Lead Cyanide: A Deep Dive into Danger, Discovery, and Dilemma
From Historical Curiosity to Modern Concern
Lead cyanide stands as an unsettling product of our tireless pursuit to push chemistry into new frontiers. Its earliest roots took hold in the shadow of the industrial revolution, at a time when chemistry’s boundaries received steady kicks from explorers more concerned with process than peril. Early 19th-century texts mention its preparation—a marriage of lead salts and potassium cyanide—mostly in the context of curiosity or metallurgical experiments. Over the years, the blend of keen interest in synthesis and the thirst for extracting value from minerals led industries to tinker with cyanides, lead included, unworried about side effects. The world grew wise to the hazards only after accidents and toxic legacies forced people to see the true color of the products they had helped into the world. Few compounds now raise eyebrows more than lead cyanide—an echo of both technological achievement and chemical aftermath.
Product Complexity and Chemical Identity
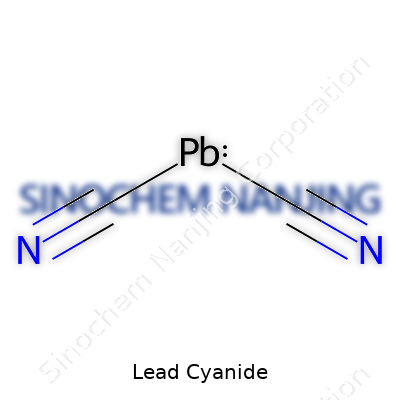
On the periodic table, this stuff is written as Pb(CN)_2, a white crystalline powder that tries to look harmless, masking the dual threat baked into its formula. Cyanide, already cursed with a reputation for swift toxicity, mingles with lead, a heavy metal whose poisonous effects need no introduction. This compound, weighty and powdery, dissolves in some acids and presents low solubility in water or ethanol, which makes it deceptively persistent in soil or waste. Chemists usually describe its reactivity as moderate under ambient conditions, but, exposed to acids or strong bases, it can unleash volatile and deadly gases. The cyanide ion inside brings risk, though it’s the pairing with lead that turns each gram into a long-term environmental threat. I remember handling compounds like this in my laboratory days—donned in protective gear, not daring to touch anything outside the fume hood, always double-checking cords and seals.
Technical Standards and Labeling Practices
Labeling lead cyanide isn’t optional, and regulators have spent years tightening the standards. Properly marked containers must scream their contents—hazards, handling precautions, response actions. Producers and warehouses abide by rules forced through after countless lessons in neglect. Safety data sheets for lead cyanide warn about wearing gloves, goggles, and storing it locked away from acids, food, and untrained hands. Where I worked, no one could accept or move a jug of this stuff without paperwork and emergency drills fresh in mind. OSHA and similar agencies flagged it as high-priority: serious toxicity, and severe environmental persistence. Regulatory oversight keeps most use highly restricted; transport falls under “hazardous material,” with full documentation for every step. Carelessness here finds little forgiveness—one slip spells contamination or a medical emergency, and the world now knows better than to ignore those risks.
Preparation and Chemical Reactions
Production has always relied on a basic approach: reacting a soluble lead salt with a potassium or sodium cyanide solution. Industrial-scale synthesis means measured, slow addition in ventilated reactors—no chemist wants a burst of hydrogen cyanide to end their day. Even small mistakes during preparation can generate cyanide gas, asphyxiating and rarely forgiving, so safety must trump speed. Once you have lead cyanide, chemical modification possibilities exist, although most labs avoid tinkering out of caution and tight regulation. It reacts violently with acids to yield hydrogen cyanide—a compound infamous for its lethal impact. Contact with oxidizers spells trouble too, risking fire or hazardous byproducts. Every bottle becomes a reminder: behind mundane chemical equations, real danger calls the shots.
Other Names, Same Warnings
In journals and trade catalogs, lead cyanide pops up under several names: plumbous cyanide, lead(II) cyanide, or even the less-used dicyanoplumbate. These aliases change little; all routes lead to the same health warnings and the same restrictive access. Whether poured from a bottle stamped ‘Pb(CN)2’ or requested by synonym, the end result stays the same—special training, airtight storage, and nervous respect for toxic legacy.
Living with Danger: Safety Measures and Operation
All workplaces handling lead cyanide face unyielding scrutiny. Safety isn’t just a rule; it is an unspoken culture built from stories of past accidents. Gloves, face shields, respirators, and chemical hoods become routine. Draining liabilities mean few modern labs risk its presence without absolute necessity. Emergency plans require neutralizing agents, anti-cyanide kits, swift access to medical care, and regulated disposal protocols. I’ve seen firsthand a team’s collective sigh of relief whenever scheduled inspections ended with no findings and no exposures. Waste comes labeled for hazardous removal, and cleaning crews trained to deal with accidental spills—not a job anyone seeks, but indispensable to the system.
Application Areas Shrinking with Time
If you look for reasons to use lead cyanide, you’ll find the list short and growing shorter. Earlier decades saw it in metallurgy and electroplating—applications that valued its cyanide for complexing stubborn metals. Industrial chemists once eyed it as a reagent or an intermediate, useful for extracting precious metals from tailings or for making pigments, though public health outcry curbed these avenues. Most societies now discourage these roles, pushing for substitution with less hazardous alternatives. Legislation keeps tightening, and emerging technology in electronics, plating, or pigment involves new compounds, designed with lower toxicity in mind. I’ve followed the pivot in academic journals, as researchers now devote their energy to removing or neutralizing toxins once considered essential.
Research: Hazards, Health, and Innovation
Toxicity gets more scientific attention than any practical value that lead cyanide brings today. Studies detail its interference with enzymes, its willingness to cross the blood-brain barrier, and its legacy of developmental harm—especially in young children. Cyanide poisoning remains well known for rapid onset; lead toxicity, slower but relentless, causes organ and neural injury over months or years. The compound’s fate, once released, troubles environmental chemists—soil retention spreads the risk far beyond original sites. Research in remediation and rapid detection outpaces any hunt for additional applications, as scientists seek trusted ways to find and destroy lead cyanide, not harness it.
Toxicity and Long Shadow
Toxicity research pulls little punch. Chronic exposure, even at trace amounts, can mean neurodevelopmental regression and kidney disease. Acute cyanide poisoning brings immediate respiratory shutdown. In fieldwork, I saw mitigation efforts turn into endless rounds of soil and water testing. Children bear the brunt, with low concentrations translating to lower IQs, shortened attention spans, and a lifetime of health care needs. Communities downwind or downstream from careless use still pay the tab for mistakes made decades ago. Peer-reviewed studies continue to confirm what local doctors observe: no safe level, no meaningful escape once widespread contamination occurs.
The Future: Beyond Legacy Chemicals
There’s a broad movement to phase out lead cyanide, joining a wave of chemical bans worldwide. If there’s hope, it lies in better detection technology—using sensors capable of reading minute traces in water or soil, and offering cleanup solutions that destroy, not just contain, the compound. Advancements in green chemistry look promising, as labs race to create complexing agents from organic feedstock that mimic the use-case without inheriting the toxic baggage. Industry gets real pushback when suggesting continued handling in non-essential processes, and insurers shy away from underwriting operations that keep lead cyanide on hand. Regulatory frameworks, community activism, and economic cost converge to cut this compound from the roster of commonly accepted chemicals. In a few decades, the only place for lead cyanide may be behind a locked glass case: an artifact, a warning, proof that even the most useful breakthroughs demand humility and accountability from their stewards.
What is Lead Cyanide used for?
What Is Lead Cyanide?
Lead cyanide’s name turns up every so often in chemical supply catalogs. It’s a pale, yellowish powder, made by mixing lead salts and cyanide compounds. Folks might ask: where does this stuff even fit into the picture? In my years following industrial chemistry, this compound rarely pops up in your average lab or workshop. Most regular people—outside of mining, metallurgy, or perhaps an old-school chemistry class—won’t ever see it in person.
Why Do Some Industries Use Lead Cyanide?
Lead cyanide finds legitimate use mainly in the mining sector, especially in extracting gold and silver. It helps dissolve precious metals from ore, allowing miners to separate out the valuable bits from the rest of the rock. There’s a legacy to its use in early 20th-century mining during the expansion years in North America and Australia. Old handbooks and journals talk about it, but today’s professionals tend to pick safer and more efficient chemicals where they can.
It crops up every now and then as a reagent in specialized laboratories. For example, some experimenting with advanced inorganic syntheses or electrochemical applications might encounter it. Still, lead cyanide’s toxic nature combined with the push for safer processes keeps it out of most hands.
Risks Tied to Lead Cyanide
Handling lead cyanide requires vigilance. It is toxic in two ways: both the lead and cyanide components cause serious harm. Cyanide interferes with cells’ ability to use oxygen, leading to organ failure. Lead accumulates in bones, nerves, and blood, interfering with brain function, growth, and every major organ. Anyone working with this compound needs real safety gear, tight protocols, and must never cut corners on disposal.
Tales float around industrial circles of accidents from mishandling—lab workers exposed without realizing, communities left with contaminated water, or wildlife poisoned downstream of old mines. These stories show the gap between paper guidelines and human mistake. Even short exposure can have lifelong effects.
Environmental and Human Health Challenges
Decades of mining left soil and rivers polluted in regions dependent on extraction for jobs. Cleanup costs run high. Entire communities pay the price through higher cancer rates, neurological issues in kids, and generational poverty tied to health problems.
The science clearly shows that runoff containing lead cyanide damages aquatic environments. Fish and insect populations drop. Birds eating those fish suffer next. These changes ripple up the food chain, eventually affecting people living nearby.
Solutions for Today
Stronger rules help: regulators now demand tighter controls, regular inspections, and phase-outs of the worst chemicals. Companies embrace alternatives like thiosulfate or other non-cyanide options in gold processing when possible. Education pushes back against shortcuts, making sure workers understand the consequences of carelessness.
In my work, I’ve spoken to professionals who see technology as the answer—better detection for leaks, precise management of waste, and robots or remote systems handling riskier steps. It shifts the conversation from “how do we work with lead cyanide?” to “can we do this without it?” That’s real progress, cutting future generations a break from today’s bad habits.
What are the safety precautions when handling Lead Cyanide?
What is Lead Cyanide and Why Should We Care?
Lead cyanide carries a scary reputation for good reason. Touch or breathe it in, and you’re dealing with two notorious toxins at once. Lead can damage organs and hang around in your body, while cyanide can cut off your cells’ ability to use oxygen. The stuff finds its way into industries like mining, chemistry labs, and sometimes even metal plating shops. Anyone in the vicinity of this compound finds themselves facing real danger.
Risks Around Exposure
Most folks may never cross paths with lead cyanide, but for those working in certain industries, the risk is real. Both skin contact and inhalation can put someone in serious trouble, sometimes with symptoms showing up hours later. Lead builds up in bones and tissues, increasing risks long after any actual exposure. Cyanide’s effects come fast, hitting the heart and brain the hardest. Ignoring safety would be reckless, not only for workers, but for families and the wider community downwind or downstream.
Practical Safety Steps
Safety gear matters every time: sturdy gloves, full-face shields, and industrial respirators block out both dust and vapor. I remember pulling on thick, chemical-resistant gloves in my early lab days; there are no shortcuts worth the risk. Even the smallest cuts or tears render that barrier useless. If you work near this compound, stick to splash-proof lab coats and closed shoes, never sandals or shorts.
Ventilation keeps air safe. Any workspace using or storing lead cyanide needs high-quality extraction fans and sealed-off work areas. No open beakers or haphazard mixing. Having worked in a facility with poor ventilation once, headaches and sore throats spread quickly—even with less toxic materials. Managers should run frequent checks on air quality, rather than relying on warning smells or symptoms.
Clean-up routines matter. No one should assume a quick mop will do the job. Specific protocols—HEPA vacuums for dust, approved chemical spill kits for liquids—protect everyone. Work surfaces get wiped down with proper neutralizers, not just soapy water. Once a spill happens, everyone needs to be trained to block off the area and report up the chain immediately.
Training and Storage
Knowledge saves lives. Every worker should know the full dangers—not just the immediate symptoms but the slow build-up and long-term risks. Hands-on drills stick better than pamphlets or online quizzes. In my own experience, the muscle memory gained from real practice means you act faster and make fewer mistakes.
Lead cyanide must stay in tight, clearly labeled containers, away from anything acidic, wet, or reactive. Containers can’t show rust, dents, or cracks. I’ve seen storage rooms with unlabeled bottles and mystery dust, and have experienced the panic that comes from not knowing what’s inside. Locked cabinets with logs and regular inspections set a much higher bar for everyone’s safety.
Wider Impact and Solutions
Safe handling doesn’t just protect workers. Runoff and dust can contaminate soil, water, and crops far beyond the plant fences. Neighbors and ecosystems suffer in silence when corners get cut. Regulators need to inspect sites more often, and communities can push for tighter standards and transparent reporting. Smart policies, combined with a serious respect for safety routines, keep disasters at bay.
Keeping people safe starts with taking lead cyanide seriously every single day. Equipment, training, storage, and oversight all come together. No shortcut or excuse is worth a lifetime of health damage.
What are the chemical properties of Lead Cyanide?
The Heft of Heavy Metals: Lead Cyanide on the Bench
Lead cyanide, with the formula Pb(CN)2, doesn’t make headlines until something goes wrong in a lab or an industrial yard. Clear, white or slightly off-white, powdered, and certainly toxic, this compound rarely makes an appearance outside chemistry circles. The dangerous duo of lead and cyanide puts it in a unique category where chemistry, safety, and real-world risk overlap in messy ways.
How Lead Cyanide Reacts
I remember from my student years that its solubility isn’t impressive—drop some into water, and most sits stubbornly at the bottom. The cyanide part offers a slight solubility boost compared to some lead compounds, but not enough to forget it’s hazardous. Solutions stay transparent unless something reacts with the cyanide ions.
Add a strong acid and things turn dangerous fast. Cyanide ions form hydrogen cyanide gas, which brings an intense almond smell and risk that no one wants in the air. The acid test in the lab proves why so many protocols start with “work under a fume hood.” On the other side, if you throw in an excess of cyanides (for example, potassium cyanide), you get soluble complexes like potassium dicyanoleadate. This trait plays a part in mining, where chemists use cyanide's tendency to grab onto metals to pull them away from ores.
Stability and Decomposition
This powder doesn’t just break down for no reason, but hit it with heat and decomposition looks ugly. Lead cyanide splits, releasing cyanogen (a poisonous gas) and leaving behind lead oxide. Both result in toxic exposure, making incineration or uncontrolled thermal events risky. Environmental incidents involving fires at facilities storing metal cyanides show just how hazardous everything gets if controls fail.
Where’s the Danger?
Sitting at the junction of two big-league toxins, exposure brings the worst of both worlds: lethal acute toxicity from cyanide and chronic damage from lead. Cyanide goes right after enzymes involved in cellular respiration, blocking energy in living cells. Lead collects in organs over years, damaging nerves, kidneys, and more. Laboratories keep lead cyanide behind lock and key, in dry, labeled cabinets, for a reason.
Spills call for more than paper towels. Sodium hypochlorite, the active ingredient in bleach, helps to break down spilled cyanides. Still, no cleanup happens without good gloves, face protection, and, where possible, full-on respirators. Even trace amounts entering municipal water raise alarms. The EPA and similar agencies in other countries do not play around with allowable levels.
Toward Safer Handling and Reducing Risks
Hard-learned lessons in chemical safety keep strengthening the regulations around manufacturing, moving, and disposing of substances like lead cyanide. I’ve seen tighter tracking and greater emphasis on engineering controls even in university labs. Digital logs, double containment, emergency stations, and mandatory reporting of every incident form a safety net. Sourcing non-toxic alternatives whenever possible continues to improve practices in industry and academia alike.
It’s easy to dismiss a compound like lead cyanide as rare and unlikely to cause harm, but history shows that chemicals in storerooms often find surprising paths into air, water, or living tissue if ignored. Keeping chemistry rooted in respect for the power of materials and clear-headed risk management matters every step of the way.
How should Lead Cyanide be stored and disposed of?
A Dangerous Compound With Serious Consequences
Lead cyanide doesn’t belong anywhere near a home garage or a regular toolbox. Handling something like this brings risks that run beyond what most folks expect. In labs and certain factories, people use it because it works for very specific jobs, mostly in making chemicals. Nobody wants to end up with contamination or exposure, because the health problems caused by lead and cyanide together can last for years—affecting nerves, organs, and even passing through soil to get into groundwater.
Putting Safety First—Starting With Storage
Safe work with lead cyanide starts with keeping it away from everyone except trained professionals. Sturdy, airtight containers with chemical-resistant seals protect workers and the environment. The old paint can from the hardware store won’t cut it. Containers should feature clear hazard labeling that warns about both the lead and cyanide risks. There’s no room for mistakes—mixing up containers in a crowded storeroom can spell big trouble.
Materials like polyethylene or glass stand up to harsh chemicals and keep their shape in a fire or if dropped. Locks and limited access keep unauthorized hands away. Storage rooms need serious ventilation—it only takes a tiny whiff to start causing harm. Temperature control helps too; high heat can break down some chemicals, turning a small risk into a big one.
Disposal—Not A DIY Project
I’ve seen too many stories of toxic waste dumped in ditches, only to cause health problems for whole neighborhoods years later. Lead cyanide sits high on the list of substances that demand a hands-off approach. Facilities with hazardous waste permits take the lead here. Workers use personal protective gear—gloves, masks, eye shields, the works—before they get anywhere near the stuff.
According to agencies like the EPA, every step must be documented. Waste handling outfits use special containers and ship them with tracking forms so nothing gets lost. Chemical treatment at licensed plants neutralizes the cyanide; lead ends up locked inside containers sealed for long-term burial in secure landfills. This way, nobody can dig it up or accidentally open it decades later. Sending lead cyanide down the drain or out with the garbage leads to criminal charges and destroys public trust in the system.
Rules and Good Habits
Plenty of laws back up these careful steps. OSHA lays down rules for workplace safety. The EPA and state agencies spell out disposal laws in detail. I’ve learned along the way that following the law only gets you so far, though. Building a culture where people speak up about unsafe storage or sloppy disposal means fewer accidents and better health outcomes.
Finding Smarter Solutions
The best way to cut risk comes from using less of these toxic chemicals in the first place. Research and development can switch out lead cyanide for something safer in manufacturing. Until those shifts happen, people working with it need equipment, training, and the right attitude—the kind that keeps hazards away from themselves and their neighbors. Preventing disaster isn’t fancy. It means showing up and getting the small things right every time, long after headlines move on to other topics.
Is Lead Cyanide toxic or hazardous to health?
Understanding the Risks
Lead cyanide sits quietly on lab shelves and inside industrial storage drums, but its quiet reputation masks a dangerous secret. Both lead and cyanide pose threats by themselves, and together, they pack a double punch. Anyone working around chemicals knows the importance of respecting what’s in a bottle or barrel—sometimes, the first mistake can be the last.
What Makes Lead Cyanide Toxic?
Lead cyanide contains lead, a heavy metal infamous for damaging developing brains, kidneys, and blood cells. You start to see the harm unfold not just in factory workers, but in neighborhoods near sites where lead-containing compounds get released. As a child growing up in a city with peeling paint and industrial leftovers, I saw classmates miss school days and struggle with learning. Later, we learned about blood lead levels, not playground tag.
Cyanide adds to this trouble. It can block cells in the body from taking up oxygen—a cruel trick that brings quick, sometimes lethal results. Put simply, a dose of cyanide can rob tissues of the breath of life. Even when symptoms don’t knock someone out right away, the leftovers can linger and weaken the body over months or years.
Industry Practices and Oversights
Factories and research centers use lead cyanide for a reason: it works well in processing metals and making some pigments. The gear and safety checks matter a lot. Gloves and masks keep workers safe for a shift, but trouble starts when practices get sloppy or gear breaks down. Dust or solution left on skin, forgotten spills, or poor ventilation in small labs leads to breathing or absorbing the stuff.
Many places still lack strict enough rules on monitoring exposure. Some sites rely on old fans and faded warning signs instead of testing rooms for airborne lead or cyanide. Folks bring fine dust home on clothes. Kids and pregnant women face even larger risks when contamination seeps into houses and water pipes.
Data Paints a Stark Picture
Lead’s damage stacks up fast. In the United States alone, CDC keeps sounding the alarm over children exposed to even small traces of lead. Symptoms like learning trouble and mood swings can follow for years. Cyanide poisonings, often from accidents or improper waste handling, show up in emergency rooms with the worst outcomes when help comes too late. The trouble is, both toxins can fly under the radar until someone collapses or symptoms linger without clear cause.
Long ago, the solutions seemed simple—just ban lead and cyanide where possible. Yet, industries hold on because switching brings cost and complexity. Fortunately, there’s been movement toward better substitutes and tougher oversight. But change only sticks when workers, families, and local officials push for better protection and regular health checks.
Moving Toward Safer Futures
Training brings knowledge, but routine blood testing and air monitoring give real-world backup. Companies with strong safety cultures keep thorough logs, offer health screenings, and clean up forgotten corners. Community groups show up to meetings and press for answers when they worry about strange odors or rashes.
Trust comes from openness—posting test results, cleaning up old sites, and helping workers understand the dangers. Meanwhile, technology brings new tools. Sensors get smaller and cheaper every year. More companies can now track chemical leaks in real time, giving everyone a little more peace of mind.
Lead cyanide proves that a hazard doesn’t need to shout to be deadly. Respecting the risk, investing in cleaner alternatives, and pushing for honesty in reporting—these are the shields that can protect tomorrow’s kids and workers. Ignoring these lessons comes at a cost no community can afford.
| Names | |
| Preferred IUPAC name | dicyanolead |
| Other names |
Plumbous cyanide Plumbum cyanatum Cyanogen plumbate(II) |
| Pronunciation | /ˈliːd saɪəˌnaɪd/ |
| Identifiers | |
| CAS Number | 592-05-2 |
| Beilstein Reference | 358993 |
| ChEBI | CHEBI:86447 |
| ChEMBL | CHEMBL3340505 |
| ChemSpider | 22816 |
| DrugBank | DB11543 |
| ECHA InfoCard | EC 208-056-1 |
| EC Number | 209-084-0 |
| Gmelin Reference | 85424 |
| KEGG | C18744 |
| MeSH | D007857 |
| PubChem CID | 166834 |
| RTECS number | OU9100000 |
| UNII | WY6I6QSY38 |
| UN number | 1588 |
| CompTox Dashboard (EPA) | `DTXSID4044705` |
| Properties | |
| Chemical formula | Pb(CN)₂ |
| Molar mass | 283.25 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 6.44 g/cm³ |
| Solubility in water | insoluble |
| log P | -0.71 |
| Vapor pressure | Negligible |
| Acidity (pKa) | Approximate pKa = 9.2 |
| Basicity (pKb) | 11.58 |
| Magnetic susceptibility (χ) | Diamagnetic |
| Refractive index (nD) | 2.19 |
| Dipole moment | Zero Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 199.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -174.8 kJ/mol |
| Pharmacology | |
| ATC code | V03AB56 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or in contact with skin; may cause cancer; contact with acids liberates very toxic gas (hydrogen cyanide). |
| GHS labelling | GHS07, GHS08, GHS06, Danger, H301, H331, H341, H350, H360, H373, H410 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H302, H332, H350, H360Df, H373, H410 |
| Precautionary statements | P273, P280, P302+P352, P305+P351+P338, P310, P330, P501 |
| NFPA 704 (fire diamond) | 3 2 2 |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 oral rat 10 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 10 mg/kg |
| NIOSH | CY7350000 |
| PEL (Permissible) | 0.15 mg(Pb)/m3 |
| REL (Recommended) | 0.015 mg/m3 |
| IDLH (Immediate danger) | 25 mg/m³ |
| Related compounds | |
| Related compounds |
Lead(II) nitrate Lead(II) acetate Lead(II) carbonate Lead(II) oxide Potassium cyanide Sodium cyanide |

