The Real Story Behind Lithium Perfluorooctanesulfonate: Unpacking the Science, Risks, and Prospects
Historical Development
Lithium Perfluorooctanesulfonate—often called LiPFOS among chemists—carries a legacy stretching back to the middle of the twentieth century, trailing behind the story of perfluorinated compounds that helped launch the modern age of chemistry. This compound came on the scene thanks to the early enthusiasm for perfluorinated surfactants, driven by the search for chemicals resistant to heat, acids, and bases, ones that just wouldn't quit whether used in electronics or firefighting foam. By the 1960s, research into the versatility of these molecules, especially their ionic forms like lithium salts, picked up steam. Industry players realized these products had some seriously useful properties—hard to ignore, but even harder to manage given their stubborn persistence in the environment. Over the decades, regulators and researchers started raising pointed questions. Communities living near manufacturing sites called out contamination, demanding answers and accountability as more data on PFOS and its relatives—LiPFOS included—emerged.
Product Overview
At its core, Lithium Perfluorooctanesulfonate is a surfactant. Its structure features a lithium ion paired with a perfluorinated octane sulfonate anion. This mouthful just means that the molecule balances a lithium atom against an extraordinarily stable carbon-fluorine tail linked to a sulfonate group. Unlike common detergents or soaps, LiPFOS doesn't break down easily when dumped into a river or left out in the sun. Its resilience makes it tough—exceptionally persistent everywhere it goes. Industry latched onto these features to develop products ranging from electronics to chemical processing, banking on its thermal stability and ability to keep surfaces free of static, grime, or dangerous sparks.
Physical and Chemical Properties
Anyone handling LiPFOS up close knows it exists as a white powder or crystalline solid, practically odorless and usually dusts up easily when poured. It dissolves in water, producing a clear to slightly hazy solution, and holds up even under punishing temperatures. Chemically, the carbon-fluorine bonds inside the molecule refuse to break, even when exposed to most acids or bases. This stubbornness might sound appealing—until it appears in groundwater after a factory spill. The molecule’s lipid-phobic and water-loving ends give it the excellence needed for specialized coatings, but that's the same quality that lets it linger in natural ecosystems.
Technical Specifications and Labeling
Most manufacturers supply this compound in grades intended for industrial, rather than consumer, use. Labeling requirements demand hazard warnings due to persistence and toxicity, typically classifying it as hazardous under most chemical safety systems. The product often comes marked with its Chemical Abstracts Service (CAS) number and warning symbols dictated by global standards. The compound’s solubility, melting point, and information on safe storage must show up right on technical datasheets, reflecting not just regulatory rules but also a measure of transparency owed to those regularly in contact with the compound.
Preparation Method
As with many notorious synthetic chemicals, the path toward LiPFOS leads through complex fluorination chemistry. The traditional route relies on assembling the perfluorooctanesulfonyl fluoride backbone, then exchanging its terminal group for lithium, usually through a simple acid-base reaction with lithium hydroxide. Plants producing this compound handle harsh fluorinating reagents and precise reaction conditions—lab coats and respirators a must, unless you like playing with fire. Synthesis does not produce a tidy product either; the process chugs out byproducts and contaminated streams that need specialized treatment before anyone can even think of discharge.
Chemical Reactions and Modifications
LiPFOS demonstrates high resistance to almost everything thrown its way—meaning it barely reacts unless pushed through severe chemical processes like high-energy incineration. Attempts to degrade or transform the substance have challenged chemists for years. Standard oxidative degradation methods, such as incineration above 1100°C or advanced electrochemical oxidation, show moderate effectiveness at best. Environmental chemists spend their days testing catalysts, seeking new ways to pry apart the carbon-fluorine bonds, but success comes slowly. The fortunate upside, in a tightly controlled setting, is this chemical doesn't turn into unknown hazards easily. The drawback crops up in the wild, where it just refuses to quit.
Synonyms and Product Names
Beyond its systematic name, this compound goes by LiPFOS, lithium perfluorooctyl sulfonate, and a handful of acronym-heavy alternatives in patent filings. Most chemical supply catalogs state its molecular formula C8F17LiO3S. Its PFOS backbone sets off loud alarm bells for anyone familiar with environmental safety bulletins, while the lithium part hitches its legacy to battery technology and electrochemical research. In some technical reports or older studies, the compound appears under perfluorooctanesulfonic acid lithium salt or similar translations.
Safety and Operational Standards
I can't overstate the need for robust operational safeguards around compounds like LiPFOS. Ever since perfluorinated chemicals grabbed regulators' attention, workplaces dealing with them have faced tougher occupational exposure limits. Employees must wear personal protective gear to avoid inhaling dust or handling contaminated gear barehanded. Airborne particle monitoring, regular surface cleaning, and dedicated waste management protocols all anchor daily routines in these settings. Specialists who respond to spills must work fast, as LiPFOS binds tenaciously to soils and surfaces, setting tight windows for mitigation. Listening to health experts and following government-mandated tracking systems don't just tick compliance boxes—they help build a culture where worker safety and environmental stewardship become non-negotiable routines.
Application Area
The usefulness of LiPFOS crops up in several advanced fields. Electrochemists use it as an electrolyte ingredient in specialty batteries, especially ones where standard salts degrade under high loads or fail to deliver cycle stability. In semiconductors, device manufacturers sometimes include LiPFOS for antistatic and cleaning treatments, leveraging its surfactant power in contamination-sensitive environments. For a while, some found it handy in fire suppression foams, where only the most robust surfactants stop high-octane fires in their tracks—a practice that communities and regulators questioned sharply once groundwater readings started showing unwelcome levels downstream from airports and military bases. Its enduring role in niche research, especially in labs churning through advanced materials science, underpins a dilemma: balancing the need for specialized performance against undeniable environmental and health risks.
Research and Development
The scientific world keeps investing in understanding and perhaps replacing this stubborn molecule. Post-2000, growing awareness around PFOS-related compounds, including LiPFOS, kicked off a gold rush in analytical chemistry to detect even trace levels in water, soil, and blood. Investigators focused on unraveling its long-term movement through the environment, bioconcentration in wildlife, and consequences in exposed human populations. R&D teams across public and private sectors continue the hunt for alternative chemistries—molecules with the perks of perfluorinated surfactants, but without the unchecked persistence or toxic legacy. Progress appears in fits and starts. Some new surfactants escape the regulatory crosshairs for now but nobody wants a repeat of the PFOS saga. Vigilant peer review and open sharing of testing data now form essential pillars for any R&D built on this foundation.
Toxicity Research
Tales from the field highlight the toxic footprint of perfluorinated chemicals, LiPFOS among them. Animal studies, everything from fish to rodents, capture a pattern: buildup in blood or liver over time, impacts on immune systems, disruption of hormones, and, in some grim scenarios, declines in reproductive success. Human epidemiology points at worrying links with certain cancers, high cholesterol, and developmental delays in children born to exposed families. No one claims every molecule produces disaster, but enough biological red flags have surfaced for global health agencies to advocate for restraint. The fierce stability of carbon-fluorine bonds spells long half-lives in human bodies. In the communities I’ve spoken to near manufacturing sites, parents and local officials rarely take reassurance from “trace exposure” arguments; they want clean water and answers.
Future Prospects
Looking at the road ahead, the conversation around LiPFOS spotlights tough trade-offs for policymakers, industries, and society. Advanced manufacturing may still rely on compounds in this class for years, mostly where no workable substitutes deliver. The demand for shorter supply chains and “greener” products creates pressure, but lesser-known substitutes sometimes skate through testing gaps. On the regulatory front, countries have started banning or tightly restricting perfluorinated substances—including LiPFOS—while supporting trials on remediation and destruction technologies. Getting rid of legacy contamination, in soil and aquifers, could take decades—sometimes, new chemicals wind up echoing the same problems if scrutiny wavers. Real progress only comes from listening to frontline communities, promoting transparent science, and pushing industry to accept upstream responsibility rather than passing future damage down the line. Our lasting responsibility sits at the intersection of chemistry, ethics, and public health.
What is Lithium Perfluorooctanesulfonate used for?
Manufacturing with More Than Just a Salt
Lithium perfluorooctanesulfonate isn’t a household name, but it sits quietly on the shelves of companies working on electronics, firefighting foams, and specialty coatings. Industries rely on its unique properties—mainly the dramatic surface tension lowering and chemical stability. This chemical often lands in the electrolyte formulas for lithium-ion batteries. Battery makers are in a race, fine-tuning every element to squeeze out longer runtimes and better safety. Adding lithium perfluorooctanesulfonate can help batteries handle higher voltages and work through bigger temperature swings. In the search for durable electronics, nothing quite behaves the way this compound does.
In Coatings and Firefighting
Lithium perfluorooctanesulfonate can enhance the performance of specialty coatings. I’ve seen it used to prevent industrial-scale corrosion, especially on metal pipes and storage tanks out in harsh weather. These coatings spread better across surfaces and resist water, oil, and other hazards once the compound joins the mix. Traditional chemistry rarely achieves this balance between slickness and toughness. For anyone working near the ocean or in high-humidity plants, those factors spell real consequences—less downtime, safer workers, equipment that can last longer than usual.
Firefighting foam relies on this chemical’s surface activity. Fires fueled by oil or gas throw major challenges at first responders. Specialized foams can smother those fires quickly, and this compound helps the foam spread thin and fast, forming a barrier that shuts out oxygen. Growing up near a chemical plant, I watched crews run monthly foam drills. They had to trust their gear. The quick action of these foams gives people a fighting chance. At the same time, environmentalists and public health experts are paying closer attention to what happens after the fire is out. Traces of these chemicals can linger in water or soil once the foam washes away.
The Tough Question: What Happens After Use?
My biggest concern and probably everyone else’s who follows environmental news is about contamination. Perfluorinated chemicals, including lithium perfluorooctanesulfonate, don’t break down easily. They can drift into water systems and build up in fish, animals, and even people. Scientists now watch for perfluorooctanesulfonate and related compounds in groundwater near airports, military bases, and factories. Documented health problems range from hormone disruption to cancer risks.
Some governments push for stricter rules. They call for improved containment methods, alternatives that degrade naturally, or even switching to non-fluorinated chemistries where possible. My experience looking at how these policies play out shows it takes years, but some companies are making the switch. A few have piloted new battery additives based on boron or silicon, or foams using plant-derived surfactants. The technology isn’t perfect yet, but change often starts on the fringes before it takes off. Veterans in chemical manufacturing know half the battle involves getting everyone—regulators, suppliers, users—to agree that the long-term risks outweigh the short-term performance gains.
Thinking About Solutions
The story of lithium perfluorooctanesulfonate reflects a broader challenge. People want better products and more safety, but demand keeps clashing with clean water and air. Real transparency about ingredients and investments in green chemistry make a difference. I’ve met scientists testing enzymes that digest persistent chemicals, venture capitalists funding non-toxic alternatives, and workers retraining for safer jobs. That’s progress worth supporting, even if the path isn’t smooth or fast. Recognizing this chemical's impact puts the focus on communities and industries working to balance innovation with responsibility.
Is Lithium Perfluorooctanesulfonate hazardous to health?
The Science Behind Lithium Perfluorooctanesulfonate
Lithium Perfluorooctanesulfonate falls under the class of chemicals known as per- and polyfluoroalkyl substances (PFAS). People often refer to PFAS as “forever chemicals” because these compounds barely break down in nature. I remember hearing this term on the news and thinking, if these compounds stick around for decades, that spells trouble for health and the environment.
This compound sees use in many industries, from electronics to firefighting foams. Over time, everyday activities—like using non-stick cookware or washing water-resistant fabrics—can add trace amounts to air, dust, and drinking water. Even those trying to avoid chemicals might already have some PFAS in their blood.
Health Concerns Linked to PFAS
Growing up near a manufacturing town, I often wondered about the weird tastes in tap water and the odd fish warnings on riverbanks. Studies now link PFAS exposure to a laundry list of health concerns, and Lithium Perfluorooctanesulfonate sits among that group. Research points toward increased risks of certain cancers, hormone disruption, immune system issues, and developmental problems in children. The Centers for Disease Control and Prevention (CDC) and the Environmental Protection Agency (EPA) both warn about cumulative exposure to such chemicals. They note even low levels can build up in the body over years, with effects that stretch into adulthood.
I’ve read studies from communities too close to PFAS factories. People living in those neighborhoods report higher than average rates of thyroid disease and high cholesterol. Some children experience higher rates of asthma and behavioral problems. Health authorities investigate these outbreaks, and more often than not, they find traces of PFAS in drinking water or soil.
Industry Response and Regulatory Challenges
Companies have pushed to replace especially persistent PFAS chemicals, but sometimes the substitutes raise their own safety questions. Lithium Perfluorooctanesulfonate often comes up in technical papers as a “safer” alternative, but regulatory researchers urge caution. Europe moved to restrict the use of perfluorooctanesulfonate and related compounds years ago. The United States lags behind, but some states set their own stricter levels for water contamination. These policy moves don’t always keep up with emerging science or shifting industry practices.
What Can Help Address the Problem?
Filtering out PFAS at water treatment plants requires new equipment and investment. I’ve seen community groups pool together resources to fund testing and install household filters. This grassroots approach works in the short term but only covers a handful of homes at best. Broader testing, public notification, and better cleanup measures all matter. Health professionals need to keep patients informed, not just about possible risks but also about signs to watch for, especially in children and pregnant women.
Scientists continue to call for clearer rules on reporting PFAS use and stronger action on cleanup. Pushing manufacturers toward safer alternatives can make a real difference. Consumers armed with knowledge drive demand away from products linked to long-term harm. Everyone has a role in solving this problem—from policymakers crafting enforceable standards, to industry leaders adopting safer materials, to families learning more about what’s in their water and products.
What are the storage requirements for Lithium Perfluorooctanesulfonate?
Understanding What’s at Stake
Lithium Perfluorooctanesulfonate falls under a class of chemicals known for their persistence and unique properties. In the lab or out on the shop floor, improper storage can turn a useful chemical into a safety headache. Every good chemist or facility manager knows that storing chemicals right is about more than just keeping labels visible on a shelf—it’s a big part of keeping people safe, protecting the environment, and making sure that investment in supplies doesn’t end up wasted.
Why Storage Gets Complicated
With Lithium Perfluorooctanesulfonate, you’re dealing with a substance that reacts poorly if you let it soak up moisture. Humidity in storerooms is every bit as dangerous as a cracked seal on a bottle. From personal experience, I’ve seen what a few unnoticed drops of water can do in a storeroom: corrosion, ruined samples, and, in the worst cases, chemical reactions that put people at risk. A dry, climate-controlled space is essential.
Common Dangers and How to Watch Out for Them
The compound doesn’t just quietly sit if its environment isn’t sorted out. Besides humidity, there’s the issue of temperature swings. Many substances, and this one included, don’t belong anywhere near a heat source. Excess heat can encourage breakdown or evaporation, turning a stable powder into an airborne hazard. Over the years, I’ve noticed everything from sunbeams sneaking in through a window to poorly-placed radiators giving headaches—both to people and to chemicals. Keeping storage away from heat and out of direct sunlight is a straightforward step, but it gets missed too often.
Proper containers make a difference. Sturdy, airtight bottles made from compatible materials—think HDPE or glass with PTFE linings—lock moisture out and prevent leaks. Shoddy containers invite accidents. From a safety standpoint, clear and consistent labeling matters as well; a hastily written note or a faded sticker can have real consequences if someone misreads it during a busy morning. I’ve seen confusion in shared labs where two nearly identical containers ended up swapped. Routine checks and tight rules on how everything’s labeled and shelved cut down on that risk.
Thinking Long-Term
PFOA-related chemicals earned a reputation not just for their performance in industry, but for their toughness in the environment. Every time a container leaks, the risk isn’t just for the worker on shift—the mess can leave traces for years, complicating cleanup and influencing future use of that space. This makes secondary containment a smart investment. A simple tray or bin on a shelf, just big enough to hold a spilled bottle, can make all the difference if a seal fails or a bottle tips over. In facilities using larger quantities, dedicated chemical cabinets with spill containment offer extra peace of mind.
Documenting Everything
Documentation ties it together. If you track inventory, note expiration dates, and keep a log of inspections, surprises drop off. Good records make audits smoother and put teams in a much better spot if regulators ask questions. From what I’ve seen, digital logs cut down on headaches—no digging through binders, no missing paperwork. It’s simple: tighter control equals fewer emergency calls and less wasted product.
Room for Improvement
No storage setup stays perfect forever. Facilities should train new staff regularly, update policies, and watch for signs of changing conditions, like humidity spikes in summer. Ventilation checks, fresh absorbent pads, and routine cleaning keep the risk low. That’s how you protect staff, secure material investments, and make sure communities downstream don’t pay the price for small mistakes in a chemical cupboard.
How do you safely handle Lithium Perfluorooctanesulfonate?
Real Risks Deserve Real Respect
I’ve worked around dangerous chemicals more than once, and Lithium Perfluorooctanesulfonate, or LiPFOS, sits among the trickier materials. It’s easy to treat chemicals as just another lab supply, but LiPFOS highlights why habits matter. This compound combines lithium’s high reactivity with the persistence and toxicity of perfluorinated substances. My colleagues and I learned quick that shortcuts invite trouble.
Direct Contact: Protection Starts With Gear
Putting on gloves and goggles is the low bar for handling LiPFOS. The stuff can cause harm if it touches skin or splashes in the eyes. I always check the gloves for pinholes and make sure my sleeves overlap. A lab coat with snug cuffs makes a difference, especially if you get startled mid-task. Nitrile gloves work—latex won’t block everything, and the risk of skin absorption never quite vanishes.
Breathing Easy: Air Quality Matters
Fumes sneak up. LiPFOS, in dust or solution, drifts and finds its way everywhere without good airflow. A well-maintained fume hood helps more than most imagine. I keep the sash low, stay mindful of splashes, and never stir up powders outside the hood. Respirators seem bulky, but after coughing through a simple experiment years ago, I do not skip that mask if any dust escapes.
Spills and Storage Shape Safety Culture
I remember cleaning up a minor spill years back. Someone grabbed a dry paper towel, forgetting how quickly LiPFOS can migrate if not addressed in the right way. Absorbent pads and dry sand keep the spill from spreading. It’s important to scoop up everything, seal the waste, and keep it far from regular trash. Those cleanup steps seem tedious, but soil and water in the area do not recover from perfluorinated contamination. I always label the waste container with every detail imaginable—no guessing games for the next crew.
Why MSDS and Real Training Beat Guesswork
Every time a new technician joins, I pull out the Material Safety Data Sheet and walk through the hazards. It’s not just a formality. The MSDS calls out risks such as environmental toxicity, long-term persistence, and acute health impacts. Anyone who’s watched fish die in a waterway knows these warnings carry weight. Practical drills and walk-throughs help more than a checklist ever could.
Respecting the Material—And Each Other
After fifteen years around labs, I’ve seen how attitudes shape outcomes. Overconfidence or complacency leads to accidents—the sort nobody wants on their record. Teams that treat LiPFOS like everyday salt rarely last without issues. People working together, checking one another’s gear, and sharing close calls build real safety culture.
Looking for Safer Alternatives
Several companies have started switching away from perfluorinated compounds, especially as regulations tighten worldwide. Green chemistry doesn’t offer a fast fix, but every substitution reduces long-term harm. Pressure from workers and the public speeds up change, so speaking up in staff meetings and listening to real concerns always nudges progress.
Trust Grows from Consistency
LiPFOS isn’t kind to mistakes. Small habits—gloves, spill procedures, good ventilation—keep people from learning lessons the hard way. Every time I set up an experiment with LiPFOS, I remember that safety isn’t about rules; it’s about respect for each other and for the world outside the lab.
What is the chemical structure of Lithium Perfluorooctanesulfonate?
What’s Hiding Behind That Long Name?
Saying “Lithium Perfluorooctanesulfonate” out loud feels like a brain teaser. Most folks haven’t run into this compound unless they're deep into chemistry or materials science. But peel back the name, and you see a story about how modern industries lean heavily on specialty chemicals for performance and innovation. The molecule itself isn’t some vague or shapeless thing. It’s got a hard-and-fast formula: C8F17SO3Li.
What Does That Look Like?
Start by picturing eight carbon atoms strung together—think of a sturdy chain. In place of the usual hydrogen atoms that cling to carbon, you’ll find fluorine at every link, except where the chain ends with a sulfonate group. This full fluorination gives the chain its “perfluoro-” status. The last carbon hands off to a sulfonate group, which holds onto lithium. It’s this strong embrace with fluorine that makes the molecule so stable, while the sulfonate head loves to interact with water and ions. Chemists call this structure amphiphilic—the tail keeps away from water, but the head seeks it out. That split personality shows up in how companies use it.
The Role in Technology and Environment
Lithium salts pop up all over the world of batteries and specialty chemicals. This one goes further, popping up in everything from electronics to firefighting foams. That slick fluorinated carbon backbone makes the molecule stubborn against heat, acids, bases, and just about anything else you can throw at it. It keeps equipment running clean under tough conditions. But the same stability that powers performance also means it sticks around once released into the environment. These “forever chemicals” don’t break down easily. Recent reports have found traces of them in streams, soil, and even blood samples far from any chemistry lab. The Environmental Protection Agency and European Union have sounded the alarm about their persistence and possible health risks.
Health and Safety: The Real Consequences
People worry about long-lasting chemicals for good reason. Research links long-chain perfluoroalkyl substances to hormone disruption, developmental problems, and some cancers. The sulfonate versions—like this lithium salt—spark particular concern, since they seem to move into living things more easily. Nobody wants to see a world where cleaner technology brings unexpected side effects decades later. Prevention starts with knowing the structure and tracking how molecules like these flow from the factory into the wider world. Sharing this knowledge drives better choices at every step, whether crafting safer substitutes or fine-tuning treatment in wastewater plants.
What’s Next?
Innovation won’t slow down. Modern society depends on molecules that go above and beyond—lasting longer, resisting heat, turning unstable processes into things you can count on. At the same time, the demand for “greener” chemistry is growing louder. Concrete steps can limit the impact from these compounds. Some labs already chase after shorter, less stubborn chains. Others work on filters to pull out even tiny traces before they spread. It’s on scientists, companies, and policymakers to keep the conversation moving, using the hard facts—like that structure C8F17SO3Li—as ground zero for smarter, safer chemical design.
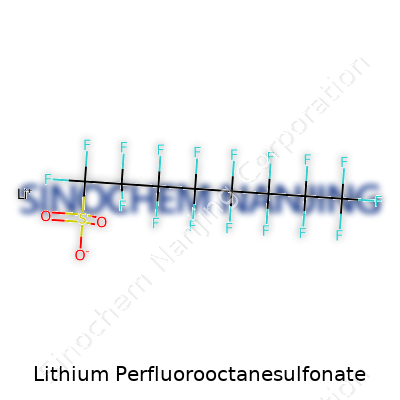
| Names | |
| Preferred IUPAC name | lithium 1,1,2,2,3,3,4,4,5,5,6,6,7,7,8,8,8-heptadecafluorooctane-1-sulfonate |
| Other names |
Lithium PFOS Lithium perfluorooctylsulfonate Perfluorooctanesulfonic acid lithium salt Lithium heptadecafluorooctanesulfonate |
| Pronunciation | /ˈlɪθiəm pɜrˌfluːəˌrɒkˌteɪnˌsʌlˈfəʊneɪt/ |
| Identifiers | |
| CAS Number | 29457-72-5 |
| Beilstein Reference | 4432115 |
| ChEBI | CHEBI:47499 |
| ChEMBL | CHEMBL4296977 |
| ChemSpider | 21792022 |
| DrugBank | DB11266 |
| ECHA InfoCard | 20c6e8b2-015a-444d-9e67-ec4b8227273e |
| EC Number | 206-991-8 |
| Gmelin Reference | 1622080 |
| KEGG | C18605 |
| MeSH | D017567 |
| PubChem CID | 11382817 |
| RTECS number | OV8775000 |
| UNII | B33U8P827I |
| UN number | UN3077 |
| Properties | |
| Chemical formula | C8F17LiO3S |
| Molar mass | 538.11 g/mol |
| Appearance | White powder |
| Odor | Odorless |
| Density | 1.64 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -0.18 |
| Vapor pressure | Negligible |
| Acidity (pKa) | -3 |
| Basicity (pKb) | -6.2 |
| Magnetic susceptibility (χ) | −52.2×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.3500 |
| Viscosity | 2.08 mPa·s (at 25 °C) |
| Dipole moment | 2.56 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 652.8 J·mol⁻¹·K⁻¹ |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and serious eye irritation, may cause respiratory irritation, may cause damage to organs through prolonged or repeated exposure, very toxic to aquatic life with long lasting effects. |
| GHS labelling | GHS05, GHS07 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H302, H318, H361fd |
| Precautionary statements | P261, P273, P280, P301+P312, P305+P351+P338, P337+P313, P501 |
| NFPA 704 (fire diamond) | 3-0-3-W |
| Lethal dose or concentration | LD50 (Oral, Rat): >2000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50: >2000 mg/kg |
| NIOSH | NIOSH: LU5600000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Lithium Perfluorooctanesulfonate: Not established |
| REL (Recommended) | 0.1 mg/m3 |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Perfluorooctanesulfonic acid Potassium perfluorooctanesulfonate Sodium perfluorooctanesulfonate Ammonium perfluorooctanesulfonate |

