The Story and Reality of Lead Silicate: More Than Just a Chemical Compound
Historical Development and Context
Lead silicate has walked a tricky line in human industry ever since its discovery. Back in Europe’s early efforts to color glassware, artisans found that adding lead compounds gave their products strength and a sparkly richness no one had seen before. Chemists dug a little further, testing different blends, and landed on lead silicate as a star player. Its use exploded through the 19th and 20th centuries, particularly as the ceramics boom brought color and shine to everyday items. These days, thanks to what we know about toxicity, its role has changed. The modern world doesn’t embrace lead in quite the same way—less in homes, more in controlled factories—but behind the scenes, it’s still part of the complicated dance between tradition and safety.
What Lead Silicate Is and Why It Matters
This compound doesn’t show up in nature ready to use. Lead silicate usually comes as a bright yellow, orange, or red powder, each shade signaling a slightly different recipe—think lead(II) silicate or lead(II) ortho-silicate. These powders blend lead oxide and silicon dioxide in certain ratios, which means you have to know your chemistry to keep things consistent. In glassmaking and ceramics, this chemical mix lets manufacturers fine-tune melting points, color fastness, and how light bounces off surfaces. That explains why stained glass windows and fine porcelain carry lead’s fingerprint. The unique properties of lead silicate change the rules for artists, engineers, and manufacturers, letting them create the sort of tough and beautiful objects that last for centuries.
What Science Tells Us About Its Properties
Lead silicate stands out because its structure throws a wrench in the usual patterns we see in mineral chemistry. The dense, crystalline shape means it holds up under heat well above 800°C—far higher than many ceramic alternatives. It doesn't dissolve easily in water, which makes cleanup and disposal a hassle. Mix it in with glass or ceramics, and you’ll see rich, stable colors that don’t fade, even after repeated heating and use. Chemically, lead pairs with silicon to offer a lattice that’s tough and inflexible, helping vessels resist drops or bumps. Over the years, labs checked and rechecked properties like refractive index, melting point, and chemical durability, finding lead silicate offered more predictability than cheaper substitutes. That has kept it a staple for demanding industrial applications, even with ongoing health debates.
Technical Details and Labeling: Why Labels Can’t Be Ignored
It takes clear labeling to keep workers and consumers out of trouble with lead silicate. Regulations in North America and Europe demand warnings about lead content, plus instructions for safe handling and disposal. Technical specifications outline exact percentages of lead and silica, expected particle size, and purity—important numbers when the product ends up in factory settings. Governments push for labeling as the first defense, especially since the line between safe and unsafe exposure can get blurry, depending on how a factory runs. Without precise labeling and full transparency, entire industries risk health claims, product recalls, and relentless regulatory crackdowns.
From Raw Chemistry to Useful Material: How Lead Silicate Gets Made
Making lead silicate isn’t just dumping lead oxide and sand into a kiln. The chemical dance starts by reacting lead(II) oxide with silicon dioxide at high temperatures, sometimes topping 1000°C, until a uniform melt forms. Cooling turns that hot solution into a fine powder or glass frit. Each manufacturer tweaks temperature, time, and ratios to hit a specific blend—too much lead, and the result turns brittle; too little, the color and melting profile miss the target. Getting the chemistry right matters in glassmaking, where a batch disturbed by a few percent can ruin an entire production run. That hard-earned precision is why the basic recipe hasn’t changed all that much over generations, but labs do keep searching for new ways to reduce contamination and waste.
The Chemistry of Lead Silicate and Paths to Improvement
The reactions don’t stop once lead silicate comes off the production line. When mixed with different fluxes or oxides, its behavior opens doors for experimentation: metallic finishes, unique surface textures, enhanced strength, or stealthier colors that would be impossible with simple iron or copper-based minerals. Academic researchers keep looking for tweaks that might blunt lead’s toxicity without losing those prized effects—some tinkering happens on the atomic level, swapping lead for tin or bismuth in the framework, others try to trap lead inside less soluble matrices. Modifiers like alumina get tested for their ability to cut leaching rates, especially in tableware glazes. Still, the challenge stays the same: keep performance high, but risk low.
The Many Names and Uses of Lead Silicate
People working in industry may know lead silicate by half a dozen names: lead glass frit, lead(II) silicate, plumbous silicate, or under various manufacturer codenames. Ceramics shops will say “lead glaze,” while chemists note the precise ratios. In some catalogs, you’ll spot references to CAS numbers—a sort of chemical fingerprint—but in conversation, shorthand rules the day. This patchwork of names reflects its long, tangled history. Navigating these synonyms grows more important now that regulators and importers scan supply chains for restricted chemicals. Clear naming ensures workers aren’t surprised by an unexpected ingredient, and helps researchers connect old studies with new trends.
Health, Safety, and Smart Practices: Lessons Learned the Hard Way
Lead poisoning isn’t some footnote in chemistry textbooks. Early glassblowers and potters often paid a steep price for their craft, suffering from chronic exposure without knowing why their hands trembled or stomachs ached. Today, safety standards demand air filtration, protective equipment, regular blood testing, and thorough training. Factories committed to reducing workplace risks swap in ventilation hoods and automate the most hazardous steps to keep dust levels low. The most effective places partner with medical professionals who understand lead’s subtle effects—neurological changes, kidney problems, and developmental risks for children. Big improvements show what’s possible when workers and managers take these concerns seriously, but occasional headlines about cottage industries remind us that gaps remain, especially in small operations or developing regions where rules go ignored and information moves slowly.
Where Lead Silicate Finds a Role: Today’s Application Areas
Ceramic factories and specialty glass producers form the strongest market for lead silicate right now. Its durability doesn’t just serve fine china; electronics manufacturers use it in glass solders, protective coatings, and seals for everything from light bulbs to microprocessors. Artists reach for it when after centuries they want that unmistakable “rich yellow” found in stained glass. Conservation scientists trust original lead silicate recipes to repair historic windows and pottery without changing their authentic appearance. Still, the shadow of toxicity means that most food contact items now skip lead-based glazes entirely, nudged along by tougher international laws and consumer awareness. Some industries search for safer alternatives, but where unmatched optical or chemical properties matter, lead silicate keeps showing up.
Research, Development, and the Persistent Problem of Toxicity
Scientists spend long hours hunting for lead-free options that perform as well as lead silicate but don’t threaten workers or end up in children’s bloodstreams. Bismuth and zinc-based compounds lead the charge, showing promise in early tests, but many replacements cost more and perform worse where precision color or toughness counts. University labs and corporate R&D teams routinely share studies on reducing lead release from finished wares, aiming to keep leaching below global regulatory limits. Toxicity research, meanwhile, drills into how tiny doses harm the nervous system or get into groundwater. Discoveries about low-dose exposure and long-term risks have pushed regulators to keep cutting exposure limits. In the field, product recalls and lawsuits punish companies that don’t stay up to date on safer formulations. Some research projects point toward encapsulation methods or composite materials that could one day let artists and manufacturers keep the best properties of lead silicate without the liability.
Where Will the Story Go Next?
Debates over lead in art, industry, and electronics won’t end soon. For centuries, lead silicate packed color and toughness into objects from stained glass to dinnerware, driving innovation in chemical engineering and design. Even with health dangers so well documented, the search for a complete substitute drags on, slowed by technical hurdles and cost. Researchers and manufacturers who confront these realities—rather than downplaying them—do a service for both workers and the public. Keeping honest about risks, investing in safer processes, and sharing knowledge will decide whether lead silicate’s future is marked by more bans and replacements, or a safer balance between past practice and new discoveries. Until science finds a safer match, lead silicate stands as a reminder that progress demands hard choices, both in the workshop and the lab.
What is Lead Silicate used for?
What Lead Silicate Brings to Modern Manufacturing
Growing up in a family of ceramicists, I spent countless hours around pottery kilns and glazing tables. Through these experiences, I discovered how much science hides behind the scenes. Lead silicate is one of those invisible workhorses that shapes not only art but also practical products. In ceramics, manufacturers count on it for the brightness and smooth texture it gives to glazes. It allows colors to pop on plates, tiles, and vases, turning a plain object into something vibrant. When you run your hand along a shiny bathroom tile, there’s a good chance lead silicate had a hand in that finish.
Lead Silicate’s Role in the Glass Industry
Factories forging specialty glass—such as crystal glassware or decorative pieces—use lead silicate to enhance the clarity and sparkle. It helps produce a glass that refracts light beautifully, making chandeliers dazzle and fine stemware gleam. Technical glass, such as X-ray shielding panels in hospitals, often contains this compound too. Its unique density and structure help block harmful radiation, keeping patients and workers safe.
Risks That Cannot Be Ignored
Lead compounds, despite their usefulness, pose real health risks. Experts point to the dangers of lead exposure—brain damage in children and kidney dysfunction in adults stand as well-documented outcomes. The public’s growing awareness has pushed manufacturers to investigate safer alternatives and modernize their practices. For instance, strict workplace controls and air filtration systems have become routine in glass factories. I’ve heard from friends in the business who wear personal monitors and work under rigorous safety checks, reflecting an industry that can’t afford to look away from these hazards.
Pushing for Safer Alternatives and Solutions
Finding substitutes for lead-based compounds remains a huge challenge. Chemists experiment with barium and zinc compounds, which bring their own quirks but lack the same toxicity. Switching over often means reworking entire production lines, retraining staff, and tweaking firing temperatures. Some ceramic studios now use lead-free glazes, labeling their creations for the safety-conscious buyer. I’ve made the switch myself in small pottery batches, and while colors change and surfaces feel different, peace of mind takes priority.
The Importance of Transparency and Regulation
Government agencies keep a close eye on products made with lead silicate, and for good reason. Regulations demand clear labeling, limiting its use in items destined for food contact or children’s rooms. Some countries have phased it out from everyday dishes, thanks to strict safety standards. As a consumer, I pay more attention to product labels these days, especially with children in the house. This isn’t just about following the law—it’s about trust between makers and families who use these products every day.
Looking Ahead: Balancing Tradition and Responsibility
Lead silicate carries a legacy in art and science, making certain things possible that other materials simply can’t match. Yet the costs of convenience and tradition stand in clear view now. Striking that balance between keeping what works and protecting public health lands on all our shoulders—workers, artists, regulators, and shoppers alike. As the industry leans toward safer practices, our choices keep the conversation moving forward.
What are the safety precautions when handling Lead Silicate?
Understanding Lead Silicate’s Hazards
Lead silicate crops up a lot in ceramics, glass production, and enamel work. It’s no secret that this compound packs a punch in industrial processes, but it also brings real health risks. Lead has a nasty habit of targeting the body’s nervous system, kidneys, and blood cells. Breathing in the dust or getting it on your skin can mean trouble, and I’ve seen colleagues get careless and pay the price with headaches, fatigue, or worse—long-term lead poisoning is no joke.
Practical Safety Steps from the Shop Bench
Getting comfortable with lead never made sense to me. I’ve always kept my safety routine strict, and I recommend these steps for anyone handling lead silicate:
- Wear proper personal protective equipment (PPE): Gloves, safety goggles, and a solid dust mask or respirator keep lead particles out of your body. I use nitrile gloves—latex doesn’t stand up as well to repeated exposure.
- Work with good ventilation: Open windows don’t cut it in an enclosed studio or lab. Use a fume hood or an industrial-grade ventilation system. When our shop finally upgraded its airflow setup, the air quality and my peace of mind both shot up.
- Keep your workspace clean: Lead dust settles fast and ends up on surfaces, tools, and even clothing. Wet-wiping surfaces beats dry dusting every time. If you sweep, skip the broom and go for a vacuum with a HEPA filter. This keeps the fine particles in the bag, not your lungs.
- Practice good hygiene: Lead has a way of sneaking home with you. Wash your hands with cold water and soap before eating or drinking, and change out of work clothes before heading home. I set aside a pair of shoes and an old shirt just for the workshop—it takes a few extra minutes, but it keeps my family safer.
Why Training Matters
Book smarts can’t replace hands-on experience with materials like this. Every crew I’ve worked with does annual training on hazardous materials. That training sticks because it comes with stories—someone spills a container, or finds out their favorite snack picks up dust from an unwashed hand. Sharing those stories reminds us that these rules never exist in a vacuum.
Regulations and Accountability
OSHA sets pretty clear legal limits on workplace lead exposure, and companies get cited or even shut down for falling short. In our shop, regular air and blood lead testing keep everyone on track. Ignoring these isn’t just risky—it brings real consequences, from heavy fines to serious health problems. No one wants to be the one responsible for coworkers getting sick.
Technology Lends a Hand
Downtime used to mean cleaning up by hand, but now better filters, sealed containers, and real-time air monitoring make safety a shared project. Each new investment pays off in higher morale and fewer sick days. It feels good to know that the work we do for safety today keeps paying off long after we clock out.
Better Choices for the Future
Some newer art studios and manufacturers look for lead-free alternatives. These don’t always match the color or finish that lead silicate provides, but health comes first. Keeping the conversation going about substitutions, safety drills, and equipment upgrades gives everyone more control over their environment. That’s where long-term trust grows, worker to worker and company-wide.
What is the chemical formula of Lead Silicate?
Breaking Down Lead Silicate
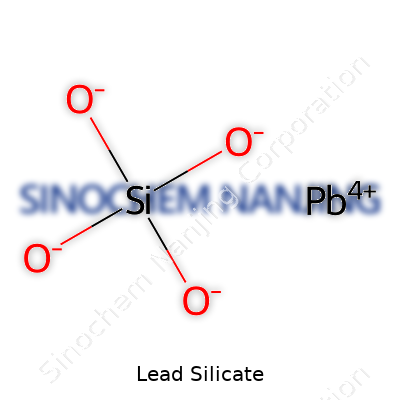
Lead silicate is something I’ve crossed paths with during my time studying both basic pottery glazing and the less glamorous side of heavy industry pollution. The compound, often listed under the chemical formula PbSiO3 or sometimes 2PbO·SiO2, pops up in ceramics, glass making, and a few niche corners of electronics. Anyone who’s ever spent time in a ceramics studio or peeked behind the scenes at a glassworks has probably encountered pigments or glazes relying on this material.
Why Chemical Formulas Matter
Whenever I talk with folks outside science, formulas seem like just a pile of letters and numbers. Yet, they show the ratio of elements that come together for a specific purpose. For lead silicate, the recipe usually involves one part lead (Pb), one part silicon (Si), and three parts oxygen (O). When written as PbSiO3, it tells workers and scientists exactly what balance to expect on an atomic level. Sometimes, you see manufacturers specify the ratio with 2PbO·SiO2—they just stir together two moles of lead(II) oxide with one mole of silicon dioxide before firing.
Lead in Everyday Objects and Safety Concerns
From the early days of glass windows to bright, durable ceramic glazes, lead silicate’s been a go-to choice for centuries. I grew up in a town with an old lead smelter on the outskirts. Generations of workers, including some of my relatives, dealt with lead compounds every day. These materials deliver color and durability but bring health problems no one wants to experience. Studies link lead exposure to serious neurological harm, especially for kids. Even trace common exposure matters.
In recent decades, regulatory agencies like the U.S. EPA and the European Chemicals Agency enforced limits on using lead in paints, water pipes, and children’s toys. Yet, glass and ceramics often still contain lead silicate because it delivers unique optical qualities and lowers firing temperatures—cutting energy costs for manufacturers.
Efforts Toward Safer Alternatives
It’s not enough to simply swap out lead silicate and hope for the best. Substitute materials need to hold up during repeated kiln firings, stay stable in sunlight, and avoid costly production changes. In my experience, artists and craftspeople care about these transitions but often don’t trust substitutes will act the same or look as good. Zink-based or barium-based glazes show promise but cost more, and their long-term performance sometimes falls short. Researchers keep testing combinations using safer materials, including boron or calcium silicates.
What Producers and Consumers Can Do
Responsibility sits partly with companies, for sure. Yet consumers play a role, too. Folks looking for dishware, art supplies, or even hobby glazes ought to check for “lead-free” labels. Art teachers can show students how to work safely, using choices that don’t gamble with their health. Funding for independent testing matters, so claims on packaging hold up in the real world.
Putting Health First Without Losing Craft
If you ask artists, most don’t want to jeopardize their communities. It’s possible to protect craft traditions by supporting research and giving creators genuine replacements that feel and look right, not just on paper but in each finished piece. Only with honest information and real partnerships can the world balance creativity, industry, and well-being.
Is Lead Silicate hazardous to health?
What Lead Silicate Does in Industry
Factories use lead silicate to help make ceramics, certain types of glass, and even glazes that cover the things we eat off every day. It improves color, melting point, and finish. I worked summers at a ceramics plant during college. Safety sheets about every material covered the walls, and the lead-based ones always got more attention. Workers wore respirators and went through special training. The company strictly controlled how lead moved from warehouse to workroom.
Health Risks of Lead Silicate
Lead’s hazards aren’t a mystery. Scientists have known for decades that it damages health. Lead silicate, like other forms of lead, gets into the body through dust, fumes, or accidental contact. Lungs, digestive tract, and skin all act as doors for exposure. Lead builds up in bones, brain, and organs. The U.S. Centers for Disease Control and Prevention states that no safe blood level for lead exists, especially for children.
Most adults exposed on the job feel early symptoms. Headaches, stomach pain, fatigue, and memory trouble can signal something’s wrong. Over time, kidneys, nerves, reproductive functions, and the cardiovascular system get hit hardest. Growing up, my uncle worked twenty years in a battery plant. Decades later, lead still galloped through his blood, causing problems his doctors struggled to treat.
Children get hit hardest. A Harvard study published in 2020 linked childhood exposure to lower IQ, attention problems, and slower growth. Kids can touch contaminated objects or breathe in dust at home, don’t have to step inside a factory. Simply washing hands after gardening or before meals slashes risk.
Regulations and Modern Practices
Most developed countries now limit occupational lead exposure to specific levels. In the United States, the Occupational Safety and Health Administration set a permissible exposure limit of 50 micrograms per cubic meter of air for an eight-hour shift. Companies running kilns or glassworks must test air, train staff, and monitor blood lead levels. Workers log exposure, and many employers provide regular medical exams and strict hygiene policies. European Union rules also pressure companies to move away from lead-based compounds.
Despite these improvements, exposure still happens, especially in developing economies. Factories sometimes lack the money for new equipment or protective gear. Dust control and worker training slip through the cracks. Informal recycling operations in some parts of Asia and Africa pose serious risks for families, especially children.
What Can Make a Difference
People stay safest when manufacturers switch to less toxic alternatives, like barium or zinc-based compounds in ceramics. At home, avoid old, chipped cookware and imported brightly-colored ceramics unless clearly marked lead-free. Wash hands and keep dust down in homes and classrooms. Communities need more education on lead’s dangers. Health agencies should fund blood testing and provide rapid support for anyone exposed.
Lead’s legacy runs centuries deep, but simple actions today cut risk for everyone.
How should Lead Silicate be stored?
Why Proper Storage Matters
Lead silicate may seem like just another chemical on the shelf, but those in manufacturing, ceramics, or battery work know how dangerous it gets if handled carelessly. Inhaling dust or letting it leach into the workspace doesn't just threaten health on the spot—it seeps into long-term wellbeing. Overexposure can mean real trouble: nerve damage, reproductive harm, and more. For workers in industries with daily contact, health guidance from OSHA and local authorities remains vital. Long story short: loose handling often leads to lasting problems.
Storage Starts with Container Choice
A lot of mistakes happen at the basic level—open bags, damaged bins, and reused older containers ask for disaster. I once saw a workplace using cracked plastic bins for lead silicate. Dust would drift whenever the wind kicked up, and you could taste it on the air. Metal or tightly sealed, chemical-resistant plastic containers work much better. No leaks, no cracks, no rust. Lids shut tight and labels clear enough for anyone—from a new hire to a safety inspector—to know exactly what's inside.
Picking the Right Location
Where the material sits in a building matters. Enough space, good air, no risk of flooding. I’ve seen shops shove toxic powders in dark corners to “keep them out of the way,” but hidden dangers do not keep anyone safer. A dry place keeps clumping and container rot away. Avoid stacking containers above shoulder height, since drops increase risk almost as much as spills. Keep everything off the floor and away from exits—easy to access, but not blocking safety walks in case of emergency.
Ventilation and Dust Control
Even with sealed containers, dust still turns up near workspaces. Proper ventilation keeps what you can’t see from hurting you. Open windows or fans without filters can just move the problem further down the hall. Local exhaust systems, like those seen in some paint or lab rooms, pull particles straight away. Wet-clean the storage area, never sweep, and skip compressed air which only kicks up more dust.
Personal Responsibility and Training
People make the difference. Even with perfect storage systems, one careless move spoils everything. Simple training and repeating the basics prevent accidents. Wash hands after each contact, avoid food and drink near storage, and keep protective gear like gloves and masks ready to use, not buried.
Proper Disposal and What Not To Do
Too many try shortcuts—throwing containers with tiny residues into the trash or pouring leftovers down drains. These acts turn a workplace risk into a community hazard, especially in cities or older buildings with fragile plumbing. Most regions offer hazardous waste disposal programs. Following their guidelines keeps fewer people at risk, especially kids and wildlife who suffer most from careless disposal.
Moving Forward: Real-World Solutions
Good storage demands both space and discipline. Some companies add visible checklists. Regular inspections, even unannounced, reveal problems before they become health scares. Sharing stories about actual close calls at work helps, too—real consequences stick in people’s minds better than empty warnings on a wall chart.
Every step taken to control where and how lead silicate rests means fewer headaches down the line. Nobody benefits from a shortcut here. The investment pays back in safer workplaces, better health, and less trouble with inspectors and legal issues. Even tough economic times don't justify putting people at risk over something so easily controlled with a bit of forethought.

| Names | |
| Preferred IUPAC name | Lead(II) silicate |
| Other names |
Lead Monosilicate Silicic acid, lead(2+) salt Lead(II) silicate Lead silicate glass Lead glass |
| Pronunciation | /ˈliːd sɪˈlɪkeɪt/ |
| Identifiers | |
| CAS Number | 11120-22-2 |
| Beilstein Reference | 3560466 |
| ChEBI | CHEBI:53325 |
| ChEMBL | CHEMBL612367 |
| ChemSpider | 20393448 |
| DrugBank | DB14516 |
| ECHA InfoCard | ECHA InfoCard: 100.031.004 |
| EC Number | 215-235-6 |
| Gmelin Reference | 11980 |
| KEGG | C15988 |
| MeSH | D007857 |
| PubChem CID | 166873 |
| RTECS number | OG9275000 |
| UNII | YO7N9R91P7 |
| UN number | UN1477 |
| Properties | |
| Chemical formula | PbSiO3 |
| Molar mass | 430.13 g/mol |
| Appearance | Yellow Powder |
| Odor | Odorless |
| Density | 2.5 – 3.6 g/cm³ |
| Solubility in water | Insoluble |
| log P | 1.3 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 8.5 |
| Magnetic susceptibility (χ) | `-56.0 × 10⁻⁶ cm³/mol` |
| Refractive index (nD) | 1.92 |
| Dipole moment | 2.54 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 198.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -879 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -644 kJ/mol |
| Pharmacology | |
| ATC code | V03AB35 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or in contact with skin; causes damage to organs through prolonged or repeated exposure; harmful to aquatic life with long lasting effects. |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS07,GHS08,GHS09 |
| Signal word | Danger |
| Hazard statements | H350, H360Df, H373, H410 |
| Precautionary statements | P201, P202, P260, P264, P270, P272, P280, P301+P310, P302+P352, P304+P340, P308+P313, P330, P405, P501 |
| NFPA 704 (fire diamond) | 2-1-0 |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 (oral, rat): 7,000 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 1000 mg/kg |
| NIOSH | SN4550000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Lead Silicate: "0.05 mg/m3 (as Pb) |
| REL (Recommended) | 0.05 mg/m³ |
| IDLH (Immediate danger) | 100 mg/m3 |
| Related compounds | |
| Related compounds |
Lead(II) oxide Silica Lead tetroxide Lead glass |

