Lithium Nitrate: Past, Present, and Future
Historical Development of Lithium Nitrate
Lithium nitrate has a history that winds through early chemistry labs and major industrial processes. Chemists stumbled onto it in the nineteenth century, adding another chapter to the story of lithium compounds. Long before smartphones and electric vehicles became household essentials, lithium salts, including lithium nitrate, shaped medicine and glassmaking. Today, this compound continues to climb in importance, largely because of the growing demand in energy storage and specialty manufacturing. Its story runs parallel to shifts in battery technology, specialty chemicals, and environmental applications, making it more than just a footnote in the expanding world of lithium economics.
Product Overview
A colorless, crystalline salt, lithium nitrate appears at first glance like many other inorganic nitrates. It stands apart because of its unique reactivity and solubility. Chemically, it follows the formula LiNO3 and slips easily into water, driving many of its industrial uses. Manufacturers offer lithium nitrate in powder or granular form, typically with a purity above 99%. Deliveries show up in sealed, moisture-proof containers, with tight controls over impurities such as sodium, potassium, and magnesium. The product’s shelf life tracks closely to how tightly it’s stored away from moisture and air. Large-scale suppliers label each batch with lot numbers, production dates, and purity readings to help trace the source and quality.
Physical & Chemical Properties
Lithium nitrate melts at 264°C and breaks down at higher heat, producing toxic nitrogen oxides. Its cubic crystal structure dissolves with ease in water and mixes well with ethanol, which gives it a hand in both laboratory work and industry. While most people focus on the more famous lithium carbonate for batteries, lithium nitrate sticks around as a stabilizer in heat-transfer salts and oxidizer in pyrotechnics. The compound holds a relative molecular mass of 68.95 g/mol and settles into white, odorless crystals at room temperature. Measuring solubility, it far exceeds some of its cousins—this property steers its popularity in processes like molten-salt reactors and desiccants.
Technical Specifications & Labeling
Bags and drums of lithium nitrate come stamped with technical details: net weight, manufacturer, batch number, expiration date, and hazard classifications. Purity drives most specs, and customers check for total sodium under 0.01%, minimal calcium, and clear limits on sulfate and chloride. Labels warn of its oxidative nature and the risk of skin or eye irritation, reflecting regulations such as REACH and US OSHA HazCom. Some suppliers even print out COA (certificate of analysis) sheets for every consignment, aiming for full traceability. With modern demands from battery and materials industries, technical data sheets have grown to include particle size distributions, moisture content, and detailed impurity breakdowns.
Preparation Method
Traditional preparation of lithium nitrate draws from neutralization or exchange reactions. Mixing lithium carbonate or lithium hydroxide with nitric acid is the usual route in large-scale production. The process tosses out carbon dioxide or water along with lithium nitrate and demands careful temperature management; overheating triggers hazardous decomposition. Recent improvements help minimize waste and boost yield. Closed systems, automated feeding, and multi-stage filtration heighten consistency for modern electronic-grade needs. Newer research looks for routes that minimize acid use, cut effluent, and recover spent lithium from batteries—a growing necessity given the recycling push seen in global policy frameworks.
Chemical Reactions & Modifications
Lithium nitrate steps in as both an oxidizer and a source of NO3- groups in various syntheses. Reacting with strong reducers or metals often yields exothermic results, useful in pyrotechnics and propellants. In the lab, it shows up as a nitrating agent or helps drive the exchange of lithium ions into complex structures. Modified versions, such as doped or co-crystallized lithium nitrate, support advanced battery and electronic materials research. Its reactivity with organics leads to specialty nitrations and stabilization of other active chemicals, giving it a wide footprint beyond traditional industries.
Synonyms & Product Names
Lithium nitrate answers to several names: Nitric acid, lithium salt; LiNO3; UN2722; and sometimes pharmaceutical references, thanks to its older medical use. Product catalogs often list it under special grades like “Electronic Grade Lithium Nitrate,” “Battery Grade LiNO3,” or “ACS Reagent.” Labels will also highlight whether the product targets laboratory, technical, or industrial sectors—a nod to purity demands across user bases. Chemical suppliers standardize codes to match CAS registry number 7790-69-4, smoothing over sourcing confusion.
Safety & Operational Standards
Working with lithium nitrate calls for care typical of strong oxidizers. Direct skin contact can cause irritation, and swallowing pulls in toxic effects like nausea, abdominal pain, and in severe cases, kidney damage. Dry product is stable, but mixing with combustible materials sparks fire risk at surprisingly low ignition points. Safety Data Sheets press the use of nitrile gloves, eye shields, and dust masks during handling. Industrial operators supply local exhaust ventilation and teach staff to manage small spills with inert absorbents, avoiding any contact with organic matter. Waste disposal obeys strict hazardous waste protocols, usually involving neutralization and expert oversight.
Application Areas
Lithium nitrate runs a double shift in metallurgy and energy. Specialty glassmakers add it to improve clarity and alter refractive indices. In heat-transfer salts, mixes of lithium nitrate with potassium or sodium salts raise thermal stability for solar power installations. Battery researchers use it as an electrolyte additive, aiming to boost lifetime and safety. As a green chemistry agent, some labs pick lithium nitrate for organic syntheses meant to dodge more aggressive or polluting reagents. Explosives and rocket fuel designers use it to tweak burn rates and produce consistent ignition, thanks to its reliable oxygen release. Water treatment teams explore its use to knock down ammonia emissions and improve denitrification, reflecting tighter emission rules across urban infrastructure.
Research & Development
Across academia and industry, research on lithium nitrate stands active and diverse. Battery scientists chase new electrolyte blends, hoping lithium nitrate can limit dendrite formation and extend cell longevity. In solar thermal energy, researchers model mixed-salt cycles with the aim of pushing storage temperatures above 500°C to match advancing turbine technologies. Materials chemists investigate nanoscale interactions where lithium nitrate acts as a template or activating agent for porous carbon and metal-organic frameworks. The emerging field of battery recycling also finds new roles, using lithium nitrate in leaching and selective precipitation, both for spent battery regrowth and environmental protection.
Toxicity Research
Toxicologists review lithium nitrate’s biological impacts closely, especially as lithium supplies tighten and environmental concentrations rise. Acute toxicity studies place it in the “low to moderate” range, but chronic effects from long-term exposure still require clarity. Early studies point to kidney and neurological risks at high doses; occupational safety agencies call for strict workplace exposure limits. Wastewater researchers examine how lithium nitrate influences aquatic systems. It shows some bioaccumulation in fish and plant tissues, and regulators now track effluent concentrations near mining and battery-processing sites. Animal studies continue, with a goal to map full metabolic fate and cross-reference with real-world industrial loading.
Future Prospects
On the horizon, lithium nitrate stands to grow along with renewable power and battery installations. The shift toward electrification pulls lithium chemicals deeper into supply chains, with product innovations likely in next-generation batteries. Environmental applications hold promise, especially in water treatment and cleaner oxidant systems for chemical manufacture. Circular economy principles drive investment into closed-loop lithium systems, including nitrate-based recovery from electronic waste and end-of-life batteries. Researchers focus on scaling processes that cut waste and shrink the product’s environmental footprint, while keeping purity high enough for tomorrow’s technologies. As lithium nitrate’s role expands, strong supplier transparency and eco-conscious sourcing look set to define both its market success and public trust.
What are the main uses of Lithium Nitrate?
Fireworks, Flares, and Colorful Shows
Anyone who’s ever watched fireworks light up the night knows those vivid reds grab the eye. Lithium nitrate supplies that deep crimson tone, just as table salt colors flames yellow. Firework makers bank on it to get brilliant, stable reds in everything from city displays to roadside flares. This isn’t just for beauty; flares save lives whenever rescue crews need to signal through the dark, fog, or at sea.
Industrial Pyrotechnics and Heat Transfer
Factories need reliable heat. Lithium nitrate doesn’t just end up in the sky—engineers mix it into heat transfer salts used in big solar plants and specialized manufacturing. Mixing lithium nitrate into salt-based heat storage systems lets those setups reach high temperatures without breaking down. This keeps green energy plants pumping out power long after the sun drops below the horizon. More than a flashy compound, lithium nitrate helps cut costs and boosts energy storage tech, two things sorely needed as the world races toward cleaner electricity.
Chemistry’s Building Block
In labs, precise results can make or break whole product lines. Chemists pick lithium nitrate to help steer chemical reactions in the right direction. Sometimes it’s a mild oxidizer. Engineers and researchers use it as a catalyst in making specialty glasses, ceramics, and coatings. It proves useful for breaking the tough bonds in cellulose, which helps the paper and textile industries make stronger, purer materials. My own time in a university lab drove home that specialty chemicals like lithium nitrate often punch above their weight in keeping experiments on track.
Electrolytes and Batteries
Everyone craves longer battery life, whether it’s for their laptop or the neighborhood grid. Lithium nitrate doesn’t headline battery headlines, but it still matters. Some companies mix lithium nitrate into experimental lithium-sulfur and lithium-ion batteries to boost performance and stabilizing life cycles. This means batteries can last longer, run safer, and power more devices. In a world tilting further towards renewable energy, every small advance in storage chemistry matters.
Mining, Metal, and Beyond
Lithium nitrate gets called on in the mining industry as an additive to boost extraction and separation processes. In some cases, metal refiners add it to leaching solutions to help retrieve rare metals from ores. Better yields mean less waste, which translates to lower costs and less damage to the environment. That usually comes home to communities who live near mines and worry about pollution.
Environmental Questions and Alternatives
With lithium demand hitting new highs due to electric vehicles and renewables, every part of the supply chain draws scrutiny. Shifting more chemical manufacturing to limit water and soil impact becomes as important as improving battery life. Researchers, regulators, and manufacturers can help by developing recycling systems and safer handling practices for lithium nitrate byproducts. Rethinking extraction and waste can trim pollution now, sparing communities from the cost of future cleanup. Industry—and everyday buyers—can push for smarter policies and technologies so chemical essentials like lithium nitrate power progress without piling on new problems.
Is Lithium Nitrate hazardous to health or the environment?
Lithium Nitrate in Everyday Use
Lithium nitrate belongs to the family of chemical salts used by industry for several decades. Factories mix it into heat transfer fluids or use it as an oxidizer in pyrotechnics. Labs also reach for it during experiments in chemistry classes. Some people might see the word “lithium” and think only about batteries, but lithium nitrate feels like a different beast. Curiosity about its safety seems justified, especially since it doesn’t show up much in consumer goods.
Health Hazards: What Happens on Exposure?
Not all chemicals warrant panic. Lithium nitrate, though, shouldn’t be taken lightly. Inhaling its dust or fumes can irritate the nose, throat, and lungs. Workers in factories, who routinely handle bags of the stuff, sometimes complain about coughing or wheezing after a long shift.
Eating or swallowing a significant amount of lithium nitrate could raise blood pressure, interfere with the heartbeat, or even cause kidney trouble. Symptoms like nausea, vomiting, or abdominal pain have shown up in cases of accidental ingestion. Regular exposure, especially in jobs where safety rules go ignored, can bring worse news for health down the line. Chronic effects remain less studied, which doesn’t help put minds at ease.
Skin contact, especially if left unwashed, sometimes causes rashes or mild burns. I remember a college lab partner getting a small splash during a chemistry experiment. The white patch didn’t look scary, but it stung for hours. Basic safety—gloves, and goggles—keeps problems small.
Environmental Footprint
Industrial chemicals often make us wonder where the leftovers end up. Waterways take the brunt when rules go ignored. Lithium nitrate dissolves well in water, and fish or algae don’t seem to handle high concentrations very well. It can add to the general mess of nutrient pollution, helping harmful blooms set in. Local ecosystems can shift, not always in ways people notice right away.
Landfill disposal of lithium nitrate also creates headaches for waste managers. Rainwater can pull it out of discarded containers. The risk of groundwater contamination keeps regulators watching. Lithium ions might not build up in food webs like mercury, but that doesn’t mean they disappear without trace. Crops watered with contaminated sources could add measurable lithium to leaves and vegetables.
The risk of fire increases around large piles or storage tanks, since lithium nitrate speeds up combustion for other materials in an accident nearby. Firefighters try to keep powdered stores isolated from flammable goods for this reason.
Practical Ways Forward
Routine chemical handling never gets easier through carelessness. Gloves, masks, and tight storage serve as the frontline protection for workers. Better labeling and training reduce confusion, especially when chemicals have similar-sounding names but real differences in danger.
Environmental protection starts with industrial accountability. Safe disposal, using approved hazardous-waste channels, trims down the odds of water or soil pollution. Regulators inspect facilities to keep dumps out of rivers and fields.
Replacement options get attention, but trade-offs show up quickly. Some heat-transfer salts drop the lithium, but their performance lags. Meanwhile, regular monitoring at disposal sites and factories keeps problems from spiraling.
Lithium nitrate isn’t for casual use, but panic doesn’t serve anyone. Hard-won safety habits let us handle it with a measure of confidence, both for personal health and for the world outside.
What is the proper storage method for Lithium Nitrate?
A Matter of Safety and Sense
Lithium nitrate sits quietly among chemical supplies in research labs, factories, and sometimes educational storage rooms. Despite its calm, white-crystalline appearance, this salt deserves extra respect. Having worked in a university chemistry stockroom and collaborated with industry partners, I've seen small overlooked mistakes snowball into bigger headaches. Failing to store chemicals properly often tops the list of what goes wrong, and lithium nitrate tends to crop up on chemical safety review forms for good reason.
Why Worry About Storage?
Storage mistakes go beyond violating regulations. Lithium nitrate pulls moisture from the air, which can turn clumped or even dissolve into a messy puddle. It reacts strongly with organic materials. If stored next to something flammable, it boosts any fire from bad to disastrous. Sodium nitrate and potassium nitrate get similar warnings, but lithium nitrate packs more punch in high-heat reactivity. Accidents may start as confusion—grabbing the wrong bottle, ignoring a torn label—but the fallout can involve real danger, expensive cleanup, and possible legal trouble.
Setting Up Safe Storage
Best practice means storing lithium nitrate in a tightly sealed, clearly labeled container, made of glass or certain plastics—no metal lids, because nitrates and metals form a bad partnership. My old lab kept it on a lower shelf in a locked corrosives cabinet, far from paper towels, solvents, or acids. Physical separation matters here; don't stick lithium nitrate next to anything even slightly reactive or combustible. In hot or humid regions, desiccators or sealed bins with silica gel help keep it dry and prevent solid cake formation.
The Right Environment
A cool, dry place works best for lithium nitrate. High temperatures increase the chance of unwanted chemical changes and drive up pressure inside containers, risking a cracked jar or pop-off lid. You can't always control the building's temperature, but insulating chemical storage closets from steam pipes or direct sunlight goes a long way. Humidity can quietly erode chemical stocks over months, so checking on old storage spots or using portable dehumidifiers helps prevent disasters before they start.
Labeling for Longevity
A mystery bottle on the shelf, faded sticker, or label scrawled with a ballpoint—these details create more trouble than curiosity. Proper storage begins with readable, detailed labeling. Include the date received, chemical name, hazard class, and initials of the person responsible. In one chemical stockroom I managed, regular audits kept mystery bottles from hiding out for years. Scrap the “store out of direct sunlight” shortcut—clear instructions beat cryptic codes every time.
Smart Solutions
Digital inventory systems track expiration dates and alert staff to check for leaks or crusty lids. Training everyone—professors, students, and maintenance staff—in proper chemical handling lowers risks across the board. Sometimes, users get careless during a busy project. I’ve seen more accidents happen from overlooked bottles or returned containers than any dramatic spill. A culture where everyone respects storage rules, no matter how experienced, sets the tone for safety.
A Final Word to the Wise
Lithium nitrate isn't waiting to explode every day, but careless storage opens the door to problems. A bit of vigilance saves time, money, and confidence in any workspace dealing with chemicals. Storing chemicals right, and constantly reminding each other why it matters, creates safer labs and fewer emergencies.
How should Lithium Nitrate be handled in case of a spill or exposure?
Why Care About Lithium Nitrate?
Lithium nitrate ranks as an important chemical, found in laboratories, battery production facilities, and some specialty applications. Years spent working in chemical storage and industrial safety taught me that even “routine” chemicals deserve respect, and lithium nitrate is no exception. Its strong oxidizing nature and definite toxicity create safety demands that can’t be shrugged off. Mishandling, whether from a dropped beaker or a bag rupture in storage, sets up risk for people, property, and the wider community. I’ve seen workplace complacency turn a simple cleanup into a near-emergency.
What Spills Actually Mean
A spill never happens at a “good time.” White crystals scatter on concrete, or maybe a fine powder coats a benchtop. In my time, colleagues felt tempted to sweep up and move on, but the reality demanded different actions. Lithium nitrate can irritate the skin, eyes, and lungs. With enough exposure, more serious symptoms like headaches or dizziness pop up, and long-term health hits are possible. To ignore these facts invites a mess that’s bigger than what meets the eye.
Immediate Steps After a Spill
The right response starts fast. First, anyone not geared up should step away. My personal mantra: “Nobody proves toughness by skipping their gloves and mask.” Proper protection looks like chemical-resistant gloves, safety goggles, and a dust mask or respirator if powder hangs in the air. I remember someone laughing at how “geared up” I looked for a small cleanup—until the wind caught a pile and blew it back at them.
Block off the area by taping or posting signs to keep others away. Spilled material shouldn’t get swept up dry. Instead, use damp disposable towels; for larger spills, scoop up with a dedicated dustpan, avoiding brooms that send more particles flying. Careful containment keeps the chemical from migrating into drains or getting trampled into carpet.
Collected waste should go into labeled, tightly sealed containers. Regular trash cans won’t do. Following up with a wet wipe of the area—using detergent, never just water—removes remaining traces. Any contaminated cleaning items go right into hazardous waste.
Exposure: Taking It Seriously
During my years as a safety trainer, the worst outcomes came from hesitation. Splash in the eye? Rinse with water for at least fifteen minutes, aiming for a gentle stream. On skin? Same routine: plenty of water. If someone breathes in dust, stepping out into fresh air gives the first relief. Any sign of breathing trouble, confusion, or rash justifies immediate medical attention—hospital trips seem scary, but trust me, recovery beats regrets. Always report exposures to the on-site supervisor so follow-up can happen.
Preventing Next Time’s Panic
The best stories are the ones you don’t have to tell: no spill, no exposure, no emergency. Strong storage practices—sealed dry containers, labeled clearly, on shelves with catch trays—cut risks drastically. Training sessions with real-life scenarios stick with workers much better than dry lectures. Fast access to spill kits and personal protection gear means action happens quickly. From personal experience, leadership sets the tone: supervisors who model best practices inspire better daily habits down the line.
Big Picture: Valuing Caution
Lithium nitrate deserves thoughtful handling not because it’s the most dangerous thing in a workplace, but because it sits in a gray zone—common enough to become invisible, yet risky enough to leave real harm. Relying on habits that respect the chemical, prioritizing smart storage, and responding to spills with trained confidence all help people go home safe at the end of the shift. That’s the piece I care about most, shaped by years of seeing both the best and worst outcomes.
What is the chemical formula and appearance of Lithium Nitrate?
The Chemical Structure and Formula
Lithium nitrate goes by the formula LiNO₃. It’s a simple compound and a staple in many labs. The formula tells you what’s inside—one lithium atom, one nitrogen atom, and three oxygens all joined through ionic bonds. Lithium sits in the group of alkali metals, and nitrate brings in that extra punch of reactivity because of its oxygen-rich structure. The nitrate part gives it its oxidizing properties, which can be both useful and hazardous. Chemists keep the formula handy because lithium nitrate often steps in during synthesis and reaction studies.
Physical Appearance of Lithium Nitrate
Anyone working in a lab or dealing with materials engineering will recognize lithium nitrate’s look right away. It shows up as a white crystalline solid. The crystals are often tiny, but sometimes you’ll find larger lumps if they haven’t been broken down. They look similar to table salt, so it’s important to keep the label clear. The powder sometimes feels slightly grainy between the fingers, much like sugar, though not nearly as sweet or safe. In air, the crystals tend to attract moisture because the material is hygroscopic—meaning the granules will get sticky and clumped up if left out, especially in humid environments.
Why It Matters
Lithium nitrate has more going for it than just simple appearance. The characteristics influence where and how it fits in with real-world tasks. Back in my student days, I remember the first time I mixed it into a beaker for an experiment. It melted quickly in water and left a clear solution, telling me right away how soluble it really is. That solubility means lithium nitrate can easily be used in aqueous reactions. Folks working in the field find this helpful for heat transfer applications and even in pyrotechnics. Its oxidizing nature lets fireworks burn brighter red—a trick used at big stadium shows and local celebrations alike.
On the flip side, you need to watch out for storage. Those same properties that make lithium nitrate attractive in industry also bump up the risk. The oxidizing element means it reacts strongly with certain materials, sometimes even causing fires if mishandled. Storing it in airtight containers, away from anything flammable, keeps accidents off the books. That’s a lesson learned after hearing about too many close calls from other chemists.
Improving Safety and Best Practices
Safer storage and use come from knowing the risks and preparing accordingly. Label containers carefully and keep them away from heating elements or sunlight. Always wear gloves and eye protection, since granular lithium nitrate can irritate the skin and eyes. If you’re working outdoors or on larger projects, consider local regulations and be respectful of environmental impacts. Labs and workshops where I’ve spent time take this seriously, and it makes all the difference for long-term health and safety.
The Role of Lithium Nitrate in Society
Beyond the lab, lithium nitrate steps into battery technology and ceramics production. Industries look toward lithium compounds because they boost conductivity and help control melting points. That pushes forward everything from electric cars to heat-proof glass and tiles. The chemical’s unique footprint reaches far beyond appearance alone, shaping advances in clean energy and manufacturing. Anyone looking at the lithium-ion revolution should keep an eye on substances like this, as they play a quiet but essential role in new technology and everyday products alike.
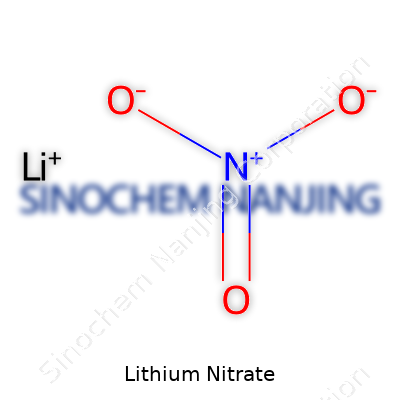
| Names | |
| Preferred IUPAC name | lithium nitrate |
| Other names |
Nitric acid, lithium salt Lithium salt of nitric acid Lithium mononitrate Lithium(I) nitrate |
| Pronunciation | /ˈlɪθiəm ˈnaɪ.treɪt/ |
| Identifiers | |
| CAS Number | 7790-69-4 |
| Beilstein Reference | 1811343 |
| ChEBI | CHEBI:63038 |
| ChEMBL | CHEMBL1200807 |
| ChemSpider | 57932 |
| DrugBank | DB14562 |
| ECHA InfoCard | 03b29730-4f7c-4f6a-b2b8-569e6fcd6dfe |
| EC Number | 231-550-4 |
| Gmelin Reference | Gmelin Reference: 14795 |
| KEGG | C14154 |
| MeSH | D008090 |
| PubChem CID | 10130502 |
| RTECS number | OG2100000 |
| UNII | NLV863Q7XE |
| UN number | UN2722 |
| Properties | |
| Chemical formula | LiNO3 |
| Molar mass | 68.946 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 2.38 g/cm³ |
| Solubility in water | 52 g/100 mL (20 °C) |
| log P | -2.5 |
| Vapor pressure | Negligible |
| Acidity (pKa) | -1.9 |
| Basicity (pKb) | -10 |
| Magnetic susceptibility (χ) | +7.2·10⁻⁶ |
| Refractive index (nD) | 1.505 |
| Viscosity | 3.01 mPa·s (at 25 °C) |
| Dipole moment | 0.0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 95.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -483.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | –1387.0 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | N05AN01 |
| Hazards | |
| Main hazards | Oxidizing, harmful if swallowed, causes serious eye irritation |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS03, GHS07, GHS09 |
| Signal word | Danger |
| Hazard statements | H272, H319 |
| Precautionary statements | P210, P220, P221, P280, P370+P378, P264, P270, P301+P312, P330, P501 |
| NFPA 704 (fire diamond) | 3-0-1-OX |
| Autoignition temperature | 540°C |
| Lethal dose or concentration | LD50 (oral, rat): 1190 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral, rat: 390 mg/kg |
| NIOSH | NA7906 |
| PEL (Permissible) | PEL: 10 mg/m³ |
| REL (Recommended) | 5 mg/m³ |
| IDLH (Immediate danger) | 30 mg/m³ |
| Related compounds | |
| Related compounds |
Sodium nitrate Potassium nitrate Rubidium nitrate Caesium nitrate Lithium nitrite |

