M-Phenylenediamine Hydrochloride: A Close Look at Its Story, Features, and the Road Ahead
Historical Development
Roots of m-Phenylenediamine hydrochloride, sometimes known in the lab as meta-phenylenediamine hydrochloride or 1,3-benzenediamine hydrochloride, go way back to the early breakthroughs in synthetic dyes. The late nineteenth century saw chemists chasing new methods to modify aromatic rings. Curiosity around benzene rings, joined with the search for new coloration techniques and reactive intermediates, brought scientists to derivatives like this diamine. What stands out is how technology and industry shaped the availability, guiding its role from small-batch research chemical to larger-scale manufacture. Lab notebooks full of purple, brown, and yellow stains bear testament to hands-on discovery decades before safety goggles and modern fume hoods became laboratory standards. Many of those early syntheses seeded entire families of compounds, weaving this chemical into the broader story of analytical reagents and specialty intermediates.
Product Overview
Chemists rely on m-Phenylenediamine hydrochloride for its clear reactivity profile and accessibility. Typically offered in crystalline or powdered form, this salt of the parent diamine finds its slot in a huge spread of fields from textile dyes to rubber chemicals and pharmaceutical intermediates. Those who have spent hours at the workbench can recognize its sharp tang as the crystals hit the air. It’s the hydrochloride salt of an amine—this simple transformation keeps handling straightforward, delivering a more user-friendly solid with a longer shelf life than the base liquid. Researchers often use it for discovering synthetic routes, combining ease of use with promising reactivity.
Physical & Chemical Properties
This compound carries a white to off-white appearance, trending toward yellow if left exposed too long. Purity levels hit above 98%, with melting points clocked around 137-142°C. Solubility in water is strong, thanks to the hydrochloride group enhancing the base molecule’s ability to interact with polar environments. As someone who has handled it, the distinct amine odor stands out, along with the tendency of the material to cake if humidity levels rise. This substance resists oxidation fairly well, but like many aromatic amines, prolonged exposure to air can nudge the color darker, reminding one of its reactive nature. Looking at its chemical fingerprint, the presence of two amino groups and a hydrochloride moiety creates a platform for further modification and reaction.
Technical Specifications & Labeling
Suppliers must detail batch information, purity, water content, and residual solvents. Customers in research or commercial settings check for clear labeling that outlines hazard symbols, handling instructions, and recommended storage conditions. Given the carcinogenicity flagged with aromatic amines, strict attention to technical documentation—down to the labeling font and batch traceability—cannot be ignored. No matter how familiar the user grows with the product, regulatory compliance and clarity in documentation play critical roles. Each barrel or jar, whether destined for a pilot plant or academic lab, should shout its identity, concentration, batch number, and shipping date. Labs need this information for both traceability and protective measures, leaving no room for error during audits or safety checks.
Preparation Method
Traditionally, production follows a two-part process. Nitration of benzene generates m-dinitrobenzene, which is then reduced—often by iron and acid or through hydrogenation—to the amine. Combining the resulting m-phenylenediamine with hydrochloric acid yields the hydrochloride salt. Industrial-scale preparation keeps a keen eye on temperature, reagent ratios, and purification steps. Impurities, especially byproducts of incomplete reduction or residual nitro compounds, challenge both yield and product safety. Scaling up this chemistry demands attention to waste handling and gas scrubbing, as amine fumes pose hazards and can lead to environmental complaints in surrounding areas. Anyone who has run such a synthesis knows the value of good ventilation and gloves, learning quickly how small process errors multiply in large batches.
Chemical Reactions & Modifications
The diamine structure shines for its reactivity. Aromatic substitutions, diazotizations, and coupling reactions open doors to dyes, azo compounds, and cross-linkers. Those double amino groups anchor a slew of interesting transformations, making the hydrochloride salt a ready starting point for sulfonation, acetylation, or even polymer formation. Synthesis-minded chemists tweak reaction times and pH to tune selectivity or push conversion rates higher. From personal experience, the tendency toward side products—especially undesirable oxidation products—keeps chemists on their toes. Understanding the basicity of the amine functionalities guides deprotonation and modification strategies, which can make or break a reaction depending on the downstream goal.
Synonyms & Product Names
Academic references and supplier catalogs list it under names like 1,3-diaminobenzene hydrochloride, meta-phenylenediamine hydrochloride, and m-PDA HCl. Some older literature might call it C.I. 76040, especially in the context of colorant precursors. These different handles reflect naming conventions over time and across disciplines, from analytical chemistry to industrial manufacturing. Clarity in product nomenclature strengthens research communication and purchasing accuracy, especially as suppliers shift between regional and international standards.
Safety & Operational Standards
M-Phenylenediamine hydrochloride requires a cautious attitude, not just a set of gloves and a fume hood. Scientific reports and safety data sheets flag risks of skin sensitization, respiratory irritation, and more grave concerns about carcinogenicity. Industrial and academic environments alike must enforce chemical hygiene, with eye protection, lab coats, and secure storage away from oxidizers and acids. Labeling goes beyond regulatory compliance; it builds habits that protect people from long-term harm. Some of the more troubling incidents in chemical research have stemmed from taking shortcuts with substances like this—the lessons learned are written in safety procedures and reinforced by regular audits and training sessions.
Application Area
Uses cut across dye manufacturing, rubber antioxidants, polymer intermediates, and fine chemicals. Dye and pigment plants value the diamine’s ability to build stable colorants for synthetic fibers. Rubber producers lean on it for its role in vulcanization accelerators, helping tires hold up under heat and wear. In pharma R&D, scientists shape it into more complicated molecules, finding utility for the reactivity buried in those two amine groups. Chemical suppliers see demand from research labs spinning out new testing protocols, detection reagents, or analytical standards. Even after all these years, the versatility of its molecular framework continues to surprise, with startups and academic teams looking at further modifications for electronics, imaging agents, and advanced polymers.
Research & Development
Innovation keeps this compound in play, even as regulations around aromatic amines tighten. Environmental and health concerns drive R&D efforts to greener synthesis routes—reducing waste, reusing solvents, and minimizing worker exposure. Advances in catalysis and bioconversion show promise in streamlining production while lowering the carbon footprint. Many research groups experiment with new derivatives, aiming for less toxic, more effective alternatives in dye technology or biomedical applications. As labs build high-throughput screens and automation into organic synthesis, demand grows for pure, reliable intermediates that push chemistry into new territory. Anyone who has wrestled a stubborn side reaction into submission knows the value of a well-characterized, high-quality starting material.
Toxicity Research
Toxicity concerns continue to loom over m-Phenylenediamine hydrochloride and its relatives. Data from in vivo and in vitro studies point to dangers around skin sensitization and possible links to bladder cancer—echoing broader worries about aromatic amines in occupational settings. Regulatory agencies in Europe, the United States, and Asia keep a sharp eye on permissible exposure levels. Research focuses on exposure routes, metabolic fates, and safer alternatives in both occupational and consumer products. Practical knowledge aligns with the science; even a small accidental exposure brings an unforgettable reminder of the substance’s biological impact. With growing awareness, companies and labs improve ventilation, monitor air levels, and launch health surveillance programs for their staff to keep risks at bay.
Future Prospects
As regulations in the chemical industry get tougher and markets demand lower-toxicity materials, m-Phenylenediamine hydrochloride sits at a crossroads. Researchers push for new derivatives—hoping to blend utility with greener, safer profiles. Advances in process control, like in-line monitoring and AI-driven reaction optimization, offer the promise of greater efficiency and consistency, making it easier to enhance production quality while meeting regulatory hurdles. Partnerships between academic labs, industry, and regulatory groups push the boundaries of application, but the focus remains on balancing function and safety. Sustainable synthetic routes, improved labeling, and smarter risk management will likely determine which sectors keep using this compound and which shift to substitutes, reflecting lessons learned across decades of chemistry and manufacturing.
What is M-Phenylenediamine Hydrochloride used for?
A Closer Look at What It Does
M-Phenylenediamine Hydrochloride turns up in places most people never notice, but its impact reaches into several corners of modern living. Chemists recognize it as a key piece in making dyes, especially dyes that find their way onto fabrics and into hair coloring products. Working in a chemistry lab during college, I came across this compound during dye formulation experiments. Without it, a whole range of permanent hair color shades would not exist.
Beyond coloring textiles and hair, this compound shows up in the pharmaceutical industry. In drug development, this molecule acts as an intermediate for producing certain medicines. Research points to its usefulness in synthesizing drugs for everything from bacterial infections to more specific medical treatments. The route chemicals take from lab bench to medicine cabinet often involves steps built on chemicals like M-Phenylenediamine Hydrochloride.
The Science Behind Its Role
Talking to chemists, it’s clear why M-Phenylenediamine Hydrochloride matters. Its chemical structure helps create bonds in complex organic molecules. Industries rely on it to build bridges between molecules, laying the foundation for things like epoxy resins and adhesives. On the factory floor, this compound helps glue together everyday products, from car parts to chairs in an office. During a short stint in a manufacturing plant, I learned how much precision goes into every batch of material—chemicals like this one play a silent but crucial role.
What Makes It Tricky?
On the flip side, frequent handling of M-Phenylenediamine Hydrochloride comes with real risks. Many studies flag its action as a skin and eye irritant. A friend who worked in hair salons shared stories about allergic reactions from hair dye customers. The evidence points towards the importance of protective gloves, eye protection, and ventilation—especially for those using it daily. Animal tests and long-term exposure data have pushed regulators in several countries to create safety limits for its use.
Recycling and safe disposal prove challenging. Municipal wastewater treatment plants find it tough to break down this compound fully. Trace amounts in rivers and lakes hurt aquatic life. Environmental studies published in the past decade argue for better waste management at the source—manufacturing plants, salons, and pharmaceutical facilities. It’s become clear, even to those outside the chemical industry, that ignoring environmental health brings consequences for everyone.
How People Can Use It Safer
Regulatory agencies keep tightening restrictions on compounds like this. From labels with clearer warnings to stricter exposure limits for workers, progress is happening but needs constant attention. As a consumer, it’s worth reading labels on hair dye and understanding which ingredients carry risks. For those working in industries using M-Phenylenediamine Hydrochloride, regular training about hazards and safety gear never feels unnecessary. Responsible disposal—as simple as using special waste bins or not pouring chemicals down the drain—can make a difference.
Relying on safer alternatives in manufacturing also offers hope. Chemical engineers worldwide are testing new substances for dye and drug production that come with less baggage for health and the environment. The journey to safer chemistry is slow, but every thoughtful step counts.
What are the safety precautions when handling M-Phenylenediamine Hydrochloride?
Understanding the Risks
M-Phenylenediamine hydrochloride sits on the shelf with other chemicals that demand respect and focus. Years at the lab bench taught me that skipping precautions brings real pain. This substance doesn’t just carry an intimidating name — it brings hazards for the skin, eyes, and lungs. A splash or a speck of dust can trigger burns, allergic reactions, or breathing problems. I once saw a co-worker rush out, eyes watering, after exposing bare hands to less than a gram. Stories like that stick with you and shape habits.
Personal Protective Equipment (PPE) Isn’t Optional
Sweet-talking yourself into skipping gloves or goggles does not pay off. Nitrile gloves form a solid barrier, and I always double-check the fit before dosing out powder. Chemical splash goggles, not just reading glasses, block dust and accidental squirts. Some days, the air in the lab picks up tiny airborne particles. That’s a real risk for your lungs, so I reach for a lab coat, long sleeves, and—especially during weighing—a NIOSH-approved respirator. If you think lab coats are hot and annoying, wait till you deal with skin irritation.
Working With Good Ventilation
I never underestimate proper ventilation. Fume hoods protect breathing—not by accident, but by design. Pulling the sash down and turning the fan on always comes before opening a jar of solid chemical. One time, the extraction fan failed; we learned real quick that headaches and coughing come on fast in a stuffy space. Fume hoods and exhaust fans really make the difference between safe handling and sneaky exposure.
Spill and Waste Management
Preparation gives peace of mind. Absorbent pads and spill kits line my bench. If M-Phenylenediamine hydrochloride powder gets loose, a wet towel traps dust best, instead of dry sweeping which just lifts particles into the air. I bundle waste — gloves, wipes, contaminated gear — into clearly labeled containers. Hazardous waste pickup schedules aren’t just for show. Once, improperly stored rags set off a fire alarm after a long weekend of neglect. That memory changed my waste habits for good.
Why Training and Labeling Matter
Labels and training save accidents. Legible, taped-down hazard labels on bottles mean no guessing during busy shifts. I sit through yearly safety seminars, and each time, someone shares a fresh story: missed labels, skipped glove changes, careless storage. Chemistry doesn’t forgive shortcuts. Real talk with colleagues after close calls does more than posters.
Best Practices Are More Than Rules
I check material safety data sheets before starting any new project. Knowing where the eyewash and shower stations live isn’t dramatic, it’s the difference between a quick rinse and real damage. Keeping your face away, opening bottles slowly, and never working solo—these turn common sense into true fences against disaster.
Moving Toward Safer Labs
Building a safety culture that everyone buys into trumps any shiny equipment. I noticed that open conversations about accidents, visible PPE compliance, and accessible signage push safety forward faster than lectures. Regular drills keep muscle memory sharp. Reporting minor incidents shines a light on hidden risks before someone gets hurt. Good habits, not luck, shape long, healthy careers around substances like m-phenylenediamine hydrochloride.
How should M-Phenylenediamine Hydrochloride be stored?
Why Storage Choices Matter
I’ve seen what happens in labs when someone lets their guard down with sensitive chemicals. Years ago, a simple oversight with a less hazardous compound caused a whole research team to halt their experiments for days. That moment stuck with me. M-Phenylenediamine Hydrochloride brings a unique set of challenges, not only because it supports a range of industrial uses but because mishandling can harm people and compromise research. Safety and chemical integrity simply demand more respect than most folks give.
Protecting from Air and Moisture
With M-Phenylenediamine Hydrochloride, keeping air and water away feels like the baseline. Once this chemical picks up moisture, the product can clump or degrade. As soon as one uses a damp spatula or leaves a cap loose, the subtle change sneaks in unnoticed. Over time, the risks build. Choose a tight-sealing, chemical-resistant container—glass works, but certain plastics made for laboratory use can do the job too. Screw tops help, as do secondary containers for extra peace of mind, a habit my mentors drilled into us during training.
Temperature Control Speaks Volumes
Every chemist who’s ever waded through a pile of ruined supplies because a room ran too hot knows the misery. M-Phenylenediamine Hydrochloride keeps its shape best away from heat. Go room temperature or a bit cooler. Refrigerated storage (in a dedicated chemical fridge, not the same one with lunches or samples) gives an added buffer, especially in places that see big seasonal swings. Avoid freezing, though; repeated temperature jumps create as many problems as constant warmth.
Light Isn’t Always Your Friend
Many overlook the way light kicks off unwanted reactions, yellowing powders, and shortening shelf life. M-Phenylenediamine Hydrochloride can react to light, slowly at first, then more visibly over months. Store those containers somewhere dark—a chemical cabinet with an opaque door works. For folks who keep an inventory in open shelving, wrap the bottles in foil or use amber-colored containers.
Clear Labels and Training Build Confidence
Plenty of incidents happen because someone grabbed the wrong bottle or didn’t know the rules. Every time I step into a new lab, labeling stands out. Masks may help, but nothing replaces bold labels: product name, hazard symbols, opening date, and a notice for “keep dry, cool, sealed, dark.” Chemical suppliers follow these for a reason—they cut confusion and prevent slip-ups. Lab managers should go further, regularly walking staff through storage routines to refresh knowledge and highlight where things start to slip.
Thinking Long Term: Safer Spaces and People
Safe practice grows from experience and clear heads. Invest in fire-resistant cabinets if you work with big volumes. Chemical spill trays, secondary containment, and regular inspections don't cost much compared to lost time or emergencies. I've seen how one forgotten vial can turn into a crisis. Build routines that work in the real world: check containers, clean up spills fast, keep inventories up to date, and never treat M-Phenylenediamine Hydrochloride as “just another powder.”
Practical Steps Prevent Disaster
Good storage doesn't just protect chemicals—it shields people and research. Rely on air-tight, well-labeled containers. Keep humidity and sunlight away. Control your temperature. Train everyone, not just new hires, and set up checks to spot trouble. These straightforward habits draw a clear line between a well-run lab and an avoidable disaster. The lesson learned: chemicals don’t forgive carelessness, but safety isn’t complicated if you work from experience and respect the risks.
What is the chemical formula of M-Phenylenediamine Hydrochloride?
Getting to Know the Compound
M-Phenylenediamine hydrochloride has caught the attention of researchers, chemists, and industry professionals for years, not just because it appears in synthetic chemistry labs, but also due to its unique set of properties. Its chemical formula—C6H8N2·2HCl—reveals plenty about its composition. The molecule starts with a benzene ring, holds two amine groups, and pairs with two hydrochloric acid units.
How Structure Translates to Usefulness
Each element in its formula plays a role. The benzene ring provides a stable backbone. Two amine groups at the meta positions lend the compound its reactive personality. The presence of hydrochloride boosts solubility in water, which helps in many practical applications. I’ve mixed dozens of aromatic amines and found that these small configuration changes make a difference. For m-phenylenediamine hydrochloride, mix it in water and you get a clear solution—handy for hair dye industries, chemical syntheses, and analytical chemistry settings.
Sourcing Quality and Safety in the Workplace
The compound rarely arrives in a vacuum. Reputable suppliers track every batch carefully because impurities can skew results and, in some cases, place end users at risk. I’ve seen what happens when someone takes shortcuts during procurement: laboratory results become unreliable and product stability suffers. Chemical companies recognize this, following strict standards—ISO certifications and transparent records keep mistakes scarce.
Potential Health Hazards and Responsible Handling
Even simple mixing deserves caution. This chemical can irritate the skin and mucous membranes and may pose greater risks with repeated exposure. Researchers trust the safety data sheets, not just gut feeling. Clear labeling, gloves, and good ventilation turn routine handling into a safe routine. One time, a lapse in safety led to an entire lab being decontaminated. Nobody forgets such lessons. For larger-scale industry use, it’s common sense to ensure that training always remains current and barriers separate chemicals from unprotected skin.
Role in Industry and Everyday Products
M-Phenylenediamine hydrochloride’s footprint extends further than most realize. It takes a skilled team to use it in synthesizing pharmaceuticals and dyes. Coloring agents in textiles, permanent hair colorants, and certain photographic developers rely on this compound’s reactivity and solubility. I’ve talked with hair stylists who appreciate the hues it helps produce—though most would never know a benzene ring sits at the root of that chestnut brown color.
Toward Sustainable Solutions
Sustainability enters every modern industrial conversation. By optimizing reaction conditions, using green solvents, and reducing disposal waste, industry can keep m-phenylenediamine hydrochloride from causing unnecessary harm. There’s also momentum behind recycling and reusing materials wherever possible. Government agencies set disposal guidelines, but individuals in the lab or on the plant floor often spot inefficiencies and spark change from within.
Conclusion
Understanding the formula C6H8N2·2HCl means more than connecting atoms. It means knowing how to harness this compound safely and responsibly. With growing knowledge and commitment to best practices, its benefits can outweigh the potential risks. The real challenge comes from translating the science into safe, sustainable use across labs and industries.
Is M-Phenylenediamine Hydrochloride hazardous to health?
The Hidden Dangers We Overlook
A lot of people working in labs or factories probably recognize the name m-Phenylenediamine Hydrochloride. It’s used to make dyes, polymers, pesticides, and even plays a part in some hair coloring products. The problem is, most don't realize how hazardous this chemical can become to human health without proper handling.
Not Just Another Factory Chemical
Once, in graduate school, I spent afternoons researching industrial solvents and intermediates. Colleagues in the lab would casually handle everything—pipettes dripping, gloves barely changed. Back then, we thought as long as nothing spilled, all was safe. It’s easy to forget that inhalation, skin contact, and even minor spills can introduce dangerous chemicals right into our systems.
M-Phenylenediamine Hydrochloride has a reputation for being particularly nasty if you come into contact with it. Science backs this up—studies show that it can cause severe skin and eye irritation, and a person breathing in dust or vapors risks damaging their respiratory system. Data from the CDC and European Chemicals Agency lists allergic contact dermatitis, breathing issues, and long-term health effects as real dangers. Accidental swallowing brings even more trouble: doctors report headaches, nausea, and in bad cases, kidney and liver problems.
Why Health Agencies Sound the Alarm
Guidelines from OSHA and NIOSH set strict exposure limits for m-Phenylenediamine Hydrochloride. Agencies don’t take this lightly, because a single oversight can send someone to the emergency room. In some workplace incidents, people didn’t even realize the seriousness of a small spill until rashes and coughing showed up hours later.
Peer-reviewed studies link long-term repeated exposure to increased risks of asthma-like symptoms and hypersensitivity. In my own classes, young researchers learned about the risks through real case studies. Forgetting to secure a chemical hood or skipping a glove change led to days off with medical evaluations.
Changing Habits, Building Safety
Mitigating these risks starts on the shop floor or in the school lab. Proper PPE—nitrile gloves, lab coats, goggles—should be non-negotiable for anyone around m-Phenylenediamine Hydrochloride. Good ventilation limits how much workers breathe in day after day. Regular safety drills and spill-response training can make the difference between a minor scare and a real emergency.
Companies ought to invest in clear instructions and up-to-date safety data sheets, keeping them in reach rather than buried in digital folders. On a practical note, I’ve seen research groups develop their own color-coded labeling system for dangerous compounds. This simple step curbed confusion and helped new members spot hazards right away.
Looking Toward Safer Solutions
Safer substitutes often exist for high-risk chemicals, though switching over takes time and money. Industries using m-Phenylenediamine Hydrochloride should consult with industrial hygienists and look for less hazardous alternatives, especially in cosmetics and dyes where thousands of workers risk exposure. Daily exposure limits sometimes need review as new evidence emerges, so policies must adapt as the science grows.
Real change comes from persistent small actions—routine glove changes, rigorous training, listening to people who've experienced close calls. Protecting health in the lab or factory isn't just about following rules. It’s about remembering that every safety practice shields someone’s future.
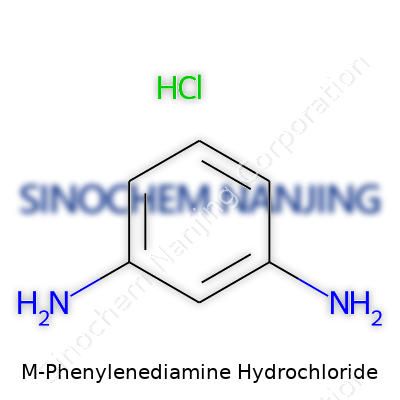
| Names | |
| Preferred IUPAC name | 2,4-Diaminobenzenium chloride |
| Other names |
1,3-Benzenediamine hydrochloride Meta-Phenylenediamine hydrochloride m-Phenylenediamine monohydrochloride 1,3-Phenylenediamine hydrochloride |
| Pronunciation | /ɛm-fɛˌnaɪliːnˈdi.əˌmiːn haɪˌdrɒxɪˈklɔːraɪd/ |
| Identifiers | |
| CAS Number | 538-58-9 |
| 3D model (JSmol) | `3D model (JSmol)` string for **M-Phenylenediamine Hydrochloride**: ``` NC1=CC=CC=C1N.Cl ``` This is the **SMILES** string representing M-Phenylenediamine Hydrochloride suitable for JSmol loading. |
| Beilstein Reference | 74237 |
| ChEBI | CHEBI:63097 |
| ChEMBL | CHEMBL15815 |
| ChemSpider | 16318 |
| DrugBank | DB14088 |
| ECHA InfoCard | 012119457040-46-XXXX |
| EC Number | 219-489-5 |
| Gmelin Reference | 57393 |
| KEGG | C01787 |
| MeSH | D017434 |
| PubChem CID | 61246 |
| RTECS number | SS9625000 |
| UNII | MDW3Z89N24 |
| UN number | UN2928 |
| CompTox Dashboard (EPA) | DTXSID5036678 |
| Properties | |
| Chemical formula | C6H8Cl2N2 |
| Molar mass | 144.59 g/mol |
| Appearance | White to light yellow or light brown crystalline powder |
| Odor | Odorless |
| Density | 1.241 g/cm³ |
| Solubility in water | soluble |
| log P | -2.1 |
| Vapor pressure | 0.01 hPa (25 °C) |
| Acidity (pKa) | pKa = 4.7 |
| Basicity (pKb) | pKb: 9.88 |
| Magnetic susceptibility (χ) | -51.0 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.695 |
| Dipole moment | 2.99 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 169.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -37.65 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1052.4 kJ/mol |
| Pharmacology | |
| ATC code | D08AX04 |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled; causes severe skin burns and eye damage; may cause allergic skin reaction. |
| GHS labelling | GHS05, GHS07 |
| Pictograms | GHS06,GHS07 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331, H317, H319, H341, H351, H410 |
| Precautionary statements | P261, P280, P302+P352, P304+P340, P305+P351+P338, P310, P321, P330, P363 |
| NFPA 704 (fire diamond) | 2-3-0 |
| Lethal dose or concentration | LD50 oral rat 387 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 350 mg/kg |
| NIOSH | WN3100000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.1 mg/m³ |
| Related compounds | |
| Related compounds |
Aniline o-Phenylenediamine p-Phenylenediamine Benzidine 4-Chloroaniline |

