M-Cresyl Acetate: A Comprehensive Look
Historical Development
M-Cresyl acetate didn't show up out of the blue—chemists first explored cresol compounds before the full industrial significance came into focus. As early as the late 1800s, researchers started to modify m-cresol, hunting for derivatives that would stay stable but react well enough for downstream chemistry. By the middle of the 20th century, large chemical companies locked in on m-cresyl acetate for its balance of reactivity and safety in both lab and industrial settings. Early applications centered on fragrance and resin manufacturing, but pharmaceutical researchers quickly picked up on its potential during the growth of synthetic drug development. Each decade brought more controlled synthetic methods, better understanding of its toxicity, and regulations that shaped safer handling.
Product Overview
M-Cresyl acetate belongs to the family of aromatic esters formed from m-cresol and acetic acid. Most supplies come as a clear, oily liquid with a distinctly sweet, phenolic scent. Its unique structure—a methyl group on the benzene ring at the meta position—sets it apart from its close ortho- and para-isomers. As an intermediate, it sees heavy use in chemical plants looking to build more complex molecules, often serving as a flavor, fragrance base, or additive in specialty resins. Manufacturers label it under several names, although the backbone chemical features remain the same: a cresol core with an acetyl group linked through esterification.
Physical & Chemical Properties
M-Cresyl acetate stands out with a boiling point near 230°C and a melting point below room temperature, keeping it liquid under typical factory conditions. It’s only slightly soluble in water but dissolves well in ethanol, ether, and most non-polar solvents, making it easy to work into various mixtures or reaction streams. The color ranges from colorless to faint yellow, reflecting purity and exposure to light or air. Its vapor carries a strong phenolic note, which comes from the underlying m-cresol. Chemically, it behaves as an ester—hydrolyzing under strong acid or base conditions—and remains stable in storage if kept away from moisture and high temperatures.
Technical Specifications & Labeling
Industrial-grade m-cresyl acetate usually meets tight purity requirements—often 98% or higher, according to international standards like ASTM or ISO. Labels list the main active compound, mention trace impurities such as m-cresol or acetic acid, and provide information on flash point, vapor pressure, and recommended storage conditions. Packaging ranges from glass containers for lab-scale work to lined drums for bulk shipments. Safety features, such as corrosion-resistant seals and venting systems, have become standard over the years.
Preparation Method
Production relies on classic esterification. Chemists react m-cresol with acetic anhydride or acetyl chloride, using acid catalysts like sulfuric acid for higher yield. Charging a reactor with m-cresol, adding the acylating agent, and controlling the temperature around 50–80°C lets the reaction proceed smoothly. Careful monitoring prevents side products and excessive hydrolysis. Once the reaction reaches completion, water washes and fractional distillation drive off impurities, leaving behind concentrated m-cresyl acetate. Technicians check the result with gas chromatography and infrared spectroscopy to confirm purity.
Chemical Reactions & Modifications
This ester takes part in a few key downstream reactions. The aromatic ring allows for electrophilic substitutions, such as halogenation or nitration, expanding its use in dye and agricultural chemical production. Hydrolysis reverts the ester back to m-cresol and acetic acid, which can help in recycling waste or adjusting purity at later stages. Reductive cleavage is rare but useful for custom syntheses in pharmaceutical labs. Structural tweaks—like alkylation or ring substitutions—offer access to dozens of novel compounds for R&D teams looking to build more complex experimental molecules.
Synonyms & Product Names
Shoppers at chemical supply houses might also see names like 3-methylphenyl acetate or meta-cresol acetate. In catalogues, CAS number 93-54-9 usually connects buyers to the right product. Brand-specific variants sometimes appear under trade names reflecting slight tweaks in purity or intended use, but the core chemical structure stays identical across all these listings.
Safety & Operational Standards
Handling m-cresyl acetate in the lab or on the plant floor takes more than gloves and goggles. The liquid irritates skin and eyes, with phenolic components making respiratory exposure risky. Training now includes regular fit checks on respirators, clear written protocols for chemical spills, and eyewash stations close to work areas. Storage containers carry hazard symbols per GHS norms, and suppliers often provide comprehensive MSDS to outline first aid and safe disposal. Regulatory agencies push for closed-system pumps and vapor extraction where possible, reducing inhalation and accidental contact risks.
Application Area
Most of the compound finds use as a fragrance and flavor intermediate, prized for notes reminiscent of smoke or leather in food and perfume markets. Chemical manufacturers tap it for specialty resin synthesis, where developers hunt for improved heat resistance or chemical durability. Agrochemical researchers test cresyl esters as potential carrier solvents for pesticides and herbicides, though not every test leads to a commercial winner. Medical researchers sometimes check modifications of the compound for antimicrobial or analgesic effects. It gets less attention as a solvent, but niche electronics applications—think plastic coatings or specialty ink—do exist thanks to its stability and chemical inertness under the right conditions.
Research & Development
Teams in industrial and academic labs continue exploring new synthesis routes for m-cresyl acetate that cut environmental impact and boost efficiency. Older methods used hazardous acids or gave off problematic byproducts; newer ones try solid acid catalysts or phase-transfer systems. Research tracks the effectiveness of m-cresyl acetate as a starting point for complex organic molecules, including pharmaceuticals or polymers with custom features like UV resistance or hydrophobicity. Analytical groups refine detection and quantification techniques, using high-performance liquid chromatography or advanced mass spectrometry for both process control and toxicology testing.
Toxicity Research
Concerns around the compound's safety showed up as soon as industrial-scale production ramped up. Toxicologists classify m-cresyl acetate as a mild to moderate irritant, warning against repeated skin or inhalation exposure. Animal studies point to liver and kidney effects at high doses, following the same metabolic breakdown observed with related cresol esters. Regulators in the US and Europe set workplace exposure thresholds based on these findings. Ongoing research checks for long-term carcinogenic and mutagenic risks, with current evidence suggesting risks climb only with sustained, careless exposure far above norms. Environmental studies focus on breakdown pathways, since slow biodegradation may lead to accumulation in aquatic systems near manufacturing sites.
Future Prospects
As demands for safer, greener chemicals rise, researchers look at m-cresyl acetate with fresh eyes. Green chemistry pushes new synthesis catalysts and recycling strategies to limit waste and byproduct contamination. Applications in smart materials and drug delivery open up with controlled-release formulations using aromatic ester cores like m-cresyl acetate. There’s steady interest in modifying the core molecule for bespoke polymers or aromatic building blocks, feeding the hunger for lighter, more robust plastics and specialty resins. Regulatory compliance and worker protection will remain at the front of R&D agendas, pushing manufacturers to design processes and packaging that lower risks at every step. The strong platform m-cresyl acetate provides for innovation keeps it squarely in the sights of both chemical engineers and research scientists mapping out the next generation of performance materials and safe work environments.
What is M-Cresyl Acetate used for?
Where You Find M-Cresyl Acetate
Walk through the halls of any electronics manufacturing facility and odds are high you'll find M-Cresyl Acetate playing a part in the process. Most people have never heard of it, but this chemical sits in plenty of chemical cabinets for a reason—it's reliable and gets results. M-Cresyl Acetate finds its way into formulations for specific resins and plastics. Its chemical structure, part of the cresol family with an attached acetate group, helps it act as a solvent and chemical intermediate.
Printed Circuit Boards and Electronics
Modern electronics would struggle to function without high-performance insulators, and that's where M-Cresyl Acetate comes in. Factories that make printed circuit boards (PCBs) need materials that keep electricity in check while allowing the device to stand up to heat and long-term use. The production of polyimide resins leans heavily on M-Cresyl Acetate as a key raw material. These resins get layered onto electronics boards and flexible wiring—think smartphones, medical devices, and automotive control systems. The use of M-Cresyl Acetate means other solvents with tougher safety and environmental profiles get left behind, which makes a difference in workplace safety.
Specialty Coatings and Adhesives
Working with adhesives and coatings calls for precise results; otherwise, products peel, fail, or don't last the way they should. Experienced chemists rely on M-Cresyl Acetate to dissolve advanced polymers so they can apply smooth, even coatings on surfaces. Imagine sealing the back of a circuit board to protect it from moisture. That protective film often relies on materials processed with M-Cresyl Acetate. Its ability to mix without breaking down improves shelf life and helps production lines avoid costly delays.
Potential Risks and Safety Concerns
No chemical comes without its drawbacks. Like many industrial solvents, M-Cresyl Acetate can irritate skin, eyes, or lungs if handled carelessly. Breathing in fumes or spilling the liquid can cause health problems, so labs and factories set up proper ventilation and provide protective gear. Long-term exposure or high concentrations pose higher risks, prompting calls for strict storage and handling protocols. Seeing labels and safety posters everywhere might seem excessive, but they save lives.
Challenges for Industry and Environment
Factories keep a close eye on the chemicals they use thanks to increasing regulation. The Environmental Protection Agency tracks substances like M-Cresyl Acetate to reduce air and water pollution. Disposing of unused stock correctly prevents chemical leaks and soil contamination. Over the last decade, more companies search for greener alternatives while still depending on M-Cresyl Acetate for tasks where substitutes fall short. Success stories from companies using recovery systems and investing in worker safety training remind us that progress is possible even with established chemicals.
Where to Go From Here
Research never stands still. Chemists who spend time in the lab tinkering with new formulations sometimes uncover solvents or production methods that reduce overall risk. Partnering with universities and sharing findings, businesses find ways to make electronics sustainably. Maintaining clear guidelines on chemical use—and sharing best practices between companies—helps everyone work smarter. Consumers might never see the bottle labeled ‘M-Cresyl Acetate,’ but the products in their pockets and cars rely on the consistent performance this chemical delivers. The push for safety, transparency, and responsible use continues.
What are the safety precautions when handling M-Cresyl Acetate?
Understanding the Risks
M-cresyl acetate isn’t the kind of chemical you want to handle without respect. Stepping into the lab, the first thing you notice is the sharp, biting scent that clings to the air when the bottle opens. This is one of those organic compounds that brings several hazards to the table. Forgetting gloves or protective eyewear can lead to serious consequences, ranging from skin irritation to respiratory trouble. During my own years in chemical research, a spill was never just a minor inconvenience — it became a learning moment about how quickly things can go sideways without good habits.
Personal Protection Makes a Difference
Anyone who’s spent time working with organic solvents knows the drill: gloves, goggles, long sleeves, and a lab coat aren’t just boxes to tick. M-cresyl acetate absorbs through the skin and can cause redness, burning, or even systemic effects with enough exposure. Disposable nitrile gloves work best here. For eyes, I found wraparound goggles more effective than basic safety glasses, since even a tiny droplet can sting like crazy and damage sensitive tissue. Standard white lab coats add a barrier, but sometimes a splash apron is worth it, particularly during transfers or weighing.
Ventilation: More Than Just a Lab Rule
Inhaling vapors may lead to headaches, dizziness, or worse. Fume hoods offer the only reliable fix for airborne exposure. More than once, I’ve seen someone underestimate how strong these fumes can get. If a hood isn’t available, the next best thing is to set up fans and keep windows open, but nothing beats a properly working hood. Air quality sensors don’t lie — spikes pop up fast during open handling. Even with the hood’s glass sash lowered, keep your face at a distance.
Spill Control and Emergency Planning
Large or small, spills demand quick action. During a minor mishap in my early days, I scrambled for absorbent pads and found out the hard way that paper towels only spread the mess. Specialized spill kits turn a disaster into a minor speed bump. Neutralize using absorbents made for organic chemicals, and always seal contaminated materials in labeled waste bags for disposal. Every decent lab drills its staff on the eye-wash station and safety shower locations — seconds really count if someone gets splashed.
Safe Storage and Waste Handling
With reactive chemicals like m-cresyl acetate, never grab just any bottle for storage. Containers need tight-fitting caps, and labels must be clear and up-to-date. Keep bottles away from acids, oxidizers, and heat sources. Once, a loose cap sent vapors drifting throughout our chemical storage area, tripping alarms and forcing us to reorganize the shelves. Waste has its own rules: collect all residues and contaminated items in approved solvent waste containers, not the regular trash.
Training and Continuous Awareness
No one remembers everything from safety briefings, so regular refresher courses matter. I've seen even experienced chemists cut corners, leading to near-misses that could have gone much worse. Never hesitate to ask questions or double-check procedures if something looks off. Peer accountability lifts everyone — someone noticing an open flask or a missing label can save a big headache later.
Mistake-Proofing with Better Habits
Working with chemicals like m-cresyl acetate means developing habits that stick. Rushing leads to slips, so slow down and check everything twice. Clean up as you go, not just at the end of a session. These lessons, learned after a few too many close calls, have shaped how I approach not just m-cresyl acetate, but every bottle on the shelf.
What is the chemical formula of M-Cresyl Acetate?
A Close Look at a Common Ester
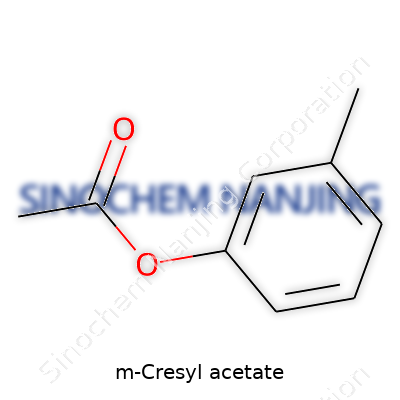
M-Cresyl acetate doesn't show up in headlines, but it plays a quiet role in chemistry labs and manufacturing plants. For anyone wondering what it actually is, the chemical formula is C9H10O2. In plain terms, this compound results from an acetyl group joining up with m-cresol. Chemists refer to it as meta-cresyl acetate, which means the methyl group sits on the middle carbon of the benzene ring.
Personal Perspective in Workspaces
After spending countless afternoons in the lab, one of the first lessons I learned involved double-checking chemical structures before reaching for glassware. Meta-cresyl acetate always stood out as one of those esters with a clear, sharp smell—hard to miss on the bench but not dangerous to work with if you follow basic ventilation and glove policies. Once, mixing it in a small-scale synthesis for a fragrance intermediate, I appreciated how a bottle of colorless liquid masked a lot of complex science underneath the surface. The structure gives you a hidden story: nine carbons, ten hydrogens, and two oxygens, hooked together in a specific configuration that allows it to act as both a solvent and a chemical intermediate.
Why Chemical Structure Matters
Every atom in that formula counts. Having a chemical formula on hand means knowing what to expect if it interacts with another substance or heats up above room temperature. Breathing in fumes from any aromatic ester, including this one, becomes a health risk if you skip proper precautions. Agencies like the National Institute for Occupational Safety and Health remind workers that aromatic esters can irritate skin and eyes, especially in production quantities. That’s why personal protective equipment isn’t just a suggestion—good practices keep accidents from turning into problems. From pesticide manufacturing to making fragrance compounds, the formula keeps chemists safe and precise.
Health, Safety, and Responsibility
Remember the time a colleague ignored the safety datasheet and handled m-cresyl acetate with bare hands? His skin tingled for hours—clearly a lesson in respecting the molecule and its formula. Even small amounts deserve respect. Detailed handling guidelines have always made the rounds in research and industry newsletters: keep containers sealed, work in ventilated spaces, and use chemical splash goggles. Knowing the numbers on the formula helps labs and plants design good ventilation and spill response plans without guesswork.
Paths Toward Safer Workplaces
Some changes can help everyone breathe easier. Digital inventory systems and barcoding chemicals by formula prevent mix-ups. Regular training keeps seasoned workers and newcomers wise to fresh risks. I’ve seen industries invest in better fume hoods and switch to less hazardous solvents for processes where possible. Even with familiar compounds like m-cresyl acetate, transparency and clear labeling support a safe environment for everyone—from the warehouse floor to the research suite.
Shared Knowledge, Shared Safety
Every bottle marked with C9H10O2 didn’t get there by accident. Teams working together and passing on their experience go a long way. The formula gives clarity in a world where confusion can bring harm. Looking out for each other, sharing what works, and never assuming you know enough—that’s how labs thrive, no matter what’s in the flask.
How should M-Cresyl Acetate be stored?
Why Storage Matters
Over the years in both labs and warehouses, plenty of people have seen accidents turn into expensive headaches just because the basics of chemical storage got ignored. M-Cresyl Acetate brings its own set of risks, and, just like any solvent-based material, calling for respect and straightforward safety precautions.
Understanding the Hazards
Keeping M-Cresyl Acetate safe starts with knowing what you’re dealing with. This compound can release strong fumes if left open, and even small spills are enough to irritate eyes or skin. Working with it in poorly ventilated spaces could fill the air with vapor, which does no favors for anyone’s lungs.
Choosing the Right Container
M-Cresyl Acetate belongs nowhere near leaky jars or old food containers. Heavy-duty glass or metal bottles with tight-sealing caps handle this chemical best. I’ve seen accidents where those cheap, brittle plastics crumbled within weeks of use, leading to leaks and sticky cleanups.
Temperature and Lighting
Heat ramps up evaporation rates and boosts pressure inside containers. Direct sunlight also breaks down many chemicals over time. Tucking M-Cresyl Acetate onto a cool, shaded shelf—away from heat and electrical sources—keeps things stable. Standard guidelines put the sweet spot for storage under 25°C, with extra care in summer if you’re flashing through hot spells without air conditioning.
Organization and Labeling
Mislabeled bottles have started more than a few emergencies. Clear, chemical-resistant labels with the chemical’s name and hazard symbols protect everyone, including the late-night shift. Shelving that separates M-Cresyl Acetate from acids, strong bases, or oxidizers blocks the chance of violent reactions. I’ve watched experienced techs catch their mistakes in time, all because an obvious tag gave them pause.
Ventilation and Spills
Keeping M-Cresyl Acetate in a well-ventilated chemical cabinet shrinks risk. Fume hoods and local exhausts pull harmful vapors away from worktops. Absorbent spill kits and proper gloves offer a reliable backup in case of accidents. Someone once told me, ”You only forget your goggles once,” and the same goes for gloves in these situations.
Long-Term Storage Safety
Checking inventories every couple of months helps spot cans that might be bulging or corroding. Marking dates on bottles keeps stock fresh, and following expiration dates means you don’t get caught by surprise with degraded material. Disposal through certified hazardous waste centers stops chemicals from leaking into drains or ending up in regular trash, where they clearly don’t belong.
Training and Awareness
Training turns safe storage from a rule on a wall into a real habit. I’ve seen teams who talk through possible scenarios, and they handle surprises with calm. Simple drills build confidence in youthful workers and remind seasoned staff of details—like how just a drop or two outside the bottle can become a slip hazard.
Practical Solutions for a Safer Workplace
Enough stories point to one truth: good habits, not luck, keep accidents at bay. Simple storage steps—tough containers, clear labels, well-ventilated spots—cut the real risks to people and property. Add regular training, and the odds of a chemical accident drop even further. That’s how anyone manages M-Cresyl Acetate responsibly.
What are the physical and chemical properties of M-Cresyl Acetate?
Understanding M-Cresyl Acetate Beyond the Lab
M-Cresyl Acetate shows up in industries that rely on precise reactions and outcomes. Its chemical formula is C9H10O2, which means it carries a distinct combination of carbon, hydrogen, and oxygen. At room temperature, this compound presents as a clear liquid, slightly yellow, sometimes carrying the faint, medicinal note known to many lab workers. It isn’t explosive or particularly volatile, but safety remains crucial due to its aromatic nature.
Boiling, Melting, and Miscibility
My early lab days often involved adjusting distillation processes, so boiling points became more than just textbook trivia. M-Cresyl Acetate boils around 230°C, putting it firmly into the range where heating must be watched closely to avoid accidents. This stability at higher temperatures adds value for producers in dyes or specialty chemicals.
On the lower end, the melting point hovers right around room temperature—sometimes dipping just below. This tells us storage in cooler environments calls for attention, as unexpected solidification can mess with automated systems. The liquid state makes transfer easier, but it asks users to handle it in tight, well-sealed vessels to guard against evaporation and contamination.
Chemical Behavior Counts
Chemically, M-Cresyl Acetate stands out for its resilience. It's an ester formed by the reaction of m-cresol and acetic acid. I remember more than one rushed experiment with acetic acid, where improper ventilation left the unmistakable tang in the air. M-Cresyl Acetate keeps most of that under control but does release a mild aroma when left open.
It's relatively resistant to breakdown under normal conditions. Still, exposure to strong acids or bases can lead to hydrolysis, splitting it back to its original cresol and acetic acid forms. I learned that spill cleanups need more than just water and towels—using a basic neutralizer works better, cutting down both risk and cleanup time.
Solubility and Usage Realities
In terms of solubility, water isn’t its friend. M-Cresyl Acetate doesn’t mix well with water, which explains why it tends to separate out in waste streams. Cleaning up after spills from glassware or equipment means reaching for solvents like alcohol or ether. This reluctance to mingle with water keeps it useful in organic syntheses, where accidental mixing causes more problems than it solves.
The real magic of M-Cresyl Acetate unfolds in its role as a solvent or intermediate. Anyone who’s spent time in paint or ink labs knows these are high-stakes environments for purity and consistency. Its moderate volatility lets it carry pigments smoothly yet evaporate at a convenient rate, giving paints and coatings that reliable finish.
Managing Risks and Moving Forward
Safe handling and stewardship mean wearing gloves, goggles, and ensuring airflow in the workspace. I’ve watched seasoned techs skip the gloves and regret it later—skin exposure can lead to irritation, so no shortcuts. Fire risk isn’t extreme, but in industrial quantities, vapors call for responsible storage away from sparks or open flames.
Finding alternatives with less environmental impact remains a goal, but as long as M-Cresyl Acetate’s properties stay essential for certain chemical pathways, improving handling and waste treatment are steps that matter. Strong protocols keep people and the environment safer, letting industries make use of this compound with fewer worries down the line.
| Names | |
| Preferred IUPAC name | 1-Acetyloxy-3-methylbenzene |
| Other names |
m-Cresyl acetate 3-Methylphenyl acetate Acetic acid m-cresyl ester meta-Cresyl acetate 3-Methylphenyl ethanoate |
| Pronunciation | /ˈɛmˌkriːsɪl əˈsiːteɪt/ |
| Identifiers | |
| CAS Number | [101-39-3] |
| Beilstein Reference | 0638417 |
| ChEBI | CHEBI:77819 |
| ChEMBL | CHEMBL517324 |
| ChemSpider | 16719 |
| DrugBank | DB14158 |
| ECHA InfoCard | 100.041.586 |
| EC Number | 211-889-1 |
| Gmelin Reference | 8083 |
| KEGG | C07327 |
| MeSH | D003404 |
| PubChem CID | 89387 |
| RTECS number | GO7875000 |
| UNII | N1I66F73F7 |
| UN number | UN1993 |
| Properties | |
| Chemical formula | C9H10O2 |
| Molar mass | 164.20 g/mol |
| Appearance | Colorless to yellow liquid |
| Odor | Faint, pleasant odor |
| Density | 1.08 g/cm³ |
| Solubility in water | insoluble |
| log P | 1.98 |
| Vapor pressure | 0.05 mmHg (20°C) |
| Acidity (pKa) | 14.45 |
| Basicity (pKb) | 15.0 |
| Magnetic susceptibility (χ) | -62.5e-6 cm³/mol |
| Refractive index (nD) | 1.530 |
| Viscosity | 2.35 cP (25°C) |
| Dipole moment | 2.87 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 362.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -380.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3940.0 kJ/mol |
| Pharmacology | |
| ATC code | D02AE02 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | Harmful if swallowed. Causes serious eye irritation. Causes skin irritation. May cause respiratory irritation. |
| Precautionary statements | P261, P280, P305+P351+P338, P304+P340, P312 |
| Flash point | 110 °C |
| Autoignition temperature | 490°C |
| Explosive limits | Explosive limits: 0.9–6.6% |
| Lethal dose or concentration | LD50 (oral, rat): 1960 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1,770 mg/kg (oral, rat) |
| NIOSH | WI0450000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for M-Cresyl Acetate: "No OSHA PEL established |
| REL (Recommended) | 5 ppm |
| IDLH (Immediate danger) | 250 ppm |
| Related compounds | |
| Related compounds |
Phenol Cresol M-Cresol P-Cresol O-Cresol Phenyl acetate Anisole |

