Magnesium Bisulfite: A Closer Look at Its Past, Present, and Future
Historical Development
Curiosity guided chemists to magnesium bisulfite centuries ago, mostly through paper-making. Early factories hunted for ways to break down tough wood fibers, and magnesium bisulfite offered a less corrosive approach than sodium-based alternatives. In the early 1900s, magnesium salts entered pilot pulp mills in Europe and North America, offering a practical route to sulfite pulping. These techniques spread as the printing industry skyrocketed and demanded higher-grade paper with fewer impurities. Direct experience in a mill sometimes revealed the differences between pulping agents—fewer scale deposits, improved recovery of chemicals, and lower maintenance costs drew many to magnesium chemistry. Over decades, labs tinkered with additives, tried different pressure and temperature settings, and found more efficient routes for producing and recycling the bisulfite liquor, gradually building up knowledge still referenced in today’s process manuals.
Product Overview
Magnesium bisulfite shows up as a clear, acidic solution or in solid form as a white to yellowish salt. Industry uses it for delignification in paper, water treatment, and food preservative applications. The compound dissolves in water and reacts in controlled ways with wood chips, removing lignin efficiently. Technical grades often carry names like “pulping-grade magnesium bisulfite” or “magnesium hydrogen sulfite,” and industrial buyers often recognize their product by supplier labels such as “Mag-Sulfite” or “Magnesium Acid Sulfite Solution.” Product selection depends on concentration, impurity levels, and the packaging format, reflecting end-user expectations for smooth batch operations, reliability, and regulatory compliance.
Physical & Chemical Properties
Magnesium bisulfite appears as a moderately soluble salt. Its odor points unmistakably to sulfur dioxide, which matters from both handling and process safety viewpoints. Physical handling exposes operators to a white granular powder or a slightly turbid liquid, both being highly acidic—with a pH in solution well below 5. Typical technical material boasts strong reducing properties and releases sulfur dioxide gas if heated or left in contact with acid. These properties make it attractive for both chemical synthesis and pollutant control, but also demand solid containment and ventilation. Density, solubility, and melting points differ with water content; most plant settings use it dissolved in water at concentrations ranging from 30% to 60% by mass, which impacts both storage requirements and feed dosages.
Technical Specifications & Labeling
Labels and material safety data sheets publicly list magnesium bisulfite solutions by their percentage content, pH range, specific gravity, and storage temperature requirements. Facility operators often keep reference cards handy, since the solution can lose SO₂ gas if left unsealed. Standard labels cite its UN number for corrosive substances and warn about contact hazards. Major suppliers test for iron and heavy metals, since presence above set limits leads to off-color products and risk downstream. Genuine technical product comes packaged in coated drums or tanks, each batch carrying lot numbers to match to quality records. Inspection certificates help inventory managers trace the chemical’s origin and usage in regulated industries.
Preparation Method
Manufacturers create magnesium bisulfite by bubbling sulfur dioxide gas through a slurry of magnesium carbonate or hydroxide. This reaction yields a magnesium salt solution with plenty of SO₂ dissolved in water, resulting in a stable acidic liquid if kept under cool, airtight conditions. Old-school pulp mills once generated SO₂ on-site by burning elemental sulfur, passing the gas through scrubbers, and squeezing every bit into the reacting tanks. Most operations these days automate this step, feed precise magnesium oxide quantities, and recirculate heat and water to boost efficiency. Equipment layouts and process controls impact final concentration and purity, as handling cold, reactive solutions with corrosive vapors brings its own operational headaches.
Chemical Reactions & Modifications
Magnesium bisulfite enters into redox reactions—especially with lignin in wood or dissolved oxidants. It acts as a reducing agent, scavenging oxygen in both pulp and environmental settings. If exposed to oxidizers, it flips into magnesium sulfate, and exposure to base converts it to less reactive magnesium salts, which changes its value in process streams and final product. Labs modify magnesium bisulfite with stabilizers or blend with other sulfites for specialized dechlorination or microbiological controls. Overdosing, impurities, and temperature spikes trigger off-gassing, which complicates system design and worker safety needs, particularly where venting and scrubbing gear updates haven't kept pace.
Synonyms & Product Names
This compound travels through supply chains under names like magnesium hydrogen sulfite, E227, magnesium acid sulfite, or simply “Mag bisulfite.” Brands vary regionally. In North America, chemical suppliers may use “Mag-Sulfite” while European catalogs favor “Magnesium Hydrogensulfit.” Buyers learn to check CAS numbers and local trade names, since mislabeling can lead to the wrong application or missed hazard signals. Trade publications and regulatory handbooks stress these distinctions, especially for import/export paperwork and handling emergency response.
Safety & Operational Standards
Everyone handling magnesium bisulfite learns to respect the compound’s reactivity and corrosivity. Without good ventilation, SO₂ fumes cause eye and lung irritation fast. Acid-resistant gloves, face shields, and fume hoods come out on every job, and operators receive drills on chemical burns and containment leaks. Industry sets training and inspection intervals—equipment must withstand both acid attack and sudden gas release. Regulations set thresholds for allowable exposure, require eyewash stations, and spell out containment and waste handling practices. Failures risk product contamination or workplace accidents, which can grind operations to a halt and bring regulatory fines. On plant tours, I’ve seen audits focus intensely on storage temperatures, clear labeling, and maintenance plans for the venting network—a missed warning sign on any of these increases accident likelihood.
Application Area
Magnesium bisulfite dominated the pulping sector for decades, breaking down tough lignin to produce a smoother, brighter paper sheet. Its role in water treatment keeps chlorine and chloramine in check, so the drinking water flowing from taps meets quality targets. Food manufacturers sometimes use permitted grades to protect delicate fruits and reduce discoloration, although strict limits cap daily intake due to residual SO₂. Labs specializing in organic synthesis employ this salt as both reducing agent and preservative. Some environmental remediation projects inject magnesium bisulfite underground to bind heavy metals and reduce their bioavailability. Each field demands rigorous purity assurances, delivery schedules, and follow-up support—which means chemical suppliers keep busy with technical field reps chasing down satisfaction and troubleshooting problems as they surface.
Research & Development
Research circles around magnesium bisulfite focus on improving efficiency, lowering emissions, and refining byproduct streams. Industry labs investigate catalysts to boost SO₂ binding, while university groups hunt for cleaner preparation routes or greener recycling options. Some work explores blending magnesium bisulfite with other chemistries to tailor properties to paper grade, water purity, or pollutant load. In advanced material sciences, attention turns to modifications that limit off-gassing, reduce corrosion, or enhance compatibility with next-generation biodegradable polymers. Experiments track changes in molecular structure, looking for clues to reactivity under real-world conditions. This research underpins technical bulletins and best practice guides, which serve as the blueprints for safer, more cost-effective production and application.
Toxicity Research
Health questions often arise over exposure to magnesium bisulfite, particularly in confined workspaces. Low concentrations irritate airways and eyes, and chronic exposure without protective gear leads to coughs or worse in sensitive individuals. Toxicologists have documented SO₂’s effects—mild doses prompt discomfort, higher doses carry acute risks for people with asthma or other respiratory issues. Food safety scientists watch out for traces in packaging or finished products, enforcing strict “residual SO₂” testing to keep levels under legal limits. The risk to aquatic life comes up in wastewater discharge conversations, since unneutralized spills can spike acidity and deplete dissolved oxygen. Producers, regulators, and user groups all push for better monitoring, improved emergency response, and ongoing education to keep risks low and product trust high.
Future Prospects
With the chemical industry pivoting toward sustainability, magnesium bisulfite faces fresh tests. Paper mills, under growing pressure to lower emissions and cut waste, look for ways to recycle lignin-rich effluents and recover more energy from pulping streams. Water treatment operators demand next-generation sulfites that perform reliably with fewer safety tradeoffs and lower ecological impact. The food sector encourages research into less intrusive antioxidants, but old standbys like magnesium bisulfite stay in the mix thanks to proven cost and results. Academic groups continue searching for better reaction pathways, energy-saving process steps, and circular economy models that close the loop on salt and sulfur cycles. Lately, digital monitoring tools coupled with automated dosing improve operational safety, giving producers a surge of data that feeds back into technical improvements. Innovation continues to reshape the field, giving industries more tools to blend environmental responsibility with commercial practicality.
What is Magnesium Bisulfite used for?
Magnesium Bisulfite: More Than Just a Chemical Name
People don’t usually think about what’s behind a newspaper, a book page, or even a food label. In papermaking, magnesium bisulfite shapes the world of pulp and paper long before the presses roll. Years ago, I visited a paper mill on a college trip. The machines hummed, workers looked over giant vats, and I realized that all those bright white sheets started from a process full of chemistry—magnesium bisulfite front and center.
Pulp and Paper Production
The main job for magnesium bisulfite lives in pulp production. This is the chemical pulping industry’s workhorse for the sulfite process, which breaks down wood chips into a mushy mix. Adding magnesium bisulfite lets mills pull out lignin, the sticky stuff that binds cellulose fibers. This gets the pulp softer and easier to bleach later. Compared to harsher chemicals, this process handles certain woods better, gives higher yields, and makes paper with a nice finish. Sulfite pulps, made using magnesium bisulfite, help give us printing paper, tissue, and specialty packaging. Factories using this method cut down on waste and reduce energy needs. The difference in efficiency helps companies keep up with sustainability goals.
Environment and Safety: Real Concerns
People working with chemicals always think about safety. Magnesium bisulfite brings fewer hazards than some alternatives, but it still calls for respect—ventilation, masks, safeguards on tanks. Spills need quick cleanup, and water containing this chemical demands proper treatment before heading downriver. It breaks down into magnesium compounds and sulfites, so it fits in better with current environmental standards, but companies still watch runoff closely. Everybody wants clean rivers and good air. The right safeguards at pulp mills, and advances in effluent treatment, help keep accidents rare and protect surrounding communities.
Magnesium Bisulfite Beyond Paper
Outside the mill gates, magnesium bisulfite finds a place in other industries. Some winemakers use it as a preservative that stops wild microorganisms from ruining a batch. Food producers sometimes add it as a gentle antimicrobial agent for fruits and vegetables. Its ability to soak up oxygen helps protect foods from browning. Every time I see bright white dried apples, I think about all the steps in the background, working to keep them fresh.
Moving Forward: Cleaner Technology and Smarter Use
Magnesium bisulfite’s future isn’t just about making more paper or better-tasting fruit. Companies want to use less water, clean up byproducts, and find ways to keep their operations safe and efficient. Some mills recycle sulfite process chemicals to cut down costs and pollution. New sensor systems give real-time feedback so engineers can tweak the process. Regulators and workers both push for better waste handling and smarter machinery.
Chemistry in industry can sound dry, but a lot rides on how responsibly we handle it. Magnesium bisulfite, used well, helps deliver the products people rely on every day—while pushing companies to balance productivity with environmental care. That balance matters, not just for business, but for the towns and watersheds that surround every plant.
Is Magnesium Bisulfite safe for use in food or water treatment?
Looking at Everyday Exposure
Magnesium bisulfite mainly turns up in the pulp and paper industry, where it breaks down wood chips during paper production. Lately, people ask if this chemical belongs in food or drinking water. I see that question pop up partly because other bisulfite compounds—like sodium bisulfite—do land in some foods as preservatives. Still, not every cousin in the bisulfite family works the same.
The Chemistry Behind the Concern
Magnesium bisulfite shares some traits with better-known sulfites. These compounds can stop spoilage, cut back browning, or help keep things fresh. In water treatment, some sulfite compounds step in to scrub away leftover chlorine before the water enters the tap. On the surface, magnesium bisulfite offers the same dechlorination perks, which makes it appealing for treating large volumes of water.
Fact-Checking Authority Guidance
Official guidelines matter to me for safety talks. Here’s what stands out: the Food and Drug Administration (FDA) does approve certain sulfites for use in food in defined amounts. Those include sodium sulfite, sodium bisulfite, and potassium metabisulfite, all labeled as “Generally Recognized as Safe” (GRAS) for specific applications. Magnesium bisulfite does not turn up under that list. The Joint FAO/WHO Expert Committee on Food Additives hasn’t listed it either. For water, the Environmental Protection Agency (EPA) has standards for sulfate, which doesn’t match magnesium bisulfite.
No recognized health authority gives the green light for magnesium bisulfite use in food. I checked a few popular global resources on risk—nothing concrete shows up that clears its use for drinking water either. Absence of a ban does not equal safety. In my own reading, the lack of endorsement carries weight. Never saw a reputable source put magnesium bisulfite on par with approved food preservatives.
Possible Side Effects and Sensitive Groups
Sulfites can bring on reactions in sensitive people, especially those with asthma. Headaches, itchiness, or breathing trouble sometimes appear. Regular intake of any bisulfite draws concern if someone deals with allergies or asthma; with magnesium bisulfite, things only get murkier because there’s so little data.
Children, older adults, and people who rely on municipal water supplies depend on clear rules. Guesswork in food safety usually leads to regret. The risk might be small, but if people begin adding this compound to food or water without clear oversight, side effects could slip through unnoticed until someone lands in the emergency room.
Room for Smarter, Safer Choices
Plenty of established options already keep food fresh or help treat water. Each of them cleared rigorous review, such as sodium bisulfite and potassium metabisulfite for food, or even activated carbon and UV for water. I lean toward solutions that come with a track record. No one benefits from shortcuts in something as personal as what goes on the plate or runs from the faucet. Instead of reaching for a less-studied compound, the best policy sticks to what’s proven and keeps an eye on evolving science. People deserve confidence that their food and water are safe.
What is the chemical formula of Magnesium Bisulfite?
The Chemistry Behind the Name
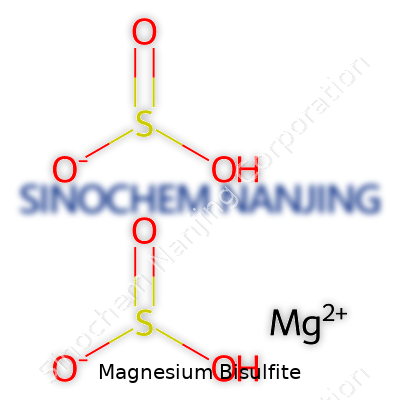
Magnesium bisulfite carries the formula Mg(HSO3)2. The name might look intimidating, but at its core, it’s built from magnesium—an element found in everything from baseball bats to chlorophyll—paired with two bisulfite ions. The “bisulfite” part means you’ve got hydrogen, sulfur, and oxygen in a specific pattern. Put two of those with one magnesium ion, and that’s the story.
Importance in Real-world Applications
Magnesium bisulfite finds a major use in the pulp and paper industry. It helps break down wood chips into something workable for making paper. It’s fascinating to see how a metallic salt, whipped up in a chemical plant, transforms logs into the pages people read every day.
After learning about papermaking through hands-on science classes, it becomes clear how much chemistry supports daily life. Papermills rely on chemicals that work efficiently and leave behind minimal environmental impact, and magnesium bisulfite fits that requirement better than many alternatives. It doesn’t add unwanted heavy metals or residues that trouble water treatment systems downstream from mills.
Health and Safety Considerations
While magnesium bisulfite provides great value, handling it comes with responsibilities. Bisulfite ions can release sulfur dioxide gas, which irritates lungs and eyes. Workers handling the compound need proper gear and training. Strict site regulations limit accidental releases. This protects not just staff but entire communities near manufacturing plants.
Doctors and emergency responders in industrial regions should know the risks tied to sulfur compounds. Sulfite sensitivity can cause powerful reactions in some people, especially those with asthma. Reviewing workplace exposure standards from agencies like NIOSH and OSHA gives context: sulfur dioxide levels must stay well below 5 ppm in the air most of the time.
Environmental Impact and Sustainable Practices
Every chemical used on an industrial scale leaves a footprint. Magnesium bisulfite stands out because it creates less troublesome waste than calcium compounds often used in older pulping methods. I’ve spent time talking with environmental engineers who study these waste streams—magnesium salts stay relatively soluble, making water treatment more straightforward. Less sludge, less hassle.
Improving sulfur management at pulp mills represents one of the easiest wins for cleaner operations. Many facilities now recycle waste magnesium, either recapturing the metal or reprocessing byproducts. Adopting closed-loop systems keeps these substances out of rivers and groundwater. Community groups near plant sites should push for independent testing and transparent waste reporting.
Room for Improvement
Even with the advantages magnesium bisulfite offers, better collection methods for emissions could drive down risks further. Mills benefit by working with local universities and research labs, exploring enzyme-based or bio-inspired treatments for pulping wood, which might eventually edge out traditional chemicals.
Sharing best practices—such as using sensors for early leak detection or investing in higher-grade vent filters—gives workers safer jobs and residents peace of mind. It also helps the industry stay ahead of regulations, which everyone benefits from. Raw chemistry knowledge and a willingness to upgrade old equipment matter far more than just following minimum legal standards.
Looking Forward
As long as there’s demand for paper and clean processes, magnesium bisulfite will stay relevant. Its formula, Mg(HSO3)2, stands as more than textbook trivia. It reflects the intersection of science, industry, and everyday responsibility.
How should Magnesium Bisulfite be stored and handled?
Practical Hazards: More Than Just a Chemical on a Shelf
Magnesium bisulfite plays a steady role in industries like paper and pulp, but storing and dealing with it turns into real work. Left alone, it releases gas and can mess with your breathing. I’ve seen pallets leaking this stuff in a corner—nobody wants that headache. So, dry, cool, and well-ventilated space should always win out. Warehouses with open doors in summer face humidity spikes, which can set off slow decomposition. Once moisture sneaks in, bags clump, labels smear, and suddenly, good product turns into a waste call and a safety incident.
Keep It Sealed, Keep It Safe
If you asked me about spills, I’d tell you: loose packaging causes more chaos than a broken machine. Seal the bags right, every time. Walk through the stockroom, check for damage, and clean residue on shovels and gloves before it spreads to door handles or lunch tables. I’ve seen slip-ups traced back to one open drum. Clear labeling with “Caution: Corrosive” or “Store Away from Acids” speaks louder than another faded sticker. Even small leaks end up as accident reports.
Why Ventilation Matters—Beyond Just Smell
Some warehouse managers focus more on temperature and less on air flow. A neighbor’s shop learned that lesson fast. Gas built up after a warm night, and the next morning nobody could breathe. Sulfur dioxide isn’t some invisible ghost; it irritates eyes and throats and sets off asthma fast. Storage where air actually moves—fans or vents, even cracked upper windows—keeps problems out of employee health logs and the company’s insurance reviews. A simple sensor for SO2 helps, especially in closed environments.
Deal With Acids Like You Mean It
I don’t trust “good enough” storage when acids are nearby. Magnesium bisulfite meets acid, and out comes sulfur dioxide, which can spread fast indoors. I recall someone kept a shelf of cleaning acids right next to a pallet of bisulfite just because space was tight—that shelf ended up at the dumpster. Smart layouts put space between chemicals. Spill trays under pallets and buckets by entryways make cleanup less frantic, should trouble come knocking.
Train With the Real Risks in Mind
Safety isn’t paperwork. Workers want backup: clear instructions, quick access to gloves and goggles, and the freedom to call out problems. Gloves and face protection mean comfort, not just compliance. Knowledge of what happens if bisulfite contacts skin or eyes—swift washing, quick reporting—keeps small errors from turning nasty. I’ve taken part in drills where even seasoned techs throw out clumsy gloves or beat-up goggles. Equipment that works, not just checked-off boxes, keeps people healthy.
Addressing Waste and Cleanup
If product gets stale or contaminated, disposal turns serious. Drains aren’t a dumping ground. Waste firms recognize magnesium bisulfite as hazardous, so coordination with local rules proves necessary. I once had to double-bag a drum, slap on labels, and make extra calls just to keep regulators satisfied. It’s not just a routine—it respects the neighbors, too, since fumes cross property lines far too easily.
Real Safety, Real People
Magnesium bisulfite asks for careful respect every day. Simple habits—good seals, proper labels, working fans, real separation from acids, and solid training—don’t just protect a bottom line. They keep people safe, keep products in spec, and let teams head home healthy. Storage plans aren’t just about shelf space—they’re about trust between every worker, from warehouse to lab coat.
What are the potential health hazards of Magnesium Bisulfite exposure?
Everyday Exposure Isn’t Always Obvious
Magnesium bisulfite often comes up in conversation around pulp and paper manufacturing. Plenty of people in industry have handled a barrel or two, but even those who haven’t can land in the same room with this chemical. It's not stocked in every home, but workplaces send it out into the world, whether through storage tanks or wastewaters. I once toured a plant where the tang in the air told its own story. Workers coughed and rubbed their eyes, and no one needed a warning sign to know something harsh lived nearby. Small exposures matter, even before you notice symptoms.
Short-Term Hazards on the Job and Beyond
Magnesium bisulfite’s biggest trick is its strong acidity. Splash a little on skin: red bumps show up, then peeling. Catch a whiff in your nose: burning, coughing, headaches soon follow. For those breathing high levels, the throat closes up. Asthma attacks happen. Safety data sheets point out eye damage that might never clear up fully. A friend once misjudged his mask seal handling a leaking pump. He couldn't shake the taste of metal for two days and had trouble swallowing food for a week. Sudden exposures force old hands to slow down and double-check PPE.
Long-Term Risks Aren’t Just Numbers
Folks who live or work near facilities sometimes ask, “What does this stuff do over years?” It turns out, health impacts go beyond just a scratchy throat. Strong acids like magnesium bisulfite wear away at body tissue, year after year. Chronic exposure eats at lung health, and people wind up with breathing problems not so easily reversed. One study from the early 2000s tracked pulp mill workers and saw more cases of chronic bronchitis in those exposed to bisulfite compounds than those who weren’t. It sounds like old news, but these problems keep popping up in modern work environments. Environmental scientists have flagged wastewater releases in small communities—trouble doesn’t stay locked inside factory doors.
Why This Deserves More Attention
Some folks ask why not just replace the stuff with safer chemicals. Turns out, in some industrial processes, alternatives either cost more or perform worse. That’s why real solutions focus on engineering and education. Every plant I’ve seen that runs smoothly trains its people tight on handling, stores the chemical in sealed and vented systems, and monitors air quality often, not just during inspections. Safety equipment only works if everyone respects it—every shift, not just after close calls. Doctors remind us too much exposure makes chronic respiratory troubles likelier, and no good job should steal your breath.
Protecting Your Health Comes Down to Action
Companies get the ball rolling with right-sized exhaust systems, real-time gas detectors, and routine medical checkups. Folks on the floor need gloves and goggles ready, and quick drenching showers close by. Community groups push for better reporting so people know what floats in their air and water. Practical steps make health manageable, not a roll of the dice. Scientists recommend even small operations test for leaks, keep emergency plans up to date, and switch to less toxic options where possible. Hazardous chemicals deserve real respect, not blind trust in luck.
| Names | |
| Preferred IUPAC name | magnesium hydrogen sulfite |
| Other names |
EINECS 232-094-6 Magnesium hydrogen sulfite Magnesium bis(HSO3) Sulfurous acid, magnesium salt (2:1) Magnesium(II) bisulfite |
| Pronunciation | /maɡˈniːziəm baɪˈsʌl.faɪt/ |
| Identifiers | |
| CAS Number | 13774-03-9 |
| Beilstein Reference | 1209261 |
| ChEBI | CHEBI:78033 |
| ChEMBL | CHEMBL1233488 |
| ChemSpider | 160391 |
| DrugBank | DB14526 |
| ECHA InfoCard | 21a1ca1a-7ed3-4bd2-bca7-845fa885770a |
| EC Number | 233-826-7 |
| Gmelin Reference | 79079 |
| KEGG | C18801 |
| MeSH | D017960 |
| PubChem CID | 24643 |
| RTECS number | SA9625000 |
| UNII | LT60V0302D |
| UN number | UN2693 |
| Properties | |
| Chemical formula | Mg(HSO3)2 |
| Molar mass | 120.38 g/mol |
| Appearance | White or yellowish crystals or powder |
| Odor | Sulfur dioxide odor |
| Density | 1.42 g/cm³ |
| Solubility in water | Very soluble |
| log P | -3.7 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 3.6 |
| Basicity (pKb) | 8.7 |
| Magnetic susceptibility (χ) | -22.6e-6 cm³/mol |
| Refractive index (nD) | 1.462 |
| Viscosity | 10-20 cP (at 25°C) |
| Dipole moment | 3.49 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 130.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1386.8 kJ/mol |
| Pharmacology | |
| ATC code | A12CC04 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes severe skin burns and eye damage. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | Harmful if swallowed. Causes serious eye damage. |
| Precautionary statements | P264, P270, P273, P280, P301+P312, P305+P351+P338, P330, P337+P313, P501 |
| NFPA 704 (fire diamond) | 2-1-2-W |
| Lethal dose or concentration | LD50 (oral, rat): 520 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 1600 mg/kg |
| NIOSH | MGY |
| PEL (Permissible) | 5 mg/m3 |
| REL (Recommended) | REL (Recommended): 5 mg/m³ |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Magnesium sulfite Magnesium sulfate Sodium bisulfite Calcium bisulfite |

