M-Isopropylphenol: Unpacking an Understated Chemical Building Block
A Journey from Labs to Industry Shelves
M-Isopropylphenol, sometimes called 3-Isopropylphenol or 3-Hydroxycumene, travels a long road from its early identification in chemical catalogs to its quiet but crucial role across industrial and research applications. Researchers first encountered compounds like m-Isopropylphenol while experimenting with alkyl substituents on aromatic rings in late-19th-century European laboratories. The work provided a foundation for later advances in synthetic organic chemistry. Over the decades, organic chemists found uses for this compound’s unique reactivity and moderate volatility. Though overshadowed by relatives such as p-cresol and thymol, m-Isopropylphenol carved out a place in the crowded field of specialty aromatic compounds by virtue of its balance of activity and stability.
Getting Acquainted with m-Isopropylphenol
This molecule steps onto the scene as a clear, oily liquid at room temperature, carrying a faint yet spicy, phenolic smell that hints at its potential in fragrance chemistry. Unlike some of the more aggressive phenols, its measured solubility in water and strong affinity for organic solvents signal a versatility that appeals to chemists working with both hydrophobic and hydrophilic systems. Its chemical structure, featuring an isopropyl group in the meta-position on the benzene ring, tweaks the electronic environment just enough to open new doors in syntheses and modifications.
Properties with Practical Impact
A boiling point around 220°C gives m-Isopropylphenol robustness under moderate heating, while its melting point ensures handling as a liquid under most conditions found in warehouse or pilot plant environments. The molecular weight, hovering just over 136 g/mol, makes it substantial enough for weight-based dosing but light enough to keep logistics straightforward. From a chemical standpoint, that meta-isopropyl group directs substitution patterns and influences how the compound reacts with oxidizers or acids, which helps shape scalable synthetic routes. Compared to ortho or para isomers, the meta variety avoids certain problems with sterics in coupling reactions, giving it a slight edge where selectivity matters.
How It’s Made and Why That Matters
Traditional syntheses of m-Isopropylphenol lean on Friedel–Crafts alkylation, usually starting with phenol and propylene or isopropyl chloride, catalyzed by Lewis acids like aluminum chloride. Over the years, process chemists adjusted reaction conditions to reduce unwanted byproducts, often para or ortho derivatives, and to keep waste streams manageable. As environmental priorities take center stage, greener conditions—using milder catalysts or solventless operations—attract more attention. Not all solutions come easy. Pilot plant operators know the challenge of keeping selectivity high without sacrificing throughput or operator safety, a tricky balancing act that pushes process development teams to rethink traditional playbooks.
Names and Labels—What’s in a Name?
Scan a typical chemical supplier’s catalog and m-Isopropylphenol appears under a handful of names. The core identifiers—m-Isopropylphenol, 3-Isopropylphenol, CAS 99-89-8—are the ones that turn up most often in purchasing systems. In some research circles, you might hear “m-cumenol” as a shorthand. Regulatory bodies around the world notice this overlap, which sometimes causes confusion when ordering for pharmaceutical or cosmetic development, where precision in nomenclature keeps orders moving and prevents mix-ups in handling and storage.
Staying Safe, Staying Productive
Lab professionals who work with m-Isopropylphenol follow established chemical hygiene principles to keep risk in check. Although less hazardous than more reactive phenolic cousins, this compound still asks for basic precautions: good gloves, chemical goggles, and working in a fume hood always count. Skin and eye irritation turn up in most supplier safety data sheets, so casual attitudes tend to disappear once someone gets a whiff of its distinct aroma or experiences minor contact issues. Storage in amber bottles, away from strong oxidizers and acids, keeps both sample integrity and worker safety intact. Over the past decade, companies have transitioned to more robust leak-detection systems and improved training, which helps reduce the casual exposures sometimes seen in older facilities.
Chemical Reactions and Modifications—Unlocking Utility
The reactivity of m-Isopropylphenol centers around its phenolic hydroxyl group and the isopropyl side chain. It pairs well with halogenation, etherification, and alkylation protocols without needing exotic conditions. The meta orientation of the isopropyl group—three carbons away from the hydroxyl—shapes which direction further reactions go. In research, teams often introduce protecting groups, then use this compound as an intermediate for more complex pharmaceuticals or agrochemicals. The moderate electron-donating effect from the isopropyl moiety influences aromatic substitution patterns, giving product makers some finesse in targeting aromatics not easily accessed with other starting materials.
Applications—Why Industries Keep Coming Back
Demand for m-Isopropylphenol spans several sectors. In the flavor and fragrance world, its spicy-sweet notes show up as a component in specialty scents, sometimes as a base for synthetic natural flavors. In materials science, its chemical backbone acts as a stepping stone in the crafting of resins or stabilizers. Laboratory researchers leverage its ability to undergo controlled modifications, using it as a model substrate to probe new catalytic reactions. Pharmaceuticals draw on this molecule’s side chain to generate scaffolds that mimic or block biological activity. Agricultural chemists see value in its use as both an intermediate and as part of eco-friendly pesticides or growth regulators. With regulatory shifts demanding less persistent environmental impact, compounds like m-Isopropylphenol—easier to degrade than heavier phenols—earn a closer look for new roles.
Research, Toxicity, and Finding Balance
Studies on m-Isopropylphenol’s toxicity paint a picture of moderate caution. Researchers have mapped out acute toxicity levels for guidance in occupational hygiene, showing it tends not to bioaccumulate but does prompt temporary irritation after direct contact. Animal studies indicate manageable risk profiles at low concentrations, though high oral doses cause signs typical of phenolic compounds: central nervous system effects and potential organ stress. Surveys in occupational medicine haven’t flagged major chronic effects so far, but long-term, high-level exposure data remain limited. This signals a need for ongoing vigilance, especially as industrial volumes inch higher. Current regulatory standards call for air monitoring and waste management in larger facilities, with a push toward closed-loop processes to keep emissions down and off-site impact minimal.
Looking Ahead—How Prospects Are Taking Shape
The role of m-Isopropylphenol is shifting as chemists in academia and industry chase greener chemistries and tighter control on side products. Machine learning and computational modeling enter the picture, guiding teams to tune reaction conditions more precisely and trim down off-target isomers. Growing demand for sustainable pesticides, specialty polymers, and pharmaceutical building blocks signals new territory for this compound, while stricter environmental rules spark creative thinking in both synthesis and end-use applications. Current research aims to engineer seismic improvements in step economy and energy input, nudging bulk production closer to carbon-neutral status. Smaller startups and university groups keep probing bio-based alternatives, inspired by natural phenolic compounds, as part of the push for a safer, less wasteful chemical future. M-Isopropylphenol stands as a steady tool in this changing landscape, not grabbing headlines but doing just enough heavy lifting to keep wheels turning behind the scenes.
What is M-Isopropylphenol used for?
What Makes M-Isopropylphenol Important?
M-Isopropylphenol sounds like a name you might only encounter in a textbook. In daily life, though, it takes on roles that most folks never notice. This molecule sits in the middle of industries like pharmaceuticals, perfumes, and disinfectants. As someone who grew up in a farming family, cleaning equipment and worrying about bacteria wasn't just for hospitals. Farmers and factory workers deal with germs all the time, and that's where M-Isopropylphenol wears one of its hats—as an antimicrobial.
Diving into Everyday Uses
Many manufacturers use this compound to add antibacterial muscle to household cleaners and personal hygiene products. Think soaps promising to kill germs—not every ingredient pulls its weight, but M-Isopropylphenol gets results. Research in the Journal of Medical Microbiology has shown how it tackles troublesome bacteria like Staphylococcus aureus. When using a soap or handwash with this molecule, you’re not just scrubbing for the sake of it. There’s science working in the background.
In the pharmacy, you'll find this ingredient in antiseptic creams and ointments. It steps in to keep cuts and scrapes from turning into something more serious. Hospitals and clinics often need a reliable way to protect both patients and staff, and here, the proven performance of M-Isopropylphenol stands tall. The Centers for Disease Control and Prevention (CDC) recognizes the role of phenolic compounds like this one when it comes to fighting infections on surfaces and skin.
More Than Cleaning: Fragrance and Flavor
Walk through a shopping mall and the scents can be overwhelming. Perfume makers hunt for chemicals that linger, blend well, and stick to the skin. Some components act as anchors—keeping delicate, fleeting smells from evaporating too quickly. M-Isopropylphenol plays this anchor role in perfumes. Its faint medicinal odor might not sound appealing on its own, but mixed correctly, it adds staying power to floral or musky scents.
In the world of flavor, this chemical appears in low concentrations in products that need a sharp, medicinal note. Food scientists draw strict lines here—too much brings a harsh edge, but in small amounts, it rounds out flavors in chewing gum or certain candies.
Environmental and Health Considerations
Handling chemicals carries its own concerns. Anyone working with phenolic compounds should know about skin irritation and potential toxicity with overuse. OSHA sets limits, and those working in production plants use protective gear for good reason. As a community, it's important to respect safety guidelines and avoid taking unnecessary risks with exposure.
Wastewater management is another factor. M-Isopropylphenol doesn’t just vanish after use. Environmental agencies, like the EPA, monitor levels and set guidelines for disposal. Investing in better filtration and treatment at factories pays off—not just for regulators, but for communities downstream. It’s one of those places where company profits and local health line up on the same side.
Room for Better Solutions
Talking with people in manufacturing and healthcare, it’s clear there’s room for safer, greener alternatives. Research teams now look at plant-based extracts or biodegradable molecules that promise the same punch against germs but with fewer side effects. Sometimes, these don’t work quite as well or as cheaply, but the push for better health and a cleaner environment keeps moving forward. As consumers, every purchase tells companies what the market values. Demanding safer products and supporting research for greener chemicals makes a difference.
What are the safety precautions when handling M-Isopropylphenol?
Understanding the Risks Up Close
Working with chemicals like M-Isopropylphenol has always forced me to pay attention to the kind of risks many overlook. This compound finds its way into labs for a reason, but it quickly reminds you not to cut corners with safety. You feel it in the sting at the back of the throat from a single whiff or that sudden irritation when it hits exposed skin. For me, it stands as a clear case: chemistry punishes carelessness.
No Substitute for the Right Gear
I’ve learned firsthand, personal protective equipment is not something to ignore. Splash-proof goggles block the compound before it touches your eyes. A fitted lab coat covers skin and won’t leave gaps. Gloves—nitrile options work—keep fingers safe from burns and rashes. Even the air needs watching: good ventilation pulls vapors away before you breathe them in. If I ever doubted the power of fumes, one headache in a stuffy room fixed that.
Storage Isn’t Just About Shelves
Letting a bottle sit on any shelf leaves too much to chance. M-Isopropylphenol belongs in a clearly marked, chemical-resistant container with a tight lid. Heat and sunlight speed up reactions nobody wants to see. Even the location counts; keep this stuff far from acids and oxidizers, because a small mistake mixes into an emergency. In any workspace I’ve set up, a locked cabinet becomes the rule, not the exception.
Spills and Accidents: Fast Action Wins
Some days, spills just happen. I never walk away, not for a second. Spilled M-Isopropylphenol needs to get covered with absorbent material—kitty litter does the job. Don’t scoop anything bare-handed. Double-bag the waste and label it as hazardous before tossing it into the approved collection spot. Fresh air in the room clears out vapors, and cleaning up with water and soap wraps things up. Supervisors and coworkers expect a report because leaving hidden leftovers behind guarantees future problems.
Don’t Wait for Symptoms
Toxic effects sneak up. Shortness of breath or a skin rash hints you’ve gone too far. Emergency eye-wash stations and safety showers must be easy to reach; I check them before starting. Training every member of the team means recognizing what trouble looks like—even days later, since toxicity can show after a delay. No one benefits when someone pretends they can tough it out.
Clear Labels Save Minds and Health
I’ve seen disasters caused by misplaced or missing labels. Every bottle needs its contents and hazards spelled out, no exceptions. Safety Data Sheets give everyone a map for what to avoid and what actions to take if things go south. Complacency isn’t just a bad habit—it piles risk on top of risk in an instant.
Tips That Stand the Test of Time
I always keep a spill kit and extra gloves nearby, because it’s never a wasted step. Buddy systems work—someone notices the smell you miss, or sees a stain you overlook. Practice drills beat reading about emergencies. Chemical processes demand personal responsibility, and trusting the right habits keeps simple mistakes from becoming headlines.
What is the chemical formula and structure of M-Isopropylphenol?
Understanding M-Isopropylphenol
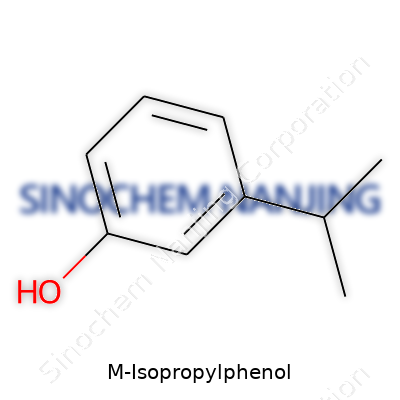
M-Isopropylphenol carries a sense of familiarity for folks who have spent time around organic chemistry. Some know it better as 3-isopropylphenol, a name that hints at how each part of the molecule connects. Its formula stands as C9H12O, which tells a practical chemist about the number of carbons, hydrogens, and that tucked-in oxygen.
Its molecular structure stacks up as a benzene ring holding onto a hydroxyl group at position 1 and an isopropyl group bolted on at position 3. Just to draw the picture: that ring of six carbons sits at the core, one spot gears up with an -OH (the phenol part) and two carbons attached to a single central carbon branch off somewhere else, making the isopropyl group. So you get a molecule that looks simple but turns complex in the right company.
The Power Behind Its Properties
This isn’t just a line on a lab order form or an entry in a catalog. I’ve worked with compounds like this while testing antioxidants for food and cosmetics. M-Isopropylphenol falls into the family of alkylphenols. That means it carries some antibacterial and antifungal punch. Formulators don’t just use it for the scent—although it does provide a woody, medicinal note that cuts through many synthetic fragrances. Studies back up its role as a preservative, especially in personal care.
Many natural oils and resins contain related molecules, but the manmade version lets you run controlled tests and aim for higher purity. Chasing down contaminants in cosmetics taught me that synthetic M-Isopropylphenol usually brings fewer surprises. You know exactly what sits in your bottle, which matters for companies checking for regulatory approval.
Real-World Importance and Challenges
M-Isopropylphenol slides into more applications than most expect. Cleaners, disinfectants, shampoos—none of these seem exciting until something stops working. A surprising fact: hospital-acquired infections often stem from common bacteria hanging out on surfaces. Formulations that harness active phenols drop infection rates.
But not everything about this compound fits smoothly into the supply chain. Phenols, even ones that seem harmless, can cause allergies for some people. During my time reviewing safety data, I ran into reports of skin rashes and complaints related to extended use—not only from end users but also from workers handling bulk materials.
That brings up the push for good manufacturing practices. Factories keeping phenols under control, investing in safety equipment, and switching out older formulations for safer blends—the benefits go straight to workers’ health and product quality. I’ve witnessed first-hand how one factory that upgraded its ventilation ended up cutting accident reports by half while improving the consistency of their finished goods.
Looking Ahead
Green chemistry offers a fresh way forward. Some labs have started tweaking phenol derivatives to degrade more easily in wastewater. Smarter stewardship also means companies now look for ways to recover solvents, minimize batch sizes, and train their teams to respect each chemical, no matter how familiar.
Government watchdogs, like the US EPA and Europe’s ECHA, have tightened up oversight. As a result, every batch gets tracked, and customer labels must clearly signal potential hazards or uses. This kind of transparency didn’t always exist, but people are safer for it.
M-Isopropylphenol stands as a good reminder: each ingredient in the products we use every day represents choices made by chemists, safety engineers, regulators, and the hands-on workers, all connected across a global supply chain.
How should M-Isopropylphenol be stored?
Understanding the Risks
M-Isopropylphenol, known for its sharp smell and clear appearance, may seem straightforward to work with. In my years around chemical storage, I’ve noticed that people sometimes overlook everyday chemicals because they don’t strike them as overtly dangerous. This can be a costly mistake. Even mild exposure to vapors may lead to headaches or irritation. Direct contact brings a real risk of skin or eye harm. Workers and anyone who comes in contact with the substance have to take these risks seriously, which makes proper storage essential—not just for toolbox talks, but for real safety outcomes.
Temperature and Light Matter
Most labs and warehouses keep a running checklist for temperature-sensitive materials. M-Isopropylphenol demands a cool, dry spot, free from sunlight. Heat speeds up evaporation and increases the chance of vapors, which raises both health and fire hazards. From my time managing chemical stocks, I’ve found that storing small bottles in a locked, vented cabinet, away from radiators and windows, makes life easier for everyone. If left in a warm, bright place, you get a stinky mess, and worse, a fire risk.
Ventilation Can’t Be Ignored
A closed cabinet without proper airflow can turn a minor leak into something dangerous. Vapors may accumulate and cause harm even without anyone knowing right away. I’ve seen storage rooms give off a strong chemical odor just because a single bottle wasn’t sealed tight. Simple fixes like using cabinets with exhaust fans help cut down on vapor buildup. This keeps the storage area—and the people around it—safer.
Container Quality
Cheap plastic bottles won’t cut it with solvents like M-Isopropylphenol. Glass containers with screw caps work best. In my own handling, plastics sometimes become brittle or dissolve, leading to spills. As obvious as it sounds, using only containers meant for this chemical rules out accidents. Tight seals matter just as much; any slack lid gives vapors an escape route. Tight bottles mean no waste and fewer headaches for workers.
Labeling and Segregation
An unlabeled bottle is a recipe for disaster. I’ve walked into storage rooms and spotted bottles with faded writing or no label at all. One mix-up can turn a routine process into an emergency. Proper labeling in bold, waterproof letters prevents confusion. Stores must keep M-Isopropylphenol away from oxidizers and acids; cross-contamination creates bigger risks, sometimes leading to fires or violent reactions.
The Human Factor and Training
Trusting people to know these rules without support rarely works. I’ve seen accidents tied back to poor training more often than to hardware failures. Posting clear instructions in a language everyone understands makes a real difference. Teams who run regular drills respond with less panic, and fewer mistakes. Putting eyes on the storage area often, instead of doing yearly checks, helps spot problems early.
Solutions for Everyday Operations
Keeping spill kits nearby, along with gloves and goggles, is non-negotiable. Anyone who handles the chemical gets a briefing on what to do if something goes wrong. Even small workplaces can afford this level of readiness. Checking every incoming shipment for leaks and checking shelves for out-of-date stock keeps things running smoothly. The small stuff prevents big problems down the line.
Taking good care with M-Isopropylphenol isn’t just about ticking regulatory boxes—it protects people, property, and bottom lines. Real safety grows from daily habits, not just rules on paper.
What are the physical and chemical properties of M-Isopropylphenol?
A Closer Look at M-Isopropylphenol
M-Isopropylphenol stands out in the big world of aromatic compounds. This molecule, also known as 3-isopropylphenol, consists of a phenol ring with an isopropyl group at the meta position. Chemists settled on its structure early on because its properties open the door to a mix of interesting uses, especially in making fragrances, pharmaceuticals, and disinfectants. For people working in those fields, knowing the basics of this compound can make the difference between a successful blend and a product that falls short.
Physical Properties You Can See and Feel
M-Isopropylphenol appears as a colorless to pale yellow liquid at room temperature. Folks in the lab can tell it's in the room by its strong, medicinal smell, which sets it apart from similar compounds. The boiling point lands near 221°C, so it needs a fair amount of heat before vapor starts rising from a flask. With a melting point around 24°C, it might switch from solid to liquid depending on a room’s temperature, making storage a point to consider. The density rests at about 0.96 g/cm³, putting it just below water. This matters for anyone who has ever tried to separate out layers in a separatory funnel or plan solvent choices in synthesis work.
As far as solubility, m-Isopropylphenol dissolves clearly in most organic solvents—think ethanol, ether, and acetone—thanks to that balanced combination of a hydrophobic isopropyl group with the polar OH group. Water solubility stays limited, though. That helps keep it stable and easy to handle in organic chemistry labs but builds in limits for use in water-heavy applications.
Chemical Properties That Spark Interest
The chemistry starts with the phenolic OH group. This makes the molecule slightly acidic, which means it donates protons in water but not nearly as strongly as mineral acids. This plays into its effectiveness as an antimicrobial agent and explains why it's found in several cleansing products. People concerned with shelf life and safety care about this: phenols can kill bacteria and fungi. The presence of an isopropyl group at the meta position narrows down the sort of reactions it takes part in. It resists easy oxidation, holding up better compared to some relatives like cresols.
This resilience against oxidation also helps pharmaceutical researchers who lean on phenolic building blocks that keep their structure during tough reactions. Nitration, sulfonation, and halogenation work smoothly on this ring, allowing chemists to build more complex molecules from a reliable starting point. In fragrance chemistry, that predictability means scents stay truer to their intent over time, with m-isopropylphenol offering a steady base note.
What Makes These Traits Matter?
M-Isopropylphenol’s specific melting and boiling points, combined with its moderate acidity and ability to dissolve in common organic solvents, make it an attractive choice for formulating antiseptics. Manufacturers often choose it when phenol itself seems too harsh. The relatively high boiling point keeps it working longer in topical products, while its scent helps mask less pleasant components.
With proven antimicrobial power and a stable chemical setup, m-isopropylphenol addresses the practical needs of research labs, healthcare, and industry—not just through tradition, but by offering a sensible balance of safety and effectiveness. Chemists working with it appreciate both its consistency and its versatility. Handling protocols reflect the need for care with phenols, but anyone regularly dealing with organic compounds picks up the routine fast. All these features lock m-isopropylphenol into a unique niche—trusted, tried, and useful across a wide spectrum of everyday products.
| Names | |
| Preferred IUPAC name | 3-(Propan-2-yl)phenol |
| Other names |
3-Isopropylphenol meta-Isopropylphenol |
| Pronunciation | /ˌɛm-aɪˌaɪsəˈproʊpɪlˌfiːnɒl/ |
| Identifiers | |
| CAS Number | 599-76-6 |
| Beilstein Reference | 1209238 |
| ChEBI | CHEBI:34499 |
| ChEMBL | CHEMBL16255 |
| ChemSpider | 12155 |
| DrugBank | DB08386 |
| ECHA InfoCard | 100.120.620 |
| EC Number | 201-280-9 |
| Gmelin Reference | 8287 |
| KEGG | C01841 |
| MeSH | D007354 |
| PubChem CID | 7444 |
| RTECS number | GO9260000 |
| UNII | FHC1H0XH70 |
| UN number | UN2928 |
| CompTox Dashboard (EPA) | DTXSID6020283 |
| Properties | |
| Chemical formula | C9H12O |
| Molar mass | 136.19 g/mol |
| Appearance | Colorless to light yellow liquid |
| Odor | Phenolic odor |
| Density | 0.969 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 2.8 |
| Vapor pressure | 0.2 mmHg (25°C) |
| Acidity (pKa) | 10.6 |
| Basicity (pKb) | 10.87 |
| Magnetic susceptibility (χ) | -68.0e-6 cm³/mol |
| Refractive index (nD) | 1.517 |
| Viscosity | 3.91 mPa·s (25 °C) |
| Dipole moment | 2.33 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 129.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -209.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3597.4 kJ/mol |
| Pharmacology | |
| ATC code | D02AE02 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P273, P280, P301+P310, P302+P352, P303+P361+P353, P304+P340, P305+P351+P338, P312, P321, P330, P337+P313, P361, P363, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 1-3-0-健康-蓝 |
| Flash point | 82 °C |
| Autoignition temperature | 595 °C |
| Explosive limits | '1.1 - 5.9% (in air)' |
| Lethal dose or concentration | LD50 oral rat 820 mg/kg |
| LD50 (median dose) | LD50 (median dose): 820 mg/kg (rat, oral) |
| NIOSH | UR9230000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for M-Isopropylphenol: 5 ppm (skin) as an 8-hour TWA (OSHA) |
| REL (Recommended) | 20 mg/L |
| IDLH (Immediate danger) | IDLH: 250 ppm |
| Related compounds | |
| Related compounds |
O-Cresol P-Cresol Thymol Carvacrol |

