Magnesium Bromate: In-Depth Commentary and Overview
Historical Development
Magnesium bromate started getting attention over a century ago, back when chemists were hungry for new oxidizing agents beyond the usual potassium or sodium salts. Its story weaves through the long tradition of bromate research. Compared to the early domination of sodium and potassium bromates in analytical chemistry and industry, magnesium’s moment came later, spurred by demand for alternatives that wouldn’t carry over some of the downsides, such as high sodium levels or expensive raw material sourcing. Laboratory synthesis methods matured with advances in chemical purification, and practical production scaled up as the need for different bromates grew across labs and niche manufacturing outfits.
Product Overview
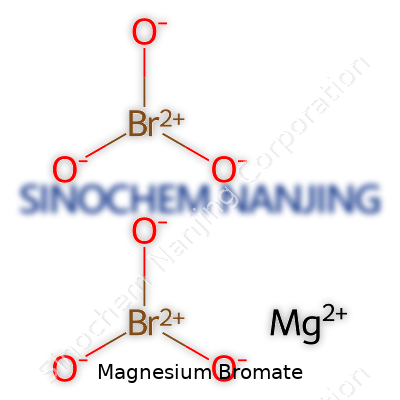
Magnesium bromate stands out as an inorganic compound with the formula Mg(BrO3)2. The choice of magnesium as the cation opens the door for uses in situations where sodium and potassium could interfere with downstream applications. Chemists and manufacturers look to it when exploring oxidation reactions. Its relatively specialized role narrows the application window compared to brute-force potassium bromate, but where tolerance to magnesium ions is critical, it finds its niche. The compound's crystalline nature and solubility in water add to its relevance in solution chemistry.
Physical & Chemical Properties
Magnesium bromate forms white, odorless crystals. The compound dissolves readily in water, handing over bromate ions into solution, but resists dissolving in most organic solvents. Its molecular weight sits at 344.11 g/mol. Like most bromates, it functions as a powerful oxidizer—something that stirs both opportunity and caution in chemists’ minds. It holds its own at ambient temperatures but decomposes before melting, which limits processing options and affects storage. Handling the powder always draws focus to its oxidizing character and the associated reactivity risks, particularly with combustible materials.
Technical Specifications & Labeling
Buyers check for purity when ordering magnesium bromate—industrial grades usually fall above 99%, and analytical grades promise even tighter specs. Product sheets dive deep: residual magnesium or sodium, moisture content, pH profile of a 5% aqueous solution, plus trace metals like iron or lead. Safety labeling carries standard oxidizer marks and signals airway, eye, and skin exposure concerns. European and US chemical labeling systems—GHS and OSHA—call for precaution statements about mixing, storage away from reducing agents, and strict personal protection protocols. Bulk packaging leans toward polyethylene-lined fiber drums or HDPE containers, with tamper-resistant seals to prevent moisture ingress and accidental spillage.
Preparation Method
Production usually starts with reacting magnesium sulfate or chloride with a bromate source, most often sodium bromate, in an aqueous phase. The reaction leverages the solubility differences to encourage magnesium bromate to crystallize out, leaving sodium chloride or sulfate in solution. Purification follows as the mother liquor gets decanted, and the crude product repeatedly recrystallized to strip away any lingering contaminant ions. This method makes the process straightforward but demands close control over reactant ratios and filtration to hit high-purity targets. Scaling up introduces issues with waste salt management and water consumption, so sustainability stays front and center for established producers.
Chemical Reactions & Modifications
Magnesium bromate sits in the toolbox as a dependable oxidant. Organic chemists reach for it when they need to oxidize certain alcohols and aromatic compounds where selectivity trumps brute force, since magnesium as a counter-ion shows less interference compared to sodium or potassium in some cases. Side reactions remain a risk—heat or acidic conditions decompose it explosively, liberating toxic bromine gas, so lab work always calls for robust ventilation and monitoring. Inorganically, swapping out magnesium with lithium or calcium to tweak properties leads to comparative studies on solubility and stability, helping researchers fine-tune applications from electrochemistry to analytical spot tests.
Synonyms & Product Names
Magnesium bromate also answers to names like bromic acid magnesium salt and magnesium dibromate. It shows up as an entry in chemical catalogues under its CAS number (usually 24474-00-6), with variations for hydrated forms like magnesium bromate hexahydrate. Commercial suppliers sometimes tack on house codes or proprietary branding, but the underlying chemical stays the same. Knowing the synonyms pays off for researchers who cross-reference international databases, regulatory documents, or multilingual safety sheets.
Safety & Operational Standards
Handling magnesium bromate demands a culture of care—there’s no skating past its oxidizing nature. OSHA and European directives classify it as hazardous, focusing attention on dust control, containment, and direct exposure minimization. Workers in manufacturing gear up with goggles, gloves, and lab coats. Facility managers design workspaces with explosion-proof ventilation and dedicated chemical-resistant storage, never stacking bromates with fuels or organics. Spill training drills and chemical hygiene plans make up part of routine operations. Fire response plans exclude traditional extinguishers; special oxidizer-rated agents get written into protocols. Clean-up procedures revolve around inactivating small spills with dilute sodium thiosulfate solution, letting industry experience set the pace for practical, real-world safety.
Application Area
Magnesium bromate finds fans in niche analytical chemistry labs. Its oxidizing drive works in spot tests and specific titrations, where a non-sodium matrix can make or break accuracy. Environmental labs turn to it for studies that track bromate as a disinfection byproduct, especially in municipal water treatment settings where magnesium outpaces sodium as a tolerated ion. Some research collects data on its use in organic synthesis, lifting up bromate as a clean oxidant for specialty reactions where interference complicates yields. Most large-scale industrial processes prefer cheaper, more accessible bromates; magnesium bromate’s higher cost and limited supply push it toward quality-over-quantity operations instead of brute force applications.
Research & Development
Research trends around magnesium bromate haven’t exactly lit up the literature like batteries or semiconductors, but the past few decades saw incremental gains. Teams dig into its reactivity, especially under exotic solvent systems or catalyzed by transition metals. Analytical chemists document detection limits, instrument compatibility, and cross-reactivity for bromate ions using magnesium as the carrier. Environmental scientists keep a watchful eye on bromates in drinking water, worried about health impacts when ozonation skews toward excess bromate production. With each study, better analytical techniques trickle into water monitoring protocols and guide regulatory actions.
Toxicity Research
Health warnings around bromates aren't new, and magnesium bromate doesn’t sidestep the concern. Toxicology studies point out bromate’s tendency to cause oxidative damage to biological tissues, with animal studies showing negative impacts on kidneys and red blood cells. Inhalation of powders or accidental ingestion carries acute risk, prompting regulatory bodies to clamp down on permissible exposure limits, especially in workplaces. Chronic exposures warn of potential carcinogenicity; as a result, food and beverage uses get shut down hard by safety agencies. With stricter water testing, bromate content stays closely monitored, steering municipal authorities to explore alternatives in water disinfection to keep drinking water safe.
Future Prospects
Looking ahead, magnesium bromate’s future will likely hover around specialty chemical markets and research. Demand could pick up if greener oxidation methods tip the scales back toward inorganic oxidants, especially when purity, low sodium, or magnesium compatibility becomes critical. Water treatment research regularly circles back to bromate detection and mitigation, creating opportunities for analytical innovation. Labs might leverage advances in automated synthesis, better crystal engineering, or greener manufacturing to reduce waste and improve safety. The main drag on wide adoption continues to be toxicity and regulatory caution—until those are tackled, magnesium bromate will walk the tightrope between useful tool and managed risk, pressed into service by those who understand its strengths and limitations.
What is Magnesium Bromate used for?
The Real Uses Behind Magnesium Bromate
Magnesium bromate doesn’t pop up in everyday conversation. The name alone rings of chemistry class, rather than the aisles in a grocery store or the gadgets we use daily. Yet, it sits tucked away in labs, manufacturing plants, and sometimes research, nudging progress along quietly.
Standing Out in Lab Life
Most people never see magnesium bromate up close, but many researchers notice its value when they run experiments. Its biggest use lies in chemical analysis and as a strong oxidizing agent. In simple terms, it helps reactions along, pushing other compounds to change form, break apart, or combine. My time running small chemistry setups during college lab days made that pretty clear—sometimes, finding a pure, reactive agent can save hours of work. Magnesium bromate does that job well since it reacts smoothly under controlled settings.
While you won’t see it on the shelf at the corner store, advanced testing work sometimes calls for it. Once, while helping in a water quality lab, I saw bromates included as a benchmark for determining the effectiveness of filtration systems. Not magnesium bromate specifically—more often potassium bromate, but the principle carried over. Labs treat such compounds with care, both for the useful results and due to their possible hazards to health and environment.
Why It Raises Safety Concerns
Magnesium bromate grabs attention for reasons beyond its chemistry. The compound is no friend to our bodies: it can irritate skin, and both bromate ions and their breakdown products can introduce toxic effects if mishandled. Researchers flagged certain bromates as possible carcinogens, a term few ignore once it enters the conversation. The EPA and FDA placed tight limits on bromates in water and food precisely because persistent exposure could harm people over time. I remember a case during a public health project where we had to investigate a spike in bromate content in a local water supply—community trust dropped overnight, and it took serious transparency and action to restore it.
Environmental Impact and Responsibility
Magnesium bromate doesn’t just vanish after use. If disposed of carelessly, oxidizing compounds linger in soil or water, triggering unexpected reactions. Careless disposal can cause problems for plants, animals, and even air quality. Years ago, I volunteered at a recycling day where people brought in old chemicals. The organizers stressed the importance of safe disposal. Mishaps were not an option—a spill or a mix with organic material could escalate fast, with contaminated runoff or even fire.
Finding a Safer Path Forward
Anyone handling magnesium bromate needs solid training and respect for lab safety. Tech improvements brought better controls for handling these types of chemicals in both research and industrial settings. Still, every time I open a news site and see another chemical spill, I think about how we all play a part in reducing risks. Investing in education for handling, storing, and disposing of lab materials protects not only workers but everyone in the surrounding community. People design processes now to catch spills early, monitor waste, and even substitute safer alternatives where possible.
Magnesium bromate tells a story that goes beyond scientific data sheets. Every chemical has a consequence—positive when used right, risky when care slips. The conversation needs to stay grounded in the facts, and our responsibility to each other, because chemicals like this don’t just vanish once the bottle empties.
Is Magnesium Bromate safe to handle?
Handling Chemicals Isn’t Like Grabbing Table Salt
Sometimes, people see a chemical name like magnesium bromate and think, “How risky could it be?” Maybe it comes from watching science videos or dealing with similar-sounding compounds in school labs. But not every white powder in a bottle is safe to handle with bare hands. I’ve learned to approach chemical safety with a mindset shaped by experience and a little skepticism. The stakes are higher than they seem – we’re not just talking about a sneeze or a skin rash.
The Real Hazards: More Than Skin-Deep
Magnesium bromate isn’t as widely talked about as household cleaners or acids, but that doesn’t make it harmless. This compound falls under oxidizers – a category that brings plenty of caution tape. An oxidizer accelerates burning, making fires more severe. Drop a bit of the dust around organic material, and the risk of ignition increases. Some labs and seasoned chemists even refer to it as “fire’s little helper,” not in a good way.
Direct exposure to magnesium bromate can harm skin and eyes. Based on published research and chemical safety resources, bromates in general can cause irritation on the skin, lead to eye redness, and trigger respiratory issues if dust gets inhaled. If you ever caught a whiff of strong chemicals and ended up with a sore throat, you’ll understand how even a small amount can make life uncomfortable.
Not All Hazards Shout Warnings
The dangers don’t always show themselves right away. Bromates are known for toxicity in more than one way. Swallowing even a small amount leads to nausea, vomiting, or more severe poisoning. There’s enough documentation out there showing that bromate compounds have caused kidney damage in animal studies. The EPA and other agencies have issued strong guidance on keeping bromates out of food and water. That’s not a mistake — it’s a big red flag.
Chemistry students often feel confident in the classroom, following instructions and using gloves or goggles like a badge of honor. But take those same habits out of the controlled environment, and risks climb. In commercial labs, the protocols looking strict are rooted in accidents people want to avoid repeating. Chemical burns, ruined equipment, and even hospital visits become real possibilities without proper diligence.
Facts At The Core: Safety Guidelines
Standard procedure calls for gloves, lab coats, and good ventilation. A dust mask or respirator helps avoid breathing in the fine powder. Magnesium bromate should stay far away from heat sources and organic material. Chemical storage guides recommend keeping it in a cool, dry place, separated from flammable substances. Spills must be cleaned carefully—not swept or vacuumed—since dry sweeping can send more particles into the air or make a cloud that delivers harm in seconds.
Forget about kitchen sinks or ordinary trash cans for disposal. Local waste disposal rules usually cover oxidizer chemicals and for good reason. Pouring them down the drain can hurt water systems and wildlife. It’s worth checking the safety data sheets and following statements from recognized agencies. Relying on a friend’s anecdote or online shortcut recipes puts more than personal safety at risk.
How We Can Address These Risks
Access to clear, science-backed education helps reduce accidents in both labs and classrooms. Practicing strict handling processes, labeling containers, and running through safety drills prepares anyone working with chemicals to respond, not panic, when things don’t go as planned. Wearing safety gear and respecting the warnings on the label will always outweigh the inconvenience. Investing in proper training pays off when mistakes cost more than money.
Chemicals like magnesium bromate deserve caution and respect. Real safety doesn’t come from shortcuts. It comes from good habits, teamwork, and a commitment to the facts over assumptions.
What is the chemical formula of Magnesium Bromate?
The Real-Life Importance of Getting Formulas Right
Magnesium bromate might not be the first thing people talk about at dinner, but knowing its chemical formula has plenty of real-world value. In my lab days, jotting down the correct formula on a data sheet meant the difference between getting results and ruining an experiment. Magnesium bromate, by the way, carries the formula Mg(BrO3)2. You’ve got one magnesium ion and two bromate ions holding hands in this compound.
How Structure Shapes Understanding
Chemical formulas can seem like a bunch of letters and numbers mashed together, but they're more useful than a scribble on a chalkboard. Let’s break down this one. Magnesium shows up as Mg2+, a metal from the alkaline earth family. Bromate appears as BrO3-, which means bromine teams up with three oxygen atoms and snags a negative charge. Nature keeps things balanced. Magnesium’s two positive charges need two bromates to keep the compound neutral. This isn’t a “pick any number” situation. If you try mixing up the proportions, you will change what you’re working with— sometimes dangerously so.
Accuracy Matters Beyond the Classroom
Misreading or guessing at something as simple as a formula can upend chemistry projects. Take it from someone who’s cleaned up a mess or two: confusing magnesium bromate with potassium bromate or magnesium sulfate means lost time, off-kilter results, and a possible scramble for safety gear. Labs expect accuracy. So do manufacturers, especially those in water treatment or chemical supply. This formula identifies not only what’s in the bottle, but also how it reacts, stores, and transports. Engineers build process guidelines around the exact ratios in formulas. Mixing up the charges or skipping a bracket means a failed process and wasted batches.
Trust and Responsibility in Chemistry
People rely on science more than they realize. Safe municipal water, medical research, and agriculture all depend on the tiniest details, like getting the right combination of ions. One time during a summer research project, my crew double-checked every single compound entry. Extra time spent reading, writing, and confirming formulas like Mg(BrO3)2 helped us avoid setbacks that would’ve made headlines for all the wrong reasons. That healthy fear led to good habits and better end results.
Solutions for Avoiding Mistakes
Textbooks, chemical databases, and digital tools give people ways to check their work. I’ve used apps that spit out formulas after plugging in the charges, and I’ve watched students flip to the periodic table posters on the classroom wall. Double-checking feels routine for professionals, but recreational chemists, teachers, and students need to pick up those same habits. Building these checks into every project, no matter how simple, keeps mistakes from snowballing.
Why Correct Formulas Still Matter
Sometimes the simplest equations form the backbone of bigger systems. Magnesium bromate’s formula points to its basic building blocks, but those blocks can cause chain reactions if mishandled. Trust in industries and research labs sure doesn’t grow without handling these details well. My time in group projects and late-night lab sessions convinced me that chemistry’s small steps—like noting the correct formula—are critical for building trust, achieving results, and keeping people safe. The right answer isn’t just about points on a quiz; it’s part of what makes science work in the real world.
How should Magnesium Bromate be stored?
Real Dangers Hiding in the Details
Magnesium bromate sits in a tricky category—strong oxidizer, white crystal, no-nonsense hazard. I remember walking into a supply room years ago, seeing a drum labeled with that bold red “OXIDIZER” diamond. Seeing that label, you know you’re dealing with a chemical that will boost the power of a fire or make even small mistakes a big deal. Firefighters have cited cases where improper storage turned a manageable incident into something that turned deadly fast.
Keep It Dry, Keep It Cool
Experience tells me that moisture is enemy number one for compounds like magnesium bromate. A spill on a humid day led to caked, sticky material and a stressful cleanup—the substance absorbs water, and, worse, wet material sets up the risk for decomposition or more violent reactions. So, always pick a dry, well-ventilated space. Humidity levels should stay low—old-timers keep desiccants in storage rooms for a reason. If you store magnesium bromate near any water source, you’re gambling with everyone’s safety.
No Sparks, No Flames, No Mix-ups
Heat accelerates trouble. I’ve seen colleagues skip checks on air conditioning or insulation, and come back to find labels warped and containers crusted by chemical changes. Prevent these headaches: never store magnesium bromate near heat sources. Lock it away from boilers, radiators, or even sunny windowsills. Keep away every flammable or combustible substance—paper, grease, wood. Even tiny leaks or dust on wood shelving can mean a small error turns explosive.
Only Original, Airtight Containers
Factories send magnesium bromate out in specific containers for a reason—they resist chemical damage, seal tightly, and don’t react with the product. I’ve seen people pour leftovers into repurposed plastic buckets or glass jars. Those can fail. Stick to strong, chemical-safe packaging, keep lids sealed, and label clearly. It is easy to forget a half-used jar shoved to the back of a shelf, but unlabeled leftovers have caused confusion and accidents, both in labs and in schools.
Keep It Away From Acids and Organic Stuff
One of the fastest ways to see magnesium bromate misbehave is to let it mingle with acid or anything organic. Acids break down the compound and can start a fire or release toxic gases. In 1997, a warehouse accident in New Jersey involved an oxidizer and an unknown acid spill, and the cleanup made national news. That shows that acid must live on a separate shelf, preferably in an entirely different room.
Routine Checks Save Lives
Storage conditions change with the seasons. What felt dry in January could feel damp by July. Regular checks matter; inspect seals, look for leaks, note signs of clumping or color change. Address issues right away. An unchecked leak in a back storeroom once forced an entire facility shutdown for three days.
Education Prevents Accidents
Every worker handling magnesium bromate ought to know the risks. Simple safety briefings work. The more familiar you get with what can go wrong, the easier it becomes to spot a risk before it grows. Stock spill kits and make sure everyone knows evacuation routes. Safety is not a box on a checklist—it’s a habit that keeps you and your coworkers going home at the end of the day.
The Bottom Line
Storing magnesium bromate safely is not complicated, but it punishes shortcuts. Dry air, cool surroundings, sealed containers, and strict separation from anything reactive—these are the basics that protect people, property, and peace of mind. Responsible storage reflects how much you value safety, and that is never just a formality.
What are the possible hazards of Magnesium Bromate?
Understanding the Risks Around Us
People walk past bags of lab chemicals every day, never thinking twice about strange compounds like magnesium bromate. It’s not a name you find in most households, but it’s used in industry and labs, and there’s a catch: it poses some risks that deserve attention. I worked in a research setting for a few years, and just seeing this compound on an inventory list meant we’d need special handling protocols. Some chemicals come with a nasty bite, and magnesium bromate happens to be one of them.
Digging Into the Dangers
Magnesium bromate falls into the category of strong oxidizers. Oxidizers help other stuff burn—sometimes much faster than expected. A simple mistake like storing it near flammable materials can set the stage for fires or even small explosions. When you hear stories of warehouse fires in the news, often it’s an oxidizer that tipped the scales. Mix magnesium bromate with things like paper, sawdust, or even regular household chemicals, and you can spark a real crisis.
There’s more than just fire risk. Inhalation or skin contact brings another set of problems. The dust can irritate airways, eyes, and skin. Several safety data sheets, including those published by the European Chemicals Agency, say magnesium bromate has toxic properties. It isn’t something you want on your hands, and it definitely doesn’t belong in the body. If care lapses in a lab or storage area, accidental exposure can cause symptoms like nausea, headaches, or breathing troubles. Ingesting it—on purpose or by accident—could lead to severe health consequences such as kidney or nervous system problems.
Environmental Concerns
Environmental hazards often fly under the radar, but they matter too. Improper disposal leads to bromate ions reaching water sources. Once in groundwater or lakes, bromate threatens aquatic life. These ions don’t rapidly break down, so plants and fish can take a hit. The US Environmental Protection Agency has set strict limits on bromate in drinking water for good reason—persistent toxins don’t just vanish with a little sunlight and fresh air.
Learning From Experience
Handling risky chemicals taught me to respect every label and storage instruction. Gloves, goggles, and closed containers weren’t negotiable. I remember one emergency drill where a small oxidizer spill turned into chaos because someone put a wet cloth on it—a big mistake with an agent like magnesium bromate. Water doesn’t always neutralize; it can even react.
Steps Toward Safer Use
An honest look at these hazards leads straight to safety training. Every place that stores or moves magnesium bromate should run regular drills and feature clear, honest signage. Safety data needs to be more than just a binder collecting dust. I found that making hazard information visible changed coworkers’ habits faster than a stack of emails from management.
Substitution is another real option. Many industries exploring greener chemistry options frequently swap out high-risk oxidizers for less hazardous ones. This approach has lowered incident rates in some labs without killing productivity.
Staying Ahead of the Risk
No matter how seldom you encounter magnesium bromate, knowing how it acts and what it can do makes a difference. Experience says complacency invites disaster, especially with reactive chemicals. With honest information, clear steps, and the right gear, it’s possible to keep workplaces and the wider environment safer—one careful decision at a time.
| Names | |
| Preferred IUPAC name | Magnesium dibromate |
| Other names |
Bromic acid magnesium salt Magnesium dibromate |
| Pronunciation | /maɡˈniːziəm ˈbrəʊ.meɪt/ |
| Identifiers | |
| CAS Number | 7789-78-8 |
| Beilstein Reference | 3567933 |
| ChEBI | CHEBI:78033 |
| ChEMBL | CHEMBL1201617 |
| ChemSpider | 21232018 |
| DrugBank | DB14695 |
| ECHA InfoCard | ECHA InfoCard: 100.031.914 |
| EC Number | 236-184-5 |
| Gmelin Reference | 83906 |
| KEGG | C18759 |
| MeSH | D008264 |
| PubChem CID | 24636 |
| RTECS number | EA8686000 |
| UNII | 78F97177J1 |
| UN number | UN1476 |
| Properties | |
| Chemical formula | Mg(BrO3)2 |
| Molar mass | 184.113 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 3.72 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | -0.52 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 8.6 |
| Basicity (pKb) | 8.67 |
| Magnetic susceptibility (χ) | +1340.0e-6 cm³/mol |
| Refractive index (nD) | 1.652 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 190.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1532 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1427.7 kJ/mol |
| Pharmacology | |
| ATC code | V04CM03 |
| Hazards | |
| Main hazards | Strong oxidizer, may cause fire or explosion; harmful if swallowed, inhaled, or in contact with skin; causes irritation to eyes, skin, and respiratory tract |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Danger |
| Hazard statements | H272, H302 |
| Precautionary statements | P264, P270, P273, P301+P312, P330, P501 |
| NFPA 704 (fire diamond) | 3-0-2-OX |
| Autoignition temperature | 580°C |
| Lethal dose or concentration | LD50 (oral, rat): 1800 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 180 mg/kg |
| NIOSH | MWG75000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 5 mg/m³ |
| Related compounds | |
| Related compounds |
Magnesium bromide Magnesium perchlorate Magnesium sulfate |

