Magnesium Arsenate: A Comprehensive Look at Its Journey, Properties, and Role in Science
Historical Development
The discovery of magnesium arsenate stretches back over a century, rooted in the search for useful inorganic compounds during the late 1800s. Scientists working through periods of industrial revolution explored mineral deposits, often stumbling across magnesium arsenate while extracting other arsenates for dyes, pigments, and analytical reagents. Chemists soon realized the value of combining magnesium’s lightness with the unique reactivity of arsenic. Early documentation referenced its crystallization habits and ease of precipitation, especially in geochemical studies trying to decipher the stability of arsenic minerals in natural waters. Magnesium arsenate entered scientific conversations not because it became a popular ingredient in industry, but due to its applications in soil chemistry, environmental testing, and mineralogy research.
Product Overview
Magnesium arsenate commonly appears as a white powder or crystalline solid. Its main formula, Mg3(AsO4)2, hints at a relatively simple structure, but under the microscope, researchers notice the intricate arrays of magnesium and arsenate ions bound into rigid frameworks. Unlike many widely used mineral products, magnesium arsenate only shows up in specialized markets—laboratories, geological surveys, and some synthetic applications. Investigators in analytical chemistry have used it to calibrate tests or assess arsenic contamination, leveraging its defined stoichiometry and solubility profile.
Physical & Chemical Properties
Dense and stable at room temperature, magnesium arsenate barely dissolves in cold water, which pulls its utility toward controlled-release scenarios instead of biological or agricultural applications. It forms orthorhombic crystals, reflecting its ordered internal lattice. The compound has a melting point hovering above 900°C, allowing it to resist most common thermal decomposition tricks. Some acids, including hydrochloric and nitric acid, break down magnesium arsenate, supplying arsenate ions into solution. In my experience handling it in a teaching lab, magnesium arsenate carries little to no odor and resists moisture absorption unless left exposed for prolonged periods, making it manageable with standard lab protocols. Its high density and poor electrical conductivity make it easy to separate from lighter, more reactive arsenates.
Technical Specifications & Labeling
Standard-grade magnesium arsenate, as supplied by specialty chemical vendors, clocks in at greater than 99% purity. Bottles bear hazard warnings tied to arsenic’s reputation for toxicity, plus storage suggestions that call for airtight containers and segregation from acids or reducing agents. Labels display the chemical's CAS number, molecular weight (383.03 g/mol for the basic salt), and basic regulatory designations from OSHA, REACH, and the German Water Hazard Class. For scientific research, trace contaminants like iron, calcium, or phosphate draw close scrutiny, with reputable suppliers issuing batch-specific purity analyses. Storage in cool, dry places—not near food or animal feed—remains standard advice across global suppliers.
Preparation Method
In most laboratory syntheses, magnesium arsenate arises from the controlled mixing of magnesium sulfate or magnesium nitrate with sodium arsenate solutions. The chemist stirs at room temperature, adjusting the pH as a faint cloudiness signals precipitation. After filtration, careful rinsing removes sodium and sulfate ions, yielding pure magnesium arsenate. Scaling up demands caution, as the process always involves soluble arsenic and the risk of environmental release. The main byproduct, sodium sulfate, can often be used in other reactions, reflecting the practical mindset of minimizing waste. In the teaching environment, we always worked under a fume hood, avoiding inhalation or skin contact.
Chemical Reactions & Modifications
Magnesium arsenate stays stubbornly unreactive under neutral or basic conditions, resisting reduction or oxidation by gentle reagents. Strong acids convert it back to soluble arsenates, liberating magnesium as a salt. Classic qualitative analysis for arsenic often relies on its conversion into magnesium arsenate as part of a confirmatory sequence. Some researchers have explored doping magnesium arsenate with rare earth metals, hoping to change luminescence or magnetism, but so far, such efforts remain in niche academic corners. Chemical reactivity remains hemmed in by the stability of the arsenate group, which lends a certain reassurance during storage but limits modifications.
Synonyms & Product Names
A handful of synonyms appear on product lists: magnesium orthoarsenate, magnesioferrite (in specific mineral forms), and in some places, magnesium diarsenate. Pharmaceutical or pure analytical suppliers sometimes tag the product with “ACS Reagent Grade” or “Lab Use Only” notation. Trade names rarely stray far from the straightforward chemical naming system, minimizing confusion during procurement. In older literature, you might see “arsenic magnesium salt,” but that term lacks the precision required today.
Safety & Operational Standards
Arsenic-containing compounds always set off alarm bells, and magnesium arsenate deserves the same high level of respect. Direct skin or respiratory tract exposure leads to arsenic poisoning. Gloves, safety goggles, and dust masks or respirators stay mandatory in handling areas, with clear spill protocols written on every lab wall. Waste disposal channels arsenic residues through licensed contractors, who either immobilize or safely incinerate the waste under controlled conditions. Policies across the United States and Europe align closely, reflecting the universal risks associated with arsenic chemistry. Every staff member working with magnesium arsenate receives regular hazard training.
Application Area
Magnesium arsenate never broke into consumer markets, nor did it enter agriculture at scale, thanks to environmental awareness about arsenic. Its uses persist in analytical and geochemical laboratories, where it serves as a primary standard or reference material in certain arsenate analyses. Environmental scientists have studied it to understand the fate of arsenic in soils and mine drainage, using artificial magnesium arsenate to calibrate their field equipment or simulate geochemical transformations. Some mineral collectors prize well-formed magnesium arsenate crystals for their beauty and rarity. A smaller group of researchers investigates them as potential templates for inorganic synthesis, where the steric and charge properties of the arsenate group help control crystal growth.
Research & Development
Most recent research explores magnesium arsenate's behavior in natural and contaminated environments. Geochemists look for traces in aquifers, trying to predict whether arsenic will leach into drinking water supplies. Some environmental studies test the use of magnesium salts to bind arsenic in mining waste, reducing bioavailability under field conditions. Academic labs experiment with hybrid materials based on magnesium arsenate, seeking new electronic or structural behaviors through subtle doping or nanostructuring, but applications remain distant. Ongoing scrutiny by toxicologists contributes to the body of knowledge around arsenic cycling in biota, refining our understanding of long-term risk.
Toxicity Research
Arsenic compounds show acute toxicity even at low doses, and magnesium arsenate’s insolubility only modestly reduces the risk in most routes of exposure. Ingesting or inhaling fragments introduces arsenic ions, wreaking havoc on enzyme systems and causing oxidative stress. Lab animal studies tie arsenic mineral ingestion to cancers, neurological disease, and developmental delays. Innovative toxicology assays now use sensitive biomarkers to trace arsenic bioaccumulation from minerals like magnesium arsenate, pressing regulators to impose even stricter controls over permissible exposure. The consensus among medical researchers remains clear: avoid direct human or animal exposure, and maintain robust barriers and monitoring whenever magnesium arsenate enters a process.
Future Prospects
Tighter regulations on arsenic-containing materials limit growth in major markets, but magnesium arsenate’s role in academic research and environmental technology remains secure for now. Some chemists look toward novel uses in inorganic templates or sorbents for selective ion removal, believing the structural stability could prove useful in niche scenarios. Any future expansion depends on advances in safe handling protocols and breakthroughs in neutralizing arsenic toxicity after use. Responsible stewardship by labs and suppliers, combined with ongoing toxicological research, may open paths for safer utilization in the years ahead. The legacy of past arsenic contamination lingers in public memory, shaping both the opportunities and boundaries for this rare but scientifically important compound.
What is Magnesium Arsenate used for?
Magnesium Arsenate in the Modern World
Magnesium arsenate is no household name, yet it has a few places in science and industry that deserve a closer look. Its chemical formula, Mg3(AsO4)2, tells part of the story: this compound holds both essential and dangerous qualities in a single package. I remember my early days working in a materials lab, the way magnesium compounds always seemed useful, and arsenic-based ones always set off alarms. The lesson: always ask what a compound brings to the table and what risks ride along.
Industrial and Laboratory Use
Magnesium arsenate shows up in laboratories mainly as a reagent. Analytical chemists have used it for years to test for the presence of certain ions and to run experiments that need a reliable, relatively stable arsenate source. The reason? It dissolves in acids and helps create controlled arsenic environments in water samples.
In teaching labs, magnesium arsenate lets students see precipitation reactions that offer real insight into how ions interact. Its use in practical demonstrations sticks with a young chemist, probably because arsenic has always demanded respectful handling and brought in lessons about limits, careful planning, and personal protection.
Toxicity and Environmental Warnings
Any compound containing arsenic raises eyebrows for good reason. Arsenic ranks right up there with mercury as an element the human body doesn’t tolerate well. In magnesium arsenate, the arsenic sits tightly bound to oxygen and magnesium, yet break that bond and the danger surfaces. Long-term exposure, even in industrial settings, links to skin, lung, and other cancers, plus acute poisoning risks.
Agriculture once tried out various arsenate salts for pest control. The downside became clear: these compounds stay in soil and water a long time, and runoff threatens human health for decades. Magnesium arsenate shares in this problematic legacy, so current laws keep it out of fields in places where food grows. The science tells us arsenic lives long in the environment—clean-up costs and health fallout keep mounting.
Modern Use: Shrinking and Specialized
Demand for magnesium arsenate shrank as industries found safer, friendlier chemicals for most tasks. Today its use falls mostly on scientists running controlled tests, historians reconstructing past industrial processes, and health experts looking to trace sources of arsenic in old dumps. For large-scale activity, better choices exist—ones that won’t poison rivers, plants, or people.
A handful of researchers still turn to magnesium arsenate for its unique chemical properties. For example, mineralogists look at it to help understand arsenic’s movement in the earth, especially in groundwater studies. Every so often, environmental labs spot traces of magnesium arsenate as evidence of past contamination, and these findings guide decisions on cleanup and water safety.
Solutions and Safer Futures
Strict oversight protects workers and the public. Labs run regular risk assessments and use sealed environments for tests. Anyone handling magnesium arsenate gets clear instructions, gloves, goggles, and access to wash stations. In my experience, that culture of control around hazardous materials stays essential. Teaching safety by example, with stories of accidents averted by quick action, does more for new workers than a thousand dry warnings.
Better environmental monitoring has become the rule. Testing water around old mine sites gives early warnings. Laws block dumping and track chemical use from start to disposal. Every few years, scientists review which chemicals are still safe to use—and magnesium arsenate faces that scrutiny each time.
What matters: blending a love of chemistry with a respect for the world’s limits. Investigating magnesium arsenate opens deeper conversations about what we make, what we waste, and how we care for each other as we work with the raw stuff of nature.
Is Magnesium Arsenate toxic or hazardous?
The Real Risks Behind the Name
Magnesium Arsenate doesn’t pop up often in daily conversation, but its reputation still carries weight thanks to that word: arsenate. Arsenic has a long, troubled history as a notorious poison. I remember the first time I studied chemical compounds in school, a teacher held up a chart and said, “If you hear arsenate or arsenite, be careful.” My curiosity led me to research, and what I found matches the concern experts express today: Magnesium Arsenate packs a punch that’s less about magnesium and more about arsenic doing damage.
What Science Says About Exposure
Arsenic, whether in organic or inorganic forms, brings toxicity by getting into cells and disrupting how bodies use oxygen. Studies have shown that long-term arsenic contamination in water leads to serious health issues, including skin lesions, cancers, and neurological problems. Magnesium Arsenate, with arsenic at its core, poses similar risks if it ever winds up in your drinking water, on your crops, or in the air you breathe. There’s enough research to confirm arsenic can bioaccumulate in plants and animals, which makes its way onto our plates. Breaking that chain remains incredibly difficult once contamination starts.
Handling and Storage: Not Your Everyday Task
In industrial settings, workers might handle Magnesium Arsenate under strict protocols. Lab safety manuals treat compounds containing arsenic with respect, requiring gloves, fume hoods, and careful waste procedures. My friend who worked in an environmental analysis lab told me about a day they cleaned out long-ignored bottles. Opening a bottle marked “magnesium arsenate” meant setting off a chain of safety checks and double-bagging for disposal. Nobody wants to risk direct exposure or let powder drift into HVAC systems.
The Occupational Safety and Health Administration (OSHA) sets tough limits for arsenic exposure on the job. Chronic exposure doesn’t just risk sickness; it can ruin careers and spur lawsuits. Strict air and wastewater controls help lower those odds, but only if companies follow the rules. Having seen reports on illegal dumps or forgotten stockpiles, the danger feels all too real to ignore.
Environmental Hazards Don't Stay Hidden
Magnesium Arsenate doesn’t break down in the environment simply. Rain can dissolve the compound and wash arsenic into groundwater, where it can travel and concentrate far from its original source. Rice crops in Asia have picked up arsenic from contaminated water—leading to routine food recalls and ongoing monitoring. The lesson hits home: once contamination starts, cleaning it up drains community resources for years to come.
Prevention Works Better Than Treatment
Once a community faces arsenic poisoning, solutions turn into massive infrastructure projects. Reverse osmosis, ion exchange, and specialized filters offer some hope—but none guarantee a quick fix. For those growing food or managing water systems, regular testing stands between safe water and a public health nightmare. Teaching the next generation of chemists, engineers, and local officials about compounds like Magnesium Arsenate matters because prevention saves more lives and dollars than cleanup ever could.
So, Is Magnesium Arsenate toxic or hazardous? Absolutely. If arsenic forms the center of a compound, trust your instincts and practice caution. Where there’s risk, smart handling and tight controls give us a fighting chance to keep workers, families, and towns safe. People deserve nothing less.
What are the handling and storage requirements for Magnesium Arsenate?
An Overlooked Danger on the Shelf
Magnesium arsenate isn’t a household name, but its risks shouldn’t get swept under the rug just because only a few people have seen it. A salt containing arsenic, this compound brings both magnesium, which plays its role in health, and arsenic, which has lived up to its toxic reputation for centuries. If you’ve ever worked in a lab or a warehouse for chemicals, just hearing “arsenate” probably triggers an instinct to step back. That’s a good instinct to have.
Risks That Don’t Cut Corners
Years ago, during my brief stint in a college chemistry stockroom, the storage area for arsenic compounds had a bright red door. We joked about the warning stickers, but honestly, those were the most sensibly labeled chemicals we had. Any compound with arsenic demands real respect. Even in small doses, arsenic exposure adds up, targeting the nervous system, kidneys, and liver. Inhalation or skin contact with dust should get avoided at all costs. Avoiding food and drink in work areas isn’t just an old rule for its own sake—it’s the difference between a safe shift and one that ends at the ER.
Recent reviews from agencies like the CDC and OSHA keep hammering home the value of personal protective equipment. Gloves, lab coats, and especially dust masks play a big part in day-to-day handling. In one lab exercise, a student once brushed arsenic-containing powder onto their hands. Even though it was just a speck, the whole class had to stop and go through a decontamination drill. Simple chemical respect shows up in everything from careful weighing to double-bagging leftovers—measures meant to keep any loose powder from going airborne.
Sensible Storage that Works
Safe storage doesn’t mean locking things in a box and walking away. Magnesium arsenate belongs in a rock-solid, sealed container. No cloudy, cracked jars from the back of the shelf. Keep it away from any acids or reducing agents. A spill can turn routine cleanup into a toxic mess fast. I remember one case where an old cap failed, releasing white dust onto shelves and forcing us to replace a good chunk of that stockroom cabinetry. That time, the label said everything you needed to know: keep it cool, keep it dry, and never let the container sit open any longer than needed.
Most stockrooms store these kinds of chemicals far from direct sunlight and far from high-traffic hallways. Regular checks—by actual people, not just spreadsheets—matter more than fancy facility upgrades. If containers look damaged, it’s time to replace them, not just tape them up. Teams who oversee these stores often set up inventory checks to track quantities, matching usage with ordering, and that routine can save lives—tracking down any vanished grams before they become a news story.
What It All Adds Up To
Talking about magnesium arsenate means talking about prevention, not just procedure. I always remind myself: the point isn’t to fear the compounds, but to put safe habits before convenience. Put gloves on before unsealing; shut doors and lids right after finishing a step. Teaching new staff the “why” behind these habits sticks better than drilling them on policy. Respect for risk doesn’t mean paralyzing fear—it means every person takes responsibility, with the right knowledge. Good science demands it.
What is the molecular formula and structure of Magnesium Arsenate?
Understanding Magnesium Arsenate in Everyday Terms
Magnesium arsenate isn’t a common name on household cleaning products, but its chemical makeup matters for more people than just those in a lab coat. With so many headlines circling about chemical safety and environmental health, it becomes clear why learning the basics behind compounds like this isn’t just something left to textbooks.
The Building Blocks: Identifying the Formula
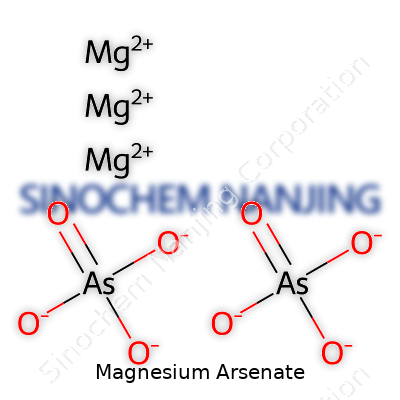
At its core, magnesium arsenate’s molecular formula sits as Mg3(AsO4)2. This means three magnesium (Mg) ions join forces with two arsenate (AsO4) groups. Each arsenate group carries a negative charge, balanced out by the positive charge of magnesium ions. Seeing these numbers on paper helps us recognize the ratio of different atoms inside this compound, something that shapes its chemical behavior.
Visualizing the Structure
Explaining the structure of magnesium arsenate calls for a closer look at how atoms connect and create form. Arsenate itself features one arsenic atom bound tightly to four oxygen atoms, creating a tetrahedral shape. Think of a small pyramid, with arsenic at the center and oxygen atoms making up the base corners. Two of these arsenate groups pair with three magnesium ions, which each act a bit like bridges, linking up with the negative charges of the oxygen atoms. All in all, it creates a solid crystal lattice if you peer close enough with the right tools. This stable arrangement keeps the whole compound from falling apart under normal conditions—one of the main reasons it’s found in mineral forms rather than as loose powder in nature.
Why Formula and Structure Matter Both In and Out of a Lab
Paying attention to magnesium arsenate’s formula and structure isn’t just splitting hairs for chemists. Many natural mineral deposits contain this compound, and its makeup influences water hardness, soil quality, and even the durability of certain rocks that get used in construction or landscaping. On the downside, arsenate-based compounds can pose environmental risks. Arsenic’s history is crowded with health warnings, from groundwater contamination to crop safety. Knowing how magnesium bonds with arsenate helps scientists design better filtration systems or safer disposal methods, and it gives community leaders more information to trace possible sources of contamination if something shows up during a local water test.
Real-Life Solutions and Responsible Chemistry
Experience with water testing back home during a well inspection opened my eyes to the importance of knowing which compounds move through our ground and waterways. Our local lab flagged magnesium arsenate levels in a report, and understanding its structure made it easier for community members to talk with public health officials. Instead of just panicking at the mention of “arsenic,” knowing how it connects with magnesium in this context helped shape smarter questions about risks and action steps.
More and more, universities and research labs turn to techniques like X-ray diffraction and spectroscopy to map structures and keep track of trace minerals in the environment. Clear formulas and tangible structures give those methods a starting point, add reliability to environmental testing, and support efforts to minimize harmful exposure. If communities push for transparency around mineral testing and ask about the underlying chemistry, more focused solutions come into play. Options like adding iron to bind arsenate, or using advanced membranes, emerge from a deep understanding of how compounds like magnesium arsenate come together.
Basic knowledge of molecular formula and structure lets families, schools, and policymakers keep an eye on both risks and benefits—and drive safer, better-informed decisions about what’s in our water, soil, and everyday lives.
How should Magnesium Arsenate be disposed of safely?
Reality of Magnesium Arsenate Waste
Magnesium arsenate, used in industrial chemistry and sometimes as a laboratory reagent, brings serious risks because arsenic is a notorious poison. Stories about arsenic winding its way into drinking water haven’t faded for a reason. In my own childhood, living near a mining town, warnings about arsenic in dust or wells felt commonplace. If a chemical laced with arsenic goes down the drain or landfill without care, that legacy only worsens.
Not for the Local Landfill
Tossing magnesium arsenate in with regular trash makes little sense; doing so risks contaminating groundwater and soil. U.S. Environmental Protection Agency (EPA) reports link arsenic to cancer, heart trouble, and nerve damage. Safe levels rank in the parts per billion, nothing more, and it takes only a small amount to tip the scales. Those working in laboratories or industries who handle this compound rarely get a casual attitude from regulators — and with good reason. Even trace contamination matters.
Direct Disposal: Time for a Rethink
People sometimes believe that neutralizing a chemical before disposal solves the issue. In this case, it doesn’t. Arsenate ions stay toxic even after mixing with plenty of reagents. Pouring waste down the sink, even after dilution, puts treatment plants under stress because municipal systems rarely destroy such harmful metals. Local water authorities know this. In fact, many communities now track chemical signatures and take action when they detect unexpected spikes.
Turning to Professionals
Turning waste like magnesium arsenate over to licensed hazardous waste companies gives the best protection. These outfits specialize in identifying, packaging, and transporting chemicals using rules set by agencies like the EPA and OSHA. Shipping even small bottles of this material means paperwork and secure containers, and drivers carrying hazardous waste certificates. I once watched a hazmat crew pack up lab leftovers, and the protocol stuck with me. Personal protective equipment, spill trays, labels, waste tracking logs — every step matters.
High-Temperature Incineration: A Gold Standard
Incineration at specialized facilities, using temperatures high enough to break down arsenic compounds, becomes the next step. Incinerators designed for hazardous waste don’t resemble ordinary garbage burners. They have strict controls and advanced air scrubbers that capture fumes and trap any escaping particles. Ash collected at the end gets treated as hazardous too, and never meets open landfill. The EPA lists these sites online, keeping the public aware of disposal chains.
The Human Factor: Training and Awareness
Even so, technology only does so much if people cut corners. Drug labs and illegal dump sites cause plenty of the arsenic trouble communities face. Training, clear labeling, and rigid procedures change outcomes. In my own experience, the safest labs don’t just rely on written plans; they practice what to do if something spills or leaks. Success builds on habits, not just rules.
Community Solutions
Communities near factories and labs benefit from local hazardous waste days, offering a way for households or small businesses to hand in dangerous chemicals for safe handling. This sort of program matters. Regulators, businesses, educators, and environmental groups working together form a stronger shield than any one group alone. In the end, real stewardship over magnesium arsenate, or any poison, rests with people choosing care over convenience every single time.
| Names | |
| Preferred IUPAC name | magnesium arsenate |
| Other names |
Trimagnesium arsenate Magnesium(II) arsenate |
| Pronunciation | /maɡˈniːziəm ˈɑː.sə.neɪt/ |
| Identifiers | |
| CAS Number | [10103-58-9] |
| Beilstein Reference | 77434 |
| ChEBI | CHEBI:131379 |
| ChEMBL | CHEMBL1201564 |
| ChemSpider | 20640971 |
| DrugBank | DB14626 |
| ECHA InfoCard | 100.029.035 |
| EC Number | 234-722-8 |
| Gmelin Reference | 77955 |
| KEGG | C15965 |
| MeSH | D015808 |
| PubChem CID | 24563 |
| RTECS number | OM3850000 |
| UNII | 4H5Q2W6P6B |
| UN number | UN1477 |
| Properties | |
| Chemical formula | Mg₃(AsO₄)₂ |
| Molar mass | 342.11 g/mol |
| Appearance | White powder |
| Odor | Odorless |
| Density | 3.52 g/cm3 |
| Solubility in water | Insoluble |
| log P | -0.434 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 12.1 |
| Basicity (pKb) | 2.89 |
| Magnetic susceptibility (χ) | -23.0e-6 cm³/mol |
| Refractive index (nD) | 1.672 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 167.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -2510 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2347.2 kJ/mol |
| Pharmacology | |
| ATC code | A12CC06 |
| Hazards | |
| Main hazards | Toxic if swallowed, suspected of causing cancer, causes damage to organs through prolonged or repeated exposure. |
| GHS labelling | GHS07, GHS08, GHS09 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. |
| Precautionary statements | P261, P264, P270, P271, P272, P301+P312, P301+P330+P331, P302+P352, P304+P340, P308+P313, P314, P405, P501 |
| NFPA 704 (fire diamond) | 2-0-0 |
| Lethal dose or concentration | LD₅₀ oral rat: 2 mg/kg |
| LD50 (median dose) | LD50 (median dose): 2.6 mg/kg (intravenous, rabbit) |
| PEL (Permissible) | PEL: TWA 0.01 mg(As)/m³ |
| REL (Recommended) | 0.002 mg As/kg bw |
| IDLH (Immediate danger) | 15 mg As/m³ |
| Related compounds | |
| Related compounds |
Magnesium phosphate Magnesium sulfate Magnesium arsenite Sodium arsenate Calcium arsenate |

