Lead Chromate: A Closer Look at Its Story, Science, and Impact
Historical Development of Lead Chromate
Lead chromate, known for its bold yellow hue, has roots grounded in the 19th century. Early pigment-makers discovered its bright, sunlight-stable color offered artists richer options. History tells us that Antoine Jean Antoine A. B. Guimet introduced “chrome yellow” around 1809, and painters like Van Gogh loaded it onto their canvases for vivid sunflowers or luminous skies. Factories grew busy, mixing lead nitrate with potassium chromate to produce this pigment on an industrial scale. Back in those days, few people worried about the long-term effects of such vivid colors—what mattered was the gleam and staying power on the surface. As industrial chemistry evolved, lead chromate moved from artists’ palettes into road markings, plastics, and even children’s toys. Laws caught up much later, but the pigment’s story is tied as much to creativity as it is to cautionary science.
Product Overview
A staple in the world of pigments, lead chromate stood out because it delivered an intense, opaque yellow unmatched by simple iron oxides or natural earth tones. Pigment makers prized its covering power and ability to produce color shades from greenish yellow to deep orange, which led to its adoption in paints, plastics, ceramics, and industrial finishes. The chemical sits in the family of chromates, meaning it contains both a lead atom and a chromium atom in its structure, which are both heavy metals with environmental relevance.
Physical & Chemical Properties
Lead chromate presents itself as a bright yellow to orange powder, gritty and insoluble in water. At a practical level, it resists the effects of sunlight and moisture better than most older natural pigments. Its density is high, owing to the lead content. Chemically stable in neutral conditions, it breaks down in strong acids or bases, releasing chromium and lead ions—this makes contamination and exposure a significant concern. Its color comes from specific electron arrangements in the chromate ion, absorbing in the blue-violet part of the spectrum so our eyes see yellow.
Technical Specifications & Labeling
Today, it’s hard to find lead chromate without warning labels—hazardous substance markings, risk phrases, and instructions for safe handling are common requirements. Regulators demand clear hazard communication because this pigment contains lead and hexavalent chromium, two substances with long records of toxicity. Labeling never stays the same; more research usually means tougher wording down the road. Professionals who work with it keep exposure limits in mind and treat powder spills and dust clouds as major safety hazards.
Preparation Method
Traditional synthesis involves mixing solutions of lead(II) nitrate and potassium chromate, causing yellow lead chromate to precipitate. Chemists learned to tweak the method, changing temperature, pH, or the use of additives, to adjust particle size and color shade. All of these tricks matter when manufacturers want a specific brightness or tint. The process generates waste streams containing chromate and nitrate ions, which call for proper treatment. This preparation route is straightforward, but every step kicks up regulatory oversight and disposal needs.
Chemical Reactions & Modifications
Lead chromate reacts with hot acids, particularly hydrochloric or sulfuric, breaking down into lead salts and soluble chromate. Alkaline conditions can turn yellow lead chromate into orange-red basic lead chromate or even green chromium oxides after further reactions. Modification usually means surface coating or stabilization, giving the pigment a tougher time leaching harmful ions or fading outdoors. Researchers often focus on ways to coat the particles with inert substances, like silicates, to slow down environmental problems.
Synonyms & Product Names
People in industry or art might know lead chromate as “chrome yellow,” “C.I. Pigment Yellow 34,” or basic lead chromate when describing redder shades. Each name circles back to the same challenging group of chemicals: the ones that brought color and then brought concern. Sometimes paint tins skip the direct chemical name, but every artist and professional who’s worked with “chrome yellow” recognizes its peculiar weight and boldness.
Safety & Operational Standards
Factories and labs working with lead chromate follow strict occupational safety rules. Personal protective gear—respirators, gloves, goggles—isn’t optional. Dust control and air filtration keep airborne particles in check to protect workers from inhaling lead and chromium. Proper disposal routes reduce the risk of these metals contaminating soil or water. Over my years in laboratory settings, the lesson has always been clear: you make the job safer through engineering controls and training, not just paperwork.
Application Area
Lead chromate earned a long roster of uses once because no alternative matched its brightness and weather resistance for decades. It found its way into bold paint for road markings, signs, and heavy machinery. Even beyond paint, this pigment showed up in plastics, ceramics, and even some older latex products, leaving its mark on both infrastructure and the environment. Today, plenty of companies have moved on, swapping in organic pigments or safer oxides, but in some developing markets, lead chromate still shows up—usually where bright yellow is the standard or regulations aren’t as tough.
Research & Development
No scientist ignores the dangers tied to the pigment anymore. Research teams now put major effort into creating alternative yellow pigments that lack lead and hexavalent chromium. Work also focuses on stabilizing lead chromate through coatings or binding methods that keep it from leaching into air, water, or soil. Some researchers have tackled waste treatment, running tests on chemical agents that convert dangerous ions to safer forms, or studying natural bacteria that detoxify chromate. Funding and published studies from Europe, North America, and Japan show a clear direction: safer is always the goal.
Toxicity Research
Evidence continues to stack up against lead chromate. Scientists know both lead and hexavalent chromium affect nervous systems, damage kidneys, and create cancer risks in workers and communities. Case studies of paint workers and pigment makers display higher rates of lead poisoning and cancers like lung and stomach tumors. Toxicologists have measured pigment persistence in soil and water, showing how long-lived and mobile exposed chromate ions become. As monitoring technology improves, even low-level environmental traces draw attention. Regulatory bodies, including the EPA and European Chemicals Agency, have flagged this pigment for nearly every type of hazard rating.
Future Prospects
Fewer industries lean on lead chromate these days because public health laws have gotten sharper, alternatives have improved, and lawsuits reset risk calculations. The future likely brings a continued drop in production and more aggressive replacement by organic and mixed-metal oxide pigments, which offer brightness without serious health risks. In tougher regulatory climates, manufacturers avoid it entirely, not just to meet the law but to build consumer trust. In other parts of the world where regulations haven’t caught up, environmental groups and NGOs push for change. As the history of lead chromate shows, sometimes a pigment can become a case study for the costs and benefits tied to industrial chemistry—shining bright at first, but leaving tough questions for generations.
What is lead chromate used for?
Why Do Industries Choose Lead Chromate?
Lead chromate paints a really bright picture—in a literal sense. This chemical gives yellow road lines their harsh, unmistakable color. It also gives school bus paint that unmistakable yellow. Factories have relied on it for well over a century because few substances match its color strength or shine. Artists once reached for “chrome yellow” whether they were making signage, advertisements, or old posters. It sticks to surfaces in a way that makes wear and tear less likely, which matters if you’re painting roads or vehicle parts.
It stretches beyond just paint. Plastics companies use it to make everyday items look cheerful—think of yellow toys, kitchen tools, or electrical parts. If plastic needs to stand out, lead chromate can pack a punch. Folk working with pigments also know that it holds color well when mixed with certain chemicals. Ceramics industries tap this property for tiles and decorative glazes. Printing houses—especially back in the day—saw it as a go-to for eye-catching inks and packaging.
The Real Trouble with Lead Chromate
Things start to get more serious with a look at health. Lead is notorious for messing with the brain and nervous system. It stunts children’s growth, triggers learning problems, and causes lasting harm even with small-scale contact. Chromate contains hexavalent chromium, which troubles the lungs and causes cancers when inhaled or ingested. Paint chips, dust, or fumes from manufacture drift into the environment and end up inside people, water, and animals. Several years ago, road paint in developing countries turned out to be a hidden source of both these toxins, poisoning workers and bystanders alike.
Experts agree—there’s just no safe way to handle substances like this for the long haul. The challenge is that old infrastructure still contains lots of it. Scraping paint from bridges or ships, or recycling plastics or metals, kicks up contaminated dust. Many workers struggle with equipment and safety clothing. Regulations aren’t always enforced well, either. It creeps into food when pottery with bright yellow glazes chips or breaks. Even some turmeric imported from abroad tested positive for lead chromate, added to punch up the spice color.
Time for Safer Choices
Luckily, progress doesn’t just mean sacrifice. Alternatives aren’t science fiction anymore. Many factories now use organic pigments for paints, plastics, and printing. Pigment chemists keep refining these replacements to match the vivid look of lead chromate, minus the risks. More roads get painted with titanium dioxide blends and other safe options. The European Union cracked down—banning most uses of lead chromate—based on expert evidence. Many paint companies pledged to change formulas after pushback from environmental and public health groups. In the U.S., limits on usage keep growing stronger as cities and states look at the evidence.
Personal experience matters too. Anyone who has seen the impact of lead poisoning—kids missing out on potential, families facing bills and heartbreak—knows why the switch is overdue. Vigilant labeling, routine product checks, and top-line workplace safety gear make a big difference. Community education helps consumers ask questions about where and how products get made. Standing up for safer options means fewer kids or workers suffer, brighter futures for neighborhoods, and more trustworthy businesses on store shelves.
Is lead chromate hazardous to health?
Bright Pigment, Dark Price
Lead chromate gives us bold yellows and oranges in paints, plastics, and road markings. That flash of color might catch your eye on the street or in old building trim. Years ago, I used to walk past construction sites splashed with these colors and never thought twice. The trouble with pigments like lead chromate hides behind its shine.
Lead and Chromium: Both Pack a Punch
There's no dodging the facts here: lead is a neurotoxin. Even small exposures can slow brain development in children. Lead builds up in bones over time and causes headaches, memory problems, stomach pain, and even kidney failure. Adults suffer too—blood pressure, heart, and fertility can all get hit. The CDC doesn’t mince words on this. Any exposure adds risk.
Lead chromate doesn’t just stop at lead. Chromium in this compound, especially hexavalent chromium (Cr(VI)), increases cancer risk. The International Agency for Research on Cancer put hexavalent chromium on its list of known human carcinogens. Factory workers dealing with pigment dust have shown higher rates of lung cancer. A simple paint job becomes a gamble when old coatings start to chip away.
Environmental Mess Lingers
Once this stuff peels, it ends up in the soil. Heavy metals don’t break down. Water runoff can carry them to rivers and plants. I’ve seen urban gardens thriving on old industrial lots while researchers find traces of toxic metals in the veggies. Neighborhoods near old factories often report higher childhood blood lead levels. The link doesn’t come from statistics alone—you see the aftermath in health clinics and emergency rooms.
Why Bother With Lead Chromate Anymore?
It sounds obvious to drop such a hazardous material, but lead chromate sticks around in lower-income countries and in products that bypass regulation. Banning it in the US and Europe brought childhood lead poisoning cases way down. Removing it from gasoline and paint, scientists found, saved billions in future health costs according to the EPA.
I once helped repaint a school built in the 1950s. You see kids playing under eaves lined with old lead paint, dust swirling every time a ball slams against the wall. Even today, plenty of renovations uncover these hidden dangers. Local governments often scramble for funds to remediate or inform families.
Safer Options and Action Steps
Lead-free pigments work just as well for plastics, traffic markings, and household paints. They cost a bit more, but regulators and big purchasers can demand safer substitutes. In my experience, the difference shows up most when you look at health spending—lead cleanups and medical bills eat far more money than safer materials ever would.
The simplest step is checking labels—look for “lead-free” whenever buying paint for home projects. For bigger jobs, cities can do more by tracking old uses and warning the public where lead chromate may lurk. When workers do need to handle hazardous coatings, training, proper gear, and regular testing protect lives and families.
Lead chromate poses risks we don’t need to take. The evidence stands clear. Pushing for safer materials, enforcing limits, and staying informed not only protects individual health but gives communities a shot at a safer, brighter future.
What are the main properties of lead chromate?
Color, Stability, and Use in Everyday Products
Bright yellow grabs attention the moment you see lead chromate on paper or in paint. Artists and industrial designers have relied on that punch of yellow for generations. One thing about this chemical—its ability to keep color from fading in harsh sunlight led to its use in outdoor paints and road markings for decades. Old school street signs, even early model toy cars, held their shine thanks to this pigment.
Chromate minerals don’t dissolve easily in water. Rain can pound on lead chromate lines for years, and they will stubbornly resist fading or washing away quickly. That stubbornness comes from a tight crystal structure. The pigment’s high density gives it heft, so it sticks where it lands and provides solid coverage. If someone’s painting wood, metal, or even concrete, it lets them get a clear, bold line in one pass.
Hazards and Tighter Rules
Lead brings a different chapter to the story. People and animals are at risk if dust or flakes of lead chromate get swallowed or inhaled, and this proved true in workplaces over time. Workers have shown high blood lead levels from breathing in pigments, leading to damage to nerves, kidneys, and even young children’s development. I grew up in a house with chipped yellow trim, and later learned a lot of those paints used lead chromate. Parents like mine had to bring in professionals to check, scrape, and seal old woodwork, just to keep their kids safe.
Chromate also brings carcinogenic properties. That threat led public health groups to push for strict bans. The European Union and many U.S. states clamped down, with paint and plastics industries moving away from lead-based compounds. Newer rules limit exposure and promote safer substitutes, pushing companies toward organic and iron oxide-based yellows. Companies that once turned out drums of lead chromate every year now focus on collecting, recycling, and sealing away the old stuff.
What’s Next for Old Paint and Industrial Sites?
Community groups face long-term cleanup jobs. On some city blocks, lead chromate flakes from paint seep into soil. Kids still get exposed—especially in older neighborhoods—so regular soil testing helps. Swapping out contaminated dirt costs money, but it’s necessary. On the positive side, more states and cities invest in awareness campaigns so owners can recognize hazards and hire pros to handle removal.
Factories that once produced lead chromate deal with buried waste. I met a team in Michigan dealing with an old pigment plant. Rather than ignore the issue, they tested groundwater, tracked shifting soil, and used thick barriers to keep toxins from spreading. Sharing information helps, whether someone’s a homeowner in a historic district or a city inspector on a tight budget.
The long reach of lead chromate serves as a reminder about using chemicals with care and looking beyond just the quick payoff of a vivid yellow. Bright paint loses its charm if it lingers in the wrong places. Ending wide use calls for continued investment in cleanup, education, and forward thinking from industries and local leaders. Tracing the path from pigment to public health teaches lessons about responsibility—ones worth carrying forward as we invent and build around us.
How should lead chromate be safely handled and stored?
What Makes Lead Chromate Dangerous?
Lead chromate offers a bright, reliable yellow pigment, but both lead and chromium in it threaten human health. Breathing dust or touching surfaces contaminated with lead chromate puts workers at risk for lead poisoning and cancer. Even low exposure builds up over time and can cause brain and kidney trouble, especially in kids. Regulations exist for good reason: no one wants to bring these problems home or see a lifelong health issue caused by a shortcut.
Workplace Controls That Matter
People can say "just wear gloves," but simple solutions break down. Real safety depends on a proper system. In manufacturing spaces where I’ve worked, good ventilation always makes a difference. Dust from pigments like lead chromate finds ways to float and settle, so local exhaust fans and negative pressure keep it from reaching the rest of the shop. Old-timers tell stories about jobs where yellow pigment covered their arms. Today, rules require keeping personal protective equipment on — gloves, long sleeves, eye protection, and at times a fitted respirator. It’s not about paranoia, just being honest about what lead exposure does to families.
Storage Isn’t Just Locking It Away
Safe storage isn’t only for keeping unauthorized hands out. Proper containers matter—the kind designed to seal tightly, made of material the pigment won’t corrode. Marking every container in clear lettering keeps those on night shift or new hires from accidental contact. I’ve seen a few close calls over the years when bins lost labels or slipped out of sight. Rigid inventory logs back up the physical barriers by tracking every container from delivery to disposal. No shortcuts, even if someone insists it wastes time.
Caught Spills: Quick, Not Sloppy
If a spill happens, letting it sit tempts fate. I learned early on to treat every spill as if it’s in the worst possible spot—floor cracks, worktables, near drains. Dry sweeping kicks up dust, so trained teams use HEPA vacuums or wet cleaning methods. Waste heads right into hazardous material bags, then those go in drums for professional disposal. Workers need to know where these supplies live and must feel permitted to stop production to clean up. Too many places skimp here, thinking cleanup will “just take a minute.” That’s not how you build lasting safety culture.
The Law Steps In for a Reason
Government rules don’t show up in a vacuum. Agencies like OSHA cite specific exposure limits and spell out steps for handling lead chromate. These guidelines stem from decades of hard-earned science and accident reports. Ignoring them gets companies fined but, more importantly, puts real people at risk. I remember a supervisor getting visibly upset once when he thought a shortcut got by under his nose—not out of bureaucracy, but because he’d watched a friend’s health decline after years of exposure.
Training and Culture Keep People Safe
Every new worker needs the story, not just the written checklist. Periodic safety training gives people the confidence to speak up and spot hazards. Supervisors deserve a thankless job: reinforcing rules and checking protective gear isn’t missing. Rewarding clean work habits, not just fast output, trains everyone to value safety as much as profit. This mindset has to last longer than one quarter or inspection cycle—word spreads fast in an industry about which shops care about their people and which treat workers like numbers.
A Safer Future in Chemistry
Companies keep reviewing alternatives to lead chromate, since legal and health pressures mount worldwide. Some newer pigments compete on brightness but don’t bring the same health risks. Until then, workplaces handling lead chromate owe their teams the most careful protocols in the playbook. Protecting workers shouldn’t be a favor; it’s the floor, not the ceiling, for any operation worth respecting.
Is lead chromate banned or regulated in certain countries?
Understanding Lead Chromate and Where It's Banned
Lead chromate’s bright yellow and orange shades once colored everything from road lines to candy wrappers. The pigment’s vibrant look isn’t the only thing that catches attention—its chemical makeup contains both lead and hexavalent chromium, two substances that can damage health and pollute communities. Researchers and health officials have spent decades piecing together what happens when people meet lead chromate in everyday life, linking it to developmental problems in children and certain cancers for those working with it day after day.
Governments in many places responded to these dangers with strict bans and heavy regulation. In the European Union, laws like the REACH regulation put lead chromate on the list of substances nobody can use without a special permit—hard to get and rarely granted. Canada chose to ban its use in consumer paints. In the United States, lead chromate is one of several lead compounds facing limits under the Toxic Substances Control Act, though some loopholes remain, especially for industrial and military uses.
Why Regulation Still Matters
I’ve lived in areas where old buildings have shed layers of paint for years. Kids play nearby and nobody thinks twice about the dust swirling around. When lead chromate turns up in those flakes, trouble follows. The pigment breaks down into dust, spreading lead and chromium particles into the soil and the air. Just a small amount in the bloodstream can stunt a child’s growth or cause behavioral issues that last a lifetime. Workers spraying paint or recycling car batteries breathe in those same particles, sometimes without realizing the long-term price they’ll pay.
The story with regulations reads less like a one-time fix and more like an ongoing fight. Some manufacturers argue they just can’t match the durability or colorfastness of lead chromate with substitutes. Paints meant for marking roads or outdoor metal structures seem especially tricky to replace. It’s easy to find articles from trade associations pushing for continued exemptions, hoping to avoid costly changes to old formulas. Yet every delay means another generation living with tainted soil and risky workplaces.
Global Trade and Enforcement Challenges
Countries with strict bans can still end up importing products finished elsewhere using lead chromate. It happens in plastics, children’s toys, and paints for toys. Enforcement doesn’t always keep up with global trade, leaving gaps that companies can slip through—sometimes without even knowing. Customs officers face a tough job checking for chemicals hidden in finished goods. In markets where enforcement lacks teeth, industries keep turning to cheap pigments, ignoring health warnings. Not every country has the funding or political will to test, fine, or recall on a large scale.
Paths Toward Healthier Alternatives
Some companies succeed in phasing out lead chromate, switching to safer pigments. They spend time and money on research, tweak manufacturing methods, and share what works with others. In my experience, progress moves faster when governments set clear deadlines. Firms carve out resources for compliance, knowing that business as usual just won’t cut it. Community groups step up to pressure officials for tougher checks, especially when local hospitals start reporting lead exposure among kids.
It changes the game when citizens care enough to ask about paint ingredients, question building materials, and demand action from leaders. The risk that lead chromate brings doesn’t respect national borders, so real action means collaboration across industries and governments. Following through looks like replacing hazardous pigments, holding companies responsible, and testing consumer goods before they hit store shelves. Without that, the legacy of lead chromate continues far longer than anyone planned.
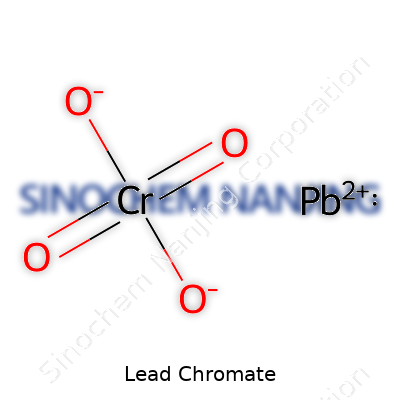
| Names | |
| Preferred IUPAC name | dioxido(dioxo)chromium; lead(2+) |
| Other names |
Chrome yellow C.I. Pigment Yellow 34 Chromic acid lead(2+) salt Lead(II) chromate C.I. 77603 |
| Pronunciation | /ˈliːd ˈkroʊ.meɪt/ |
| Identifiers | |
| CAS Number | 7758-97-6 |
| Beilstein Reference | 8541322 |
| ChEBI | CHEBI:83155 |
| ChEMBL | CHEMBL511170 |
| ChemSpider | 21515 |
| DrugBank | DB14066 |
| ECHA InfoCard | ECHA InfoCard: 014736236368-44 |
| EC Number | 231-846-0 |
| Gmelin Reference | Gmelin Reference: 2089 |
| KEGG | C14072 |
| MeSH | D007857 |
| PubChem CID | 24413 |
| RTECS number | OV8740000 |
| UNII | VWR59812AJ |
| UN number | UN1479 |
| CompTox Dashboard (EPA) | CompTox Dashboard (EPA) of product 'Lead Chromate' is **"DTXSID8020825"** |
| Properties | |
| Chemical formula | PbCrO4 |
| Molar mass | 323.19 g/mol |
| Appearance | Yellow crystalline powder |
| Odor | Odorless |
| Density | 6.3 g/cm³ |
| Solubility in water | insoluble |
| log P | 1.74 |
| Vapor pressure | Negligible |
| Basicity (pKb) | Basicity (pKb) of Lead Chromate: 6.24 |
| Magnetic susceptibility (χ) | '−47.0·10⁻⁶ cm³/mol' |
| Refractive index (nD) | 2.31 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 332.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -357.0 kJ/mol |
| Pharmacology | |
| ATC code | V09MX04 |
| Hazards | |
| Main hazards | May cause cancer, is toxic if swallowed or inhaled, and causes damage to organs through prolonged or repeated exposure. |
| GHS labelling | GHS02, GHS06, GHS08, GHS09 |
| Pictograms | GHS07,GHS08,GHS09 |
| Signal word | Danger |
| Hazard statements | H301+H331, H311, H350, H360Df, H372, H410 |
| Precautionary statements | H261, H301 + H331, H350, H360Df, H372, H410, P202, P210, P260, P264, P270, P273, P280, P301 + P310, P302 + P352, P304 + P340, P308 + P313, P321, P330, P332 + P313, P337 + P313, P363, P391, P405, P501 |
| NFPA 704 (fire diamond) | 2-0-1-OX |
| Lethal dose or concentration | LD50 oral rat: 12,000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 12.8 mg/kg |
| NIOSH | 0106 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Lead Chromate is 0.05 mg/m³ (as Pb, OSHA PEL) |
| REL (Recommended) | 0.012 mg/m³ |
| IDLH (Immediate danger) | 15 mg/m3 |
| Related compounds | |
| Related compounds |
Chromium(III) oxide Lead(II) oxide Lead(II) chromate Barium chromate |

