Magnesium Chlorate: Past, Present, and Future Under the Microscope
Tracing the Path: Historical Development
Magnesium chlorate doesn’t grab headlines, yet its story weaves into some of modern chemistry’s biggest ambitions and tough lessons. Back in the late nineteenth century, chemists tinkered with chlorates for their powerful oxidizing punch. They saw potential for uses from explosives to disinfectants. Magnesium chlorate drew interest for its solubility and reactive nature. Its promise seemed clear: strong oxidizer, decent stability, simplicity in synthesis. Academic curiosity and the search for better propellants lit the initial spark. Scientists elbowed through practical challenges—production, purification, safety—learning along the way that oxidizers don’t forgive mistakes. Ever since, classrooms and industrial labs have kept magnesium chlorate around for its unique qualities and as a launching point for teaching more advanced chemistry.
Product Overview: More Than Just a Compound
Anyone who’s encountered magnesium chlorate in the lab understands that it stands apart from basic salts. Forget kitchen or road salt; this pale, crystalline powder wields much stronger chemical influence. Technicians and researchers value it for reliable oxidation, strong solubility, and clear-cut reactions. Beyond textbook curiosity, magnesium chlorate shines in certain specialty sectors—pyrotechnics, some disinfectant blends, and as an oxygen source in chemical syntheses. The compound sometimes goes by other labels: magnesium perchlorate can turn up in conversation by mistake, though the chemistry isn’t the same. In many settings, abbreviations and generic terms get thrown around, leading to confusion unless everyone knows the language and risks.
Physical and Chemical Properties: What Sets It Apart
Pile magnesium chlorate on a dish and the differences jump out. The crystals glisten, easily pulling moisture from the air. The hygroscopic quality matters; sloppiness during storage leads to clumping or, worse, decomposition. Laboratorians remember the precise, almost dramatic reaction with heat or friction—one wrong step and the whole batch turns hazardous. Reactivity defines this compound. Heat it too fast and decomposition can run out of control, releasing toxic chlorine oxides. As a magnesium salt, its solubility allows for straightforward preparation of solutions—a trait that often streamlines intricate syntheses. Yet, the very same properties mandate strict respect; smoke, gases, and the chance to accelerate combustion situate magnesium chlorate squarely in the “handle with care” category.
Technical Specifications and Labeling: The Devil’s in the Detail
In the chemical world, ambiguity breeds accidents. Handling magnesium chlorate relies on clear labeling and up-to-date documentation. Safety advocates remind teams to check chemical identity, hydration level, purity, and storage advice every time they reach for the jar. Simple oversights, like unclear concentration or improper containers, easily escalate to workplace incidents. Labels must flag major hazards: strong oxidizer, possible skin and respiratory irritant, unstable if contaminated. Conversations about labeling now run deeper than just regulatory compliance; after a few mishaps, even the most confident researcher becomes meticulous.
Preparation Methods: Precision Outweighs Speed
Making magnesium chlorate isn’t glamorous, but it reveals chemistry’s unforgiving side. One common technique involves reacting magnesium oxide or carbonate with aqueous chloric acid. The process seems simple on paper; mix, stir, then crystallize from solution. Real lab life introduces impurities, annoying side reactions, and unpredictable yields. Hard-won lessons teach that slow addition, tight temperature control, and rigorous purification set the difference between a safe operation and disaster. Sloppy technique invites self-destruction—excess chloric acid, rough filtration, and residual contamination increase explosion risks. Patience, ventilation, and strict adherence to routines keep the process from spiraling out of control.
Chemical Reactions and Modifications: Chemistry’s Opening Move
Magnesium chlorate rarely exists alone in the laboratory. Its role as a strong oxidizer means it often starts multi-step reaction chains. Introduce it to organic materials and the reaction turns energetic, sometimes violently so. In pyrotechnic work, it’s mixed with fuels to produce vivid colors or intense flashes. It also helps in analytical chemistry by providing a reliable oxidizing environment. Chemists often explore derivatives or attempt to temper reactivity by pairing with stabilizers or shifting the pH. Each attempt brings trade-offs: modify the compound too much and you sacrifice performance; leave it unaltered and you trade efficiency for higher risk.
Synonyms and Product Names: Language Matters
Magnesium chlorate hides behind a patchwork of names—magnesium chlorate hexahydrate most often, or just "mag chlorate." Some regions list the CAS number in catalogs, others slap on cryptic codes for inventory tracking. Even veteran chemists get tripped up when language blurs, especially where perchlorate and chlorate get mixed. The practical takeaway: always double-check. I’ve seen projects delayed and equipment ruined simply because the wrong label slipped through on a shipment. Manufacturers sometimes market it under their proprietary blends, which adds more confusion unless end users keep a critical eye on product information.
Safety and Operational Standards: No Room for Shortcuts
Magnesium chlorate draws a bold red line under safety. Its oxidizing potential turns every spill, every scrap of incompatible material, into a potential ignition source. Labs that use it must train workers to never mix it around organic debris, flammable liquids, or strong reducers. Separate storage in cool, dry, secure containers isn’t up for debate. Dust control, routine inspection, and incident drills remain part of the workspace where this chemical appears. Regulatory agencies set tight rules, not just for transport but for workplace limits, handling signage, and emergency response. Even the most routine use grows complicated; complacency quickly invites disaster. I’ve seen labs outlaw its use outright after minor incidents, while others rely on rigorous compliance culture and constant vigilance.
Application Area: Where Magnesium Chlorate Makes Its Mark
Talk of magnesium chlorate seldom leaves the walls of specialist circles, but its footprint shows up in pockets of real-world technology. Firework and pyrotechnic specialists covet it for color and intensity, exploiting its oxidizing ability for dramatic effect. Industrial water treatment and disinfectant producers sometimes use it where other chlorates won’t dissolve or function as efficiently. Agricultural controls slip it into herbicides, though shifting safety standards push users toward alternatives. I’ve watched industry cycles upend favorite compounds in favor of new ones, often balancing cost, safety, and environmental aftertaste. Chemists hold on to magnesium chlorate for niche purposes—test reactions, oxidizer studies, rapid synthesis—often swapping it out as soon as something less hazardous gets proven up.
Research and Development: Evolving Understanding
R&D labs treat magnesium chlorate as a benchmark for oxidizer science. It offers a straightforward model for exploring reaction mechanisms, teaching students the practical art of redox chemistry. Recent years brought more focus on stabilizing the compound and controlling decomposition, drawing lessons from past lab accidents. Some research probes how it interacts with soil and water, mapping out environmental persistence and transformation products. Advanced fields, like rocket propellant design, dig into magnesium chlorate’s properties in controlled settings, looking for performance improvements or new safety tricks. My own experience tells me most chemists pursue alternatives for large-scale work but circle back to magnesium chlorate for specific proof-of-concept studies and educational demonstrations.
Toxicity Research: Unwrapping Risks
Science stopped taking chlorates lightly decades ago. Toxicology research warns that magnesium chlorate, like its cousins, can cause acute poisoning if mishandled. Inhalation, skin contact, or accidental ingestion bring real dangers—oxidative stress, methemoglobinemia, kidney and liver impact. Animal studies highlight the risk of cumulative exposure, particularly in sensitive groups. Environmental risk studies dig into water solubility and runoff risks; aquatic organisms prove especially vulnerable. This isn’t alarmism. I remember reading case reports from old agricultural accidents where improper use led to harm. Today’s training and safety protocols exist to head off these repeat stories. Progress marches on, pushing toward better monitoring and substitution wherever possible.
Future Prospects: Searching for Balance
Magnesium chlorate faces a crossroads as technology and regulation evolve. Scientists and regulators balance its strong performance as an oxidizer with a drive for safer, more sustainable alternatives. Research continues on stabilizing modifications, improvements in synthesis, and ways to contain or remediate contamination. New chemical industries spring up overnight, often with greener mandates that rule out chlorates and similar oxidizers. Some see magnesium chlorate’s role shrinking in step with advances in green chemistry, safer pyrotechnic formulations, and next-generation disinfectants. For now, it remains a touchstone for classroom learning, specialized chemical processes, and proof-of-concept studies. The chemical community eyes the horizon, seeking reduced risk and environmental impact, but the lessons learned from magnesium chlorate’s past remain stitched deep into laboratory culture.
What is Magnesium Chlorate used for?
What It Brings to the Table
Magnesium chlorate might not roll off the tongue, but its presence makes a real-world difference across several fields. Used mostly in agriculture and industrial applications, this compound shows up in places you’d least expect.
Agriculture Leans on It
Out in the fields, magnesium chlorate acts as a herbicide. Weeds can choke out crops and destroy harvests, so farmers reach for strong tools to manage them. This chemical doesn’t just stop weeds at the surface; it penetrates through and interrupts their growth cycles. I’ve seen older farmers in small towns recalling how a simple sprayer moved through rows of cotton or soybeans, knocking back unwanted plants and giving their crops some breathing room.
The Food and Agriculture Organization recognizes magnesium chlorate as effective on annual and perennial weeds. Unlike some weedkillers, it doesn’t break down instantly in the soil, so results stick around for a while. Yet, this power comes with risks. Its high oxidizing ability means that mixes and storage require serious respect. Poorly stored magnesium chlorate near organic materials can trigger accidents—there’ve been mishaps in rural sheds when someone ignored safety instructions.
Helping Science and Industry
In the lab, the compound surfaces in analytical chemistry. Chemists use it when preparing solutions or testing reactions where strong oxidizers fit the bill. Magnesium chlorate has helped researchers pull off syntheses that would be trickier without it around.
Paper mills have also used small amounts for bleaching wood pulp. Its oxidizing force pulls color out of wood fibers, brightening the paper and prepping it for printing. The process is tightly controlled, though—the possibility of explosive reactions means factories must follow strict protocols.
Environmental and Safety Questions
Once you recognize its utility, concerns about runoff and exposure can’t be ignored. Magnesium chlorate doesn’t vanish harmlessly. Rainfall can spread traces from treated fields into streams, putting aquatic life at risk. At high doses, it’s toxic to fish, which threatens more than just farm profits—it puts whole ecosystems on edge.
The U.S. Environmental Protection Agency lists it as hazardous waste. Disposing of leftovers or contaminated gear means following guidelines designed to keep neighborhoods and workers safe. Even for home gardening, I’d steer clear of magnesium chlorate and look for safer alternatives, since not enough research addresses long-term exposure.
Fire departments have flagged the compound because of its oxidizing strength. Sulfur, phosphorous, and many organic materials turn volatile when stored anywhere near this salt. Farmers, warehouse owners, and lab managers must know their inventory and teach new workers old lessons learned the hard way.
Searching for Safer Paths
Better weed control options exist now. Integrated pest management, crop rotation, and targeted herbicides can help keep weeds at bay with less fallout. The same goes for bleaching agents: modern methods use less aggressive chemicals, lowering accident risks and protecting rivers and groundwater. It matters that companies and growers take environmental stewardship seriously and adapt when new options become available.
What Experience Shows
Magnesium chlorate answered a need in agriculture and industry, but experience and evidence show that careful handling is the only responsible approach. Progress in chemistry doesn’t mean leaving old solutions completely behind, but it does demand we weigh results against risks every season. Choosing safer routes when possible ensures that crops thrive, workers head home safe, and landscapes aren’t the ones paying the price in the end.
Is Magnesium Chlorate safe to handle?
Real Risks People Overlook
Magnesium chlorate doesn’t show up much in normal life unless you’re working in a lab, dealing with chemistry, or some niche industrial process. Its use gets pretty limited compared to things like baking soda or table salt. Still, the risks involved aren’t minor. Anyone searching for a fact sheet on safety can quickly learn it carries the “oxidizer” label, which should give pause for thought. In my own experience working with chemicals, “oxidizer” is always a red flag that demands respect.
What Happens If You Get Casual?
Here’s something you won’t find on any fancy label—magnesium chlorate can really surprise you when mixed with flammable stuff or exposed to heat. I watched a demonstration once where just a sprinkle onto a paper towel, accidentally done, caused a flare-up. Unlike table salt, magnesium chlorate wants to give off oxygen, so anything that’s even remotely flammable gets an unwanted boost. It doesn’t scream danger from the outside, but chemistry doesn’t care about appearances.
Handling and Human Health
No one likes breathing dust, and magnesium chlorate is no friend to lungs either. A few of my colleagues developed coughing fits after handling powders without proper masks, believing a simple dusting wasn’t serious. The body doesn’t have clever tricks for getting rid of chlorates. Repeated skin contact or inhalation quickly turns into headaches, irritation, and even nausea if you push your luck. I always made sure gloves and goggles were mandatory—skipping protection just never seemed worth the risk of accidental splashes or dust in the eyes.
Straight Talk About Fire
The bigger concern isn’t just personal health. Magnesium chlorate brings fire danger into places where people least expect it. I’ve seen well-meaning folks sweep magnesium chlorate into the regular trash, then panic when the trash started smoldering. It reacts with everyday organic material and can even cause spontaneous combustion if mixed with something like paper, sawdust, or oily rags. A forgotten spill left in the open sun turns into an accident waiting to happen. Keeping it away from anything remotely flammable, and keeping containers tightly closed, kept our work sites incident-free.
Responsibility in the Workplace
Magnesium chlorate isn’t for casual experimenters or anyone who shrugs off labels. The folks who handle it daily depend on the reliability of their training and protective gear. Strong oversight, regular safety drills, and honest discussions about risks make all the difference. Industry standards highlight the need for special storage away from heat and sparks. Compliance with regulations isn’t just a paperwork headache—real injuries show up if shortcuts become routine.
Safer Alternatives and Smarter Solutions
Sometimes the smartest route skips magnesium chlorate altogether. Offices and labs have turned to greener chemicals where possible, searching for options that manage the same task with a lot less hazard. Asking questions—do you really need this chemical, or could you switch to something safer—brings down risk for everyone. Even with the right training and equipment, magnesium chlorate is never something you’d call “safe as houses.”
Looking Forward
Decades of chemical use have shown that treating magnesium chlorate with respect saves lives and prevents ugly surprises. Cutting corners or underestimating risk never ends well. The lesson stands: protection and knowledge go hand in hand, keeping tough substances in their place so they don’t harm people or the environment.
What is the chemical formula of Magnesium Chlorate?
The Formula Unveiled
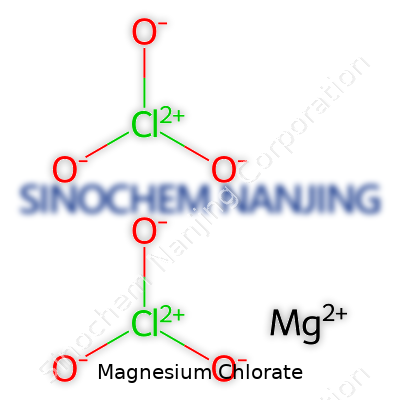
Magnesium chlorate shows up in the chemistry world as Mg(ClO3)2. You’ve got one magnesium atom side by side with two chlorate ions. Each chlorate brings three oxygen atoms along, so you end up with quite an oxygen-rich compound. Working in a school lab years ago, I remember how quickly students caught the link between the formula and its oxidizing punch. You don’t forget the extra oxygen packed into those chlorates after seeing a small demonstration.
Why Getting the Formula Right Matters
Misreading a chemical formula throws off experiments and can open the door to dangerous outcomes. With magnesium chlorate, the stakes stand a little higher because it fuels strong oxidizing reactions. If you tried to swap out magnesium sulfate or calcium chloride, you wouldn’t get the expected results. Precise formulas mean reliable results and safer spaces in classrooms and industries. That’s how you keep people out of trouble and ensure honest work.
Uses Beyond the Classroom
Looking at real-world uses, magnesium chlorate’s formula plays a role in more than academic drills. Gardeners have tried it as a defoliant. Its knack for feeding oxygen into chemical processes led to research into explosives, signal flares, and pyrotechnics. Handling it around open flames or incompatible chemicals calls for sharp attention, especially with the rising focus on safety after recent high-profile chemical accidents. Incorrect handling can trigger fires or explosions—no small risk for anyone in agriculture or research.
Pitfalls and Lessons Learned
Scouring reports from chemical safety boards highlights gaps where workers mixed up magnesium chlorate with magnesium chloride.Two very different behaviors, one spelling slip. Making sure every team member double-checks chemical labeling and recipes stops those mistakes before they spark. Those habits stem from a culture that values checking over assuming. As someone who’s had to clean up after an unexpected reaction, I know taking an extra minute up front pays off later.
Staying Safe with Magnesium Chlorate
Storing and using this compound takes respect. Sealed containers, no greasy rags or dust nearby, and strict attention to incompatible substances go a long way. Safety data sheets might not grab attention with their technical layout, yet they offer the best window onto risks. A strong safety culture helps prevent those “it’ll never happen to me” moments that cost money and, too often, lives. Regular training—with real examples rather than just slides—keeps people alert.
Improving Chemical Education and Industry Practice
Schools and companies can step up by bringing hands-on demos, clear signage, and decision-making scenarios into training. Quick quizzes and walk-throughs help information stick, especially for those new to chemistry. Tackling confusion between compounds early means less risk downstream. Anyone dealing with chemicals, from lab techs to warehouse staff, needs straight talk and a little humility in their approach.
Final Thoughts
Magnesium chlorate’s formula just looks like a collection of symbols to the unfamiliar eye. Dive deeper, and it reminds us that clarity—on paper and in practice—shapes results and safety every day. Scrutinizing details, asking questions, and sharing lessons learned turn chemical knowledge into trustworthy action out in the world.
How should Magnesium Chlorate be stored?
Why Proper Storage Matters
Accidents don’t start with a bang. Most come from ignoring the basics. Think of magnesium chlorate—an oxidizer with a track record for being reactive and sometimes risky in the wrong hands. Chemical storage never felt theoretical to me. Years ago, I worked briefly in a lab where small lapses led to big regrets. On my first week, a senior researcher drilled a simple rule into everyone: always respect the hazards, especially the ones you can’t smell or see. That lesson stayed with me.
Understanding Reactivity and Hazards
Magnesium chlorate isn’t the friendliest compound on the shelf. It kicks off oxygen and pairs up trouble when in contact with organic material or heat. One historic example took place decades ago—a misplaced container got damp, and the resulting explosion served as a textbook warning for countless chemists. Even at room temperature, the risk stays present. The facts speak for themselves. Chlorates, especially magnesium chlorate, have caused several warehouse and laboratory fires over the last century. The National Fire Protection Association (NFPA) flags this compound as a strong oxidizer. Once, I handled a similar oxidizer, and even with every precaution, the uneasy feeling stayed until the last bit was safely locked away.
The Essentials: Cool, Dry, and Isolated
Heat and moisture bring out the worst in magnesium chlorate. Temperature swings lead to decomposition. Humid air? A recipe for clumping and dangerous byproducts. At every facility I've visited, technicians insist on storing compounds like this well below room temperature, shielded from sunlight and heat sources. In damp climates, running a dehumidifier in the storage area is standard practice. Ordinary steel shelving does not cut it, either—a chemical spill eats away at the metal and taints the shelf. Most institutions stick with sealed glass or high-grade polyethylene containers, and always use tamper-proof lids.
You can’t treat magnesium chlorate like laundry detergent. Keep it away from wood, paper, oils, and even dust. I learned the hard way that cross-contamination isn’t rare; a forgotten rag once nearly caused an evacuation where I worked. That near miss hammered in a habit: wipe down the bench, separate every substance, and log every movement of reactive chemicals.
Limited Quantities and Regular Checks
It’s tempting to stockpile, especially for big projects or schools with tight budgets. Experience suggests otherwise. Storing only what you plan to use within several weeks cuts risks down. Institute regular inspections—some labs schedule a monthly walk-through, checking for leaks, corrosion, and unauthorized items. At my last job, every Friday included a five-minute chemical storage check, and it spared us more than one headache.
Label every container, and match it to a clear inventory log. Fuzzy memory has no place in chemical safety. One missed label caused a fellow researcher to mix up compounds and ruin a year’s worth of samples.
An Ounce of Prevention
Simple steps work: ventilation prevents gas build-up, secondary containment trays catch leaks, and staff training helps everyone spot problems early. Organizations like OSHA and the CDC post detailed guides, and seasoned workers have their own checklists. Listening to old hands in the field beats glossy manuals most days. Mistakes shrivel when respect for the risk is real.
Storing magnesium chlorate isn’t just about following rules. It means accepting that safety, once overlooked, rarely gives second chances.Is Magnesium Chlorate soluble in water?
What Happens When Magnesium Chlorate Meets Water?
I’ve tinkered with a fair bit of chemistry—spilling and dissolving all sorts of powders on kitchen counters and in classroom beakers. Drop a pinch of magnesium chlorate into a glass of water and you’ll soon see it start to disappear. This compound dissolves quite well. Even at room temperature, magnesium chlorate becomes a clear solution, making it easy for chemists and hobbyists to use in laboratory settings.
The reason magnesium chlorate behaves this way comes down to the structure of its molecules. Chlorate ions have a knack for interacting with water molecules. Once magnesium chlorate touches water, these ions break away from the solid salt and mingle with the water, splitting into magnesium ions and chlorate ions.
The Science Behind Easy Dissolving
There is a baseline in chemistry classrooms: all chlorates tend to dissolve easily, no matter which metal they're paired with. It doesn’t matter if you work with sodium, potassium, or magnesium—the trend sticks. Water molecules can pull these chlorate ions away from the solid, and the process barely takes a nudge. Magnesium ions are reasonably small and carry a strong charge, which means the water molecules latch onto them just as eagerly.
Experiments have shown that at room temperature, one liter of water can hold dozens of grams of magnesium chlorate. This quality made it useful in the past for creating oxygen: some older chemical kits even included magnesium chlorate, where mixing it with a bit of heat could release tons of oxygen gas. Today, with more safety knowledge, this isn’t a recommended home project, but the idea taps into how easily this compound lets its ions go.
Handling and Environmental Concerns
Easy dissolving sounds like a blessing, yet it comes with risks. Highly soluble salts wander far if they get spilled. Dropped onto soil, magnesium chlorate washes straight through with the rain. It doesn’t stick to earth or clay, unlike other chemicals. In large enough doses, this can impact plant growth, as chlorate ions can interfere with how plants absorb nutrients. That’s a lesson I learned during a failed attempt at clearing weeds from a garden—nothing would grow back for seasons.
Chlorates in water supply linger longer than many expect. Because it dissolves so fast, traditional water filters barely catch it. Municipal water systems need advanced treatment steps, like activated carbon or advanced oxidation, to remove such contaminants. Lots of industries look for greener options precisely because chlorates dissolve so quickly and travel so far in waterways. Farm runoff and improper disposal in towns push authorities to monitor for these chemicals, protecting aquatic life and drinking water quality.
What Works for Safer Use?
For anyone working with magnesium chlorate, careful storage becomes more than an afterthought. Drums with tight seals and proper labeling stop accidental spills from reaching drains or gardens. Routines that include secondary containment—think trays and barriers—are worth the time, especially in labs or warehouses. For larger industries, switching to alternative chemicals where possible brings peace of mind.
Schools and hobbyists run safer projects by limiting experiments involving any chlorates and disposing of waste liquids responsibly. It’s easy to tip a small salt like this into the nearest sink, but the bigger picture asks for better choices. Educators and professionals get valuable results with simple chemistry, yet they respect the reach magnesium chlorate has once it meets water.
| Names | |
| Preferred IUPAC name | Magnesium dichlorate |
| Other names |
Chloric acid, magnesium salt Magnesium dichlorate |
| Pronunciation | /mæɡˈniːziəm ˈklɔːreɪt/ |
| Identifiers | |
| CAS Number | 7775-99-5 |
| Beilstein Reference | 3589903 |
| ChEBI | CHEBI:78033 |
| ChEMBL | CHEMBL1201791 |
| ChemSpider | 12657 |
| DrugBank | DB15460 |
| ECHA InfoCard | ECHA InfoCard: 100.028.903 |
| EC Number | 233-102-7 |
| Gmelin Reference | 6355 |
| KEGG | C18704 |
| MeSH | D008258 |
| PubChem CID | 24586 |
| RTECS number | OM3850000 |
| UNII | TY347NC467 |
| UN number | UN1476 |
| Properties | |
| Chemical formula | Mg(ClO3)2 |
| Molar mass | 191.206 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 2.32 g/cm³ |
| Solubility in water | 193 g/100 mL (20 °C) |
| log P | -2.6 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 6.37 |
| Magnetic susceptibility (χ) | Paramagnetic |
| Refractive index (nD) | 1.423 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 246.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -963.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1359.5 kJ/mol |
| Pharmacology | |
| ATC code | A12CC06 |
| Hazards | |
| Main hazards | Oxidizing agent, may cause fire or explosion; harmful if swallowed or inhaled; causes skin and eye irritation. |
| GHS labelling | GHS02, GHS05, GHS07, GHS09 |
| Pictograms | GHS03,GHS07 |
| Signal word | Danger |
| Hazard statements | H272, H302, H318 |
| Precautionary statements | P210, P220, P221, P264, P280, P370+P378, P501 |
| NFPA 704 (fire diamond) | 2 0 2 OX |
| Autoignition temperature | 450 °C (842 °F) |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 oral rat 1540 mg/kg |
| LD50 (median dose) | LD50 (median dose) of Magnesium Chlorate: Oral-rat LD50: 2500 mg/kg |
| NIOSH | MWG |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Magnesium Chlorate: "Not established |
| REL (Recommended) | 2 mg/m3 |
| IDLH (Immediate danger) | IDLH: 10 mg/m3 |
| Related compounds | |
| Related compounds |
Magnesium perchlorate Calcium chlorate Magnesium sulfate |

