Magnesium Hypophosphite: Development, Properties, and Future Directions
Historical Development
Magnesium hypophosphite didn’t enter the chemical scene until the need for specific phosphorus and magnesium compounds picked up in the early industrial period. Researchers took an interest after seeing how sodium and potassium hypophosphite worked in both metalwork and as reducing agents. By the mid-20th century, magnesium hypophosphite grabbed attention in labs searching for new nutritional additives and flame retardants. Scientists started parsing out its unique properties, drawn by its solubility and reactivity. Through the years, its use expanded from small research projects to commercial production as industries looked for new phosphorus sources and magnesium compounds that balanced reactivity and safety.
Product Overview
Magnesium hypophosphite wears a few hats as a compound: it shows up as a white, crystalline powder, usually without much smell. Recognizable to any chemist who deals in phosphorous derivatives, the material appeals to both chemical manufacturers and researchers. Suppliers deliver it with clear labeling, showing chemical identity, hydration state (anhydrous or hexahydrate), and grade. With regulatory demands tightening, companies call out both purity and heavy metal content to reassure food and pharma buyers.
Physical & Chemical Properties
In the lab, magnesium hypophosphite pops out thanks to its good solubility in water and low solubility in alcohol. Chemists note its melting point falls below 100°C for the hydrate, breaking down and eventually decomposing to form magnesium pyrophosphate as temperature climbs. It usually takes the formula Mg(H2PO2)2, with molecular weight shifting based on hydrate state. The material brings together magnesium’s stability and hypophosphite’s reducing power, which opens up some neat reaction pathways. At room temperature, it tends to stay put, but give it a kick with acid or heat and fresh phosphorus compounds start spilling out.
Technical Specifications & Labeling
Producers keep specs tight for magnesium hypophosphite—purity often exceeds 98% for high-end grades, and they flag limits on chloride, sulfate, iron, and other heavy metals. The appearance gets checked: it must be white and crystalline, no yellowing or caking allowed. Moisture content rules the product category, especially for the hydrate form. Modern labels spell out country of origin, lot number, recommended storage, hazard statements, and directions for handling—errors here end up costing both time and safety. Compliance with REACH, GHS, and other global standards features heavily, since magnesium hypophosphite travels in both domestic and export markets for feed, food, pharma, and specialty chemical use.
Preparation Method
Producers usually go with a simple but reliable way to manufacture magnesium hypophosphite. Start with hypophosphorous acid (H3PO2), which reacts directly with magnesium oxide or magnesium carbonate under controlled temperature. The solution gets filtered and evaporated until white crystals appear. Quality hinges on the purity of starting materials and careful control of temperature and pH, especially to avoid forming unwanted magnesium salts. After precipitation, repeated washing, filtration, and drying knock down impurities and get ready for packaging. Factory workers keep an eye on every batch, since deviations in pH or contamination show up fast in the finished powder.
Chemical Reactions & Modifications
In daily chemistry, magnesium hypophosphite shows its most interesting side through redox reactions and as a phosphorus donor in specialty processes. It can give up electrons to certain metal salts—handy in electroless plating. Mix it with oxidizers and it breaks down, especially at higher temperatures, and releases phosphine gas—a known toxin in higher doses. The compound acts as a reducing agent in the synthesis of other hypophosphites and supports modified flame-retardant compositions when paired with polymers and related materials. One big draw is its ability to create other phosphorus-containing chemicals cleanly, lowering unwanted byproducts.
Synonyms & Product Names
Chemists and manufacturers use a handful of names—magnesium hypophosphite, magnesium dihydrogen phosphite, and less commonly, magnesium phosphorous acid salt. CAS numbers and standardized codes on SDS forms keep confusion at bay. In some regions, older chemical catalogs use names closer to local language conventions or legacy trade marks. For commercial supply, companies still stick with the recognized chemical name to win trust with buyers and regulators.
Safety & Operational Standards
Most lab accidents stem from poor attention to detail. Magnesium hypophosphite brings hazards mostly through dust inhalation and combustion risk if mixed with strong oxidizers. Inhaling the dust is not something any worker forgets—so sealed containers and dust masks matter on the factory floor. Its decomposition releases phosphine gas, especially if exposed to acid or intense heat. Safety Data Sheets flag these risks clearly, and training focuses on spill cleanup, safe disposal, and storage. Staff keep it away from acids and oxidizers, storing it in cool, dry conditions far from incompatible materials. Regulatory agencies, including OSHA and ECHA, set limits based on toxicity studies, pushing for strong labeling and incident reports. I’ve seen more than one lab invest in dedicated ventilation systems just because phosphorus side reactions tend to go wrong fast.
Application Area
Most magnesium hypophosphite serves two masters: as a phosphorus source in nutritional studies and as a specialist phosphorus compound in flame retardant systems. Agriculture researchers have played with it for fertilizer add-ins, although more established magnesium phosphates own that field. In electronics and plastics, it’s used for flame retardancy, especially in places where halogenated compounds draw regulatory heat. Some nutraceutical groups look at it as a magnesium or phosphorus supplement, though uptake and trace impurity levels still need more study. Chemical manufacturers sometimes use it as a reducing agent in plating or surface preparation. Its range looks set to grow, balancing between cost, handling, and regulatory approval in new markets.
Research & Development
Development teams have started putting more resources into understanding both the long-term safety and the niche uses of magnesium hypophosphite. Some groups target improvements in electroless plating, aiming for surfaces that resist bacterial growth or guarantee higher reflectivity. Other labs look at functional polymers: when magnesium hypophosphite gets added to plastics, engineers get new options for smoke suppression or halogen-free flame retardancy. Animal nutritionists dig into absorption rates and trace contamination, with some positive results, although more robust trials need to clarify any biological impacts. Academic chemists zero in on the synthetic flexibility of hypophosphite ions, unlocking new organophosphorus reactions. With every new publication, more eyes land on this compound’s unique blend of safety and reactivity.
Toxicity Research
Toxicity research still pushes forward, especially as more countries line up food or supplement registration requirements. Most animal studies suggest magnesium hypophosphite sits at low acute toxicity, with higher doses bringing mild digestive disruption. Longer-term studies examine both phosphorus load and magnesium content in feed or supplements, looking for kidney and metabolic impacts. All hypophosphites share a watch-list warning—at high temperatures or accidental acid contact, they can form phosphine gas. Researchers run median lethal dose (LD50) tests, inhalation exposure, and skin contact trials, tracking for chronic issues. Global agencies collect the results, continually reviewing exposure standards to set workplace safety and environmental limits.
Future Prospects
Magnesium hypophosphite stands on the edge of broader application as more industries rethink phosphorus and magnesium sourcing. Its promise in flame retardancy, driven by bans on older halogenated chemistry, looks bright—especially in Europe. As processing quality improves, the supplement sector keeps nudging standards higher, balancing purity, bioavailability, and regulatory green lights. Industry partnerships with universities drive new uses, especially in cleaner coatings, biomedical targeting, and sustainable agriculture. Future research marches into greener synthesis, with feedstock recycling and energy-saving production drawing attention from both innovators and investors. Each improvement brings new interest in a compound that once hung quietly in old lab notebooks but now reads as both workhorse and opportunity for modern chemical science.
What is Magnesium Hypophosphite used for?
A Chemical With a Job to Do
Magnesium hypophosphite doesn't often make headlines, but this white, crystalline powder keeps popping up in places you wouldn’t expect. Its main job? Working as a reducing agent. That’s science-speak for something that donates electrons, basically helping other substances change their chemical structure. This trait pushes magnesium hypophosphite into some pretty interesting industries, especially in electronics and surface finishing.
Supporting Electronics: A Backbone for Circuit Boards
Modern gadgets rely on intricate layers of metal built up, atom by atom, onto plastic or glass. That's possible because of a process called electroless nickel plating. Instead of zapping metal onto a surface with electricity, manufacturers use chemicals to kick off the reaction. Here’s where magnesium hypophosphite comes in. It helps start and carry the chain of chemical events that plates a thin, even layer of nickel onto electronic parts. The payoff? Devices like smartphones, tablets, and laptops that last longer and work better because of that smooth, protective metal shield.
Delivering Results Without a Lot of Fuss
One thing I appreciate about magnesium hypophosphite: it does the job without releasing a bunch of toxic fumes or leaving behind nasty waste. The chemical’s reduced phosphorus, which means fewer unwanted byproducts. That’s good for people on the production floor and the surrounding environment. It also follows modern manufacturing trends—safer conditions, clear air, less need for expensive cleanups.
Strengthening Medical and Industrial Products
Magnesium hypophosphite does more than just coat electronics. It steps into medical device manufacturing as well, helping plate stents, surgical tools, and even dental braces with corrosion-resistant layers. These tough coatings protect devices from breaking down inside the human body. In food processing and water treatment, the same properties fight off rust and chemical attacks on metal parts, reducing maintenance headaches and keeping equipment running longer.
Why Purity and Handling Matter
Over the years, working with fine chemicals taught me that purity isn’t just a brag—it’s the difference between smooth production and constant troubleshooting. Impurities in magnesium hypophosphite can mess up plating, causing spots or uneven surfaces. That can mean whole batches get scrapped. To prevent this, manufacturers enforce strict quality testing. Reliable supply chains and good handling practices keep the material dry and uncontaminated, making the difference between a world-class product and one that ends up in the reject bin.
Opportunities for Cleaner Manufacturing
Industrial chemistry isn’t always known for clean practices, but magnesium hypophosphite changes that landscape. It reduces the need for harsh chemicals, protects workers, and can be recovered from waste streams for recycling. Some companies go even further, investing in closed-loop systems that reuse chemicals again and again. These efforts keep costs down and limit pollution, answering calls from both investors and communities for responsible manufacturing.
Looking for Answers, Not Trouble
Making safer, longer-lasting products doesn’t mean workers or the planet pay the price. Magnesium hypophosphite helps businesses hit that balance. By understanding the details—from chemical reactions to workplace handling—we can innovate without cutting corners. The future of this compound sits in continuous improvement and keeping both safety and quality in focus, every step of the way.
Is Magnesium Hypophosphite safe for human consumption?
What Goes Into Food Matters
People keep a closer eye on food labels these days. Folks check every additive before buying a box of cereal or a bottle of electrolyte water. Magnesium hypophosphite pops up every now and then – often in nutritional supplements, rarely as a direct food additive. Its appearance sparks questions: is it actually safe to eat? Does it have any real benefit? No one wants something unproven hiding in their pantry.
Understanding Magnesium Hypophosphite
Chemically, magnesium hypophosphite delivers magnesium and hypophosphite ions. Magnesium itself helps with nerve function, muscle contraction, and bone structure. Hypophosphite salts have a different story: they’re mostly used in industrial settings, often as reducing agents – a far cry from your afternoon snack.
Magnesium from leafy greens supports well-being. Dieticians point out that almost half of US adults don't reach the daily requirement for this mineral. Magnesium supplements on the store shelf have a proven safety record in doses found in foods and pills. But swapping magnesium hypophosphite for naturally sourced magnesium isn’t the same thing. Novel compounds need careful study.
What Do Regulators Say?
Looking at the FDA, European Food Safety Authority, and Health Canada, you won't find magnesium hypophosphite on any recognized list of approved food additives or mineral supplements for humans. Regulatory green lights usually come after thorough safety evaluations. If a substance isn’t on those lists, scientists haven’t completed those studies or authorities haven't seen enough data to prove it’s harmless for daily use.
Studying safety means more than looking up an entry in a handbook. Researchers test for risks like kidney or liver toxicity, effects on blood markers, and any buildup of breakdown products. If something in a compound raises kidney stress or disrupts calcium balance, it fails the test. Food scientists and toxicologists always ask: what does this chemical do inside the human body over weeks and months?
Limited Evidence Doesn’t Mean Proven Safety
Magnesium hypophosphite hasn't been on the radar for long in nutrition science. A handful of animal studies look at related compounds, with some showing changes in blood chemistry at high doses. No well-conducted, peer-reviewed studies in humans exist. Without those, safety just can’t be assumed. Every mineral compound might break down differently in the body. Sometimes, the byproducts do more harm than the original chemical.
Manufacturers sometimes point out that both magnesium and phosphorus have a place in human metabolism. That ignores how the body processes each form. Magnesium hypophosphite could behave unlike the safe, tested salts already in the food supply. History reminds us that introducing an unfamiliar compound brings real risk. Melamine in baby formula and too much sodium cyclamate both showed dangerous effects that didn’t appear right away.
Addressing Consumer Concerns
For nutrition, the best path always heads toward ingredients with a documented history of use, clear dosing guidance, and oversight by independent regulators. Magnesium citrate, oxide, and chloride set that standard for supplements. Shoppers with questions about food additives should watch for substances that remain unapproved by major health agencies. Nobody wants to be a test case.Open communication with healthcare providers and direct reporting to regulatory bodies can spotlight unapproved ingredients. Keeping products off shelves until scientists do the hard work of safety testing protects everyone.
What are the storage conditions for Magnesium Hypophosphite?
Overview
Magnesium hypophosphite finds its way into labs and chemical storage rooms for its reducing properties and specialty roles in chemical synthesis. Its white, powdery appearance looks harmless, but looks don't tell the full story. Unstable chemicals deserve extra care because a slip in storage puts both people and facilities at risk. It’s not just about compliance — chemical safety really hits home when you realize what can go wrong with something as ordinary-looking as magnesium hypophosphite.
Direct Experience: Lessons from Storage Rooms
After years of managing university chemistry storerooms, I’ve seen what works and what fails. Poor labeling, humidity, and heat combine as troublemakers with chemicals like this. Colleagues who mistakenly ignored desiccators or control of light exposure learned quickly that some chemicals will react steadily over weeks, leading to slow degradation or, worse, dangerous byproducts. Instead of letting students guess, we built habits of transparency and method — every jar lived in the right spot, under conditions checked by everyone who walked into that closet.
The Best Approach to Storage
Good storage for magnesium hypophosphite starts with a dry space. Even trace moisture brings on slow decomposition, reducing the chemical’s strength and, over time, producing phosphine gas, a serious hazard in poorly ventilated spaces. Sealing containers matters more than most realize because powders tend to absorb moisture from regular room air — not just spills. A cool area, preferably below room temperature, prevents slow breakdown and adds a layer of insurance against environmental slips or equipment failures.
Direct sunlight also creates problems. UV light speeds up unwanted reactions. Cabinets away from windows and heat sources always give better results. In older buildings, I’ve often had to move chemicals after noticing temperature swings near steam pipes, which led to caked powders. Reliable labeling with date of arrival and last inspection cuts down on accidental use of ruined stock. Checklists help: every inspection means opening boxes, checking seals, and logging the date. This simple practice saves headaches later.
Fact-Based Safety
The National Institutes of Health highlight the risk of magnesium hypophosphite producing irritating fumes and possible phosphine if heated. Respiratory problems and chemical burns remain real hazards, making storage far from acids or sources of ignition key. OSHA explains that dry storage, non-reactive containers — think amber glass with plastic lids — lead to longer shelf life and fewer accidents. Not storing this chemical near food or water lines prevents cross-contamination, which keeps the workspace safe.
Regularly reviewing storage protocols matches up with Responsible Care® principles adopted across the chemical industry. I’ve seen auditing paired with proper record-keeping catch small leaks or broken seals before real trouble started. Quality storage rules are not just checkboxes for audits or inspections; they build trust among colleagues who depend on each other to handle risk with skill and respect. Chemical accidents cost money, time, and sometimes more. Every stored jar carries responsibility for everyone on site.
Building Better Habits
Basing storage decisions on research and past incidents means safer, longer-lasting chemicals and healthier workplaces. Building a safety culture, not just compliance, matters in every lab and storeroom. Small disciplines — using desiccators, checking labels, respecting heat and sunlight controls — prevent large problems. Chemical science, after all, relies on detail, and the quiet corners of storage rooms deserve just as much attention as lab benches and instruments.
What is the chemical formula of Magnesium Hypophosphite?
Understanding Magnesium Hypophosphite
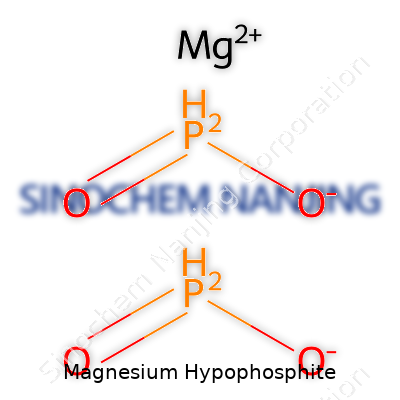
Magnesium hypophosphite, with the chemical formula Mg(H₂PO₂)₂, shows up in some interesting places in the world of chemistry. Even though it doesn’t have the household recognition of table salt or baking soda, anyone familiar with labs or certain industrial processes has probably seen its name pop up on a bottle or chemical list. Getting to know the right formula means more than just passing a quiz—accuracy here keeps people safe and helps work move forward without mistakes that cause wasted time and resources.
Breaking Down the Formula
The formula Mg(H₂PO₂)₂ gives us a window into the nature of this compound. Magnesium, that trace element people often associate with vitamins or supplements, bonds with hypophosphite ions, each carrying a (-1) charge. The balance of two hypophosphite ions to one magnesium ion creates a stable compound ready for use in a variety of settings. Recognizing this structure prevents confusion with other phosphorous-containing substances, which may behave very differently in chemical reactions.
Facts That Shape the Importance
Digging into textbooks or scientific papers shows that magnesium hypophosphite goes beyond theory—you can find it used as a reducing agent, especially in the world of electroless nickel plating. In this process, metal coatings form on objects without the need for electricity, which makes things easier and sometimes safer. The wrong formula means reactions won’t work as planned or could even cause dangerous byproducts. That’s a mistake with real-world consequences, one no one wants to see play out in a laboratory or factory.
There’s also a public health side to the story. If someone handling this material can’t confirm its identity by formula, the chance of accidental mix-ups increases. Small chemical differences can turn routine handling into a risky proposition. This isn’t theoretical—reports of lab accidents often trace back to basic details being overlooked. Everyone working with chemicals benefits from taking these basics seriously, no matter how routine the task seems.
Building Knowledge for Workplace Safety
Solid, hands-on experience in chemistry labs drives home the point that chemical formulas are more than strings of letters and numbers. Any time I worked with unfamiliar substances, double-checking the chemical formula was part of the routine. Mistakes often started with something as simple as a missed subscript or a misplaced letter. Training others, I stressed looking up formulas before reaching for the chemicals—to avoid embarrassment, yes, but more importantly, to protect the health and safety of the whole team.
Resources like peer-reviewed journals, databases from chemical suppliers, and government safety guidelines keep everyone on the same page about substances like magnesium hypophosphite. Organizations such as the Occupational Safety and Health Administration (OSHA) and National Institutes of Health (NIH) make it easy to find up-to-date information, including the correct chemical formulas, safe handling practices, and emergency procedures.
Better Accuracy, Fewer Problems
Lab efficiency grows when everyone respects the details. Encouraging a culture that values double-checking and fact-finding makes slip-ups less likely. Posting reference charts and using shared digital tools for chemical information puts clarity within easy reach. Teams benefit from a habit of open questions, where no one feels awkward about asking for a formula check. These small steps add up, helping both veterans and newcomers build reliable habits that last through their careers.
Are there any known side effects or hazards associated with Magnesium Hypophosphite?
Digging Into the Details
Magnesium hypophosphite doesn’t show up much in everyday life. It usually lands in labs or pops up in scientific settings, so most people never cross paths with it. But anyone working around chemicals knows it pays to understand what you’re handling, whether you’re mixing compounds or just refilling the stockroom.
Looking at the Safety Data
Companies and scientists take material safety very seriously. I’ve spent time reviewing safety data sheets for dozens of chemicals, and magnesium hypophosphite comes with its own set of concerns, even if information is a bit more scarce compared to everyday substances. The main documented risks focus on irritation: dust from the powder can bother the eyes, nose, and throat. Direct skin contact sometimes leads to mild discomfort or redness for workers, and breathing it in feels rough on the lungs. Nobody wants to spend an afternoon with watery eyes and scratchy skin, so it’s safest to use gloves, eye protection, and a dust mask, just to shut down the chance of an accident.
No one wants chemicals hanging around in their body either. Long-term data on magnesium hypophosphite is thin, so you won’t find a long list of chronic side effects like you do for lead or asbestos. But new research hints that some hypophosphite salts might stress out the kidneys or shift the balance of phosphorus in the body, which can create new health headaches over time. So even if people rarely get exposed at home, factory employees or researchers should still take exposure seriously.
Environmental Hazards
I grew up in a region where factory runoff sometimes leaked into rivers. It made me hyper-aware of how chemicals affect wildlife. Magnesium hypophosphite, like every phosphorus compound, puts aquatic life at risk if dumped into waterways. Phosphates and hypophosphites feed algae, leading to those nasty algal blooms that choke out fish and suck up oxygen. It doesn’t take gallons to tip the scales—just a little bit over time can set off a chain reaction. So containment and disposal plans need more than the bare minimum. Drain covers, spill kits, and thorough wastewater treatment protect both workers and ecosystems from accidental releases.
How to Stay Safe
Anyone who works around lab chemicals learns to respect the unknown. Practices like double-checking labels, keeping extra gloves on hand, and double-bagging waste aren’t just for rules—they’re about having everyone walk out healthy at the end of the day. When it comes to chemicals like magnesium hypophosphite, agencies like OSHA and the EPA set pretty clear rules on exposure limits and disposal. Relying on those guidelines, along with regular health checks for anyone who’s regularly exposed, helps catch problems early.
Potential Ways Forward
Sometimes it takes a shakeup in company culture to upgrade chemical safety. I’ve seen teams switch over to safer alternatives after a few near misses, and that step usually starts with honest discussions. Substituting less hazardous chemicals, automating processes, and investing in training all lower the odds of trouble. Safety isn’t just about avoiding fines; it’s about making sure that no one gets a trip to the ER just for showing up to work. If magnesium hypophosphite’s on your inventory list, keeping a close eye on safety stays just as important as tracking the purity of your samples.

| Names | |
| Preferred IUPAC name | magnesium phosphonate(1-) |
| Other names |
Hypophosphorous acid, magnesium salt Magnesium phosphinate Magnesium dihydrogen hypophosphite |
| Pronunciation | /mæɡˈniːziəm haɪpəˈfɒsfɪt/ |
| Identifiers | |
| CAS Number | [10377-57-8] |
| Beilstein Reference | 3589466 |
| ChEBI | CHEBI:32503 |
| ChEMBL | CHEMBL1201642 |
| ChemSpider | 14101 |
| DrugBank | DB09275 |
| ECHA InfoCard | ECHA InfoCard: 18eb307f-3f7b-4ad2-8952-61a86342f3b3 |
| EC Number | 232-036-3 |
| Gmelin Reference | 33314 |
| KEGG | C22999 |
| MeSH | D048520 |
| PubChem CID | 23666352 |
| RTECS number | OM2975000 |
| UNII | 322I6G6A7W |
| UN number | UN1458 |
| CompTox Dashboard (EPA) | DTXSID4040360 |
| Properties | |
| Chemical formula | Mg(H₂PO₂)₂ |
| Molar mass | 104.27 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 2.153 g/cm³ |
| Solubility in water | Soluble |
| log P | -3.3 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 1.0 |
| Basicity (pKb) | 12.0 |
| Magnetic susceptibility (χ) | −20.2×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.539 |
| Viscosity | 5 mPa·s (20 °C) |
| Dipole moment | 0.0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 110.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −1344.8 kJ/mol |
| Pharmacology | |
| ATC code | A12CC10 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H228, H302 |
| Precautionary statements | P264, P270, P301+P312, P330, P501 |
| NFPA 704 (fire diamond) | 1-0-1-W |
| Lethal dose or concentration | LD50 (oral, rat): 5,000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 5 g/kg |
| NIOSH | Not established |
| PEL (Permissible) | PEL: 15 mg/m³ |
| REL (Recommended) | 34.0 |
| Related compounds | |
| Related compounds |
Calcium hypophosphite Sodium hypophosphite Hypophosphorous acid |

