Lead Nitrate: A Comprehensive Commentary
Historical Development
Lead nitrate started making an appearance in chemical literature back in the alchemist days, when curiosity about metallic salts pushed some unlikely characters far past the limits of their own chemistry labs. This compound found its first formal mention in the sixteenth century, as early chemists experimented with different forms of lead for pigments and glazes. By the nineteenth century, factories in Europe refined processes for isolating lead nitrate to supply expanding dye and explosives industries. The industrial revolution, with its hunger for more efficient chemicals, ensured that this white crystalline powder would stick around. Lab workers noticed its high solubility early on, making it a favorite for reactions where quick, reliable results mattered. These developments didn’t happen in a vacuum. They followed social and economic triggers, including the urban demand for colors in printing, textiles, and even the dark business of ammunition production.
Product Overview
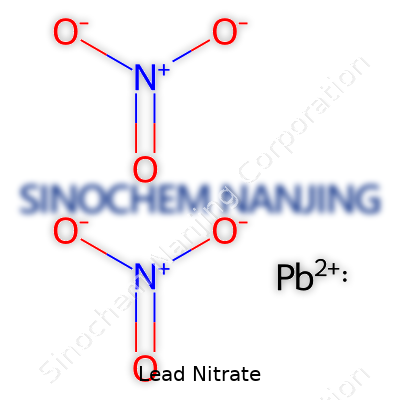
Lead nitrate shows up as a colorless or white crystal, dissolving easily in water, which isn’t something shared by all lead compounds. Its chemical formula, Pb(NO3)2, points to one lead atom for every pair of nitrate groups. In my own college laboratory days, it looked more like chunky sugar than something that could poison a river. Suppliers range from large chemical distributors to specialist firms, offering lead nitrate in grades suitable for industrial, research, and sometimes even educational use. The high purity grades command higher prices, mainly due to strict limits on contaminants.
Physical & Chemical Properties
Walk into any storeroom or lab where lead nitrate is stocked, and you’ll catch a glimpse of neat, heavy jars labeled with warnings. The compound’s crystals melt at about 470°C, and it starts decomposing before you reach boiling, releasing toxic nitrogen oxides and leaving lead oxide behind. It weighs in at about 4.53 g/cm3, a density that betrays its lead content right away. One thing that has always made it so popular—its high solubility—means you can mix it in water at nearly any temperature and watch it dissolve efficiently, topping out at around 52 grams per 100 ml of water at room temperature. Its reactivity with organic matter, reducing agents, and heat means you have to stay alert.
Technical Specifications & Labeling
The legal paperwork and hazard markings on lead nitrate drums paint a story. UN 1469, hazard class 5.1 (oxidizer) and class 6.1 (toxic), get printed right alongside the standard chemical name. The certificates that travel with the batches emphasize purity, heavy metal content, and particle size, especially for uses in mining or pyrotechnics. Not a single box leaves the warehouse unmarked—skull and crossbones, strong language, and "Keep out of reach of children" stamps everywhere. Lab supply companies make sure that batch numbers reach labs for their audits, and the best ones publish detailed impurity profiles on every shipment.
Preparation Method
Factories produce lead nitrate on a multi-ton scale by reacting lead metal or lead oxide with nitric acid. The reaction runs fairly smoothly: dump lead scrap or powder into a tank, add concentrated nitric acid slowly, watch the mixture fizz with brown fumes, and finally filter off unreacted metal. Try not to breathe in those orange clouds; those nitrogen oxides cause plenty of health problems. In smaller setups, lead carbonate—another common form in paint and pottery—sometimes stands in as a precursor. Each route winds up with a crystallization step, since you need a clean, solid product. In my experience, the purification through repeated dissolving and recrystallizing really makes a difference, stripping away copper and other metals that can cause trouble in later reactions.
Chemical Reactions & Modifications
Lead nitrate reacts with alkali and alkaline earth metals to create insoluble lead salts, a classic move in qualitative analysis. Drop it into a solution of potassium iodide and you’ll see it quickly make a yellow haze of lead iodide. In explosives and pyrotechnics, it works as an oxidizer, giving up its nitrate groups to feed combustion or drive denser, hotter flashes. Environmental chemists use it as a standard or a source of lead ions in water testing. As a student, I saw it convert into more complicated lead salts by mixing with sodium chromate or carbonate solutions. Research labs keep pushing the chemistry to produce newer catalysts and even some experimental materials for electronics, although these are niche applications.
Synonyms & Product Names
Ask for lead nitrate in an academic or industrial setting, and you might hear it called plumbous nitrate or nitrolead. European suppliers sometimes list "lead dinitrate". Labels include C&L (Chemicals & Laboratory), Merck, Alfa Aesar, and a dozen generics, depending on the region. Universally, the chemical formula Pb(NO3)2 stays the same, which helps prevent confusion. Nicknames aside, the product tracking always focuses on batch integrity and traceability, especially where health and safety laws get strict.
Safety & Operational Standards
Stories about poor handling loom large in the world of lead nitrate. Breathing dust or fumes damages the nervous system and kidneys, and long-term handling without proper gloves spreads dangerous traces over desks, doorknobs, or anything you touch. Most labs enforce double gloving and vented hoods for weighing or solution prep. Facilities storing bulk quantities maintain spill kits, and regulatory guidelines insist on separated storage from organic materials. Regular blood testing for lead exposure among workers is a routine in some jurisdictions, a practice I first saw during an internship in mining reagent supply. Waste handling became more serious in recent decades, sending all lead-laden materials off for specialist treatment rather than letting anything slip down laboratory drains. OSHA, REACH, and local equivalents keep a close watch on how this compound travels from port to workplace.
Application Area
Lead nitrate still earns its paycheck in a few big sectors. In ore processing, it boosts gold extraction rates by modifying the chemistry of leaching solutions, helping gold separate from its host rock in complex, multi-step processes. Explosives manufacturers rely on its ability to release oxygen rapidly, adding it to formulas for detonators and pyrotechnics. Some ceramics makers use it to color glazes, though that practice shrank as health concerns grew. Analytical chemists trust it to give reliable results in precipitation reactions, especially for testing halides or sulfates. I’ve seen it pop up in teaching labs, although many schools now substitute safer alternatives to avoid the lengthy cleanup and risk of exposure.
Research & Development
The search for safer, greener chemistry hasn’t forgotten about lead nitrate. Research groups investigate alternative oxidizers and leaching agents to lower environmental risks in mining and manufacturing. Some studies test whether the lead component can be captured more cleanly after industrial reactions, reducing the spread into soil and water. A few labs in materials science look at lead nitrate as a precursor for synthesizing lead-based perovskites, which show promise in next-generation solar cells and electronics. Progress speeds up when funding appears, especially if governments restrict lead use further or push for new recycling technologies. Networked databases now let researchers access decades of accident reports and toxicology findings, supporting more cautious and informed method development.
Toxicity Research
Scientists never stop studying the health impact of lead nitrate. Countless animal studies and epidemiological surveys have proven that even trace levels of lead in water, soil, or the human body can trigger developmental problems in children and chronic illness in adults. The compound’s ability to cross cell membranes means no amount of good lab practice can replace the need for airtight storage and clean handling protocols. Regulations now force manufacturers to label every drum and develop exposure reduction strategies. Major public health campaigns warn about the dangers of lead, challenging everyone from chemists to manufacturers to develop tighter controls. Even after huge reductions in the use of lead paint and petrol, the focus on industrial chemicals like lead nitrate drives ongoing reforms.
Future Prospects
Lead nitrate exists at an odd crossroad, where legacy use in established industries overlaps with growing social pressure for toxin-free production. The mining sector continues to need reliable methods for extracting precious metals, but faces mounting costs for environmental safeguards and stricter recycling rules. Environmental scientists join forces with engineers to design new capture and treatment systems—some based on ion exchange, others on bio-remediation—so that lead doesn’t keep seeping into groundwater. Legislative moves in Europe and North America may shrink industrial markets for lead nitrate, pushing up prices and spurring investment in alternatives. Some research circles believe that the future of this compound lies less in bulk industry and more in niche applications, such as microelectronics or specialized catalysis, supported by strict recycling and recovery. My generation of chemists grows wary of any work involving lead, rightly so. The next breakthroughs will likely come from teams looking to close the loop or eliminate risky materials altogether, fueled by lessons learned from the past and a genuine need for a safer chemical world.
What is the chemical formula of lead nitrate?
Putting Lead Nitrate on the Table
Lead nitrate tells a story about lab benches and old-school chemistry classes. Lead nitrate carries the formula Pb(NO3)2. You see those numbers and letters and it looks like alphabet soup, but there’s an important message tucked inside: this compound contains one atom of lead, two nitrate ions, and a whole lot of chemical history.
This Formula Isn’t Just for Memorization
People often wonder why formulas matter. From growing up seeing lead warnings on pipes, paint, old batteries, that lesson has stuck with me. Chemistry class handed out formulas, but the dangers and stories behind each compound, especially ones with lead, made me look twice. Pb(NO3)2 doesn’t just represent a lab exercise—it’s the backbone for many applications. Factories use it in dye production and explosives, and in lab settings, it tests for chloride ions and more. But just knowing its formula opens the door to understanding what it can do and where it might cause trouble.
Lead Nitrate Packs a Punch—In and Out of the Lab
The problem with lead-based chemicals comes down to the reality they can harm living things, especially children. Science groups, including the CDC and WHO, have pointed out that lead exposure affects the nervous system and brain development. It hardly takes much. The old pipes in my neighborhood, the peeling paint in historic buildings, and even imported goods remind us how common lead contamination remains. Not just in large quantities. Trace amounts become a source of lifelong health issues. Pb(NO3)2 dissolves easily in water, which means it moves around. It gets picked up by rainwater, carried through soil, or caught in runoff. If not handled carefully, it finds its way into rivers, crops, and even tap water.
Facing the Risks—Real-World Solutions
No one finds themselves wishing for more lead in their daily life. The best step? Practice smart handling and push for tougher rules. Schools, universities, and workplaces need safer storage methods and strong ventilation. Wearing gloves and using proper masks in labs matters, because skin absorption and inhalation pose real threats. Everybody in the chemical supply chain has a role to play—companies should take responsibility for how they source, move, and dispose of chemicals like lead nitrate.
On the policy side, organizations can call for better regulations and more transparency. Public safety depends on strong oversight. I’ve seen progress in community-level lead abatement projects that test water for lead and offer free replacement of old pipes—those efforts deserve wider support. If local governments get upfront and communicate health risks, families have a fair chance to avoid exposure. Education in schools can teach kids about the risks of hazardous chemicals like Pb(NO3)2, so the lessons stick for life.
Holding Chemistry to a Higher Standard
Let’s not treat a chemical formula as something to glaze over or memorize for a test. Pb(NO3)2 means something out in the world. It represents history, technology, health, and the environment all at once. Facts can’t be ignored: the lead in lead nitrate has power, but with that comes responsibility—for chemists, teachers, policy makers, and anyone else who might cross its path.
What are the common uses of lead nitrate?
The Chemical Pathways of Lead Nitrate
Lead nitrate shows up in a lot of industrial places most folks never see. Factories that make matches and explosives rely on the compound because it helps trigger certain chemical reactions. If you look inside a pyrotechnics plant or a mining operation, odds are you’ll run across barrels marked for lead nitrate. It acts as a strong oxidizing agent, which means it gives other compounds the chemical kick they need to break down or combine in different ways.
Gold Extraction and Mining
Mining outfits use lead nitrate when they need to separate precious metals like gold and silver from raw ore. In gold extraction, it teams up with sodium cyanide to pull gold from rocks during leaching. This process boosts the purity and recovery rate of gold, making each truckload of rock more valuable. The flip side is the environmental risk: lead nitrate can leak into water supplies if not handled fully responsibly. Over the years, regulations have tightened, but enforcement often falls short. My own visits to mining regions in West Africa left a mixed impression—economic growth bumping up against pollution worries, with families talking about jobs and health in the same breath.
Explosives and Pyrotechnics
Stories about fireworks don’t usually mention the chemical recipes packed inside shells. Yet, lead nitrate plays a major role in how explosions look and sound. In practice, it helps mix unstable elements in explosives for road-building or mining, acting like a middleman that smooths out the chemical reaction. Lead nitrate carries a reputation for reliability, so companies stick with it despite mounting pressure to consider safer substitutes.
Laboratory Uses and Education
Beyond big industry, lead nitrate takes a quieter role inside classrooms and research labs. Teachers use it during chemistry demonstrations to show off how compounds dissolve or to drive home lessons about solubility and chemical reactions. Researchers sometimes use it to make other lead-based chemicals. With strict handling protocols, these uses aim to expose students to real-world chemistry while keeping everyone safe. I remember my high school teacher breaking out the lead nitrate for a reaction demonstration—he wore gloves and goggles, and at the end, we’d talk about why lead dangers matter as much as the equations on the chalkboard.
Environmental and Health Considerations
The flip side of usefulness always looms. Lead nitrate, like every form of lead, raises red flags for health and safety. Inhaling or swallowing even small amounts can damage nerves, kidneys, and blood cells. Workers at facilities that process or use the compound need real training, not just warning signs taped to a wall. Filters, closed-loop systems, and cleanup routines matter as much as the chemistry of making gold or fireworks. The cost of skimping on safety shows up in real people's lives—see the headlines from industrial accidents across multiple countries.
The Bigger Picture and Safer Alternatives
Many industries hang on to lead nitrate because replacement costs money and switching brings its own risks. Scientists keep looking for new methods that work just as well but don’t carry the same health burden. Factories that have phased out lead compounds point to lower insurance bills and fewer health incidents. This step forward takes political will, investments in technology, and local communities speaking up. Lead nitrate’s long history in science and industry needs to face up to modern values, where public health tops the list.
Is lead nitrate hazardous or toxic?
What Makes Lead Nitrate Risky
Lead nitrate shows up as a white, crystal-like powder but its harmless look masks what it can really do. At work, I watched a lab tech talk about lead salts, and he told me one key thing: Touching or breathing in any form puts your health on the line. Lead nitrate, like many lead compounds, does not play around — your kidneys, brain, and nerves can pay the price. Studies have shown that even small amounts, over time, build up in your body. It impacts how children learn, it weakens memory, and it brings high blood pressure if you’re older.
Workplace Exposure: The Facts and Shortfalls
Most people never deal with lead nitrate. But in mining, battery factories, ceramic businesses, and schools, it lurks. Workers in those places need strong rules to keep things safe. There is no mystery here: Long-term exposure causes lead poisoning. OSHA and the CDC both warn against any careless handling.
A while back, a friend in metal recycling told me how hard it can be to avoid contamination. On one visit, his hands shook when he talked about colleagues sent home for blood tests. His boss had skipped providing proper gloves for weeks. I looked up statistics and noticed thousands of workers still fail regular blood-level checks every year, even where laws demand it. Carelessness and cutting corners put actual lives at risk.
Health Outcomes the Public Experiences
Kids take the worst hit. Their bodies soak up lead at a rapid rate. Even without symptoms, test results can show dangerous levels. Eating with dirty hands, breathing in dust, or taking home traces on work clothes brings a quiet crisis inside a family. In my town, a nurse once shared stories of children with attention problems, mood swings, all linked back to lead exposure from a parent’s workplace dust.
Medical journals spell it out: Lead nitrate does not just hurt you once. It collects, hangs around, and damages health years down the road. Adults see their blood pressure and risk of kidney disease climb. Women put unborn children in jeopardy if lead enters the picture during pregnancy.
How Society Can Tackle the Problem
Fixing it takes more than a warning sign. I’ve seen factories overhaul entire workflows after staff insisted on better training and newer ventilation systems. Science moves fast: Safer substitutes now exist for some tasks, and sealed benches, personal protective equipment, and routine blood testing actually make a difference. Old buildings and labs with lead nitrate need careful cleaning, not just a fresh coat of paint.
I believe education holds the key. Kids growing up near industrial sites should get regular testing and timely care. Workplaces need simple policies—wash your hands, don't eat in the lab, always change out of work clothes. The government, for all its paperwork, needs to fund inspections and respond when families speak out.
Nothing about lead nitrate’s hazard is new. But every step towards safer handling promises actual people a longer, healthier life. Mistakes cost too much to ignore the evidence staring us in the face.
How should lead nitrate be stored and handled?
The Heavy Shadow of Lead Compounds
Lead nitrate comes with a reputation. It isn’t some benign chemical that sits quietly on a shelf; its toxic and oxidizing nature has earned it a long history of strict regulation. Having worked in labs handling this compound, the memory of strong smells, white crystals, and hazard warnings sticks with you. Simple mistakes can mean heavy consequences, both for people’s health and the wider environment.
Why the Fuss?
Regulators treat lead salts as serious threats for good reasons. Once you know that lead causes damage to brains, organs, and even whole communities, you stop looking at that bottle with indifference. Nitrate only adds another layer of risk, since it accelerates combustion. At home or work, one careless move—like mixing with sawdust or storing near acids—puts people in harm’s way. It’s no mystery why so many precautions surround this chemical.
What Good Practice Looks Like Day-to-Day
No one stores lead nitrate in a lunchbox or on the same shelf as vinegar. Practical routines keep everyone safer. Those of us trained in labs or manufacturing know the drill: airtight containers, clear labels, and pads beneath bottles to catch accidental spills. Using non-reactive containers, usually glass or high-grade plastics, shields the chemical from moisture and acids in the air. Locking cabinets, well away from common traffic, keep it out of reach of the unwary.
Keeping the storage space dry matters more than most realize. Humidity draws moisture through cracks and caps, helping lead nitrate dissolve and travel where it’s not wanted. Regular checks for leaks or residues help avoid small problems turning into big ones. Security counts too: lead nitrate often factors in misuse, so tracking who’s handling it and logging every movement isn’t just paperwork—it's common sense.
Handling with Respect, Not Fear
Gloves, goggles, and even simple lab coats feel annoying, until they save your skin from burns. Plenty of experienced techs use disposable bench paper for mixing and transfers, then clean well afterwards. Fume hoods aren’t optional, since inhaling even a dusting of lead compounds can haunt people’s health for years.
Waste leaves a similar trail. No one dumps lead nitrate in a sink or trash bin—labs collect waste in labeled containers, sending them off to certified recyclers. At my own station, we checked pH and kept citations from safety audits pinned by the door as a daily reminder of what’s at stake.
Why It Matters: The Human Side
Negligence with lead nitrate isn’t abstract for old industrial towns, or for families where someone brought work uniforms home. Kids suffered brain damage from dust in corners and water down the drain. Past disasters forced communities to become experts in chemicals against their will.
Now, serious rules and careful habits have lowered the risks, but shortcuts can bring the old problems roaring back. Teams trained regularly, using real spill scenarios, build muscle memory that no written policy matches. Building a culture where someone feels free to speak up about a careless practice keeps people honest. Handing off responsibility to the next generation means teaching why these rules exist, not just how to follow them.
Lead nitrate isn’t going away any time soon. Respect for it, wrapped in practical training and visible care, keeps its dangers from reaching where they shouldn’t. That’s a lesson earned slowly—one that’s still worth repeating.
What are the safety precautions for working with lead nitrate?
Understanding Why Lead Nitrate Deserves Respect
Lead nitrate sounds like something pulled straight from an old chemistry textbook. It’s colorless, it dissolves quickly in water, and looks harmless enough. Still, folks in labs and factories know how misleading that can be. Lead, hidden inside those neat white crystals, brings real health dangers. Handling it without proper care can lead to serious trouble, and ignoring these risks never works out well for anyone down the line.
Breathing in dust or getting the powder on your hands leads to lead exposure, and over time, that’s known to damage brains, kidneys, and nerves. Problems don’t show up overnight—things get worse the more you’re around this chemical. Some of the biggest disasters in old industry came from ignoring lead’s dangers. Growing up near an abandoned battery plant, I saw families devastated when city tests found sky-high lead levels in backyard soil. People still pay for corners cut long before.
Smart Moves in the Lab
Anytime you open a container of lead nitrate, get serious about safety. Start with solid, reliable gear. That means a proper lab coat, gloves made from nitrile or neoprene, and goggles. If you splash this stuff in your eyes, damage happens fast. Don’t trust cheap “disposable” gloves—they rip too easily. I can still recall how gritty lead powder clings to sweaty skin; it’s stubborn and gets into every crease, so washing up right after use matters a lot. Some labs use special soap that binds heavy metals and removes them better than plain water.
A good lab bench setup isn’t just about neatness. Working in a fume hood, instead of out in the open air, keeps dangerous dust out of your lungs. Shops that skip hoods see higher lead readings in the air, and air monitors won’t lie if things get sloppy. Never eat or drink near your workstation—lead spreads hands to mouth without warning. No snack or coffee is worth a slow poisoning. Keep dedicated shoes for the lab, since tracking lead dust home can put your whole family at risk.
The Role of Rules and Responsibility
Rules on handling lead nitrate come written into law for a reason. OSHA and EPA regulations exist because history is full of people getting sick at work from careless chemical use. I’ve watched managers try to cut costs by skipping proper disposal or reuse of containers. That approach always backfires. Employees get sick, cleanups get expensive, and lawsuits start flying. Following the rules is the better path, no matter the budget crunch.
Storing the chemical in tightly sealed containers stops leaks and keeps things organized. Label everything, even if you “know” what’s inside—the next person may not. Having a spill kit with lead-specific binders makes sense, since lead dust never just sweeps up with a broom. Training new staff goes further than posting a few warning signs on the wall. From my years helping with safety audits, you can tell right away whose teams just memorize answers and who actually builds safe habits day in, day out.
Making Workplaces Safer for Everyone
Continuous blood lead testing is no joke, especially for those in jobs with daily contact. Catching a problem early saves years of trouble. Companies that value their crews take these measures seriously. Better ventilation, high-quality personal protective equipment, and tight inventory controls offer real protection—far beyond the bare minimum.
Building a safety culture means workers speak up when problems show up, and management actually listens. I remember a junior tech who spotted a crack in a storage jar—she got thanks, not blame, and that saved a lot of grief. Keeping everyone safe from lead nitrate works best when everyone buys in. History, science, and personal experience all line up on this one: respect the risks, and nobody has to pay a heavy price down the road.
| Names | |
| Preferred IUPAC name | dioxido(dinitrato-O,O')lead |
| Other names |
Lead dinitrate Plumbous nitrate Lead(II) nitrate Nitric acid lead(2+) salt Plumbum nitrate |
| Pronunciation | /ˈliːd ˈnaɪ.treɪt/ |
| Identifiers | |
| CAS Number | 10099-74-8 |
| Beilstein Reference | 353961 |
| ChEBI | CHEBI:75831 |
| ChEMBL | CHEMBL254001 |
| ChemSpider | 22929 |
| DrugBank | DB06751 |
| ECHA InfoCard | 03b81f47-d241-4eee-b4dc-88a845d1552a |
| EC Number | 231-841-6 |
| Gmelin Reference | 1656 |
| KEGG | C14326 |
| MeSH | D007949 |
| PubChem CID | 24924 |
| RTECS number | OG2100000 |
| UNII | 6R9QVG5HQC |
| UN number | UN1469 |
| Properties | |
| Chemical formula | Pb(NO3)2 |
| Molar mass | 331.2 g/mol |
| Appearance | White crystals |
| Odor | Odorless |
| Density | 4.53 g/cm³ |
| Solubility in water | 52 g/100 mL (20 °C) |
| log P | -0.88 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 2.8 |
| Basicity (pKb) | 11.62 |
| Magnetic susceptibility (χ) | ‘-22.0 × 10⁻⁶ cm³/mol’ |
| Refractive index (nD) | 2.01 |
| Dipole moment | 3.27 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 174.6 J K⁻¹ mol⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -451.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -451 kJ/mol |
| Pharmacology | |
| ATC code | V03AB56 |
| Hazards | |
| Main hazards | Oxidizer, toxic if swallowed, toxic in contact with skin, toxic if inhaled, causes damage to organs, suspected of causing cancer, may cause genetic defects, very toxic to aquatic life. |
| GHS labelling | GHS02, GHS07, GHS08, GHS09 |
| Pictograms | GHS03, GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H332, H350, H360Df, H373, H400, H410 |
| Precautionary statements | P210, P220, P221, P264, P273, P280, P301+P330+P331, P302+P352, P305+P351+P338, P308+P313, P405, P501 |
| NFPA 704 (fire diamond) | 2-0-3-OX |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 oral rat 450 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat 450 mg/kg |
| NIOSH | 0087 |
| PEL (Permissible) | 0.05 mg/m3 |
| REL (Recommended) | 0.015 mg Pb/m³ |
| IDLH (Immediate danger) | 100 mg/m3 |
| Related compounds | |
| Related compounds |
Lead(II) acetate Lead(II) carbonate Lead(II) oxide Lead(II) chloride |

