accidents can’t be completely avoided. Spilled magnesium tert-butoxide powders up fast and reacts quickly with water, which means cleanup needs more than just a mop and bucket. I’ve seen teams use dry sand or Class D fire extinguishers, keeping water as far away as possible. Every lab should have spill kits near reactive storage spots—no one wants to run across a building looking for supplies at the worst moment.
Pushing for Safer Habits
It’s easy to assume that proper storage means hardware and protocols—desiccators, inert gas setups, labels, gloves. But the human factor stays central. Real safety comes from a culture that values care, communication, and a lack of shortcuts. Good habits, shared stories, and reliable routines turn one hazardous powder from a headline risk into a quiet part of daily work. In my experience, the best defense doesn’t come from rules alone but from a team that’s seen what can go wrong and stays committed to getting it right.
What safety precautions should be taken when handling Magnesium Tert-Butoxide?
Why Caution Is Essential
Magnesium tert-butoxide doesn’t get the headlines like some other chemicals, but those who’ve spent time in a lab understand the risks tied to strong organic bases. The compound reacts easily, especially with moisture and air. Thinking back to my early chemistry days, lessons stuck with me not because someone read out a safety data sheet, but because I witnessed what goes wrong without real respect for chemicals like this.
Understanding the Hazards
Contact with skin or eyes brings burning and severe irritation. Inhaling dust can mess with your airways fast. This base ignites easily, especially around water, acids, and oxidizing agents. Hydrogen gas, for example, doesn’t belong in small spaces. Magnesium tert-butoxide also throws out heat when reacting with the wrong materials, creating fire risks out of nowhere.
Personal Protective Gear: Not Optional
Every chemist or tech working with magnesium tert-butoxide needs the right protection. Gloves made from nitrile or neoprene, not those regular latex gloves that look sturdy but let chemicals through after a few minutes. Chemical splash goggles stay on, even if the work “shouldn’t” splash. A lab coat covers arms, and closed shoes matter — toes can’t dodge spills.
Respirators come out for bigger quantities or fine powders. Relying on a room’s ventilation doesn’t cut it. Labs should have a fume hood in working order. Just propping open a window leaves everyone one bad cloud away from trouble.
Safe Storage Makes a Difference
Storing magnesium tert-butoxide just anywhere doesn’t work. Dry, cool locations keep it stable. Tightly sealed containers made from materials that resist strong bases keep the air and moisture out. Once opened, bottles get used up or closed again fast — too much air gets in, and the material degrades or forms clumps.
I learned to label containers with clear, readable writing. Only those familiar with the risks handle the compound. Untrained staff shouldn’t pick up a bottle just because it sits on a shelf; that leads to trouble faster than you can read the label.
Spill Response and Disposal
Spills can’t always be prevented. Small amounts on benches get swept up using materials that don’t react — synthetic scoops, not wooden tools. Water goes nowhere near cleanup unless someone wants a sudden hydrogen explosion. Spill kits kept nearby actually get used, not left in storage rooms behind locked doors.
Disposal works best following local rules. Pouring anything down a drain takes risks too far. Trained staff collect waste in separate, labeled containers and arrange proper pickup with hazardous waste handlers. Some labs try shortcuts to avoid disposal costs, but even small mistakes can bring big fines — and injuries.
Building Safer Work Habits
I’ve found the most useful safety tips grow out of routine. Refresher training helps. Checking gloves and making sure eye protection fits before starting work saves pain. Colleagues who look out for each other catch small errors before they turn dangerous. Posting clear instructions next to storage areas pushes folks to read before opening any bottle.
Constant vigilance goes a long way. Distractions in the lab lead to accidents, so phones, snacks, and unrelated conversations stay out until the work wraps up. Sharing stories of close calls encourages newer staff to respect the risks. Safety feels like an extra step until someone makes a mistake. With magnesium tert-butoxide and other reactive chemicals, taking shortcuts isn’t worth the risk.
Is Magnesium Tert-Butoxide soluble in water?
Understanding How Magnesium Tert-Butoxide Behaves in Water
Anyone who’s spent time in a lab knows that predicting whether a chemical will dissolve can be trickier than it seems. Magnesium tert-butoxide stands out as one of those compounds that don’t simply swoosh into a clear solution when dropped in water. Ask anyone who’s mixed the two: this magnesium compound tends to form a cloudy mess rather than a smooth solution. The molecule just isn’t designed for a water-friendly experience.
Many folks wonder about the details. Magnesium tert-butoxide contains magnesium bonded to bulky tert-butoxy groups. It owes much of its character to this setup. These tert-butoxy groups like to keep their distance from water molecules, which makes the entire compound resistant to mixing in. If you try dissolving it straight into water, those groups repel the water, causing the compound to clump. That’s why the Internet is full of people swapping tips for handling it: they know about the difficulties first-hand.
Practical Implications in Research and Industry
Anyone making magnesium tert-butoxide is usually preparing for reactions that only happen in special conditions. Normally, workers use it in dry, oxygen-free setups. Picture people in coats fiddling with glassware and making sure not a drop of water sneaks in. The solubility game plays a huge role in this routine. If magnesium tert-butoxide could dissolve well in water, it might make some syntheses easier. Instead, chemists often reach for organic solvents—ethers and hydrocarbons like hexane or toluene—because these keep things smooth and predictable.
For most industrial or academic applications, water just won't cut it. Those tert-butoxy arms block water molecules, and the magnesium core doesn’t offer a helping hand. At best, you get a suspension, not a true solution. The lack of solubility creates headaches for anyone eyeing aqueous processing or cleanup. If the material’s got to be removed, disposal or recycling depends on responsible solvent handling.
Why Care About These Details?
This all impacts not only researchers but also anyone interested in safe handling and environmental impacts. Many sources, such as the PubChem database and academic reviews, flag its poor water solubility. Chemists must keep their hands steady because water in the mix could mess up their carefully planned reactions. Awareness of solubility cuts down error rates and keeps big projects from going off the rails.
Environmental consequences matter as well. If magnesium tert-butoxide found its way into the water supply, it wouldn’t dissolve and mix harmlessly. It could clump or react with other materials, leading to local pollution or tricky storage problems. And because it doesn’t break down in water, cleanup can become a complicated, costly affair. Labs and factories stick to tight protocols: solvents carefully tracked, spills managed by containment specialists, not hoping the compound simply washes away.
What Scientists and Industry Can Do
Solutions for these challenges rarely come quickly, but the right approach can reduce headaches. Using gloveboxes with dry, controlled air keeps water at bay from the start. Newer labs switch to greener organic solvents where possible, and researchers chart out detailed reaction plans to avoid any water contact. Rather than forcing solubility, the focus shifts to control—managing the context, not just the reaction. Industry-wide, more effort goes into training and setting up robust environmental safeguards because a slip-up in handling isn’t just a minor inconvenience; it risks unexpected reactions or waste buildup.
Learning from these hurdles guides everyone from undergraduate lab students to large-scale manufacturers. A simple compound’s water behavior may look like a small detail, but it affects decisions about safety, workflow, and environmental protection. Magnesium tert-butoxide reminds us: in chemistry, the tiniest details can matter the most.
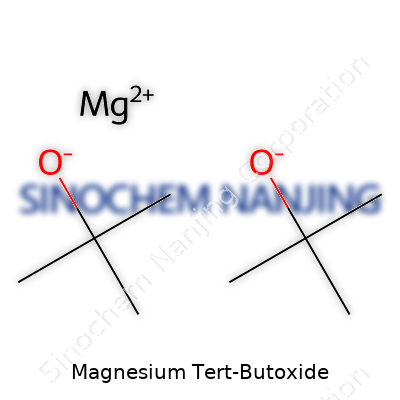
| Names | |
| Preferred IUPAC name | Magnesium 2-methylpropan-2-olate |
| Other names |
Magnesium 2-methylpropan-2-olate Magnesium tert-butylate Magnesium tert-butoxide |
| Pronunciation | /maɡˈniːziəm tɜːt ˈbjuːtəˌksaɪd/ |
| Identifiers | |
| CAS Number | 16324-26-6 |
| Beilstein Reference | 4148730 |
| ChEBI | CHEBI:37156 |
| ChEMBL | CHEMBL508160 |
| ChemSpider | 61347 |
| DrugBank | DB11497 |
| ECHA InfoCard | 07a672e2-75b8-481b-90d8-2d6b98c6688c |
| EC Number | 216-367-7 |
| Gmelin Reference | 6132 |
| KEGG | C14147 |
| MeSH | D017765 |
| PubChem CID | 159722 |
| RTECS number | OM3850000 |
| UNII | WB5K89A8NV |
| UN number | UN 2003 |
| Properties | |
| Chemical formula | Mg(OC(CH₃)₃)₂ |
| Molar mass | 166.32 g/mol |
| Appearance | White powder |
| Odor | Odorless |
| Density | 0.958 g/mL |
| Solubility in water | Decomposes in water |
| log P | 0.6 |
| Vapor pressure | < 1 mmHg (20 °C) |
| Acidity (pKa) | 26.0 |
| Basicity (pKb) | 2.94 |
| Magnetic susceptibility (χ) | -7.0×10⁻⁶ |
| Refractive index (nD) | 1.383 |
| Viscosity | Viscous liquid |
| Dipole moment | 2.86 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 216.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -696.7 kJ/mol |
| Pharmacology | |
| ATC code | A02AA10 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H260, H314 |
| Precautionary statements | P210, P222, P231+P232, P280, P305+P351+P338, P370+P378, P422 |
| NFPA 704 (fire diamond) | 3-2-1-W |
| Autoignition temperature | 430 °C |
| Lethal dose or concentration | LD50 Oral - Rat - 5,000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral, rat: >2,000 mg/kg |
| NIOSH | Not established |
| PEL (Permissible) | Not established |
| REL (Recommended) | 10 mg |
| Related compounds | |
| Related compounds |
Magnesium methoxide Magnesium ethoxide Sodium tert-butoxide Potassium tert-butoxide |

