Lithium Perchlorate: History, Properties, and the Road Ahead
A Long Road: The Story Behind Lithium Perchlorate
People sometimes overlook the stories behind everyday chemicals. Lithium perchlorate, formula LiClO₄, has roots reaching back to the early research on the chemistry of alkali metals. Since scientists began exploring oxygen-rich perchlorates over a century ago, lithium’s perchlorate salt got attention for more than just its eye-catching name. The material found its place during early advances in rocket propulsion and battery research in the 20th century. As the space race kicked off, need for better oxidizers pushed chemists to refine lithium perchlorate—part of a broader hunt for reliable, high-energy compounds that wouldn’t simply explode on the shelf or fall apart with humidity. Later, researchers gave it a new look as demand for high-performance electrolytes in batteries surged around the globe.
Getting to Know Lithium Perchlorate
Lithium perchlorate stands out thanks to a mix of unique characteristics. A white, often fine-grained crystalline solid, it easily dissolves in water and certain organic solvents. Its high solubility in polar solvents and impressive thermal stability make it a favorite in labs and industry. Some see it as just another perchlorate, but the lithium ion’s small size and charge density give this compound a completely different profile from, say, sodium or potassium perchlorate. Its melting point above 230°C offers stability that is useful in both research and industrial settings.
The Chemistry at Play
Anyone who has worked with lithium salts soon learns to treat them with respect. In its pure form, lithium perchlorate brings a potent oxidizing punch. Contact with even the smallest amounts of flammable substances can trigger rapid, sometimes violent reactions. It breaks down above 400°C to release oxygen—a trait that found it welcomed, for a time, in rocket propellant mixtures. Its hygroscopic nature, or tendency to absorb water, is worth noting as storage and handling demand strict control of moisture to avoid caking or decomposition.
Diving into Technical Details
The technical numbers often steer how lithium perchlorate gets used. Purity standards exist for pharmaceutical and battery grades, with high-end batches reaching above 99.9% LiClO₄ by weight. Its crystalline morphology ranges from powder to larger shapely grains, depending on how it’s made. Labels must warn about oxidizing power and give guidance on protective gear—goggles, gloves, and, for larger quantities, face shields and dust masks. Transport rules often keep it out of passenger aircraft, a nod to its oxidizing danger.
From Lab Bench to Factory Floor: Making Lithium Perchlorate
Production of lithium perchlorate usually follows an established route: reacting lithium chloride with sodium perchlorate, followed by purification and careful drying. Some industrial processes rely on direct oxidation of lithium chloride in perchloric acid, needing precise temperature control and continuous monitoring. Big facilities put safety systems in place to catch unexpected releases or runaway reactions. The finished product goes through several filtration and crystallization steps to shed impurities that could sabotage its later use in sensitive battery electrolytes or analytical chemistry.
The Many Faces of Lithium Perchlorate
Names pile up around this salt—lithium salt of chloric(VII) acid, lithium perchlorate monohydrate depending on hydration, and a few less common chemical handles in specialized applications. Around the world, it is listed under regulatory codes as an oxidizer and hazardous material. Different grades may simply go by “battery grade”, “analytical grade”, or “industrial,” each hinting at permissible contaminant levels and specific intended uses.
Caution in the Lab and Beyond
Every chemist who handles lithium perchlorate develops a healthy respect for its risks. Strong oxidizers can sneak up on the inattentive. While it isn’t corrosive to skin like some acids or bases, the main hazard comes from mixing with organic material or strong reducing agents. Dust in the air deserves control as inhaling fine particles shouldn’t be taken lightly—lithium compounds can cause respiratory irritation and, in some exposures, longer-term health issues. Regulatory agencies require clear hazard labeling and push for closed systems or fume hoods in industrial settings. Fire departments train specifically for perchlorate salt responses because water alone sometimes spreads hazard, particularly if fuel sources are present.
From Rockets to Clean Energy—Applications of Lithium Perchlorate
Most folks meet lithium perchlorate as an electrolyte in lithium batteries. Its solution in solvents like propylene carbonate or dimethoxyethane enables high-ion mobility, translating to higher-capacity and longer-life cells. In specialty areas, engineers have dusted off its oxidizing power for solid rocket boosters or emergency oxygen generators—when heated it releases a steady stream of oxygen, handy on submarines or for astronauts in confined space. Analytical labs use it to dry solvents, leveraging the salt’s eagerness to pull water molecules from the air or a solution. As battery technology keeps expanding—into electric cars, grid storage, and beyond—demand for safe, pure lithium perchlorate keeps climbing.
Chasing Safer and Stronger Batteries: Ongoing Research
Research into lithium perchlorate spans safety, purity, and fresh uses. Battery scientists aim to minimize trace contaminants—chlorate, chloride, or transition metals—that might poison electrodes. Others study the impact of the salt in pushing up the limits of voltage and cycle life in lithium-ion and lithium-air cells. Environmental chemists examine perchlorate persistence in the environment, as this ion resists breakdown and can make its way into water systems. Researchers do not treat its toxicity lightly; perchlorate can disrupt thyroid function in humans and animals, so research groups keep measuring environmental and biological exposure in communities near handling facilities. Factories set strict effluent policies and filtration systems to keep releases under regulatory cutoffs.
Balancing Benefits and Risks—Future Prospects
Looking at the road ahead, lithium perchlorate faces both bright promise and tough scrutiny. In the battery world, cleaner and cheaper production could cement its spot as a top electrolyte salt, especially if alternatives like lithium hexafluorophosphate show unexpected safety flaws or supply bottlenecks. In oxidizing applications, stronger regulations may tighten access, especially for amateur use. Environmental rules may push for even lower emissions and tighter lifecycle controls, driving up costs but nudging the world toward safer handling. It’s hard not to see lithium perchlorate as both a technical opportunity and a chemical challenge—one where invention and care must work hand in hand to keep progress safe, sustainable, and responsible.
What is lithium perchlorate used for?
The Gear Behind Modern Batteries
Every time I check news about electric cars or portable tech, I’m reminded lithium chemicals power these steps forward. Lithium perchlorate stands out as a key material. It works as an electrolyte salt in lithium batteries, those same batteries tucked inside electric vehicles, power tools, and backup power banks. Lithium perchlorate delivers high ionic conductivity. That means electrons get where they need to be efficiently, even if devices drain a lot of power. Ask any electronics repair tech about heat management in batteries, and they’ll talk about safety and performance. Lithium perchlorate features a wide electrochemical stability window. This lets batteries run with higher voltages. Car manufacturers appreciate extended range, and so do consumers.
Emergency Oxygen in a Pinch
I walked past a display of spacecraft life support gear once and noticed a sealed canister marked “LiClO4.” Turns out, lithium perchlorate releases oxygen quickly when heated. Space agencies stick it in oxygen candles for submarines and space shuttles. Crew members need to rely on something proven in emergencies. A single lithium perchlorate brick or cartridge lets out a burst of clean oxygen, and it doesn’t clutter the air with dangerous byproducts. Miners and divers benefit here too, since emergencies happen in places where fresh air is unreachable on short notice. The quick oxygen release provides life-saving support.
Tool for Laboratory Professionals
Back in college, our organic chemistry lab instructor brought out a jar labeled “lithium perchlorate.” We used it to dry organic solvents. The salt pulls water out of solutions, preventing side reactions that would ruin a careful experiment. Analytical chemists reach for lithium perchlorate during high-performance liquid chromatography and some specialized titrations. Its stability and low reactivity with common solvents make it popular in labs that value accuracy.
Safe Handling Still Matters
I feel some concern seeing lithium perchlorate outside a supervised setting. It's classified as an oxidizer, meaning contact with fuel or strong reducing agents can pose a danger. Accidents happen when folks treat potent chemicals like hardware supplies. Companies train their teams carefully, and regulators set limits on storage and transportation. Fire departments run drills for possible chemical incidents involving oxidizers like this one. Anyone who uses lithium perchlorate has to pay attention to safety sheets, wear the standard gloves and goggles, and keep it away from heat sources.
Supply Chain and Environmental Impact
Battery manufacturers try to look ahead, worrying about future sourcing. Lithium itself comes from mines or brine extraction, and demand keeps outpacing new discoveries. While lithium perchlorate’s use in large batteries is not as widespread as lithium hexafluorophosphate, it occupies an important niche. Proper recycling keeps perchlorates out of water systems, where they can disrupt thyroid function in people and wildlife. Waste management services and battery makers work out recovery protocols. Regulations push for safer disposal and stronger tracking over unused chemicals.
Smarter Uses Ahead
Researchers hunt for new battery chemistries, sometimes swapping out lithium perchlorate for other salts, but its shelf life and reliability keep it on the roster for certain applications. If more effort goes into recycling and safer formulations, lithium perchlorate will stay relevant. It’s not just a chemical formula in a dusty catalog—real-world uses drive the need for responsible handling and steady innovation.
Is lithium perchlorate hazardous or dangerous?
Looking at Everyday Risks
You might not spot lithium perchlorate in the cleaning aisle or on your kitchen shelf, but it pops up in science labs, battery tech, and even in space rockets. It’s no household name, but its dangers deserve some close attention from anybody who might come across it, especially those of us working with chemicals. I've spent years working around chemical storerooms and research facilities, and one thing is clear: this isn’t a substance to treat casually.
What Makes It Risky?
Lithium perchlorate, made from lithium, chlorine, and oxygen, acts as a powerful oxidizer. If you've ever watched a metal fire or heard a pop from a chemistry experiment, you get a sense of what happens when oxidizers set things off. A small spark or just the right bit of heat near lithium perchlorate kicks off a reaction. I've seen colleagues carefully label even tiny vials of the stuff—we all know that mixing it with certain fuels or organic matter can turn a science project into an emergency.
Mixing lithium perchlorate with grease, paper, or even tiny amounts of dust can spark an intense fire. Even a sealed glass container can’t always save you if heat gets to it. In factories, mistakes happen—think powdered sugar dust igniting, but on a more dangerous scale. Over the years, I’ve read more than one report where someone treated it like just another salty powder and ended up rushing for a fire extinguisher.
Breathing Risks and Water Danger
It sounds like something out of a textbook, but perchlorates—including lithium perchlorate—can disrupt health even from small exposures. Swallowing perchlorates over time affects the thyroid, especially for young kids or anyone with thyroid issues. Just a little dust in the air can end up in your lungs or the water you drink. Labs use special hoods and ventilation for that reason. I once cleaned up a shelf spill with gloves and a mask, then had to remind the new intern that even leftover traces need soap, not just a dry rag.
Environmental risks stretch further. Perchlorates stay put in soil and water long after a spill. The US Environmental Protection Agency keeps a close watch on water supplies in part because of these stubborn chemicals. Water plants in places like California have issued warnings after detecting perchlorate in wells. Cleanup is tough and slow, so real prevention beats any after-the-fact fix.
Smart Steps to Prevent Trouble
Responsible storage never felt so important. Never keep lithium perchlorate near flammable materials. Use airtight containers made for chemicals. Strong rules and labels work best, based on my experience in busy labs where it’s easy to grab the wrong bottle. If you work around it, always use gloves, eye protection, and well-ventilated spaces. And if your job means handling it in bulk, get trained on emergency procedures and have spill kits ready. There’s no shortcut around common sense here—distraction or laziness with powerful chemicals usually means someone gets hurt.
For homes and regular workplaces, lithium perchlorate almost never shows up. Most of us handle its risks best by staying alert to the hazard it brings to labs and industries—especially for the workers and communities nearby. From my years in science and safety, knowledge paired with clear rules goes a long way. You can’t fear what you understand, but you do need to respect it, every single time.
How should lithium perchlorate be stored?
The Realities of Handling Lithium Perchlorate
Lithium perchlorate sits among the more interesting chemicals a person can run into in a lab or industrial setting. Chemists get excited about its usefulness in batteries and rocket propulsion, but its hazards command respect. A white crystalline powder may look harmless, but a powerful oxidizer sits underneath that plain face. Ask anyone who’s worked with strong oxidizers and most people have their stories—accidents, breakdowns in procedural discipline, or close calls with moisture exposure.
Risks Lurking in Each Misstep
This chemical reacts rapidly with organic substances and moisture. Mix it, even accidentally, with combustible materials and you’re looking at the ingredients for a violent reaction. As it absorbs water, it makes storage tricky. If containers fail or seals break, you risk a slow creep of humidity leading to dangerous surprises. This hits home for anyone who remembers opening a forgotten container, only to find the contents clumped or dissolving from air exposure. Lithium perchlorate takes that anxiety up a notch because even a small mistake can spark a destructive reaction.
Practical Storage Lessons
Before anything else, use containers that actually keep air and water out. Glass bottles with airtight lids and proper chemical-resistant seals do the trick. I’ve seen plastic containers buckle with time or let vapors in—precision with container choice pays off. It only takes one careless moment to turn a safe chemical into a hazard, especially in mixed-use storage rooms dominated by humidity swings. Never place this stuff near organic solvents, reducing agents, or flammable materials. As far as possible, single-purpose storage spaces reduce drama. Metal shelves tend to behave better than wood, since wood can absorb stray bits of chemical. I’ve met folks who swear by double-sealing: plastic films or bags inside the main container, just in case an outer seal fails.
Labeling and Accountability
Every chemist learns the hard way that mystery jars only cause headaches. Even if you swear you’ll remember what’s inside after a week, labels matter, especially when someone else enters the picture. Large, clear warnings, hazard-specific iconography, and dates on containers make a difference. A smart storage system includes both organizational logic and living documentation. Log entries for every addition or removal keep track of how long chemicals have sat unused. Aging chemicals, especially strong oxidizers, deserve regular audits and disposal checks. Students and staff learn responsibility by seeing these systems in place—not just from theory, but by example and enforcement.
Rethinking Storage Culture
Poor chemical storage rarely comes from lack of knowledge—it’s more about shortcuts, pressure to clear benches, or inattention during a busy day. Leaders and lab veterans set the culture here. Regular training builds muscle memory, but reminders work too: checklists by the storage room door, periodic walkthroughs, and logs matched to inventory. There’s value in walking through “what-if” drills—fire, flood, or spill scenarios—to reinforce why these habits matter. Investing in ventilation and climate control adds a layer of safety, but no equipment beats discipline and honesty about bad habits. Eventually, keeping dangers like lithium perchlorate in check becomes second nature. That comfort, earned through practice, means real safety—not just for one person, but for everyone sharing the space.
What is the chemical formula and molar mass of lithium perchlorate?
The Formula Matters in Real Applications
Lithium perchlorate stands out in labs and industry thanks to a distinctive mix of chemical traits. Its formula—LiClO4—delivers quite a punch. The lithium ion, that tiny alkali metal, combines with perchlorate, a tough oxidizer. Chemists everywhere reach for this salt for batteries, rocket propellants, research work, and even making energetic materials safer. I’ve had my hands smell like ozone after working with oxidizers, and the impact sticks with you. Getting the formula right means staying safe and hitting targets in synthesis or energy storage. You do not want mix-ups at the bench or on the production line.
Breaking Down the Numbers
Molar mass guides how much to use in reactions. A little math, and the numbers clear up: lithium sits at roughly 6.94 g/mol. Chlorine offers about 35.45 g/mol. Four oxygens pack a wallop at 16 g/mol each, so 4 x 16 equals 64 g/mol. Add these together:
6.94 + 35.45 + 64 = 106.39 g/mol
Knowing this makes batch calculations much easier and helps avoid costly overuse. I once used the wrong number by half a gram scaling up a protocol—had to start over. That mistake taught me to check every sum rather than trust faded notecards or rumor across the lab bench.
Why This Salt Matters Beyond Chemistry Sets
Beyond the textbook facts, lithium perchlorate draws attention due to its role in lithium batteries. Researchers lean on this salt as a trusted electrolyte. It keeps battery reactions running by letting lithium ions shuttle smoothly. Too much or too little changes battery power, life, and even safety. Cars, solar panels, and drones all ride on the shoulders of this chemistry lesson. I’ve watched batteries grow sluggish due to poor electrolyte choice; the frustration pushes for more careful selection and better understanding all around.
It is not just about batteries either. Sporting a powerful oxidizing punch, LiClO4 gets used in air purification setups for space stations, helping astronauts breathe easier for months on end. The salt generates oxygen when needed most, a crucial detail in aerospace planning.
Hazards and Handling—Don’t Rush Headlong
Perchlorates want respect. Mix-ups or shortcuts lead to nasty surprises. Strong oxidizers like this react violently with organic materials. Anyone working with LiClO4 learns quick to keep workspace clean and double-check for spills. Wearing goggles, gloves, and not eating nearby become second nature. I have seen small lab fires during student training, often traced back to a pinch too much or stray paper. Even pros mess up without a careful mindset.
Building Better Habits and Safer Labs
Clear teaching makes the difference in chemical safety. Sharing stories of blown-up flasks or ruined battery packs means new chemists think twice. Safety data sheets and trusted supplier documents save a world of headache. Quick access to the correct molar mass and clear examples in training help avoid repeating old mistakes. Instructors who link real outcomes to these numbers set up the next generation for smarter science.
LiClO4 sticks with you—not just in its numbers but in every weighed sample, charged battery, and shared lab tale. Get the formula and mass right, and much runs smoother. Forget or fudge, and things unravel fast.
What safety precautions should be taken when handling lithium perchlorate?
What Makes Lithium Perchlorate So Risky?
Lithium perchlorate shows up in chemistry labs thanks to its role as a strong oxidizer. It helps power up reactions and even finds its way into space projects as an oxygen source. But this compound doesn’t mess around. Touch a little heat to it, or combine it with the wrong material, and things can go sideways fast. Explosions happen when people don’t treat it with the respect it deserves. Stories float around academic circles about someone thinking they could handle it bare-handed or in a cluttered space, only to learn the hard way.
Why Basic Gear Sometimes Isn’t Enough
Regular gloves and goggles help in many lab situations, but lithium perchlorate demands more. After one close call with a spill in grad school, I started doubling up on gear—using nitrile gloves under flame-resistant sleeves and swapping my standard goggles for a chemical splash face shield. Perchlorates eat through certain materials and the dust irritates pets, skin, and lungs. The chemical can leap from a spill into your bloodstream through tiny cuts. Missing even one piece of protection leaves a person open to lifelong consequences.
Storage Is Half the Battle
Lithium perchlorate doesn’t take well to sunlight, heat, or moisture. Forget about storing it near organic chemicals, old rags, or cardboard. I remember one department lost a whole storage room because someone kept it next to acetone. The right move: keep it locked in a metal container, far from anything flammable, clearly labeled, and inside a dedicated chemical cabinet. Only properly trained staff should have a key. The goal is to keep it contained, calm, and far away from potential fuel sources or work traffic.
Avoiding Tragedy with Routine and Training
People sometimes get cocky after a few successful runs with dangerous materials. That’s a recipe for trouble. Safe routines change everything. Before classes, our teaching team would walk through emergency showers, eyewash stations, and check that spill kits were stocked—with sodium metabisulfite close at hand to neutralize those nasty oxidizers. We practiced what to do if someone took a splash or inhaled dust. These drills burned the steps into muscle memory so panic wouldn’t freeze anyone mid-crisis. New lab members sat through a full orientation, with real-world stories showing what happens if steps get skipped.
Facts and Smarts Beat Luck Every Time
In 2021, the Centers for Disease Control highlighted cases where simple oversights led to burns and respiratory problems. The data doesn’t lie: the best way to keep the air clear and the work safe means only opening lithium perchlorate in a working fume hood. No exceptions. Ventilation pulls harmful dust away before it reaches your lungs. Proper labeling goes a long way in alerting anyone in a hurry to check twice before grabbing the wrong container.
Addressing Waste Without Creating More Risk
Disposal trips people up. Pouring leftovers down the drain doesn’t work and turns a chemical risk into an environmental one. Collection only happens in sealable containers, labeled clearly, handled by licensed disposal teams. Documentation matches each batch, and local hazardous waste rules decide the next step. This process protects water sources and keeps city workers out of harm’s way.
Moving Toward Fewer Harms: Training, Tools, and Trust
The safest places invest in staff training and keep their equipment up to date. Open communication beats complacency. Sharing real incidents, even the close calls, helps everyone learn. Trust comes from following up when someone sees a problem or admits a mistake. At the end of the day, no shortcut or small time saving is worth risking hands, lungs, or a life.
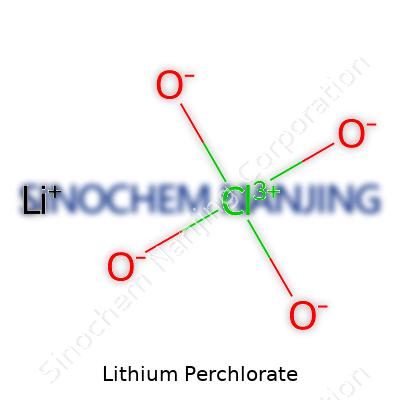
| Names | |
| Preferred IUPAC name | lithium perchlorate |
| Other names |
Perchloric acid, lithium salt Lithium perchlorate(VII) Lithium superchlorate Lithium chlorate(VII) LiClO4 |
| Pronunciation | /ˌlɪθ.i.əm pərˈklɔː.reɪt/ |
| Identifiers | |
| CAS Number | 7791-03-9 |
| Beilstein Reference | Beilstein Reference: 358833 |
| ChEBI | CHEBI:63316 |
| ChEMBL | CHEMBL499029 |
| ChemSpider | 76931 |
| DrugBank | DB11369 |
| ECHA InfoCard | ECHA InfoCard: 100.029.765 |
| EC Number | 017-006-00-8 |
| Gmelin Reference | Gmelin Reference: **1358** |
| KEGG | C18616 |
| MeSH | D008091 |
| PubChem CID | 23675676 |
| RTECS number | OU9625000 |
| UNII | 88XT1W8A9E |
| UN number | UN1473 |
| Properties | |
| Chemical formula | LiClO4 |
| Molar mass | 106.39 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 2.43 g/cm3 |
| Solubility in water | Very soluble |
| log P | -3.32 |
| Vapor pressure | Negligible |
| Acidity (pKa) | ~-10 |
| Basicity (pKb) | -3.72 |
| Magnetic susceptibility (χ) | -46.6×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.428 |
| Viscosity | 6.4 cP (25 °C) |
| Dipole moment | 0.0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 200.9 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -795.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -418.8 kJ/mol |
| Pharmacology | |
| ATC code | N05AN01 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06, GHS08 |
| Pictograms | GHS01,GHS03,GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H272, H302, H318 |
| Precautionary statements | P210, P220, P221, P280, P370+P378 |
| NFPA 704 (fire diamond) | 3-0-1-OX |
| Autoignition temperature | 160 °C |
| Explosive limits | Not explosive. |
| Lethal dose or concentration | LD50 (oral, rat): 187 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 160 mg/kg |
| NIOSH | SN2875000 |
| PEL (Permissible) | PEL: 0.05 mg/m³ |
| REL (Recommended) | 10 mg/m3 |
| IDLH (Immediate danger) | 150 mg/m³ |
| Related compounds | |
| Related compounds |
Potassium perchlorate Sodium perchlorate Ammonium perchlorate Lithium chloride Lithium chlorate |

