M-Phenylenediamine Sulfate: A Closer Look at a Vital Industrial Chemical
Historical Roots and Development
Chemistry has stories that stick with you, especially in the world of industrial compounds. M-Phenylenediamine sulfate became part of this history in the late nineteenth and early twentieth century, when demand for synthetic dyes and polymer-building blocks led scientists to work with aromatic amines. During my research years, textbooks described the early isolation and first use of m-phenylenediamine, where it stood out as a key intermediate to replace extracted or rare natural molecules. Industrial expansion in the last century, especially in polymer and dye production, transformed this compound from a mere organic curiosity to a workhorse. Unlike its para or ortho siblings, the “meta” arrangement gives its chemistry a special angle, opening routes for unique product properties and applications.
Physical & Chemical Properties with Day-to-Day Relevance
Looking at a vial of m-phenylenediamine sulfate, you typically find a white to pale tan crystalline solid, a bit like gritty table salt. It dissolves easily in water, which comes in handy for industrial batches. I always found its modest melting point notable, as it lets you handle it under moderate conditions. The compound’s aromatic ring delivers stability but the two amine groups stick reactive “handles” in prime meta spots, fueling further reactions. Its sulfate salt form tames the free amine, lowering volatility and improving shelf life. You might think these are dry facts, but I remember more than one lab technician thankful for its predictable fusion point and strong solubility, which prevent processing headaches during dye manufacture or pharmaceutical synthesis.
Technical Labelling and Real Policy
Working in a regulated lab, the way we label and track m-phenylenediamine sulfate has much to do with its hazards. You could tell safety policies matured as understanding of amine toxicity developed. Product labels have to identify the full chemical name, hazard pictograms, batch numbers, purity levels, and supplier’s traceable signature. In practice, proper labeling isn’t just a legal step—colleagues double-check labels to avoid cross-contamination or accidental dosing in batch processes. I’ve seen confusion about synonyms—some workers say “meta-phenylenediamine salt,” others use the systematic name—so it is crucial to train each new staff member while keeping old-timers up-to-date, putting the spotlight squarely on clarity and operational safety.
How M-Phenylenediamine Sulfate Is Prepared
If you ask a chemist about its preparation, the route usually starts by catalytically reducing m-dinitrobenzene, followed by adding sulfuric acid to form the sulfate salt. I remember industrial-scale synthesis brings special challenges—catalyst recovery, waste handling, energy balance for continuous operation. Skills come into play running exothermic reductions safely, then managing acid-base steps that produce a solid as pure as possible, minimizing impurities that interfere downstream. Years in R&D taught me: quality at this stage saves headaches later, whether making dyes or specialty polymers. It’s easy to overlook how each tweak—solvent choice, temperature, reaction order—changes yield and safety margins.
Reactivity and Chemical Modifications
The structure of m-phenylenediamine sulfate lets chemists branch off toward multiple value-added products. Those two amino groups, separated by a carbon ring, give a springboard for crosslinking reactions in curing agents, antioxidants, and epoxy resins. I’ve seen first-hand how controlled reaction conditions decide between polymer extension or, in the right hands, high-performance pigments. When venturing into novelty polyamides or azo dyes, the meta disposition delivers properties not seen in ortho or para analogues—changing polymers’ flexibility, toughness, or dye lightfastness. The sulfate salt adds practical value, acting as a handy delivery form in water-based synthetic routes, a trait anyone in continuous flow production can appreciate.
Recognizing Familiar Synonyms
Names can throw you off. Beyond “m-phenylenediamine sulfate,” you bump into designations like “1,3-benzenediamine sulfate,” “meta-diaminobenzene sulfate,” and in older research, shorter labels from old German and French dye patents. The proliferation of synonyms is more than a trivia point—it’s a reminder to keep communication crisp across regulation, purchasing, and research, especially for multinational teams and documentation. Overlooking this fact has tripped up colleagues more than once ordering or reviewing technical literature. Scrupulous cross-referencing of Chemical Abstracts numbers or detailed naming conventions always saves time for everyone in the lab.
Operational Safety: Not Just a Checklist
Handling aromatic diamines needs patience and serious attention. M-Phenylenediamine sulfate irritates skin, eyes, and especially the respiratory tract. In the lab, I’ve seen gloves, goggles, and strong ventilation solve many problems before they start. Large-scale operators add dust extraction, thorough training, and real-time monitoring. Not everyone appreciates the lingering toxicity risk—chronically exposed workers can experience effects on blood and internal organs. Repeated risk assessments, exposure controls, and consistent environmental monitoring matter as much as the raw safety data sheets. Over time, most companies also build in periodic blood tests for operators and stress the need for airtight process design to minimize even small leaks, especially in long campaigns or shiftwork.
Wide-Ranging Application Areas
At first glance, you might peg m-phenylenediamine sulfate as a niche specialty, but its uses stretch across industries. The most well-known spot is as a building block in manufacturing dyes and pigments—especially hair dyes and textile colorants, where molecular stability and color yield are prized. I watched in a dye house how product quality relied on this chemical’s purity, batch after batch. Moving past fashion, it supports high-performance engineering polymers, such as aramids found in personal protective equipment and advanced filtration. During my time visiting water treatment plants, I saw it included in corrosion inhibitors, adding real-world value in infrastructure. It also pops up in analytical chemistry as a reagent, and at times in pharmaceuticals, though there toxicologists keep a close watch on trace carryover.
The Ongoing Story in Research and Development
Labs continue to explore how to push m-phenylenediamine sulfate beyond tradition. In my own involvement with polymer projects, every marginal gain in process efficiency, thermal stability, or color depth feels like a win, whether in new fiber for high-speed rail or more resilient coatings for urban environments. Teams now use simulation to fine-tune each molecular tweak, looking for higher reactivity or greener processing conditions. There’s attention on byproduct reduction and recycling—pushes from both regulation and internal sustainability standards. Patents keep popping up as new applications, like electronics packaging or specialty adhesive films, demand even tighter control over molecular purity and reactivity. Watching young researchers balance environmental benefit with cost reminded me: long-term R&D calls for patience, grit, and cross-disciplinary partnership more than ever.
Unpacking Toxicity and Worker Health
Years ago, early safety notes on aromatic amines were dry and routine, but later case studies changed minds. Toxicity studies underline that m-phenylenediamine sulfate is more than a handling hazard: chronic, low-level exposure can cause methemoglobinemia, hemolytic anemia, and sometimes impacts on liver and kidney function. Those effects show up in exposed populations: textile or dye workers in poorly ventilated shops, or staff skipping personal protective gear, see health changes over months or years. Good industrial hygiene, modern process containment, and worker education help break that chain. In research, new focus drives deeper molecular-level studies, looking at genome and protein interactions for more proactive prevention. Industry and academia together can drive stricter, evidence-based exposure limits—and robust health surveillance—because each lesson from past harm ought to shape the rules for all future use.
Where Next? Future Prospects and Paths Forward
What stands out in the future of m-phenylenediamine sulfate is less about bigger volumes, more about responsible innovation. I see focus shifting from just higher output toward safer, greener production—new catalysis, closed-loop recovery, even fully bio-based routes. Regulations won’t ease up, and neither will demands from end users. To stay competitive, companies team with academic labs and digital platforms to monitor exposures, develop safer analogues, and reduce both waste and emissions. If development continues, it will be on the back of stronger data—predictive toxicology, AI-driven pathway optimization, and collaborative research. The story for this compound isn’t finished and, as any scientist with a few decades under their belt knows, the future of chemistry depends as much on what we refuse to ignore as what we chase as new opportunity.
What are the main uses of M-Phenylenediamine Sulfate?
Dye Manufacturing and Hair Colorants
Factories producing dyes rely on m-phenylenediamine sulfate as a core building block. Walk into any salon, and you’re likely to find its influence in permanent hair dye formulas. This compound helps create deep blacks and rich browns, shades that stick around without washing out after a day or two. Years ago, hair coloring felt like a risky gamble—fading, allergic reactions, sometimes even toxic exposures. As research around this chemical advanced, companies started adjusting formulas to make products safer. With evidence from repeated studies, regulatory agencies set strict usage guidelines, aiming to reduce risks while preserving the staying power customers demanded. The result is a hair dye industry still subject to scrutiny, but now much better equipped to manage possible side effects.
Polymer and Resin Production
Many engineers and chemists fit m-phenylenediamine sulfate into polymer chains for specialty plastics and resins. These materials show up in lightweight protective coatings, digital device parts, and even the finish on your favorite headphones. In my early career working with electronics production, I saw firsthand how the right chemical additive could mean the difference between a brittle phone casing and a durable one. In today’s market, where gadgets have to look slick and survive drops, companies look for additives that improve resilience without adding unwanted weight. Consistent supply and rigorous testing have become standard practice, driven by lessons from failures where inferior substitutions led to costly product recalls.
Photography
Though digital cameras have pushed film photography into niche territory, there’s still a loyal crowd keeping traditional film alive. Developers based on m-phenylenediamine sulfate help bring images to life with sharper contrasts, giving enthusiasts the look they crave. In university, I worked in a darkroom and watched how tiny tweaks in developer formulas could change the mood of a photograph. For those of us raised on film, the older processes feel almost magical, and chemistry like this continues to anchor a devoted hobbyist community. Experts tracking chemical safety highlight that these uses rely on careful handling, precise dosing, and good ventilation, to keep risks low while preserving tradition.
Research and Laboratory Chemicals
Academic and industrial labs keep m-phenylenediamine sulfate in stock for a variety of chemical reactions, especially where high-purity intermediates are needed. Organic synthesis is a game of tradeoffs and patience, and reliable reagents save costly reruns. In my limited but enlightening forays into lab work, accuracy translated to real time and money saved. The competition for reproducibility in published research means chemicals like this one are sourced from trusted suppliers, with clear documentation about toxicity, shelf life, and storage conditions. This rigorous approach ties directly to the principle of trustworthiness championed in quality research.
Concerns and Safer Solutions
Public health advocates and environmental groups continue to press for less hazardous alternatives, especially for substances with links to skin sensitization or more serious health effects. M-phenylenediamine sulfate falls under scrutiny because—when mishandled or overused—it can cause skin allergies and other adverse reactions. Some manufacturers are already investing in green chemistry, aiming to swap out ingredients that pose higher risks. Collaborative data sharing between chemical producers, regulators, and academic teams helps drive informed choices, so that innovation keeps pace with growing health and safety expectations.
Final Thoughts
M-phenylenediamine sulfate is no relic of the past. Its practical uses touch daily life through products many people overlook. At the same time, growing information about its risks means ongoing evaluation is more important than ever. The healthiest approach calls for clear labeling, protective guidelines, and a willingness to update old habits as better solutions emerge.
Is M-Phenylenediamine Sulfate hazardous to health?
What You’re Handling in Everyday Life
People working with chemicals—especially those dealing with dyes, pigments, or photographic developers—might recognize the name, M-Phenylenediamine Sulfate. This compound turns up in places from textile factories to certain hair dye formulas. I remember touring a textile plant where workers wore double gloves and kept a running fan at their workstations. Hearing chatter about headaches and skin irritation helped me realize these warnings in safety sheets aren’t just technical—real people feel these effects.
Why Exposure Worries Health Experts
Contact with M-Phenylenediamine Sulfate seldom goes unnoticed. Red skin, watery eyes, and breathing trouble crop up pretty quickly if you get exposed without proper protection. Studies link it to allergic reactions that last long after a shift ends. The risk of chronic problems goes up where proper ventilation is missing, or when people skip gloves and masks, either because they forget or because gear is too uncomfortable to wear all day. Occupational Safety and Health Administration (OSHA) reports describe cases where routine handling led to rashes and, for some, lasting respiratory sensitivity.
Digging into the Science
Researchers keep confirming that frequent contact leads to skin sensitization, evident in people who work in dye manufacturing or handle hair colors containing this ingredient. The Agency for Toxic Substances and Disease Registry (ATSDR) points out that even low doses inhaled or absorbed through the skin trigger immune responses. Reports of acute poisoning exist, too—usually among people caught off guard by leaks or accidental spills. Test data shows toxic reactions in animals as well, which makes industry watchdogs cautious about setting safe exposure limits.
What Actually Protects Workers
Basic gear—good nitrile gloves, snug goggles, fitted masks—works, but these only go so far without backup from management. I learned this while helping to write a safety manual: guidelines are only the first line of defense; daily habits and a workplace culture that values safety matter more. Regular training, well-labelled containers, and prompt cleanup of spills cut risks way down. It helps when companies invest in mechanical ventilation or air extraction systems rather than relying on open windows or fans alone, especially in older buildings.
Regulations on the Books
Governments don’t leave everything to chance. OSHA limits exposure levels, and companies must list M-Phenylenediamine Sulfate on their hazardous material sheets. The European Chemicals Agency lists it as a substance of concern, calling for tough workplace assessments. Rules like these forced companies I’ve worked with to upgrade their safety practices—not just because of the threat of penalties, but because retaining workers gets tough if people keep getting sick on the job.
Safer Substitutes and Smart Choices
Some industries moved away from using it, switching to safer alternatives. The shift isn’t always easy. Replacing a chemical in a process takes real coordination, and sometimes product quality suffers. Yet, bigger companies work with chemical suppliers to test and adopt gentler formulations. For small businesses, options include limiting tasks that involve direct contact and providing rotating assignments, spreading out the risk instead of letting one group bear the brunt.
What Individuals Can Do
If you work with M-Phenylenediamine Sulfate, read the safety sheets, but don’t stop there. Speak up if ventilation looks poor or protective gear is missing. Shortcuts on safety today cause real health problems tomorrow. Workers with existing allergies or asthma should push for extra precautions and talk to supervisors about alternatives or substitutions. Your health matters more than finishing a shift a bit faster.
What safety precautions should be taken when handling M-Phenylenediamine Sulfate?
Why Handling This Chemical Needs Respect
M-Phenylenediamine sulfate doesn’t get headlines like some bigger industrial hazards, but that doesn't mean it's safe to shrug off. From years working in labs and on job sites, I‘ve seen firsthand what cutting corners with chemicals can lead to—one splash, one missed glove, and you’re in for a rough time. M-Phenylenediamine sulfate serves as a dye intermediate and crops up in research, but it brings risk every time it’s out of the cabinet. Its toxic properties don’t always show right away, so respecting safety protocols isn’t about being dramatic—it’s about staying healthy enough to get back home after work.
Recognizing the Hazards
This compound can eat away at skin and turn simple mistakes into expensive or painful lessons. Skin exposure sometimes slips under the radar—maybe a splash lands unnoticed or a glove tears without warning. Eyes take the brunt if vapors escape or powders puff out, and breathing dust or mist is trouble. Some workers ignore the warnings, thinking a little irritation is just par for the course, but allergies, rashes, and breathing troubles can hit long after the shift ends.
Smart Precautions That Actually Matter
Safety gear isn’t for show. Nitrile gloves work well, changing them at the first tear, even if it feels wasteful. Face shields and splash goggles turn a day ruined by a stray droplet into just another routine cleanup. I stick with lab coats or long sleeves, not because spills are likely, but because tired arms and distracted minds get careless. Most chemical burns I’ve seen start small—just a tiny drop. Chemical-resistant aprons help when mixing larger batches or pouring from heavy bottles. Closed-toe shoes keep feet out of harm‘s way, and it’s best to nix open footwear entirely.
Air Quality and Ventilation
Good labs or workshops don’t just have a fan whirring in the corner. Local exhaust systems and properly maintained fume hoods pull vapors away before anyone gets a lungful. Even small-scale weighing is best inside a hood. Well-kept filters shouldn’t get skipped over on maintenance days. I’ve watched coworkers skip these steps in the name of “efficiency,” only to cough their way home—there’s nothing efficient about that.
Storage and Spills
Dry, cool shelves with secondary containment trays make life easier if a bottle tips or leaks. Keeping everything labeled keeps others safe, too, not just yourself. Mismatched or faded labels delay emergency response. During cleanup, absorbent pads and neutralizing agents should never run low. I check emergency eyewash stations every week, because cleaning up after an accident is tough enough without realizing the eyewash hasn’t worked since last winter.
Training and Team Culture
No training session covers every weird thing that can go wrong, but consistent refreshers help. Involving the whole team, talking through slip-ups without shaming anyone, and sharing real stories about close calls builds trust. I‘ve seen new employees learn more from five minutes of honest talk than from an hourlong lecture.
Final Thoughts
Avoiding shortcuts and taking every exposure seriously sends a clear message—your health matters more than any deadline. M-Phenylenediamine sulfate doesn’t forgive mistakes. By looking after each other and tending to the basics, nobody has to take unnecessary chances, and everyone gets to clock out in one piece.
How should M-Phenylenediamine Sulfate be stored?
Understanding M-Phenylenediamine Sulfate’s Risks
M-Phenylenediamine sulfate pops up across labs, workshops, and some industrial settings. Its uses stretch from dye production to polymer research. People familiar with this chemical know just handling it in an open room without controls would be inviting trouble. It gives off dust that can irritate lungs and skin contact can trigger nasty allergic reactions. Sulfate salts tend to take in moisture from the air, so leaving a jar open is asking for caking, clumping, or contamination. The trick is learning how to keep people safe while making sure quality doesn’t degrade.
What an Experienced Handler Pays Attention To
Decades of chemistry work have taught me one thing: Respect for chemicals keeps disaster away. The container really counts — it should seal tight and resist corrosion. I’ve seen labs skimp on this, repurposing jars or bottles, thinking a little moisture won’t hurt. Truth is, even short exposure in a high-humidity shop or under a leaky ceiling leads to spoiled product and more risk.
Direct sunlight changes how quickly some chemicals break down, and fluorescent light can build up heat on a shelf. For M-Phenylenediamine sulfate, the best spot stays cool and dry. My team often cleared space on lower shelves, away from vents or heaters, and double-checked storage closets for dampness.
Labeling and Segregation Go Hand in Hand
Color-coded labeling isn’t busywork. Anyone who’s faced a spill or cross-contamination event knows the headache it brings. I saw an intern reorganize shelves and set peroxide-forming agents next to amines. That almost set off an evacuation. M-Phenylenediamine sulfate deserves its own spot, away from oxidizers and especially from food or personal items. Store it low, away from your line of sight, and elevate acids or oxidizers far from any amine compounds.
Why Real Ventilation Matters
Relying on old air systems leads to dusty air and smell that lingers. Fresh air exchange helps, yes, but modern lab hoods work even better. At home or in a smaller space, I’ve seen people try storing compounds inside regular kitchen cabinets. This is a recipe for disaster. Always use well-ventilated, grounded cabinets built for chemicals, even if the cost feels high.
Sensible Protection for Staff and Cleanup
Restocking day should never feel ordinary. Gloves, goggles, and lab coats matter every time. I keep spill kits close by, with instructions posted right above the storage area. Absorbent materials, neutralizers, and sealable waste containers keep accidents limited and cleanup swift. Assigning responsibility to a specific staff member for regular inventory checks has cut down on forgotten or expired chemicals.
Solutions for Safer Storage
Automatic reminders and electronic logs helped my team keep track of container ages and conditions. It’s easy to forget when the day runs long, but digital tools flag containers before seals degrade. Encourage everyone to report even the smallest issues, like cracked lids or labels washed out from a spill.
Training means more than reading a safety sheet once a year. Real drills—walking through what to do and who to call—make theory stick. Encourage a culture where questions flow freely and reporting hazards is rewarded, not penalized.
The bottom line rests on treating every chemical with healthy respect, storing it right the first time, and revisiting protocol as new info comes to light. From new chemistry grads to seasoned lab techs, everybody should have clear access to storage standards and easy ways to do the right thing.
What is the chemical structure and molecular weight of M-Phenylenediamine Sulfate?
The Structure Behind the Name
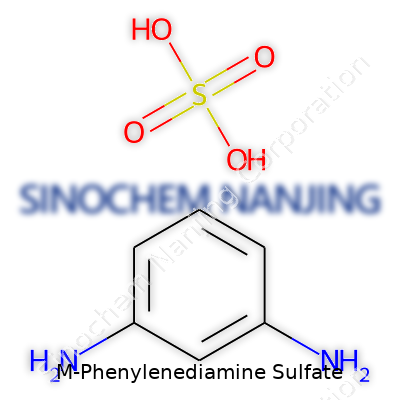
M-Phenylenediamine sulfate carries a structure that anyone working with chemical dyes or analytical labs might recognize right away. The base skeleton of this compound starts with a benzene ring. Attached to this ring, you find two amine groups, sitting at the meta positions. This simply means there’s a nitrogen group on carbon number one, and the other shows up on carbon number three if you start counting around the ring. These two amines—NH₂ groups—don’t just add some complexity; they define how the molecule behaves and what chemists can do with it.
Getting Specific: The Sulfate Aspect
Now take that core, and imagine pairing it with sulfuric acid. The sulfate salt forms when m-phenylenediamine (sometimes called 1,3-phenylenediamine) meets sulfuric acid. In this salt, two positively charged m-phenylenediamine molecules balance out a single negatively charged sulfate (SO₄²⁻) anion. The result is a white to off-white crystalline solid—a form that stores better, dissolves easier in water, and keeps less risk of quick breakdown than the raw diamine does.
Chemical Formula and Molecular Weight
Break out the periodic table for a second. The chemical formula for the sulfate salt is C₆H₈N₂·H₂SO₄, or more precisely, (C₆H₈N₂)₂·H₂SO₄, depending on exact hydration and how the salt is isolated. Most commonly in laboratory catalogs, the formula appears as for the dihydrochloride, but with sulfate it balances two cation units to one sulfate. The molecular weight (molar mass) of M-Phenylenediamine sulfate builds up from the atomic masses: two benzene-diamine molecules and one whole sulfate add up to 254.28 g/mol. This information means more in practice than it might seem—to calculate dosages, to prep solutions, or to predict results in an experimental setup, knowing the precise molecular weight saves a lot of time and error.
Real World Relevance—Why Chemists and Industry Pay Attention
This stuff finds its way into labs all over, thanks to its role as a key intermediate in dyestuff production and hair coloring formulations. The m-phenylenediamine backbone becomes a building block for synthesizing other more complex molecules—both in research and in commercial manufacturing. The sulfate form gives makers a stable, safer way to ship and handle this otherwise tricky chemical. In hair dyes, it helps deliver permanent, vivid color. Scientific texts report clear links between exposure and certain allergies, pushing manufacturers to tackle safer alternatives or tighter controls for handling.
Handling Health and Safety
For anyone working hands-on with m-phenylenediamine sulfate, the story doesn’t end at its molecular map. This chemical, like many amine derivatives and dye precursors, deserves some respect in the lab or facility. Contact can trigger skin and eye issues, and people with long-term exposure sometimes end up dealing with more serious reactions. Regulators set hazard classifications for a reason, and labs need firm protocols—proper gloves, splash goggles, and rigorous ventilation at a minimum. Cleanup and waste disposal need care, because improper handling can mean environmental harm and workplace risk.
Better Practices and Safer Alternatives
In my own experience, switching to the sulfate salt over the plain free base brought noticeable improvements in shelf stability and handling safety. Doing regular risk checks and updating training for all staff exposed to aromatic amines made a real difference in worker safety records. For teams exploring greener and safer chemistry, every incremental tweak—from using the salt form to trying new dye intermediates—contributes to progress. Keeping an eye on innovation and new research not only meets current safety standards, but often uncovers efficient ways to get better end results. The field keeps moving, and good chemistry never stands still.
| Names | |
| Preferred IUPAC name | benzenediaminium sulfate |
| Other names |
1,3-Benzenediamine sulfate m-Phenylenediamine sulfate Resorcinol diamine sulfate |
| Pronunciation | /ɛm fɛˌnaɪliːnˈdiəˌmiːn ˈsʌl.feɪt/ |
| Identifiers | |
| CAS Number | 531-85-1 |
| Beilstein Reference | 2109957 |
| ChEBI | CHEBI:86357 |
| ChEMBL | CHEMBL452458 |
| ChemSpider | 22220 |
| DrugBank | DB11378 |
| ECHA InfoCard | 100.032.310 |
| EC Number | 202-457-1 |
| Gmelin Reference | 77297 |
| KEGG | C01761 |
| MeSH | D017364 |
| PubChem CID | 24139 |
| RTECS number | SS9625000 |
| UNII | 6Q55K1I6E2 |
| UN number | UN2811 |
| Properties | |
| Chemical formula | C6H10N2O4S |
| Molar mass | 286.35 g/mol |
| Appearance | Gray white to brown crystalline powder |
| Odor | ammonia-like |
| Density | 1.38 g/cm3 |
| Solubility in water | soluble |
| log P | -1.3 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 4.66 |
| Basicity (pKb) | pKb 4.2 |
| Magnetic susceptibility (χ) | -11.6×10^-6 cm³/mol |
| Refractive index (nD) | 1.642 |
| Viscosity | 400 cP (20°C) |
| Dipole moment | 5.99 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 211.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -277.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3231 kJ/mol |
| Pharmacology | |
| ATC code | D08AX |
| Hazards | |
| Main hazards | Harmful if swallowed, in contact with skin or if inhaled. Causes severe skin burns and eye damage. May cause an allergic skin reaction. Suspected of causing genetic defects. Toxic to aquatic life. |
| GHS labelling | GHS05, GHS06, GHS08 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | Harmful if swallowed. Toxic in contact with skin. Causes severe skin burns and eye damage. May cause an allergic skin reaction. Suspected of causing genetic defects. May cause cancer. Toxic to aquatic life with long lasting effects. |
| Precautionary statements | P261, P280, P302+P352, P304+P340, P305+P351+P338, P310, P362+P364 |
| NFPA 704 (fire diamond) | 2-3-1 |
| Flash point | > 195 °C |
| Lethal dose or concentration | LD50 (oral, rat): 175 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50: 298 mg/kg |
| NIOSH | MP2875000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of M-Phenylenediamine Sulfate is "0.1 mg/m3 |
| REL (Recommended) | 0.1 mg/m3 |
| Related compounds | |
| Related compounds |
p-Phenylenediamine o-Phenylenediamine Aniline Sulfanilic acid |

