Magnesium Nitrate: A Closer Look at Its Story, Science, and Significance
Historical Development
Magnesium nitrate caught the attention of chemists back in the days when mineral exploration opened up a rush for new compounds. Nineteenth-century scientists in Europe grew curious about two things: how different magnesium salts behaved in soils, and which ones worked well as fertilizer. The hunt often led to sources such as the earth around mines and some specialized rocks. Its use as a fertilizer really picked up steam in the early twentieth century, especially as folks learned more about soil depletion and how magnesium made plants tougher and more productive. The product went from lab curiosity to practical commodity in farms, factories, and classrooms. Each generation of chemists found new ways to make it, purify it, and apply it, and much of this progress came on the back of trial and error paired with a steady rise in scientific tools. By the time modern agriculture ramped up, magnesium nitrate sat firmly among those chemicals that people who work with soil and plants trust as a problem solver.
Product Overview
Magnesium nitrate serves a simple purpose: plants crave magnesium, and folks working in horticulture found this salt provides it in a trusted form. It shows up as a white, crystalline solid that dissolves in water without fuss. Rather than posing as an ingredient in household products, it draws most of its value from agricultural and industrial fields. This salt handles tasks from fertilizing greenhouse crops to helping make specialty explosives and absorbents. In my experience, asking an agronomist about plant nutrition usually sparks a story about magnesium shortfalls making leaves yellow, and magnesium nitrate comes up as an easy fix. That’s a no-nonsense value you don’t see with every compound.
Physical & Chemical Properties
Those small crystals offer a fair bit under the microscope. Magnesium nitrate hexahydrate, a common form, melts near 88 °C and falls apart with a pop at higher heat, releasing nitrogen oxides and water. This is not the thing anyone wants to heat without precautions. It dissolves fast in water, pumping both magnesium and nitrate into solution — an advantage for folks mixing up liquid feeds for crops. Left exposed, it pulls moisture from the air and ends up sticky, so it needs dry storage and sealed bags. The material weighs about 1.46 grams per cubic centimeter, and its chemical formula writes out as Mg(NO3)2·6H2O. It also attracts attention from those focused on oxidation…I’ve worked in a lab setting where this reactivity led to some exciting flames with organic powders if left unchecked.
Technical Specifications & Labeling
Industrial magnesium nitrate usually declares a magnesium content of 9–11% in the hexahydrate form, and the nitrate part takes up most of the rest. Bags come stamped with hazard pictograms: oxidizing agent, health warning, and some brands highlight that it shouldn’t be mixed with certain reactive chemicals. The pH in water solution skews acidic, running between 5 and 7 depending on dilution. The labeling must note batch, purity, and lot number, along with a clear reminder to store the product far from flammable materials. In my time working with regulatory compliance, documentation always demanded a certificate of analysis and Material Safety Data Sheet before a supplier could deliver a pallet.
Preparation Method
Magnesium nitrate can be made by treating magnesium oxide, hydroxide, or carbonate with nitric acid, which fizzles and froths, dissolving the solid and releasing carbon dioxide if the carbonate’s in play. In industry, it’s more common to run a controlled reaction of magnesium carbonate with nitric acid, then concentrate the resulting liquid and cool it down to grow those distinctive crystals. Any leftover acid needs neutralization, and the finished crystals get separated and dried under exacting conditions. Larger factories recycle some waste streams, stripping out reusable acid and magnesium—it’s impressive to watch the process scale from small glass beakers to massive stainless-steel reactors in a proper chemical plant.
Chemical Reactions & Modifications
Mix magnesium nitrate with a base and it gives up magnesium hydroxide, while combining it with ammonium salts in controlled settings yields double salts used in fertilizer blends. Strong heating drives out nitrogen oxides and leaves behind magnesium oxide powder. In water, the salt provides free ions that move readily throughout irrigation networks and plant roots. Chemists looking to tweak its reactivity might blend it with other nitrates to alter release profiles or with chelating agents for better micronutrient mobility. Its strong oxidizing nature never loses relevance; in the wrong company, magnesium nitrate speeds up combustion and can create hazards with organic matter. This isn’t a chemical most folks store near anything remotely combustible.
Synonyms & Product Names
This product goes by several different names depending on where it’s sold and what industry uses it. You’ll see terms like nitrate of magnesia, magnesia nitrate, and magnesium dinitrate on import listings and chemical catalogues. The hexahydrate is the most common industrial grade, sometimes labeled as nitromagnes, or by trade names that hint at its use in specialty horticulture formulas. The many regional product codes can get confusing, especially for buyers new to international supply chains. My background in procurement involved quite a few hours lost to deciphering product numbers across suppliers.
Safety & Operational Standards
Anyone handling this compound wears nitrile gloves and ensures a good dust mask is handy. Because it’s a strong oxidizer, strict rules keep magnesium nitrate far from oils, sawdust, paper, and other organic materials. Storage stays cool, dry, and away from sunlight with containers sealed tight after each grab. Personal experience in a fertilizer blending plant hammered home just how strictly workers monitor humidity—it only takes one slip to turn a harmless bag into a sticky, tricky mess. Emergency eye-wash stations and clean-up protocols are as much a part of the facility as the chemical drums themselves. Regulations cover not only worker exposure, capped at levels set by workplace safety boards, but also rules for environmental discharge to prevent nitrate buildup in waterways.
Application Area
Nearly every commercial greenhouse relies on magnesium nitrate for its tomato, pepper, and leafy crops, especially in regions where soils struggle to deliver essential magnesium. It often winds up inside specialized nutrient blends designed for hydroponics, as its rapid solubility ensures plants pick it up right away. Outside agriculture, the nitrate makes itself useful in the manufacture of explosives, pyrotechnics, and certain heat-storing salts. Municipal water treatments explore it as a source of magnesium ions, and some specialty absorbents feature it because of its water-loving nature. From my perspective, supply always seems tightest during the spring rush, when growers scramble to keep their crops supplied as weather warms.
Research & Development
Research into fertilizer technology keeps finding ways to use less chemical and deliver more benefits, and magnesium nitrate catches interest for its balanced support of both growth and environmental stewardship. Several universities investigate how it interacts with various crops under stress, searching for ways to maximize nutrient uptake and minimize runoff. At the same time, chemists explore new methods to recover magnesium from industrial waste streams, using nitrate as an intermediate. There’s a strong movement towards more efficient production methods, reducing the need for energy-intensive purification steps and searching out better crystal forms that withstand humid shipping conditions. These incremental improvements matter for the bottom line and help meet regulatory demands around sustainability and safety.
Toxicity Research
Swallowing even a small amount of magnesium nitrate causes stomach problems and diarrhea, and bigger doses spell trouble for kidneys and the heart—especially if nitrates turn to nitrites inside the body, which can affect oxygen transport. Lab tests on animals set safe limits for exposure, and those findings guide strict labeling requirements for all commercial products. Extended soil tests suggest runoff from heavy magnesium nitrate use can push up nitrate levels in groundwater, threatening drinking supplies and human health. Ongoing research tracks how much of the compound ends up as nitrate in waterways after fertilizer spreads, with clear rules forcing farms to monitor runoff closely. Experience on the regulatory side means watching local and international agencies set ever-stricter maximum residue limits on food and water.
Future Prospects
Demand for magnesium nitrate likely rises in sync with trends in sustainable agriculture and precision farming. Researchers look to smarter delivery systems—think coated or slow-release granules that release nutrients right where plants need them, at the right moment. Factories continue tweaking production methods, hoping to cut waste, lower emissions, and make better use of by-products. Climate change drives more interest in resilient crops, and magnesium nitrate will keep playing a role when growers look to squeeze productivity out of challenging soils. Some start-ups design remediation techniques using the salt in soil cleanups, while others hunt new industrial uses. Watching this chemical evolve keeps me convinced it has more chapters to write, even after centuries in the spotlight.
What is Magnesium Nitrate used for?
A Clear Snapshot of Magnesium Nitrate’s Real Uses
Magnesium nitrate doesn’t get much spotlight in everyday conversation, even though its influence stretches from sprawling crop fields to some surprising corners of industry. It often comes as colorless crystals that dissolve easily in water. Growing up on a farm, I watched neighbors and my own family wrestle with soil that sometimes looked fine but simply didn't deliver strong crops. Magnesium nitrate, as it turns out, plays a key role in fixing this.
The Heart of the Matter: Fertilizers
Magnesium works as a core building block for plants, especially in helping them soak up energy from the sun. On many farms, soil can run out of magnesium after years of planting. Blended into fertilizers, magnesium nitrate steps up in fields worldwide, feeding row crops like corn, tomatoes, and citrus. Its real magic shows when crops respond to shortages with yellow leaves. Adding this mix can turn things green again, boosting both growth and yield. Food security starts in the soil, so something as simple as the right fertilizer can ripple far, ending up on dinner tables everywhere.
Industry: Keeping Safety and Efficiency on Track
People usually connect magnesium nitrate with agriculture, but it’s also a team player in manufacturing. In ceramics, for instance, it helps lower the temperature needed to fire glazed products, which trims both time and costs. Factories making fireworks and explosives rely on it not for a bang, but for its ability to control how chemicals react, especially by helping compounds burn clean without sooty leftovers. Even in wastewater treatment, magnesium nitrate cuts down odor and helps remove ammonia, making life easier for neighbors living near treatment plants.
Handling Risks: Health, Safety, Environment
No chemical comes risk-free. Getting magnesium nitrate on your skin usually just means washing it off, but large spills need fast cleanup to protect streams and wells. The main challenge arises from its ability to increase fire risk when mixed with flammable substances, so storing it far from fuel or oil tanks is crucial. Most users learn this in training, but accidents still happen. Protecting both workers and nearby communities means regular safety audits and solid protocols for storage and transport.
Building Toward Smarter and Safer Use
Experience has shown that knowledge gaps add risk. Outreach and farmer education programs from local co-ops or extension offices can tip the balance, helping people avoid missteps like applying too much to fields or mixing it with the wrong chemicals. For industry, tighter labeling standards and digital tracking help avoid mistakes during delivery or storage, especially in busy plants where chemicals can pile up.
The Way Forward: Balancing Benefits and Challenges
As researchers explore greener sources of magnesium and better recycling methods for nitrates, the future looks brighter for those depending on this compound. Communities nearby plants and fields using magnesium nitrate want transparency and trust. Open communication, grounded in straightforward facts about both benefits and risks, makes a noticeable difference. In daily life, many never think twice about the small crystals that help feed both crops and industry—but their reach stretches far beyond what most people see.
Is Magnesium Nitrate safe to handle?
Personal Reflections on Chemical Safety
My first experience with magnesium nitrate came in a dusty university lab, during a hot afternoon where the fume hoods rumbled louder than the students’ chatter. Our instructor warned us with the kind of tone that suggests “it’s fine, but don’t be reckless.” Years later, after plenty of chemical handling and a fair share of learning from close calls, I understand the value of respect when working with compounds like this one. Magnesium nitrate looks pretty tame on a shelf—white, granular, dissolves quickly in water. But safety isn’t about looks or a lack of bright warning labels; it’s about knowing what you’re touching and how it behaves.
Why the Concerns Exist
Most folks see magnesium nitrate pop up in fertilizers, fireworks, or chemical labs, not the kind of places kids explore or home gardeners keep on a kitchen shelf. The concern here stems from two key risks: its oxidizing properties and its ability to irritate skin, eyes, and lungs. If you’ve ever seen a fertilizer storage shed go up in flames, you know oxidizers don’t joke around. They can amp up a fire much faster than you’d expect.
Magnesium Nitrate and Fire RisksAccidental fires involving oxidizers like magnesium nitrate have happened before. The difference between a controllable flame and a full-blown disaster often comes down to how people store and handle the material. Even a simple act—dumping all the bags next to a heater or in a spot that leaks—can lead to big problems. It isn’t explosive by itself, but mix it with combustible materials (like sawdust or oil rags), and you’re rolling the dice.
Health Hazards Aren’t Far-Fetched
People react differently to chemical exposure. The crystalline dust can irritate eyes and trigger coughing fits. Spills on hands can cause redness. Most cases don’t call for a hospital visit, but repeated or careless contact raises the odds of more serious reactions. Inhaling the dust too often puts stress on the lungs, especially for workers who spend days moving sacks and mixing solutions. I’ve seen folks try to “tough it out” without gloves or masks, only to wish they hadn’t as the shift drags on.
Backing Up Claims
Occupational health agencies list magnesium nitrate as a hazardous substance. The US National Institute for Occupational Safety and Health reports that it irritates the respiratory system and increases fire risks in confined spaces. The European Chemicals Agency classifies it as an environmentally risky compound, too, meaning spills by water sources can impact aquatic life.
Common-Sense Safety Measures
Nobody needs a hazmat suit around magnesium nitrate, but a decent pair of gloves, safety glasses, and a dust mask make a big difference. Washing hands before eating and using protective gear costs less than medical bills. Proper storage, away from flammable substances and moisture, helps prevent disasters. Good ventilation isn’t negotiable if you’re using it inside.
Better Solutions Start With Training
Experience in labs and warehouses has taught me that clear training and a healthy respect for chemicals can do more than any sign or alarm bell. New workers benefit from watching someone who knows the ropes handle spills and storage, instead of just reading pages of protocols. Real-life stories stick better than abstract rules.
Final Thoughts
Magnesium nitrate won’t leap off the shelf to cause trouble. Given reasonable care—gloves, eye protection, a dust mask, and smart storage—it remains a useful tool with manageable risks. Taking shortcuts with safety because a chemical looks “safe enough” only invites problems, and from experience, regret lingers longer than any chemical scent.
How should Magnesium Nitrate be stored?
Why Proper Storage Matters
Magnesium nitrate’s main use shows up in fertilizers and the chemical industry. I’ve seen how easy it is for folks to overlook how important correct storage can be. It doesn’t just sit quietly on a shelf. Left unchecked, it brings fire risks, corrosion issues, and even health worries. That makes it worth looking closely at how people keep this material safe, especially with workers and facilities in mind.
Understanding the Hazards
Magnesium nitrate, on its own, won’t blow up. Still, it acts as a strong oxidizer. Mixed with flammable products or touched by a flame, it can fan up fires very quickly. People sometimes don’t notice leaks or spills. Once, in a small fertilizer warehouse, I watched an employee stack a busted bag behind a pile of boxes. It leaked onto a wooden floor, and even a week later, the wood had started breaking down. Moisture in the air speeds up this process—and breaks down packaging faster.
Choosing the Right Storage Space
Any storage area for magnesium nitrate needs a dry, cool spot—think away from water lines or steamy windows. Humidity triggers caking or can start chemical reactions. I keep mine in a dedicated cabinet, off the ground, with steady ventilation. Friends in the chemical business aim for temperatures below 30°C, away from direct sunlight, to protect shelf life and safety. Solid shelves outshine old wooden pallets, which rot and spark extra risk.
Segregation and Compatibility
Never blend magnesium nitrate storage with fuels, organic powders, or other oxidizers. Fire marshals cite plenty of incidents from this basic mistake. In the past, I’ve seen shipping containers labeled for both pesticides and nitrates. Even unopened, that’s dangerous. In the best setups, rooms for oxidizers stand physically apart with good labeling and tight inventory checks.
Packaging and Containment
The packaging matters as much as the storage location. Sealed, corrosion-proof drums or strong plastic bins give the best barrier. Light sacks or bags tear easily, especially around sharp pallet edges. At grain co-ops, sturdy containers with tamper-evident lids stop moisture entry and tamp down cross-contamination. Staff get in the habit of checking each package as it arrives—any loose seams get swapped out, no questions asked.
Accessibility and Emergency Practices
Easy access for trained staff matters, not just for convenience but for emergencies. I learned from a local depot manager to never block aisles or stack pallets above shoulder height. That cuts down spill cleanup time. Spill kits, complete with absorbents and neutralizers, sit just inside every storage room. More importantly, staff know where those kits live and how to use them on short notice. I once attended a drill where the fastest responder had the spill neutralized before anyone else had their gloves on.
Training and Monitoring Go Hand in Hand
The safest operations train everyone—drivers, handlers, even the office staff who walk through storage zones. Up-to-date safety data sheets should always sit within reach. Digital logs for temperature and humidity show trends, making it much easier to spot problems before they do real damage. In the chemical trade, each near-miss brings new rules and fresh reminders about diligence.
Lessons Learned
Over time, I’ve witnessed the difference smart storage makes. One minor shortcut could cost a company dearly, not just in dollars but in lives. Arming people with the right habits, clear procedures, and a healthy respect for chemistry keeps everyone out of trouble.
What is the chemical formula of Magnesium Nitrate?
Understanding Magnesium Nitrate and Its Formula
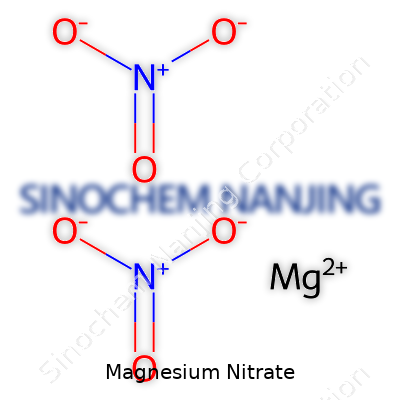
Magnesium nitrate wears the chemical formula Mg(NO3)2. Two nitrate groups, each with a hefty amount of oxygen and nitrogen, pair with a single atom of magnesium. It looks simple on paper, but there’s quite a punch behind those letters and numbers, especially for anyone working with fertilizers, pyrotechnics, or even laboratory chemistry sets. The knowledge of this formula does more than pad out a trivia night—it shapes industries and guides safe handling.
Why the Formula Matters
Anyone who’s worked with fertilizers knows the crop yield doesn’t just rest on watering and sunshine. Magnesium forms the core of the chlorophyll molecule. Plants short on magnesium start to yellow because energy can’t transfer properly in their leaves. In agriculture, the nitrate groups supply plants with nitrogen, another necessary part of green, leafy growth. Knowing that magnesium nitrate dissolves easily means it can deliver these elements right to where roots can use them.
A factory worker doesn’t think much about the periodic table during a shift. Still, anyone working with magnesium nitrate should recognize that the nitrate part isn’t harmless. Nitrates can act as powerful oxidizers. Mixed with common fuels—things found in farm storage or production sites—it speeds up fires, not just in theory but in real accidents. The chemical formula lets handlers quickly recognize the risks, guiding safe storage, labeling, and handling. In my own experience working with agricultural suppliers, clear labeling and awareness saved headaches and insurance claims more than once. Keeping the formula handy—and teaching everyone involved what that formula means—makes a difference in safety.
Reliability Through Chemistry
Trust in chemicals for key tasks doesn’t come from slick marketing. It comes from solid, reproducible science. Mg(NO3)2 isn’t just some combination a company picked to stand out. Chemists test purity and structure using this formula as the baseline. If someone tries to substitute or dilute, a simple test finds the truth. So, in industries where margins are tight, or when regulations matter, having the formula keeps everyone honest. Farmers, buyers, laboratory techs—they can know exactly what's packed in each sack or bottle.
Regulators count on formulas like this, too. Environmental officials track nitrates in soil and water to balance crop health and pollution risk. Too much nitrate runoff—often sourced from compounds like magnesium nitrate—leads to algae blooms, depleting oxygen in water and choking off fish and other life. Labs examine soil and water samples, using the formula as their guide, separating magnesium nitrate from every other blend of nutrients, and then recommending safe, practical steps.
Clear Information Fuels Good Decisions
Transparency builds trust. No one reads a label thinking of worst-case scenarios, but facts help if things go sideways. Knowing the chemical formula, its structure, and its implications keeps communities, workers, and the environment safer. It sharpens business, cuts waste, and ensures nutrients go where they’re needed most. From school science fair projects to crop trials and manufacturing audits, those letters and numbers have real-world impact every step of the way.
Is Magnesium Nitrate soluble in water?
Looking Beyond the Table
People often remember chemistry class as a blur of tables and rules, especially the solubility chart. I still remember jotting down notes, trying to make sense of which salts would actually dissolve and which would just sit stubbornly at the bottom of a beaker. Magnesium nitrate, Mg(NO3)2, always showed up under the “soluble” category. That wasn’t just a random decision—it comes straight from its structure and the way water molecules interact with ions.
The Science Behind Solubility
Magnesium nitrate dissolves easily in water. Solid magnesium nitrate breaks apart because water molecules pull the magnesium and nitrate ions away from each other. The nitrate part, with three oxygens stuck to a single nitrogen atom, spreads a negative charge over a large area, making the whole thing even easier to separate. Once dropped into water, a pile of white crystals quickly vanishes, turning the water clear again. This isn’t just a textbook statement: labs, both in school and industry, rely on this fact.
What Makes Solubility Important?
It’s tempting to treat solubility like a dry classroom idea, but anyone who has worked in agriculture, chemistry, or water treatment knows better. Magnesium nitrate provides magnesium and nitrogen—two elements essential for plant growth—right where plants can use them. Farmers use it in liquid fertilizers because plants need ions floating free in water, not clumped solids. If magnesium nitrate didn’t dissolve so easily, farmers would have to search for another source, and crops could miss out on nutrients.
Plenty of fertilizers try to deliver nutrients, but nothing beats actually getting nutrients into the soil water fast. Consider drought-prone areas where every drop counts. Magnesium nitrate’s ability to dissolve saves time, resources, and effort. In smaller hydroponic operations, hobbyists and commercial growers both appreciate how it doesn’t leave behind a stubborn scum of undissolved salts. That keeps pumps running smooth and plants healthy.
Trust, Quality, and Safety
Fake information circulates about fancy new “slow-release” fertilizers that skirt around real facts. Unless a product actually lists solubility numbers, it pays to check trustworthy sources—textbooks, university research, safety data sheets. People can read straight from the Merck Index or EPA database: magnesium nitrate dissolves in water at over 700 grams per liter at room temperature. That isn’t just pretty good, it’s nearly as high as some table salts.
Easy solubility also means the substance can draw water from the air, so storage matters. In humid environments, an open bag soon becomes a sticky mess, and the risk of accidental spillage into drains or natural waterways grows. Household and industrial users should always seal containers tightly and prevent chemicals from reaching streams or drinking water, where excess nutrients can cause algae blooms.
Solutions and Responsible Use
Farmers and gardeners can avoid runoff and overuse by applying only what soil tests recommend. Keeping containers sealed protects both the product and the environment. Factories using magnesium nitrate can control spills and treat water discharge to protect rivers and reservoirs. Simple habits, like clearly labeling containers and following safety guidelines, prevent most problems before they even start.
Teachers still put magnesium nitrate on the soluble side of the chart. Experience, not just the table, keeps proving it right every time a beaker of white powder vanishes into clear water.
| Names | |
| Preferred IUPAC name | Magnesium dinitrate |
| Other names |
Nitromagnesite Magnesium dinitrate Magnesium(II) nitrate |
| Pronunciation | /maɡˈniːziəm ˈnaɪ.treɪt/ |
| Identifiers | |
| CAS Number | 10377-60-3 |
| Beilstein Reference | 3591875 |
| ChEBI | CHEBI:32599 |
| ChEMBL | CHEMBL1201332 |
| ChemSpider | 15445 |
| DrugBank | DB14503 |
| ECHA InfoCard | ECHA InfoCard: 027-093-00-1 |
| EC Number | 233-826-7 |
| Gmelin Reference | 1414 |
| KEGG | C07438 |
| MeSH | D008271 |
| PubChem CID | 24540 |
| RTECS number | OM2100000 |
| UNII | 7OT5397T8I |
| UN number | UN1474 |
| Properties | |
| Chemical formula | Mg(NO3)2 |
| Molar mass | 148.32 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 2.3 g/cm³ |
| Solubility in water | 125 g/100 mL (20 °C) |
| log P | -2.57 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 10.7 |
| Basicity (pKb) | 7.2 |
| Magnetic susceptibility (χ) | -6.9·10⁻⁶ |
| Refractive index (nD) | 1.452 |
| Viscosity | 1.32 mPa·s (at 20°C, 50% solution) |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 138.7 J mol⁻¹ K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -802 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -455 kJ/mol |
| Pharmacology | |
| ATC code | V09CX03 |
| Hazards | |
| Main hazards | Oxidizing solid; may intensify fire; harmful if swallowed; causes skin and serious eye irritation. |
| GHS labelling | GHS02, GHS07, GHS09, Danger, H272, H319, H315, H410 |
| Pictograms | GHS03,GHS07 |
| Signal word | Danger |
| Hazard statements | Hazard statements: "H272: May intensify fire; oxidizer. H319: Causes serious eye irritation. |
| Precautionary statements | P210, P220, P221, P264, P280, P370+P378, P501 |
| NFPA 704 (fire diamond) | 1-0-1-OX |
| Autoignition temperature | 400 °C |
| Lethal dose or concentration | LD50 (oral, rat): 5440 mg/kg |
| LD50 (median dose) | 307 mg/kg (Rat, oral) |
| NIOSH | SC8915000 |
| PEL (Permissible) | 10 mg/m3 |
| REL (Recommended) | 0.2 mg/m³ |
| Related compounds | |
| Related compounds |
Magnesium nitrite Magnesium sulfate Magnesium chloride Magnesium oxide Magnesium carbonate |

