Lead Dihydrogen Phosphite: A Closer Look
Historical Development
Chemists first looked into the phosphite family over a century ago, searching for compounds to keep pace with a rapidly changing industrial landscape. Lead dihydrogen phosphite didn’t show up immediately. Earlier studies on lead compounds often circled around lead oxide or lead nitrate because those simpler salts offered obvious routes for pigments and batteries. By the early twentieth century, researchers started combining lead with less conventional anions, including phosphite. They weren’t always thinking ahead to environmental impact, but the push for new materials led the way. Over time, interest in phosphites grew, especially because they acted as corrosion inhibitors and stabilizers in plastics. Checking the old journals, a few articles from the 1930s point out the “improved stability” of lead phosphites in harsh conditions. This early work pointed industry folks toward specialty applications, and by the late twentieth century, the compound was popping up in patent databases as chemists explored fire retardants, surface treatments, and even as intermediates in electronic ceramics.
Product Overview
You won’t see lead dihydrogen phosphite sold in hardware stores or garden centers. The material offers value mainly behind the scenes—in chemical labs, manufacturing, and specialty processing. Folks who use it want to control redox chemistry or add a specific set of electronic features to a compound mix. Research groups often work with small quantities, and chemical suppliers ship it only with careful labeling and strict documentation. Walk into a lab that orders lead dihydrogen phosphite, and you’ll see bottles with hazard icons, thick safety data sheets, and every container sealed tight to avoid spills or powder trimming. Chemists know it’s not just another salt—it comes with toxicity, legal controls, and handling strictness typical for any compound with heavy metal content.
Physical & Chemical Properties
This crystalline material draws attention for its solid and stable white appearance, resisting moisture pick-up under standard lab conditions. Solubility trends matter—lead dihydrogen phosphite dissolves sparingly in cold water, more easily under heat, and less so in most organic solvents. Every batch gives off a slightly granular texture, which reminds you of powdered sugar but with extra heft. It shows chemical stability in mild acids and bases; the material doesn’t break down unless forced by strong oxidizers or intense heat. Left in an open dish, it won’t fume or melt at room temperature—a property that keeps it practical for high-precision chemical processes. The compound’s molecular structure weaves phosphorous atoms tightly alongside two hydrogen and three oxygen atoms, anchored to a lead ion, forming modest hydrogen bonds across the lattice. Basic measurements show a higher density typical of lead salts, making handling a bit heavier than other white powders.
Technical Specifications & Labeling
Any proper shipment of lead dihydrogen phosphite contains a full label with the chemical formula (Pb(H2PO3)2), purity percentage—often above 98%—and batch details for traceability. Suppliers list moisture content, particle size range, and any secondary additives or anti-caking agents to fend off clumping. Each drum or flask ships with a certificate of analysis, flagging trace metals (arsenic, mercury, cadmium) kept well below regulatory thresholds. Chemical industry standards insist on labeling the precise weight down to grams or kilograms, the production date, hazard pictograms for lead toxicity, and GHS-compliant safety instructions in at least two languages. Distributors must log exact volumes for compliance, and buying even a small amount requires clear end-use documentation. Hazard labeling always highlights acute toxicity, reproductive hazard, and environmental risk, meeting EU REACH norms and U.S. OSHA demands.
Preparation Method
The classic method for making lead dihydrogen phosphite starts with a simple, direct chemical reaction. Lab techs dissolve lead(II) oxide or lead(II) carbonate in diluted phosphorous acid, carefully controlling temperature to keep the exothermic reaction from racing ahead. Chemists keep stirring until the white crystalline product starts to settle out of solution, signaling full conversion. Filtration lets them separate the solid from any leftover solution, followed by repeated washing to clear out unreacted acids or byproducts. The washing stage matters—a little bit of excess acid can throw off purity and create inconsistent properties. Drying steps use gentle heat, usually under vacuum or in an oven set below 100°C, because higher temps might damage the product’s crystal network. For high-grade material, the whole setup happens in glassware or plastic-lined vessels, avoiding metal mixing that would shed extra ions into the final salt.
Chemical Reactions & Modifications
Lead dihydrogen phosphite takes its chemical cues from both lead and the phosphite core. Redox chemists often lean on it as a moderate reducing agent, while synthetic materials teams throw it into reactions to tune fire resistance or conductivity in complex mixtures. Under strong oxidizers like potassium permanganate, the phosphite centers can oxidize up to phosphate, releasing lead salts and increasing hazard profiles. In multi-step syntheses, this salt trades its phosphorus for other ligands, helping catalyze difficult bond formations. Adding barium or calcium salts might push ion exchange, switching lead for lighter metals and freeing up phosphorous intermediates. Thermal analysis shows it rarely decomposes below 250°C, but slow roasting edges the material toward lead(II) phosphate, with hydrogen splitting off as gas. That shift opens possibilities for ceramics doping and inorganic pigment work, areas where the special chemistry of lead phosphites keeps them valuable even as industries shy away from toxic metals.
Synonyms & Product Names
Some labs and textbooks use alternate names when tracking this compound. Common tags on stockroom shelves include “lead phosphorous acid salt,” “Pb(H2PO3)2,” “plumbous dihydrogen phosphite,” and “lead(II) phosphite, monobasic.” Chemical databases link it with EC 237-276-6 and the CAS number 19978-33-7. Larger chemical suppliers sometimes file it under PCB protection agents or flame retardant precursors, reflecting where the trade often meets laboratory demand. Patent offices occasionally record obscure variations in nomenclature, but buyers look for the same core identifiers on every certificate and shipment.
Safety & Operational Standards
Nobody treats lead dihydrogen phosphite lightly. It joins a long list of lead salts needing firm control because of the severe health risks. The compound’s dust threatens workers with neurological and developmental hazards since lead accumulates in the body over time, causing organ and nerve damage. Good labs lean on full-face respirators, gloves with real barrier protection, and double-sealed containers. Fume hoods stay down, floors stay swept, and no food or drink enters the chemical area. Waste disposal protocols send spent material directly to hazardous waste sites, and legal frameworks—like the European Union’s REACH or the U.S. EPA’s regulations—limit not only storage but also transport and ultimate end-use. Regulatory inspections demand tracking logs from receipt of raw materials through every gram consumed or discarded, reflecting the global consensus that safety trumps convenience wherever heavy metals enter the supply chain.
Application Area
Firms in plastics, fire retardancy, pigment, and specialty ceramics tap into lead dihydrogen phosphite’s unique traits. Its ability to help snuff flames makes it attractive for niche wire coatings or insulation foams, places where regulations let lead compounds operate under strict control. Electronics research teams sometimes use it to seed new types of conductive or semiconductive ceramics for sensors or capacitors, chasing properties that pure phosphates can’t match. In pigment manufacturing, its chemistry gives access to hues stable against acid or ultraviolet light—strong enough for industrial coatings, but its use faces tight restriction. Certain catalyst makers view it as a raw ingredient, sometimes as a finishing agent for anti-corrosion layers on metals, though newer, safer options squeeze those applications year on year. Generally, its role shines at the intersection of chemical effectiveness and niche technical need, not in large-volume industrial processes.
Research & Development
Researchers keep poking at the edges of what lead dihydrogen phosphite can do, especially as alternative flame retardants lose ground to regulatory crackdowns. Chemists aim to unravel the fine structure of lead phosphites by running advanced NMR or X-ray diffraction scans, mapping how atoms line up to resist thermal and UV breakdown. Collaborative research targets ways to replace lead with barium or strontium, seeking to keep core phosphite advantages while ditching toxicity. Storage stability gets careful attention—teams want to stop degradation or slow dusting, issues that drive up handling risk. Some patent applications chase synthesis tweaks, aiming to make the crystal pure while slashing synthesis waste or improving recycling options. Universities pitch ideas for immobilizing lead ions in solid-state matrices, treating the phosphite as a stepping stone for safer, higher-value ceramic phases in electronics. The R&D world pushes the technical boundaries, even if public policy urges movement away from the lead family.
Toxicity Research
The body of work on lead dihydrogen phosphite pulls together hard facts from broader lead toxicology. Scientists find that, as with other lead salts, the danger centers on its ready absorption through skin, lungs, or gut, creating risks even from a minor spill. Acute exposure causes stomach cramps, headaches, muscle weakness, and at higher doses, kidney and nervous system failure. Chronic, low-level exposure becomes the grim driver of developmental delays in children and lasting organ impacts in exposed adults—a pattern showing up again and again in population studies and workplace health surveys. Even modern, well-run plants have recorded accidental releases leading to blood lead spikes, and remediation costs for even small spills run into the tens of thousands. Ecotoxicity studies show run-off can poison aquatic life for years, bioaccumulating in river sediments and in the food chain. The regulatory world keeps limits tight—often down to parts per billion in air or water—reflecting the science showing no safe exposure threshold.
Future Prospects
Most industry insiders see usage trending downward as alternatives become safer and regulations tighten. Some small sectors, particularly those focused on fire protection for high-value infrastructure, continue to rely on the compound’s unmatched technical profile. Research investment pushes two ways—finding less toxic stand-ins, or doubling down on new handling, immobilization, and remediation technologies to contain every microgram of lead in use. A few universities and startups look at ways to convert or encapsulate lead phosphites for extended life or recycling, hoping to build success stories around circular chemistry. As global regulatory pressure builds, existing stocks see reuse under ever sharper scrutiny, and market entry for new applications seems unlikely without breakthroughs in delivering both lower risk and equivalent chemical performance. These choices reflect both the scale of human safety concerns and the stubborn technical edge some compounds bring to the table, even as the world leans away from lead chemistry.
What is Lead Dihydrogen Phosphite used for?
Inside the Chemistry of Lead Dihydrogen Phosphite
Most folks rarely hear about lead dihydrogen phosphite unless they work in chemical manufacturing or environmental safety. At its core, this compound comes from a mix of lead and phosphorous-derived compounds, ending up as a white or pale powder with a specific set of uses. When I first saw this material in a storage room at an old plastics plant, I didn’t give it much thought. It just looked like another labeled drum.
Lead dihydrogen phosphite steps onto the industrial stage as a stabilizer, especially in plastic and rubber production. Plastics tend to break down over time—ultraviolet rays, heat, and even the mixing process itself can hurt their durability. Factories lean heavily on such stabilizers to slow this process, so that pipes, cables, window frames, and many products keep their color and shape for years. Tests and field reports over decades show a clear uptick in material lifespan with the right stabilizers added.
Why Factories Keep Picking Lead Dihydrogen Phosphite
Chemists and material engineers often choose this particular phosphite for cost reasons and its ability to work under tough processing conditions. It doesn’t break down easily and blends nearly invisibly into resins that form the backbone of construction plastic. One seasoned operator in a cable plant told me, “If you want the sheath to stay white and not crack for five years, this is the stuff.” They look for something that won’t gas out or react with other additives mid-process.
On the scientific side, lead compounds like this have a proven knack for grabbing onto free radicals. These little troublemakers chew through plastic bonds and start discoloring or weakening the end product. By steering chemical reactions away from trouble and catching stray radicals, these additives keep polymer chains stronger, longer. That’s more than theory; lab teams have charted better test results for products protected by lead-based stabilizers.
Health Warnings and Environmental Tradeoffs
Here’s the rub: anything with lead in its name should make you pause. The health effects of lead exposure stack up quickly and stay in the environment longer than anyone wants. Kids are especially at risk, and that’s not just a headline—there’s a mountain of peer-reviewed evidence on developmental issues tied to trace amounts of this metal. Meanwhile, plants using these compounds need to keep a tight lid on waste streams. I’ve walked past warning signs and showers lining exits at plants just to prevent dust from hitching a ride home on workers’ clothes.
Some regions and companies pulled back from using lead dihydrogen phosphite for this reason alone. Environmental rules keep tightening. Alternatives, including calcium-zinc stabilizers and organic phosphites, have started to carve out market space, even if costs tick up or results don’t always match the durability seen with lead. Toxicity cannot be swept under the rug; numerous public health agencies call for phaseout wherever possible and proper handling where it isn’t.
Looking Toward Safer Chemistry
Upgrading chemistry labs and research budgets so that safer, affordable alternatives can hit the market calls for industry and civic leaders to share a little risk and creativity. Knowing what's in our pipes, children’s toys, or building materials should count as a basic civic right, not an afterthought. Until clear replacements become more common, plant operators and environmental inspectors carry the burden—testing, documenting, and treating every scrap with the seriousness earned by a toxic heavy metal.
What are the safety precautions for handling Lead Dihydrogen Phosphite?
Why This Chemical Matters
For folks working in laboratories, manufacturing lines, or research facilities, coming into contact with chemicals like Lead Dihydrogen Phosphite isn’t rare. We know lead isn’t friendly to the human body—pick up even the basics from a chemistry textbook, and that fact jumps out clear as day. Add a phosphate group, and you’re dealing with material that demands respect, not just familiarity.
Dangers in the Details
Lead’s effects stretch further than most realize. Breathing in powder dust or letting it get on your skin sets you up for cumulative health trouble. Chronic exposure stings hardest: headaches, nerve damage, and even kidney failure for those who shrug off the rules. Personal experience shows even veterans in the lab sometimes underestimate how quickly a stray dust cloud or careless glove can create a mess.
Phosphite brings its own set of issues. Breathing in fine particles may not sting like bleach or ammonia, but long-term, lungs can get scuffed up quietly before anyone notices. I once watched a colleague get a warning from a safety officer after dabbing sweat off with a contaminated glove—bad habits have a way of sneaking up on people.
Precaution Beats Regret
Gloves, lab coats, goggles—these aren’t window dressing. They serve up a shield against dust and accidental splatter. Not all gloves handle lead compounds equally; nitrile gloves get picked because they're tough against lead and handle phosphate chemicals without breaking down. Putting on a fresh set before every session became my personal ritual.
Ventilation matters. Not just a fan in the corner—real fume hoods draw dust and vapors away from your face. I’ve seen makeshift setups in underfunded classrooms, but cutting corners here flips a manageable risk into a health crisis. Good labs never work with this chemical outside the hood, no matter how small the sample size.
Contamination sneaks out of the lab on clothing, pens, and notebooks. Simple habits, like washing hands before and after leaving the work area, do more good than most people think. Wiping down work surfaces after each session feels tedious until you realize how much invisible dust sticks around. My old mentor drilled into us that regular cleaning saves hassle and doctor visits.
Disposal and Storage Count
After the lab work wraps up, what happens to leftover material makes a difference. Pouring waste down the drain spreads lead into local water. Standard procedure goes: seal it in clearly labeled containers, store in secure chemical waste bins, and follow the waste management service’s protocol. People who skip these steps put their whole building at risk; I’ve seen headaches and fines land on anyone careless with disposal rules.
As for storage, I learned to pay close attention to labels and dated logs. Lead compounds can’t share shelves with acids or food, and container lids stay screwed on tight. One mix-up, and the lab could face a minor emergency—or worse.
Solutions for Safer Workplaces
Training stands out as the top breaker of bad habits. Chemistry students should get more than a quick lecture about toxic compounds; labs improve safety by running regular, scenario-based drills. Supervisors don’t look for perfect compliance—they want awareness and proactiveness: people who spot hazards and speak up without fear.
Switching to less hazardous substances helps, too—some industries already swap out lead-based reagents where possible. For those who must use it, sticking with up-to-date safety data sheets ensures every worker knows what they’re handling. Over time, building a culture where everyone double-checks PPE and calls out unsafe practices has saved more than one of my colleagues from a dangerous slip. Simple habits and shared vigilance can make the difference between a safe workspace and a disaster nobody saw coming.
What is the chemical formula of Lead Dihydrogen Phosphite?
Looking at Chemical Building Blocks
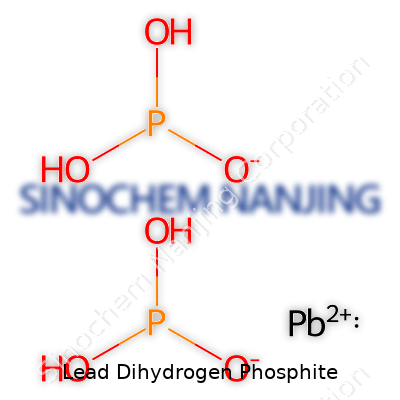
People working in material science, agriculture, and even plumbing run into chemicals with complex names that pack a punch in their day-to-day tasks. Lead Dihydrogen Phosphite stands out thanks to its structure and chemistry. Chemists often see it written as Pb(H2PO3)2. That formula tells a clear story: one lead ion binds with two dihydrogen phosphite ions. In the lab, this makes all the difference. The makeup allows certain reactivity and physical properties that have real-world consequences.
Breaking Down the Name
It’s easy to get tangled up in chemical jargon. Dihydrogen phosphite means there are two hydrogens stuck to a phosphite group (PO3). The lead in this compound shows up in a plus-two charge, a detail that influences toxicity and stability. Anyone who’s handled lead-based salts in research knows that precise formulas change everything. Subtle differences matter not just for safety sheets, but for how compounds behave out in the world.
Why This Formula Matters
You can’t talk about lead salts without talking about risk. Too many communities have seen the dangers of lead in drinking water and soil. The molecular structure of Lead Dihydrogen Phosphite means it’s soluble under certain conditions, so it has to be respected wherever it’s stored or used. Labs keep a sharp eye on labeling and containment because small mistakes ripple out to bigger impacts—both in people’s health and in the environment. Factual accuracy in naming and formula can mean the difference between a safe experiment and an accident.
Looking Beyond the Formula
Lead phosphites sometimes show up in industrial settings, especially where folks want to control corrosion or manage chemical reactions. A farmer investigating possible mineral treatments or a technician working to protect pipes from rust gets right down into the chemistry. If the formula gets muddled, mistakes happen. Supply chains can mislabel materials. Scientists and students can draw the wrong conclusions in research. For people in these jobs, understanding exactly what Pb(H2PO3)2 stands for helps keep standards high—and everyone safer.
Facts Speak Louder Than Hype
Many heavy metal salts spark tough debates—especially now, when environmental awareness rises year by year. Research shows even trace levels of lead can harm developing brains and strain public health budgets. People sometimes overlook the need for plain, factual breakdowns of compounds like Lead Dihydrogen Phosphite. Good science starts with trust, and that comes from naming things right and sharing what’s known, not dressing up the story.
Better Practices Moving Forward
The conversation has to include safe handling, ongoing monitoring, and a push for alternatives where possible. In industries where lead phosphite gets used, tighter protocols can really change outcomes. Training for those handling chemicals helps stop contamination at the source. Regulators can demand clearer labeling and more frequent audits. For students and professionals, keeping up with the literature—learning not just names but effects—grows both expertise and social responsibility.
Lead Dihydrogen Phosphite, with its formula Pb(H2PO3)2, reminds everyone that precision in science doesn’t only stay in textbooks. It finds its way into the water, land, and policies around us.
How should Lead Dihydrogen Phosphite be stored?
Why Handling Lead Dihydrogen Phosphite Matters
Lead dihydrogen phosphite catches attention because it brings real hazards to the workplace and laboratory. Too many forget that products containing lead carry serious health risks, not only for users but for anybody who shares the building. Dust from this compound causes problems if inhaled, ingested, or absorbed through the skin. Neurological and developmental damage stack up over time, especially for children and pregnant women. Phosphite ions add another layer of risk, raising the stakes in case of spills or poor ventilation.
Practical Storage Recommendations
Years of working around industrial chemicals have taught me that the best storage decisions come down to clear rules and daily discipline. Sturdy, airtight containers make the starting point. High-density polyethylene or glass with heavy-duty lids beats any cardboard box or loose bag. I’ve seen more than one lab get lazy, and every time, someone pays the price with cleanup or worse.
Keep containers out of direct sunlight and away from moisture or steam. Lead dihydrogen phosphite can react if left exposed, making both the environment and the material unstable. Temperatures matter. Store around room temperature, away from sources of heat that could cause breakdown or vaporization. If humidity gets into a container, the risk of corrosion or chemical reaction rises fast.
Segregation and Labeling
Never lose sight of the rule about keeping incompatible substances apart. Lead dihydrogen phosphite should not live near acids, strong oxidizers, or bases. An accidental mix-up with those makes for a serious accident—the sort that closes a workplace for weeks or sends someone to the hospital. I use bold, waterproof labels on every container. The best setups have dedicated shelving for each chemical type. In shared spaces, nothing beats a logbook showing who opened what, and when.
Ventilation and Containment
Never trust luck with volatile or toxic chemicals. Always pick a dry storage area with reliable ventilation. A fume hood or dedicated storage cabinet with negative pressure helps keep the room safe. I’ve talked with too many folks who thought a closet counted as chemical storage. They found out the hard way that gases will migrate, creating risks for people down the hall. Containment trays underneath shelves catch drips or leaks without delay.
Protective Measures and Accountability
Personal protective equipment, like gloves, safety goggles, and lab coats, goes hand-in-hand with good storage. No matter how careful someone tries to be, accidents happen. Frequent inspections catch cracks or aging containers long before a leak gets serious. Labs and warehouses with tight control often keep emergency gear, like spill kits and eyewash stations, within reach.
Environmental and Regulatory Considerations
Local laws shape the right approach. In my experience with regulatory audits, ignoring those rules creates only headaches and big fines. Agencies watch lead compounds closely, so records need to stay up to date. Disposal routes for any old stock or contaminated packaging must follow environmental guidelines. Not every country handles hazardous waste the same, but strong documentation always makes the process smoother and safer.
Building a Culture of Safety
Every safe storage plan relies on training everyone who could touch the chemical, from delivery to disposal. Refresher courses each year beat simple handouts. Real stories from past incidents drive the point home far better than a checklist. After seeing how serious a minor mistake can get, I always believe in a double-check, even at the end of a long day.
Is Lead Dihydrogen Phosphite toxic or hazardous?
Lead compounds raise concern
In my years working around industrial chemicals, one thing has stayed true—anything with lead gets serious attention. Lead’s reputation as a health threat isn’t just hype, either. The public health history on this metal reads like a warning label. The Center for Disease Control, the World Health Organization, and pretty much every regulatory agency out there agree that exposure to lead compounds poses a real risk, especially for children. It affects brain development, damages nerves, and harms organs. That’s not speculation—that’s medical fact.
What exactly is Lead Dihydrogen Phosphite?
Lead dihydrogen phosphite comes from the family of lead compounds. On paper, it’s a chemical with potential use in industrial settings—sometimes as a stabilizer or corrosion inhibitor. Still, none of that removes the threat that comes with the lead component. All the phosphite in the world won’t mask what lead does to the human body. If this compound gets inhaled, swallowed, or absorbed through the skin, the body can’t distinguish it from any other lead source. Workers facing regular contact with lead-based chemicals often end up with elevated blood lead levels unless they use personal protective equipment and strict hygiene measures.
Regulatory checkpoints and best practices
Government guidance doesn’t take lead lightly. The Occupational Safety and Health Administration (OSHA) sets exposure limits on airborne lead dust, fume, and mist. Europe’s REACH regulations put strong restrictions on lead use. Companies importing or using lead dihydrogen phosphite in manufacturing must comply with strict reporting, monitoring, and labelling requirements. If a spill happens, it triggers clean-up protocols that keep residues off hands, clothes, and equipment. In my own work, I’ve seen entire rooms marked off, protective suits zipped up, and equipment sealed after trust in cleaning disappears.
Symptoms and risks
It’s tough reading the charts on acute and chronic lead poisoning. Symptoms start small—mild headaches, stomach pain, irritability. Over time, loss of appetite, muscle pain, kidney strain, and memory problems tell a much bigger story. In kids, the damage comes even faster and lasts for life. Lead seeps into bones, lingers in the body, and doesn’t leave easily. According to the CDC, there is no safe level for lead exposure in children.
Safe alternatives and moving forward
Plenty of chemistry professionals now look for ways to swap out hazardous materials like lead dihydrogen phosphite. Some industries have moved toward calcium or zinc-based stabilizers, which don’t carry the same risks. Innovations keep coming as health researchers push companies to rethink the cost of using lead. Every time new tech hits the market, it signals that some companies want responsible choices—both for workers and for people living around manufacturing hubs.
A better approach is always worth the investmentIt’s easy to cut corners in the name of efficiency, but shortcuts with lead always come back to haunt. Anyone working with or near lead compounds, including lead dihydrogen phosphite, deserves transparency, training, and clear rules. Workplaces shouldn’t gamble with toxic metal exposure for the sake of convenience. In my experience, nothing beats a healthy crew going home safe at the end of the day.
| Names | |
| Preferred IUPAC name | dihydroxidophosphonato(1−)plumbum(II) |
| Other names |
Plumbous phosphite Lead(II) phosphite |
| Pronunciation | /liːd daɪˈhaɪdrədʒən ˈfɒsfaɪt/ |
| Identifiers | |
| CAS Number | [13598-12-4] |
| Beilstein Reference | 3583452 |
| ChEBI | CHEBI:86453 |
| ChEMBL | CHEMBL1201572 |
| ChemSpider | 24266145 |
| DrugBank | DB14506 |
| ECHA InfoCard | 18f04dfb-c1a9-4958-934d-25b912bc0c24 |
| EC Number | 231-263-8 |
| Gmelin Reference | 13722 |
| KEGG | C18608 |
| MeSH | Dihydrogenphosphite, lead(2+) salt (1:1) |
| PubChem CID | 165835 |
| RTECS number | OG2275000 |
| UNII | T8C90L869B |
| UN number | UN1479 |
| Properties | |
| Chemical formula | Pb(H₂PO₃)₂ |
| Molar mass | 285.2 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 6.95 g/cm³ |
| Solubility in water | slightly soluble |
| log P | -4.8 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 1.3 |
| Basicity (pKb) | 6.6 |
| Magnetic susceptibility (χ) | −47.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.574 |
| Dipole moment | 6.4 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 204.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1500 kJ/mol |
| Pharmacology | |
| ATC code | V03AB56 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause damage to organs through prolonged or repeated exposure. |
| GHS labelling | GHS07, GHS08, GHS09 |
| Pictograms | GHS07 |
| Signal word | Danger |
| Hazard statements | H302: Harmful if swallowed. H332: Harmful if inhaled. H360: May damage fertility or the unborn child. |
| Precautionary statements | P264, P270, P273, P301+P312, P330, P501 |
| NFPA 704 (fire diamond) | 2-0-0 |
| Lethal dose or concentration | LD₅₀ Oral Rat 600 mg/kg |
| LD50 (median dose) | LD50 (median dose): 220 mg/kg (rat, oral) |
| NIOSH | WA 8900000 |
| PEL (Permissible) | PEL (Permissible) of Lead Dihydrogen Phosphite: 0.05 mg/m³ (as Pb) |
| REL (Recommended) | 1 mg/m³ |
| IDLH (Immediate danger) | IDLH: Not established |
| Related compounds | |
| Related compounds |
Lead(II) phosphite Lead(II) phosphate Lead dioxide Lead(II) oxide |

