Magnesium Selenite: Commentary and Exploration
Historical Development
Looking back, researchers noticed selenium had something special as early as the nineteenth century. Magnesium selenite emerged in labs a bit after that, following the typical pattern of mixing magnesium salt solutions with those containing selenium. As synthetic chemistry matured, scientists began to study magnesium selenite in earnest, outlining its chemical structure, mapping its solubility, and digging into user-friendly production methods. Industrial chemists watched closely, seeing its potential for specialized uses where magnesium and selenium’s properties overlap—think agriculture, pharmaceuticals, and nutritional supplements. In decades past, serious attention landed on the molecule’s biological influence, as selenium’s crucial role in animal health moved it out of obscurity and into feed formulas and supplement bottles.
Product Overview
Anyone handling minerals and trace elements gets familiar with magnesium selenite’s powdery white appearance. Suppliers often offer it as a crystalline powder or sometimes granules, with easy dispersion in water. The chemical formula is MgSeO3 or sometimes MgSeO3·xH2O, depending on the water of hydration. Producers offer purity ranging from technical to reagent grade, and careful labeling tracks its selenium content—vital in feed and supplement industries, where dosing means the difference between nourishing and harming animals or people. Often, companies choose magnesium selenite instead of pure selenium or other forms (like sodium selenite), especially where magnesium’s nutritional role also matters.
Physical & Chemical Properties
Magnesium selenite presents as a white, odorless, chalky or sometimes crystalline powder. In terms of solubility, it dissolves in water at measurable rates—better than some other selenites—which helps with mixing and blending tasks. Its melting point climbs above typical room conditions, and it stays fairly stable under dry storage. Chemically, magnesium selenite holds selenium in a +4 oxidation state, which impacts how it reacts in biological systems and environmental conditions. Keep this dry, away from acids, since strong acids can turn selenite ions into toxic selenium dioxide gas. In the body and in soil, magnesium and selenium ions can separate, each going their own way—making bioavailability a distinct concern during product development.
Technical Specifications & Labeling
Suppliers who know their market label packages with clarity—often displaying selenium and magnesium content as weight percentages, along with precise batch analysis results. Labels usually include molecular weight, solubility, recommended storage temperatures, and any water-of-hydration variation. Users expect technical details on impurity limits (arsenic, lead, cadmium, etc.), as trace toxins draw regulatory scrutiny in all animal and human health products. In regulated markets, the law expects information on recommended maximum daily selenium intake, along with first-aid instructions in case of accidental overexposure. Simple, honest labeling supports safe handling, which matters most when products end up in someone’s food, feed, or supplement blend.
Preparation Method
Producers usually start with water-soluble magnesium salts—like magnesium sulfate or magnesium chloride—and react those with sodium selenite or directly with selenous acid. The result is a precipitate of magnesium selenite, which gets filtered, washed, and dried. Quality-managing this process means paying attention to pH and temperature for best yield. Some labs use a more direct pathway, bubbling sulfur dioxide through slurries of magnesium carbonate and elemental selenium, leveraging redox chemistry to form selenite in-situ. High-purity preparation sometimes means repeated recrystallization. To scale up, manufacturers automate pH control, manage filtration carefully, and track batch purity with analytical methods like ICP-MS for selenium content.
Chemical Reactions & Modifications
Though stable in dry containers, magnesium selenite reacts if mixed with acids. Acidification can readily produce selenous acid and magnesium salts. Under reducing conditions, the selenite ion drops down to elemental selenium, popping out of solution as a deep red precipitate. In the presence of strong oxidizers, the selenium in selenite can climb up to the selenate state or even selenic acid, though these reactions need well-controlled lab setups. Making derivatives means combining magnesium selenite with different cations or co-precipitating with other mineral nutrients, which opens doors for custom blends targeted at animal nutrition or soil remediation.
Synonyms & Product Names
Chemists know magnesium selenite under a few labels, including Magnesium selenium oxide and Magnesium(II) selenite. Trade names sometimes include the hydration state, like magnesium selenite pentahydrate, and companies attach internal codes or branding for specialty use. Certain feed additives or supplements include magnesium selenite as one of several “trace mineral selenite salts.” This can confuse buyers, so anyone purchasing bulk supplements or premixes needs to read certificates of analysis as well as the front label.
Safety & Operational Standards
Handling magnesium selenite carries some risk. Selenium compounds, even in small quantities, cross the line from supplement to toxin if handled carelessly. Staff in factories wear gloves and masks, and ventilated hoods stay on during weighing and mixing to prevent accidental inhalation or skin absorption. Modern standards require locked storage rooms, digital logs, and emergency response gear. Feed and supplement manufacturers work within tight regulatory windows, as excess selenium can harm animals and people through chronic accumulation. Safety data sheets point out that selenite dust, if inhaled or swallowed in significant doses, causes symptoms like garlic breath, nausea, or even nervous system effects. Safe disposal involves controlled incineration or certified waste management, never down the drain or in regular landfill.
Application Area
Magnesium selenite earns its keep in animal feed, nutritional supplements, and laboratory experimentation. Agriculture uses it to combat selenium deficiency in soils where crops and livestock otherwise suffer. In these dry and weathered soils, magnesium selenite dissolves more predictably than metallic selenium, which lets farmers supply selenium in precisely measured doses. Aquaculture and pet food suppliers value its dual magnesium and selenium content to cover multiple nutritional bases. Labs draw on it for research into the role of selenium in enzyme function, antioxidant protection, and metabolic pathways. Some antifungal formulations and glass manufacturing processes draw on magnesium selenite properties, but nutrition and research remain its largest users.
Research & Development
Scientists continue to dig into magnesium selenite’s impact on health. Animal studies link proper selenium intake with healthy immune systems, muscle development, and reproductive outcomes. Over and under-supply both produce trouble—so nutritionists spend years defining optimal addition levels for cattle, sheep, poultry, and sometimes fish. Work continues into bioavailability, as not all forms of selenium get absorbed by the same tissues or at the same speed. Research into combination minerals, where magnesium selenite acts in concert with zinc, copper, and other trace elements, aims to design all-in-one supplement blends that avoid known interactions and toxicity risks. Some clinical researchers look toward controlled magnesium selenite use for humans in selenium-scarce regions, studying metabolic and neurological benefits.
Toxicity Research
Scientists stay wary of selenium’s narrow safe window—too little and selenoproteins malfunction, too much and toxicity sets in. Workers document cases of livestock selenium poisoning (alkali disease or blind staggers), usually from excess supplementation or accidental mixing errors. Chronic exposure among humans can produce garlicky breath, hair loss, and nervous system complaints. Tests in rodents confirm toxicity at several times the daily recommended intake, but these findings support today’s strict regulatory guidance. Chemists test every batch for contamination from heavy metals, as selenium’s health effects can get muddled by toxic impurities. Long-term epidemiological studies map areas with natural selenium excess or deficiency, driving public health decisions in both crop supplementation and water management.
Future Prospects
Magnesium selenite stands to gain ground as food systems become more trace mineral-conscious. As arable land wears out, soils across regions lose the selenium reserves that once leached down from rocks and rainfall. More countries adopt mandatory selenium supplementation in feed, widening market demand for pure and reliable magnesium selenite. Researchers track soil microbe responses to repeated magnesium selenite use, checking for buildup or resistance. Food supplement companies design slow-release pills using specialized selenite complexes, looking to mimic natural dietary absorption. Next-generation research tools—like gut-microbiome modeling and nutrigenomics—may redefine dosing, personalizing selenium intake far beyond today’s population-level guidelines. This trace mineral may never get front-page attention, but magnesium selenite offers a snapshot of where science, safety, and long-term sustainability meet.
What is Magnesium Selenite used for?
Why Magnesium Selenite Matters
Some compounds get a lot of buzz, while others slip under the radar. Magnesium selenite is one of those quiet contributors in both animal nutrition and, sometimes, supplement manufacturing. Years back, I noticed it listed in ingredients for specialty livestock feed, sparking my curiosity about the role it plays.
A Boost for Animal Nutrition
Farmers and pet owners want healthy animals. Getting the right trace minerals into their diets makes a world of difference. Magnesium selenite provides selenium, a trace mineral, and pairs it with magnesium, another essential nutrient. Selenium helps with immune function and antioxidant protection in muscles and organs. Cattle, poultry, swine—many animals need careful supplementation because natural forages may not provide enough, especially in areas with low-selenium soils.
Adding magnesium selenite to feed supports growth, reproduction, and general health in livestock. Studies show that farm animals with access to the right selenium levels grow faster and produce healthier offspring. Without it, you see stunted growth, reduced fertility, and weak immune systems. For farmers facing losses from these issues, proper mineral supplementation isn't a luxury—it's a necessity.
Selenium and Public Health
Selenium deficiencies affect humans too, especially in parts of the world where the soil lacks this element. In fact, some regions in Europe and China have seen huge impacts on children’s development and immune status because of poor selenium intake. Magnesium selenite turns up in some food supplements for this reason, although not as commonly as selenite or selenate salts that dissolve better in water.
It’s important to remember that selenium's safe use depends on the dose. Too little selenium can leave people and animals vulnerable to oxidative stress. Too much tips the scale toward toxicity, causing hair loss, brittle nails and gastrointestinal distress. That’s why feed formulators and supplement manufacturers work with nutritionists and regulators to set safe limits.
Environmental and Safety Questions
Magnesium selenite, like every mineral supplement, needs careful handling. Dust from the powder can irritate lungs or skin, so safe storage and use matter to protect workers. Waste run-off from concentrated livestock operations sometimes brings selenium into local water, which is a real concern for aquatic systems—too much selenium can harm fish and wildlife.
Some countries have strict environmental regulations because of past problems with selenium run-off. One productive solution involves regular soil and water testing, precision dosing in feed, and close monitoring for signs of mineral build-up. Balancing nutrition with environmental care calls for teamwork between farmers, nutrition professionals, and environmental experts. Clear guidelines exist for safe feed formulations, and regular oversight helps keep both animals and the environment in good shape.
What’s Next for Magnesium Selenite
As animal science continues to evolve, the focus on optimal, evidence-based nutrition grows. Ongoing studies review the best forms, doses, and combinations of trace minerals for each type of animal at every life stage. Nutrition experts advocate for targeted supplementation—avoiding old habits of “more is better.” Digital tracking of feed, animal health reports, and feedback from farmers can help catch problems earlier, reducing losses while also benefiting animal welfare.
For the average person, magnesium selenite is not a household name. Yet its impact trickles into daily life every time crops thrive, animals stay healthy, and the food supply grows safer. Knowing the reasons behind such feed additives helps everyone, from producers to consumers, make more informed choices and keep food systems strong.
Is Magnesium Selenite safe for consumption?
What is Magnesium Selenite?
Magnesium selenite belongs to a group of minerals that combine magnesium and selenium, both essential nutrients. You’ll find it listed on the labels of some supplements and fortified foods, often marketed as a way to boost your selenium intake. Selenium supports thyroid health and acts as an antioxidant, while magnesium plays a role in muscle and nerve function. On paper, it sounds like a practical combo, but the exact chemistry can make a difference in your body.
The Nutritional Side
Selenium is required in very small amounts. The Recommended Dietary Allowance (RDA) for adults sits at 55 micrograms per day. Magnesium’s RDA is much higher, at around 400 milligrams per day. Magnesium selenite provides both elements, but here’s the catch—selenium in selenite form doesn’t absorb as efficiently as in organic forms, like selenomethionine you find in Brazil nuts, eggs, or fish.
Some supplement companies use magnesium selenite as a cost-saving measure. Big brands claim it “meets selenium needs.” But studies show selenite carries greater risk for toxicity if your intake climbs too high. The World Health Organization recognizes both benefits and risks, emphasizing that long-term overconsumption can cause symptoms such as hair loss, digestive upset, a metallic taste, and in extreme cases, acute poisoning.
Where Concerns Show Up
Problems can sneak up when people pop supplements without looking at doses or consider the source. From personal experience, you’ll notice that even health-conscious folks rarely check the chemical form of minerals in their vitamins. It’s tempting to think, “Selenium is selenium.” But the chemical structure can matter for safety and effectiveness.
The FDA treats magnesium selenite as “generally recognized as safe” (GRAS) up to certain limits in food, but the fine print matters. The tolerable upper intake for selenium is 400 micrograms per day for adults. Magnesium selenite supplements often provide selenium close to this boundary, especially when stacked with a selenium-rich diet.
Sourcing and Regulation
Not all supplement brands take quality control seriously. Research published in the Journal of Trace Elements in Medicine and Biology found that selenium content in supplements sometimes veers wildly from what the label promises. Poor labeling control lands people at risk for deficiency or overdose. Testing and transparency from credible manufacturers go a long way.
European and North American agencies both study selenium safety. Reports published in the EFSA Journal and by the National Institutes of Health highlight that organic selenium sources—like selenomethionine—prove safer and better absorbed. Inorganic selenites, including magnesium selenite, sit lower on the list for efficiency and margin of safety.
Smart Steps for Safer Choices
Reading supplement labels and understanding what form of selenium you’re getting could spare you some grief. Look for specifics like “selenium (as selenomethionine)” rather than selenite or selenate if you want to minimize your risk. Reach for brands that focus on third-party testing and openly share lab results.
Whole food sources usually outshine isolated ingredients for both safety and balance. But for people with clear deficiencies (confirmed by a doctor), low-dose, well-tested supplements sometimes make a difference. No one benefits from wild swings—neither in minerals nor in opinions—so cautious, informed choices keep health in check.
What are the side effects of Magnesium Selenite?
A Close Look at Magnesium Selenite
Magnesium selenite usually shows up in supplements or animal feeds as a source of both magnesium and selenium. Both minerals matter for health. Magnesium keeps muscles working right and nerves firing properly. Selenium gets a lot of attention for its antioxidant role and the way it supports thyroid function.
Possible Side Effects and Health Concerns
Dropping a supplement in your daily routine often feels harmless, but magnesium selenite brings real risks if not managed carefully. Selenium stands out as a trace mineral—the body only needs a tiny amount. Too much tips the balance and causes problems. Symptoms may not crop up right away, but they build up and can surprise people who don’t keep an eye on their intake.
Common signs of selenium overload include a metallic taste, garlic-like breath odor, nausea, diarrhea, and brittle hair or nails. Higher levels push into more dangerous territory: brain fog, irritability, fatigue, and in rare cases, nerve damage. I’ve seen people shrug off their tiredness or hair shedding, chalking it up to stress, only to find out later it traced back to selenium supplements. The National Institutes of Health marks the upper safe limit for adults at about 400 micrograms a day, and going past this line bumps up risk for toxicity.
Magnesium brings its own set of issues when taken in excess, usually through supplements. Upset stomach, diarrhea, and sometimes irregular heartbeat can follow. People with kidney problems face more risk since their bodies struggle to flush out extra minerals. Too much of either mineral over a long span sometimes leads to health problems that can be hard to reverse. I’ve seen cases where simple supplements caused real headaches for people managing chronic conditions.
Scientific Backing and Real-World Use
Experts agree that safe selenium and magnesium levels come mostly from food. The Office of Dietary Supplements and European Food Safety Authority both warn against high doses through pills. In some animal feed, magnesium selenite serves a purpose, but even livestock can suffer from too much. Over-supplementation in livestock causes hoof and coat problems along with digestive upset.
One study found that long-term use of inorganic selenium like selenite increases the risk of toxicity in both people and animals. Another review highlighted that organic sources, like selenium-rich yeast, tend to be safer and less likely to trigger harmful effects, though the dose still matters most. My years talking with nutritionists and seeing how quick fixes backfire taught me that natural, food-based sources prevent a host of worries. A Brazil nut or two delivers selenium safer than most pills.
Safer Approaches and Responsible Supplement Use
If a doctor says you need more magnesium or selenium, ask for a blood test to be sure. Avoid self-diagnosing a deficiency and reaching for supplements without guidance. Most balanced diets cover mineral needs. Pack your plate with nuts, seeds, green veggies, and seafood for magnesium, or fish and eggs for selenium. That way, the risk of overdose drops and your body absorbs the minerals correctly.
Always check ingredient lists and dosages if supplements do appeal to you. One product may pile on way more selenium than another, and brand reputation means everything. If you notice any sudden fatigue, digestive changes, or odd tastes, stop taking the supplement and check with a qualified doctor. Looking at nutrition labels and knowing your limits does more for your health than doubling up on pills ever could.
How should Magnesium Selenite be stored?
Handling a Sensitive Compound
Magnesium selenite rarely pops up in daily conversation, yet this compound stays Relevant in many labs and industries. From nutrient supplements to animal feed, people put their trust in the quality of the product used. That quality starts with effective storage. I remember a time working in a small research lab—chemicals lost their power not because of improper synthesis, but from sloppy storage. Moisture, sunlight, or even the wrong material for containers sank months of planning in just a few days.
No Room for Guesswork
Magnesium selenite demands more than basic care. Chemistry textbooks spell out those handling instructions for a reason. I’ve seen careless stacking of powders on cluttered shelves, squeezed between corrosives and acids, just because someone assumed every salt shares the same rules. This behavior can cost lives—or at best, waste money and time.
Keep this compound in a tightly closed container. I choose glass or high-quality plastic to avoid unwanted reactions. Don’t just toss it anywhere; always pick a cool, dry place, away from direct sunlight. Heat and moisture break down magnesium selenite, making it useless or, worse, dangerous. I once found containers sweating on a ledge above a steam pipe. So much risk, all from a lazy storage choice. There’s no shortcut: look for shelving away from heating vents and out of contact with water.
Health: No Excuses
Exposure isn’t something to shrug off. Selenium compounds can harm the skin, eyes, or lungs. I wear gloves and a dust mask, and urge my coworkers to take similar steps. Eye protection matters. Too many people skip over safety until something splashes or spills. Spilled powder left open in a humid room turns into a clean-up nightmare and a health hazard. Reseal containers every time. Label them clearly with hazard warnings, not just chemical names.
Contamination Risks
Cross-contamination shouldn’t be an afterthought. I’ve seen labs use the same scoops or spoons for different chemicals. That lazy habit brings trouble—not just for you, but for anyone using what’s supposed to be a pure batch. Use dedicated tools and keep them dry. Silicone desiccant packs in storage help block extra moisture, especially if the main room gets sticky in summer. Replace them often to keep the climate consistent inside the container.
Safe Storage, Reliable Outcomes
Safety and product quality walk hand in hand. Too much humidity rusts confidence, not just the product. If magnesium selenite supports animal nutrition or research, poor handling can disrupt results and waste resources. I stick to a checklist. Are there fire hazards nearby? Does the container show signs of cracking? Are all labels current and legible? Simple habits turn into reliable outcomes, whether running a small lab or a big operation. Secure, well-chosen storage isn’t just about following regulations—it's how I make sure everyone stays protected and the compound delivers what it promises every time.
What is the recommended dosage of Magnesium Selenite?
Understanding the Role of Magnesium Selenite
People sometimes overlook Magnesium Selenite in conversations about dietary minerals. Still, it shows up in trace mineral blends, animal feed, and certain supplements. The scientific journals haven't left much room for guesswork about its role: this compound provides both magnesium, which supports muscle and nerve function, and selenium, which helps the body’s antioxidant defenses and thyroid performance.
Recognizing the Right Amount
It’s tempting to pile on nutrients, especially with trace minerals. Popular wisdom says more is better, but that doesn’t work with certain minerals. Too much selenium tips the scales toward toxicity—hair loss, nerve pain, and digestive trouble all make doctors reach for the lab tests. The US National Institutes of Health pegs the Recommended Dietary Allowance (RDA) of selenium for most adults at 55 micrograms a day, rarely going over 400 micrograms unless a doctor has a specific reason. Even well-established livestock nutrition guides stick around 0.1–0.3 parts per million in feed, with Magnesium Selenite providing the selenium in a carefully calculated formula.
Magnesium doesn’t create trouble quite as fast as selenium. Adults need 310–420 milligrams each day, depending on age and sex. Food supplies most of it, from leafy greens, nuts, and beans. Supplements usually fill gaps left by diet or loss from chronic illness. Magnesium Selenite provides both nutrients at once, so you have to check both sides of the equation: not too much selenium, not too little magnesium. Some supplement manufacturers deliver doses in line with the selenium RDA, seldom topping 55–70 micrograms unless medical advice says otherwise. For magnesium, the contribution from the compound stays small, so it rarely affects the overall magnesium intake much.
Risks Tied to Dose Mistakes
A reckless approach creates dangers. Industrial accidents have poisoned livestock and wildlife with selenium. Health professionals see organ damage from long-term excess use, especially when unregulated supplements float through the market. I've treated patients who bought “mega-dose” products online, hoping for a bigger effect and winding up with shaking hands and relentless nausea. That kind of experience sinks in. Care with dosage isn’t just about following government rules—it's about respecting the body’s narrow safe zone for selenium, especially when using ingredients like Magnesium Selenite.
Getting Reliable Information and Support
Doctors and veterinarians keep up with new reports and usually recommend lab tests to check selenium and magnesium levels before handing out supplement advice. Government agencies lay out strict upper limits for selenium to protect public health. Supplement companies can earn trust by showing batch tests and sticking to these limits in their products. If you're reading supplement labels, the goal is to find clear data showing micrograms of selenium per dose, not vague marketing hype.
Safer Supplement Habits and Smarter Choices
Take supplements carefully. If your doctor prescribes Magnesium Selenite, ask questions about dosage. Look for third-party test results from reputable labs, not claims without proof. For livestock owners, match the blend to the species and local forage, since selenium levels swing across regions. Skip “one-size-fits-all” solutions. Sticking close to published RDAs steers you away from most problems, making it easier to get the benefits without the risks. Real wellness comes from information and moderation—not the biggest bottle on the shelf.
| Names | |
| Preferred IUPAC name | Magnesium trioxidoselenate(2−) |
| Other names |
Selenious acid magnesium salt Magnesium selenious Magnesium(II) selenite |
| Pronunciation | /maɡˈniːziəm səˈliːnaɪt/ |
| Identifiers | |
| CAS Number | 13123-41-6 |
| Beilstein Reference | 82326 |
| ChEBI | CHEBI:77636 |
| ChEMBL | CHEMBL61375 |
| ChemSpider | 133088 |
| DrugBank | DB11136 |
| ECHA InfoCard | 20d74008-720e-4274-8a77-3334fabdfabc |
| EC Number | 231-576-4 |
| Gmelin Reference | 84798 |
| KEGG | C18718 |
| MeSH | D008263 |
| PubChem CID | 24562 |
| RTECS number | OM3850000 |
| UNII | 3W8330755B |
| UN number | UN2630 |
| Properties | |
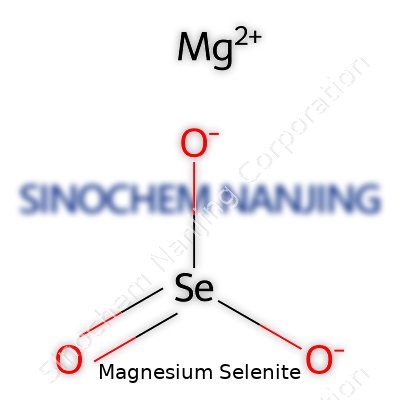
| Chemical formula | MgSeO3 |
| Molar mass | 143.27 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 3.17 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | -4.636 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 6.18 |
| Basicity (pKb) | 8.45 |
| Magnetic susceptibility (χ) | -26.2·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.651 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 121.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1242 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1187 kJ/mol |
| Pharmacology | |
| ATC code | A12CC10 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. |
| Precautionary statements | P264, P270, P273, P301+P312, P330, P501 |
| NFPA 704 (fire diamond) | 2-1-1 |
| Autoignition temperature | 700 °C (1,292 °F; 973 K) |
| Explosive limits | Non-explosive |
| Lethal dose or concentration | LD50 oral rat 11.2 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 10 mg/kg |
| NIOSH | RN 13770 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Magnesium Selenite: 0.2 mg/m³ (as Selenium, OSHA PEL, 8-hour TWA) |
| REL (Recommended) | 0.03 mg/kg |
| Related compounds | |
| Related compounds |
Magnesium sulfate Sodium selenite Magnesium selenate |

