Lead Perchlorate: Looking Past the Symbols and Into the Real Story
Historical Development
Lead perchlorate didn’t pop up in chemistry textbooks out of nowhere. Scientific curiosity over lead compounds has deep roots, stretching back centuries, but it was only in the last hundred years that perchlorate salts grabbed attention. Early researchers already knew lead compounds carried serious risks, but they didn’t always have the same approach to safety. When I was starting out in chemistry, the whispers of “danger” carried more weight with experience than any bold warning label. Laboratories once prepared lead salts for use in everything from explosives research to analytical chemistry because other alternatives couldn’t quite match their results. Methods for producing lead perchlorate evolved slowly, with each decade adding safe handling protocols, tighter purity requirements, and better storage techniques. Academic journals from the mid-1900s document the growth in use, driven by increasing study of perchlorate salts for their oxidizing power and water solubility.
Product Overview
To anyone new to the subject, lead perchlorate stands out not just as another compound, but as a blend of two elements with very different reputations. Lead, long maligned for toxicity, pairs with the perchlorate ion, a strong oxidizer. In my work, I always treated it as a serious topic—never merely as a “reagent”—because slip-ups don’t forgive ignorance. Its typical appearance as a highly soluble crystalline solid made it a staple for specialty applications, especially where water compatibility and strong oxidizing conditions are priorities.
Physical & Chemical Properties
Lead perchlorate usually forms colorless, odorless crystals that dissolve easily in water. A bottle of it looks pretty unremarkable sitting on a lab shelf, but threat doesn’t announce itself through visuals alone. The high solubility and strong oxidizing properties demand real respect. Joining that, perchlorate compounds resist easy decomposition unless provoked by heat or organic material. Handling the dry powder or concentrated solution always called for gloves, eye protection—simple but non-negotiable steps.
Technical Specifications & Labeling
In every laboratory where I’ve seen this salt, stringent labeling made clear the risks. You’d see the hazard symbols for oxidizers and acute toxicity, a clear alert to anyone who might mistake it for a routine salt. Specifications for purity almost always hit the 99% mark, as trace metals or impurities can spark side reactions. Most containers list the hydrated and anhydrous forms, each with its own handling quirks. It’s not the kind of chemical you want anywhere near food or organic solvents without double containment.
Preparation Method
Preparation rarely happens outside specialized facilities. I remember working in a lab where a small batch got prepared by reacting lead(II) oxide with perchloric acid, followed by careful evaporation and crystallization. The reaction releases heat, requiring slow addition and constant monitoring. As an experiment, it brings home the lesson that chemistry’s dangers aren’t just theoretical. Every time the acid hit the oxide, you could sense the importance of good ventilation and heavy gloves, not just as a precaution but as a matter of routine practice that separates professionals from thrill-seekers.
Chemical Reactions & Modifications
Lead perchlorate participates in classic double displacement reactions with alkali salts, precipitating various lead compounds depending on the counterion introduced. It also acts as a source of lead ions for complexometric titration, an old-school analytical method. In research settings, its value comes when oxidizing conditions are needed without the baggage of chloride or nitrate contamination. Still, most modifications involve replacing lead with other metals for similar perchlorate chemistry, aiming to sidestep toxicity issues. I’ve seen colleagues explore these substitutions, but the underlying chemistry always asks for caution and sharp attention to detail.
Synonyms & Product Names
Names on the bottle might range from lead(II) perchlorate to plumbous perchlorate, or even just “lead salt of perchloric acid.” In the real world, these all trace back to the Pb(ClO4)2 formula—differences in naming usually come down to where you source it or which catalog you pick up.
Safety & Operational Standards
Laboratory use demands strict protocols, no exceptions. Over the years, I’ve watched safety standards get tighter, and not a moment too soon. Lead compounds already carry well-known health risks—chronic exposure means damage to the nervous system, kidneys, and more. Perchlorate salts add the fire risk, making for a double punch. Working safely means using fume hoods, storing away from organic chemicals, and treating every surface as a contamination vector. Real care goes into spill kits, proper disposal, and detailed training—not just a handout, but a deep-dive into risk management every new chemist should internalize. Regulators regularly update best practices, but nothing replaces a culture where safety means more than just ticking boxes.
Application Area
Lead perchlorate found its main niche in the world of analytical chemistry and explosives research. Some older water chemistry methods measured lead by precipitation, while explosives engineers valued the strong oxidizing properties when exploring novel pyrotechnic formulations. Environmental constraints soon squeezed out many use-cases, as regulators, researchers, and companies shifted toward less hazardous alternatives. Today, you'll still find lead perchlorate in historical research and wherever legacy protocols linger. In my years of research, I’ve seen it gradually replaced by safer oxidizers, but old habits sometimes die hard in long-running experiments.
Research & Development
Modern R&D largely turns its back on lead perchlorate, searching for compounds where safety and environmental footprint rank higher. Development of greener chemistry methods stands as the big story. Researchers probe perchlorate chemistry but try to substitute less toxic metals whenever possible, and funding rarely flows toward projects involving heavy metals unless some truly unique reaction pathway exists. My own shift in focus mirrored this trend: once I left school, labs I joined put green chemistry and safety front and center. Even so, certain military and detection technology research kept tabs on lead perchlorate for specialized study, balancing risk and reward on a tightrope.
Toxicity Research
Both lead and perchlorates belong on lists of substances drawing heavy scrutiny. Toxicologists track the bioaccumulation of lead and the environmental movement of perchlorate ions, especially near old manufacturing sites. Long-term studies point to developmental risks in children and cardiovascular risks in adults exposed to lead, while perchlorates affect thyroid function at high enough levels. Health agencies recommend strict exposure controls—waste gets treated as hazardous, and medical research underlines the need for substitution wherever feasible. Real-world outcomes support these policies: we’ve seen populations benefit from lead reduction in everything from gasoline to paint, and regulatory limits keep getting stricter.
Future Prospects
Lead perchlorate’s heyday has passed, replaced by safer and more sustainable alternatives wherever possible. Still, its story offers valuable lessons. Legacy contamination means environmental scientists track its fate in old lab drains and industrial soils, while historians of science study how earlier generations balanced innovation and hazard. The big picture now centers on phasing out remaining uses, tightening clean-up efforts, and building a culture where risk isn’t just managed, but actively avoided. My own experience reminds me that good chemistry always weighs progress against responsibility, and lead perchlorate stands as one example where that balance needed hard-won lessons. Scientists, regulators, and educators keep those lessons alive to shape tomorrow’s safer lab environments.
What is Lead Perchlorate used for?
Behind the Lab Doors: What Draws Scientists to Lead Perchlorate?
Lead perchlorate isn't something you'll spot on a grocery store shelf, but it plays a notable role in scientific research, especially in nuclear physics. I’ve spent my share of late nights reading about chemical compounds, and lead perchlorate always jumps off the page because of its connection to neutrino detection. In these experiments, researchers search for ways to catch fleeting particles called neutrinos. These particles help us unravel mysteries about the universe—how stars work, why matter exists, and so on. Lead perchlorate, dissolved in water, gets used as part of the detector's solution. The lead atoms react with neutrinos, creating measurable flashes of light. That simple spark lets scientists track cosmic events that happened billions of years ago.
Beyond the stars and neutrinos, there’s a practical side hidden in this compound’s chemical formula. Some researchers use lead perchlorate when they need a concentrated lead source that will dissolve in water. It’s not every day you need something like that, but specialized labs rely on it for experiments in chemistry and radiochemistry. Portable X-ray devices and certain battery technologies once looked to similar lead salts, which shows how even niche chemicals sometimes trickle out of the lab to touch daily life, if only indirectly.
Risks That Don’t Stay in the Lab
I remember walking into my college chemistry building for the first time and spotting warning labels stuck everywhere. Lead perchlorate would have earned more than its share. The danger starts with lead—it’s toxic. The body can’t tell the difference between lead and calcium, so it sneaks into bones, blood, and organs. Over time, tiny amounts build up and set off a chain of health problems: neurological issues in kids, heart disease, and kidney trouble for adults. Even careful handling in the lab sometimes isn’t enough, as accidents and contamination can happen fast.
The perchlorate half of the mix isn’t harmless either. Perchlorates often end up contaminating water supplies, mainly from rocket fuel or fireworks, but lead perchlorate would pose the same kind of threat if mishandled. They disrupt thyroid function, which means trouble for the hormones that keep bodies working smoothly. As a parent, news stories about toxic chemicals in tap water always worry me, and lead perchlorate has the kind of profile that makes regulators sit up and pay attention. Because of the dangers, its use stays limited to close supervision inside research labs.
How to Keep Science Safe Without Halting Progress
It’s not practical to just ban tricky chemicals and call it a day. If we ended the use of every hazardous material overnight, scientific progress would collapse. Instead, the smarter move comes from strict controls, clear lab procedures, and ongoing education. The labs I’ve worked in obsessed over protocols—double gloves, proper storage, training every new recruit to treat every unlabeled bottle with suspicion. This vigilance means spills and exposure rarely happen. Regulators check inventory logs, waste gets double-checked, and exposure is kept to a minimum.
Scientists have been looking at safer substitutes, too. Alternatives without lead could someday replace lead perchlorate in detectors, but they need to deliver the same results. That kind of innovation doesn’t happen overnight. For now, health standards and solid lab practices stand as the best line of defense. If the world keeps supporting clean research facilities, invests in new detection substances, and demands transparency about what chemicals go where, maybe we’ll get those cosmic answers and healthier communities, too.
Is Lead Perchlorate hazardous or toxic?
The Toxicity Issue
Lead perchlorate raises serious concerns, especially in labs and educational settings. Growing up with stories about the troubles of leaded gasoline and pipes, it never seemed sensible to downplay the risks tied to lead compounds. Lead perchlorate stands out because it combines the health hazards of lead with the reactive nature of perchlorate. When someone hears "perchlorate," thoughts should drift to oxidizers, known for making fires more dangerous. Pair that with lead, a notorious neurotoxin, and the result isn't something to treat lightly.
Health Risks Aren’t a Guess
Scientists have linked lead exposure to developmental issues in kids and high blood pressure in adults. There’s no mystery: lead gets into the bloodstream and interferes with important biochemical processes, including those in the nervous system. Some of my friends in chemical manufacturing speak about handling lead compounds with gloves and fume hoods, always alert to the risks because the consequences stick for decades. Absorbing even a small quantity could mean long-term damage. Lead builds up, which means even small, repeated exposures add up.
Perchlorate’s Role
Perchlorate steps in as a strong oxidizing agent. In plain language, this means any accidental mixture with flammable materials or organic compounds could quickly spiral out of control. Fires, explosions, or unexpected chemical reactions aren’t just theoretical. Real incidents sit in chemical incident logs, serving as permanent warnings. I spent a summer on a team that carefully stored even tiny vials of perchlorate salts far from anything that could burn or react. Everyone double-checked labels and feared the kind of chemical mishaps that make headlines.
Environmental Impact
Beyond personal health, lead perchlorate’s journey doesn’t end in the lab. If allowed to escape into the environment, perchlorate can taint water supplies, disrupting thyroid gland function in people who unknowingly drink contaminated water. Lead, easily absorbed by soil and plants, finds its way up the food chain. I remember an old chemistry professor who said, “Lead found in dirt means lead found on dinner plates.” That rings truer every time reports surface about local water or vegetables showing contamination.
What Makes It Worth Attention
Facing facts: very little practical benefit comes from working with both lead and perchlorate together outside of niche scientific uses. Most countries implement strict rules about using or disposing of lead compounds, backed by alarming data. The United States Environmental Protection Agency, for instance, keeps tight reins on both perchlorate and lead in drinking water. Workers and students get told to report symptoms and regularly undergo blood lead level testing. Regulatory agencies provide clear exposure limits; ignore these, and the risk bites back.
Practical Solutions
One way forward calls for exhausting substitutes before bringing out lead perchlorate. Plenty of alternative chemicals can handle oxidation without dragging lead toxicity along. Better training for chemists and students—hovering close to the equipment with trusted mentors—cuts down on mistakes. If storing or disposing any chemical containing lead perchlorate, guidance must come from environmental specialists, not guesswork.
Continued education and strict workplace habits stand guard against toxic exposures. Having spent time in different labs, I can say that seeing experienced workers refuse shortcuts sets the standard. Safety gear isn’t an accessory; it’s non-negotiable.
Summary
Lead perchlorate shows the importance of respecting chemical hazards with both understanding and care. Every generation learns new lessons about the real cost of handling dangerous stuff. Staying safe starts with knowing what sits on the shelf, not assuming every risk can be managed after an accident happens.
How should Lead Perchlorate be stored safely?
Why Proper Storage Matters
Lead perchlorate is not just another chemical; it sits among the more serious hazards that demand extra respect. I’ve seen labs and industrial spaces overlook the basics of safe handling, often trading short-term convenience for long-term headaches. Its toxicity and reactivity stand out, and if you don’t get storage protocols right, you put lives and property at risk. The Centers for Disease Control and Prevention point to heavy metal poisoning as a persistent public health issue, and lead salts like lead perchlorate add fuel to the fire if handled carelessly.
Physical Security Counts
Sealed, corrosion-resistant containers should always be your first stop with lead perchlorate. Don’t use anything made of steel or soft metal. Glass with a secure plastic cap or high-density polyethylene containers keep moisture and curious hands away. Even a small leak contaminates surfaces for years—lead never gives up its danger easily. Place these containers somewhere locked, dry, and separated from other reactive chemicals. A closet that doubles as storage for cleaning solvents won’t cut it.
Moisture: The Quiet Enemy
This salt loves water, and moisture triggers a cascade of risks. Once humidity gets inside, you’re not just creating a mess; you’re laying the groundwork for complicated cleanup and unpredictable reactions. I once saw a careless storage setup where a broken air conditioner raised the humidity, leading to crystals sweating and sticking to every metal fitting nearby. Those kinds of slipups cost serious money and time to fix, and the cleanup wastes valuable research hours.
Labeling and Training—Not Optional
Too many places stash high-risk supplies on a shelf and call it a day. Training everyone who uses the storage space is just as important as the locks. Labels need glaring simplicity: clear hazard warnings, up-to-date inventory checks, and emergency contacts pinned right on the entry. Real training avoids disasters—people know what to do with a spill or accidental exposure. OSHA regulations give solid guidelines, but relying on the bare minimum invites careless shortcuts. Make it a point in routine safety briefings, not just once a year.
Preventing Exposure
Even trace exposure to lead compounds adds up in your bloodstream, so catching any issues early matters. I’ve seen good labs adopt personal protective equipment as a norm, not as a punishment. Gloves, goggles, and lab coats hang right near the door. When storage checks happen, so do hand-washing reminders and updates to safety protocols. After all, kids and community members pay the ultimate price for accidental releases, so keeping things tight benefits more than just your bottom line.
Disposal: Don’t Leave It Hanging
Old stock turns up in forgotten corners more often than people admit. Lead perchlorate isn’t the sort of thing you toss in the regular trash. Licensed hazardous waste handlers know how to package and neutralize it for safe disposal. I once saw a university handle a decades-old batch by calling a professional who traced every step, minimizing the risk for everyone involved. Local authorities keep strict oversight, so keeping a disposal log makes sense if you want zero trouble during inspections.
Room for Improvement
Not every facility gets this right. Regular audits, real-world drills, and making resources available show leadership cares about safety. The cost of ignoring smart storage will always outweigh any savings from cutting corners. In my experience, showing respect for hazardous compounds like lead perchlorate means starting with safe storage—and never letting up.
What are the handling precautions for Lead Perchlorate?
Understanding the Substance
Lead perchlorate rarely appears on lists of everyday chemicals, but its impact deserves attention. Working with it means dealing with two hazards: lead toxicity and the explosive nature of perchlorates. Lead harms the nervous system, kidneys, and blood when exposure happens over time. Perchlorates bring a risk of fire or explosion if they reach high temperatures or meet flammable materials. In practical terms, those risks add up to serious health concerns for anyone who comes in contact with it.
Why Personal Protection Matters
Direct contact with lead perchlorate, even small splashes or powder dust on skin, can raise blood lead levels. That’s not an experience anyone wants. Glove selection requires special care. Nitrile gloves offer decent protection, but workrooms dealing with corrosive or reactive materials often use layered gloves and disposable coveralls. Chemical splash goggles and face shields stop droplets from reaching eyes and face. I’ve seen researchers get rashes or headaches because they trusted “light lab work” without full protection. No shortcut works in these situations—skin, eyes, and lungs need complete coverage.
Air Quality and Dust Control
Lead perchlorate easily forms dust, and that dust travels straight to the lungs if inhaled. Fume hoods or efficient local exhaust systems do most of the heavy lifting here. Relying on open windows or fans just spreads the threat. Regular air monitoring in these labs helps flag increases in lead levels. I’ve seen labs where dust settled for months on equipment, only to lift back into the air the next time someone brushed past. Noticing that fine layer and addressing it quickly with wet cleaning makes a big difference for long-term safety.
Storage: No Place for Guesswork
Storing lead perchlorate correctly calls for more than a clear label and a dry shelf. This chemical doesn’t play well with anything flammable or reducing agents. It demands its own spot, far from potential fuel sources, in tightly sealed containers made of materials that don’t react to strong oxidizers. Lockable cabinets, preferably metal ones, reduce the chance of theft or accidental mixing. Some old storage rooms use wooden shelves, but that poses a real fire risk. Keeping an up-to-date inventory keeps surprises to a minimum and helps quickly spot leaks or spills.
Spill Response and Waste Disposal
A spill isn’t just a cleanup chore; it is a biological and chemical hazard in one. I once saw a minor spill—barely a gram—tie up an entire lab for hours. Cleanup teams wore disposable gear and used moistened pads to avoid spreading dust. All waste, solid and liquid, goes into marked hazardous waste bins. Failing to do this, even once, means someone could unknowingly handle dangerous material. Outside standard disposal channels, lead perchlorate often ends up in places where it pollutes groundwater or soil. That threat goes beyond the lab, affecting entire communities.
Training and Vigilance: Everyday Practices
People learn safe habits by doing, not just by reading a manual. Labs holding training sessions, running drills, and making chemical risks concrete keep mishaps at bay. Lead testing for staff shows who’s at risk before symptoms show up. Sign-in logs and regular audits increase accountability. None of this feels like busywork after seeing how easy it is for routine to breed carelessness. Creating a safety culture—one where every person checks, double-checks, and encourages others to do the same—protects both the present crew and anyone who enters in the future.
Can Lead Perchlorate be shipped internationally?
Tough Questions Around Shipping Dangerous Chemicals
Lead perchlorate doesn’t show up in daily life for most people. Only a handful of industries handle it. The problem is, every organization trying to ship this chemical overseas faces a maze of rules and potential hazards. Companies and regulators both know lead perchlorate brings risks, so the question isn’t just about transportation; it’s about the whole set of responsibilities that come with moving toxic substances.
Health and Environmental Concerns Take Center Stage
Breathing in or touching lead perchlorate can damage organs, especially in growing kids. The perchlorate part itself messes with thyroid function. The lead part causes cognitive and developmental issues. From cleaning up old battery sites to watching tragic lead contamination stories unfold, I’ve seen firsthand the pain and cost created when toxic materials get loose.
Shippers don’t just stick dangerous chemicals in a box and slap on a label. International laws force them to pack, track, and document every move. The threat remains, no matter how careful everyone stays. Environmental groups track these shipments, and regulators hand down huge fines when things go wrong. The Basel Convention says countries cannot simply send hazardous waste to others without consent. Customs agents in every country have the right to open, test, and even destroy risky shipments.
Strict Rules Make Errors Costly
Each country writes its own rules about importing chemicals. Most follow the UN Recommendations on the Transport of Dangerous Goods, but local differences cause major headaches. Some places treat lead perchlorate as both a chemical and a radioactive item, demanding forms and approvals from multiple government branches. I’ve witnessed entire cargo holds delayed for days over paperwork glitches or unclear labeling.
Paperwork grows so complex that many shippers work only with a few trusted logistics firms. All containers carrying hazardous products must stay tightly sealed and clearly marked. Crews on cargo flights and ships receive special training before accepting this material on board. If they spot a leak, the shipment comes to a halt, and cleanup can shut down an entire port.
Smuggling and Illegal Trade: A Shadow Industry
Despite all these controls, some still break the law. Black market brokers sneak dangerous chemicals across borders, hoping corrupt officials look the other way. Lead perchlorate shows up on online marketplaces, advertised by sellers who ignore international treaties. These sales put lives at risk, and catching the bad actors takes dedicated enforcement.
Steps Toward Safer International Shipments
Moving forward, greater transparency helps. Governments and industry watchdogs demand digital records tracing every step of hazardous shipments, including lead perchlorate. More companies invest in tamper-evident packaging and real-time shipment tracking. Clarity helps too—countries need to align their rules so legal exporters face less uncertainty. Training all port staff in chemical hazards can limit accidents.
Every viable solution starts with admitting that the dangers around lead perchlorate remain very real. Shippers, regulators, and buyers all share the job of closing gaps before people and environments suffer lasting harm.
Looking Ahead: Why Oversight Matters
Rules for moving lead perchlorate seem strict for good reason. Governments, workers, and communities feel the consequences of each mistake. If trade happens, it must stick to high standards every step. Loose controls aren’t just a legal problem—they’re a public health and environmental disaster waiting to happen.
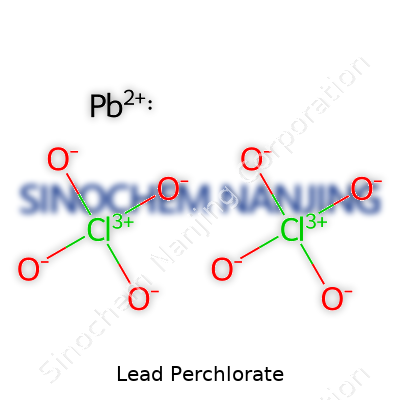
| Names | |
| Preferred IUPAC name | lead(II) perchlorate |
| Other names |
Lead(II) perchlorate Perchloric acid, lead(2+) salt Lead diperchlorate |
| Pronunciation | /ˈliːd pərˈklɔːreɪt/ |
| Identifiers | |
| CAS Number | 13453-47-3 |
| Beilstein Reference | 358937 |
| ChEBI | CHEBI:131486 |
| ChEMBL | CHEMBL4295433 |
| ChemSpider | 22977 |
| DrugBank | DB11054 |
| ECHA InfoCard | 03e6b88b-8d1a-49a1-8e03-2a3f2e7b1e12 |
| EC Number | 231-847-6 |
| Gmelin Reference | Gm. 1153 |
| KEGG | C18330 |
| MeSH | D007857 |
| PubChem CID | 24816 |
| RTECS number | OG8225000 |
| UNII | 37Q3TQN3Q9 |
| UN number | UN1479 |
| Properties | |
| Chemical formula | Pb(ClO4)2 |
| Molar mass | 406.1 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 2.74 g/cm³ |
| Solubility in water | Very soluble |
| log P | -4.36 |
| Vapor pressure | Negligible |
| Magnetic susceptibility (χ) | −25.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.690 |
| Viscosity | 18 cP (25 °C) |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 483.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -741.1 kJ/mol |
| Pharmacology | |
| ATC code | V03AB34 |
| Hazards | |
| Main hazards | Oxidizer, highly toxic, may cause fire or explosion, very hazardous to health, harmful if swallowed, inhaled, or in contact with skin. |
| GHS labelling | GHS02, GHS06, GHS08, GHS09 |
| Pictograms | GHS03,GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H272, H301, H302, H315, H319, H332, H360, H373, H410 |
| Precautionary statements | P210, P220, P221, P260, P273, P280, P305+P351+P338, P308+P311, P370+P378, P501 |
| NFPA 704 (fire diamond) | 3 0 2 OX |
| Lethal dose or concentration | LD50 oral rat 763 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 220 mg/kg |
| NIOSH | SC8750000 |
| PEL (Permissible) | 0.05 mg/m³ |
| REL (Recommended) | Suspected of causing cancer. |
| IDLH (Immediate danger) | IDLH: 100 mg/m³ |
| Related compounds | |
| Related compounds |
Lead(II) nitrate Lead(II) acetate Lead(II) carbonate Lead(II) chloride |

