Lead Bromate: A Commentary on Its Chemistry, Uses, and Impact
Historical Development
Lead bromate has been part of the larger story that tracks human curiosity with heavy metals and halogen chemistry. Its discovery goes back to studies carried out in the 19th century, as chemists started looking into the properties of both lead and bromine compounds. Laboratories in Europe, especially those in Britain and Germany, looked to combine metals with new elements freshly extracted from seawater brines. Early published papers show documentation of crystals of lead bromate forming during reactions between lead salts and bromates derived from vegetable ashes. In those years, safety guidelines did not exist and these researchers experimented without fully understanding the health implications of lead exposure. Over time, the compound gained some niche attention in the pigment industry and academic work studying oxidation reactions. Lead bromate was never mass-produced like other lead salts, yet over the decades, it remained an item for specific technical and research needs.
Product Overview
Lead bromate presents itself as a compound combining lead and the bromate anion. It does not show up in daily life or industrial catalogs as often as other lead salts do. Its main buyers are academic and analytical laboratories. Some suppliers list it for sale in high-purity grades, aimed at clients conducting chemical synthesis, photochemical studies, or niche testing for bromate ions. Other than research purposes, the compound rarely enters manufacturing lines given regulatory hurdles around lead usage, especially in Europe and North America. Any commercial presentation tends to come as a fine white or off-white powder.
Physical & Chemical Properties
Lead bromate comes with a molar mass around 367.01 g/mol. Its formula, Pb(BrO3)2, tells a clear story of one lead ion attached to two bromate ions. In air, the solid holds a crystalline form, almost always appearing as odorless white powder. It shows poor solubility in cold water, but dissolves a bit more as heat rises. Hot water brings about partial decomposition, releasing toxic bromine vapors. The crystals are dense, nearly 6 g/cm3, and stable unless exposed to reducing agents, heat, or direct sunlight for prolonged periods. Lead bromate does not burn but can encourage the combustion of organic materials because bromates act as strong oxidizers.
Technical Specifications & Labeling
Technical sheets highlight the need for tight controls over purity, usually guaranteeing lead bromate at levels over 98%. Suppliers list maximum tolerance for impurities like chloride, sulfate, and nitrate ions—each must fall below 0.1% for research-grade product. Labeling always signals clear toxicity warnings and oxidizer status, following global harmonized system (GHS) standards. Packaging text must reinforce the compound’s hazards, with skull and crossbones icons and strict storage guidance (cool, dry, closed containers). Hazard labels list risks from skin/eye contact, inhalation, and dangerous reactions with flammable goods or acids.
Preparation Method
Synthesis starts with dissolving a soluble lead(II) salt (frequently lead nitrate) in water, followed by the slow addition of a bromate salt like sodium bromate. The exchange produces a fine precipitate of lead bromate, which settles as a heavy powder at the bottom of the reaction vessel. After separation by filtration, the product is washed with cold water and dried. Anyone who has worked with lead chemistry will recall the headaches of managing highly toxic sludges and cleaning all containers thoroughly after each batch—regulations for hazardous waste apply at every step.
Chemical Reactions & Modifications
Lead bromate takes part in classic redox chemistry. Strong exposure to heat or reducing agents breaks the molecule apart, liberating lead(II) oxide and releasing bromine vapors. In aqueous reactions, it’s valued as an oxidant, sometimes used to push rare oxidation states in organic compounds. Further, it can be converted back to other lead compounds if treated with strong acids, giving up bromic acid as a byproduct. All these reactivities show why chemists respect both the technical possibilities and the hazards that come with working with such reactive oxidizers.
Synonyms & Product Names
Across catalogs and older chemical literature, lead bromate turns up under a few alternate names. “Lead(II) bromate” and “plumbous bromate” describe the same thing. Listings sometimes reference its registry numbers, such as CAS 10124-95-5, to avoid confusion with other lead or bromate salts. European suppliers have used language like “bromic acid, lead(2+) salt.” Each name points to the same chemistry, but as regulations clamped down, many former suppliers dropped lead bromate from their selections in favor of safer oxidizers.
Safety & Operational Standards
Lead bromate demands respect and vigilance in any environment. The dual threats—lead’s neurotoxicity and bromate’s established risks as a carcinogen—raise serious red flags for anyone handling or disposing of even small batches. Contact with skin, eyes, or respiratory tracts causes acute symptoms, while chronic exposure leads to cumulative harm, especially in kidneys and nervous tissue. OSHA, NIOSH, and European REACH guidelines force tight containment, proper labeling, and mandatory use of gloves, goggles, and fume hoods. Spill kits and emergency showers belong near any workspace dealing with this compound. Waste streams from experiments using lead bromate get labeled for hazardous pickup; nobody risked rinsing the remains down old laboratory drains since amplified scrutiny on environmental lead levels appeared after the 1970s.
Application Area
Daily life never encounters lead bromate directly. Most applications fit under chemical research, especially as a study reagent in redox experiments. At times, laboratories investigating oxidative processes in organic chemistry pull lead bromate off the shelf for controlled reactions. It serves as an analytical standard for bromate ion detection methods, giving a precise yardstick in calibration of testing instruments. For a while in the last century, small pilot studies looked into its use as an oxidizing pigment component or possible sensor material, but these faded in light of regulatory shifts and changing safety outlooks on lead.
Research & Development
Within academic circles, lead bromate sometimes appears in papers looking at oxidation of aromatic compounds or deep-dives into the mechanistic studies of bromate reactions. It provides a well-characterized, stable bromate source for kinetic test beds, letting researchers track sample changes with analytical precision. Some studies explored using it as a crystallographic probe due to the heavy lead atom, which aids X-ray diffraction data. Despite its serious hazards, the compound lets researchers map out chemical principles that echo outwards into safer, more mainstream industrial oxidants—bridging the gap between full-scale commercial chemistry and laboratory-scale inquiry.
Toxicity Research
Lead’s dangers remain absolutely non-trivial, and the addition of the bromate group only deepens those risks. Decades of toxicological work have documented lead’s impacts on brain growth, blood formation, and kidney function, especially in children and pregnant women. Bromate brings another set of health alarms, including kidney tumors and chronic irritation after even similar compounds in trace drinking water contamination were linked with adverse effects. Animal studies with lead bromate exposures confirm the compounding hazards—neurological symptoms stack with oxidative stress trauma. Modern risk assessments banish such compounds from food, water, and consumer goods, restricting their use to strictly managed settings with comprehensive ventilation and personal protective gear.
Future Prospects
The outlook for lead bromate as an industrial or commercial chemical appears limited, shaped by tough regulations, rising public health scrutiny, and the widespread hunt for greener, less toxic oxidizers. As rules tighten on heavy metals and compounds with proven carcinogenic activity, the chemical industry shifts research funding toward alternatives. That said, lead bromate will likely persist as an object of study for historians of chemistry and a teaching aid for oxidation reaction courses, provided its use stays inside carefully controlled laboratories. Some fringe uses could remain in high-precision analytical science, but mainstream adoption no longer makes sense. Increased attention to replacements and process redesigns keeps lead bromate out of new product pipelines, and this trend looks unbreakable as modern chemical philosophy prioritizes safety, environmental stewardship, and sustainable alternatives.
What is Lead Bromate used for?
Not a Household Name, But Still a Concern
Every so often, a chemical name pops up that most folks haven't thought about since high school science. Lead bromate falls into that category. You won’t find it in your medicine cabinet or in any over-the-counter cleaner. If you have some connection to chemistry, you know it serves as a strong oxidizer. Scientists and some niche industries use it for that property, but to most people, the material might as well be imaginary.
Industries and Experiments
Lead bromate plays a special role in labs. Chemists sometimes rely on it to spot certain ions in water testing or to carry out experiments that need a reliable way to shift oxygen atoms around. Researchers appreciate its predictability. Lead compounds have been part of analytical chemistry for a long time. I remember working with them back in college labs. The strict rules we followed highlighted just how risky the stuff can be.
In industry, lead bromate has sometimes cropped up in photography, pyrotechnics, or as a reagent for making specialty chemicals. The list keeps getting shorter as safety rules grow stricter. Factories learned long ago that safer or less toxic substitutes usually cost less in the long run, especially if the law holds them responsible for cleanup if something leaks. The shift toward greener chemistry pushed many old-school oxidizers to the back of the supply room.
The Elephant in the Room: Toxicity
People rightfully worry about lead in any form. It messes with the brain, stunts kids’ growth, and causes heart trouble. The dangers have been clear for ages. Lead bromate can bring a whole set of problems because bromates add their own risks. They show up on lists of possible carcinogens. Ingesting even small bits, or just breathing the dust, can set off headaches, nausea, and a long list of health issues. The double punch of lead and bromate raises big red flags. Most employers in science or manufacturing take extra steps to keep people safe — gloves, fume hoods, and air filters become non-negotiable for a reason.
Regulation and Real-World Impact
Government agencies in the United States, as well as in Europe and Asia, created tough limits for any workplace dealing with lead bromate. The Environmental Protection Agency (EPA) and the European Chemicals Agency update their rules as new evidence comes out. You won’t find lead bromate in food processing or drinking water without alarms going off. Some countries banned its use outright except for research. The chemical’s presence in consumer products dropped to nearly zero over the past few decades.
What Comes Next?
More scientists and policy makers now focus on phasing out lead-based reagents, especially for everyday or “bench” chemistry in school and industry. The alternatives might not always act the same way, but the lower risk to the environment and workers makes the trade-off worth it. Research centers continue looking for options that can fill the same role without dirtying water or putting workers at risk.
The story of lead bromate mirrors how society deals with old solutions that no longer fit our sense of safety or responsibility. The stuff still shows up on a few shelves where chemistry matters more than convenience, but its time under the spotlight has long passed. The lesson seems clear: pay attention to what’s behind the label, weigh the risks, and always look to do a little better.
What are the safety precautions when handling Lead Bromate?
The Hazards Are Real
Anyone who's spent time in a chemistry lab learns pretty quickly that certain compounds demand a lot of respect. Lead bromate sits high on that list, and for good reason. Its dual threat—combining the toxicity of lead with the oxidizing punch of bromate—reminds me of the early days learning about compounds that don’t forgive mistakes. Lead damages the nervous system, blood, and organs over time. Bromates ramp up cancer risk and can damage vital tissues. No experiment or manufacturing process is worth a shortcut.
Personal Protective Equipment: The Front Line
A sturdy pair of nitrile gloves doesn’t just keep dust off the skin. It keeps lead compounds and bromates from absorbing through pores. I keep safety goggles nearby, and I always put them on before a bottle even comes out of a cabinet. Even a tiny splash or particle in the eye can bring big trouble, so there's no cutting corners. A lab coat forms the next barrier, making sure nothing soaks into my clothes and comes home with me. Respiratory protection sometimes gets skipped. From what I’ve seen, many folks forget that lead bromate dust floats and lingers. Wearing a NIOSH-approved respirator isn’t overkill—it's common sense.
Handling and Storage: Small Habits, Big Difference
I never open lead bromate in a regular room. Fume hoods aren’t just a suggestion. I’ve heard older chemists tell stories about headaches and “labs with a taste of metal in the air”—a sign they handled toxic metals without the right airflow. Spending money on a proper ventilation setup pays itself back with every year free from chronic symptoms. The storage cabinet for lead compounds needs a clear, simple label. Secure doors keep everyone out who doesn’t absolutely need access. Dry, cool conditions cut down on risks of reaction and slow any decomposition. The less I move the container, the lower the chance of a spill. If I need to transfer a powder or solution, double-bagging and careful pouring keep the mess where it belongs—inside the flask.
Chemical Spills and Cleanup
Nobody likes to talk about accidents, but pretending they don’t happen gets people hurt. Spilled lead bromate should never touch rags, brooms, or shop vacs, since that just spreads the hazard. Absorbent pads and wet wiping with the right cleaning agents mean less dust. Contaminated cleaning materials go in sealed bags for hazardous waste pickup. Most places have clear instructions now, and training everyone once a year keeps the memory fresh. I write the spill steps down. Telling co-workers to call for help, not try to hide a mistake, ensures we look out for one another.
Protecting Health: Monitoring and Breaks
Long-term exposure sneaks up, especially with metals like lead. Blood lead level tests take out the guesswork. Good labs even keep a log of who handles which compounds and how often. Simple routines—washing hands after work, no eating or drinking in the prep area—make a real difference. I saw a friend develop minor symptoms after repeated low-level exposures, which changed how all of us managed our breaks and lab routines. It's not about fear, just respect for what science teaches us.
Proper Waste Disposal
Lead bromate doesn’t just disappear down the drain. Special waste collection keeps heavy metals and oxidizers out of water and soil. Complying with environmental rules protects everyone in the community, not just folks on the clock. Double-checking waste labels and sealing up containers adds a few minutes but saves headaches with regulators—and protects those who come after us.
Learning and Teaching
Staying safe becomes a habit when every worker, student, or technician has seen why these steps matter. Reading safety data sheets isn’t busywork; it’s the difference between confidence and guesswork. Real safety comes from a culture built on experience, recorded incidents, and mistakes people are willing to share. I’ve seen labs improve when folks look out for each other instead of treating risks like someone else’s problem. That attitude saves lives, not just experiments.
What is the chemical formula of Lead Bromate?
The Essentials of Lead Bromate
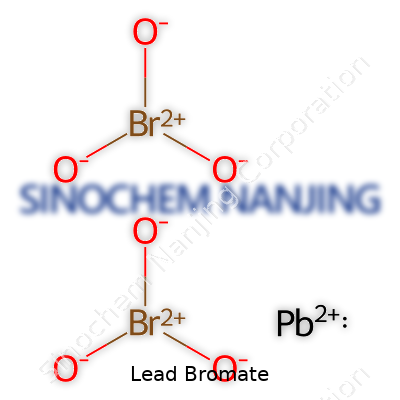
Lead bromate, often found in chemistry classrooms and discussed in toxicology circles, carries a distinct chemical formula: Pb(BrO3)2. Each molecule carries one lead atom and two bromate ions. Its structure illustrates the basic rules of ionic bonding—lead, with a +2 charge, teams up with two bromate ions, each pulling a -1 charge, keeping the compound electrically balanced.
Why Does This Matter?
Growing up around hardware stores and family-owned businesses offering water treatment, I saw firsthand the odd mix of substances coming through the backdoor. Lead stood out, not because it attracted people, but because of the concern it brought. Its reputation in environmental health circles comes for a good reason. The compound, in this case lead bromate, doesn’t pop up at your local pharmacy, but it does crop up in industrial experiments, and research often explores it for its chemical properties.
Understanding what the formula means lets anyone dig deeper into safety implications. Lead compounds in any form can cause health problems. Lead bromate contains both lead and bromate ions, which ought to send a signal to take extra care. People in academic, industrial, and scientific fields need to remember that handling compounds like lead bromate means taking care to avoid inhalation, ingestion, and skin contact.
Safety, Risks, and Real-World Perspective
Scientists know lead causes nerve damage and kidney problems when it gets inside the body. Bromates, like the ones tucked inside this formula, also carry risks. Drinking water treated with bromate-forming chemicals has caused health agencies to limit allowable levels and keep an eye on processing plants. It brings to mind past reports of communities with lead-tainted water—no parent forgets the sense of panic when a neighborhood faces warnings from the public health department.
A simple formula offers a reminder of why chemical knowledge shapes public safety rules. It’s not only an academic exercise; it touches everyday choices, from buying paint to picking plumbing parts. The lead content makes this compound unsuitable for daily household goods, and regulations reflect this reality. Watching local news, I’ve learned to spot the names of compounds that pop up in recall notices. Lead bromate hasn’t made the mainstream headlines, but given how lead-based contaminants still emerge across industries from time to time, it stays relevant.
What Can Be Done?
Training helps people who work with chemicals understand the formulas, risks, and mitigation steps, no matter if the compound feels obscure or mainstream. I volunteered once at a high school science fair and watched students suit up in gloves and goggles. Clarity around labels and formulas can create that protective habit. Enforcing the use of personal protective gear, improving labeling, and offering chemical safety classes stop accidents before they start.
Many chemical supply websites provide safety data sheets, and university labs post warning signs detailing everything from the ingredient lists to spill protocols. These steps reinforce why knowing the formula for compounds like lead bromate matters—it’s never just about memorizing symbols; it’s about making daily life healthier, workspaces safer, and science accessible for everyone.
Is Lead Bromate soluble in water?
A Closer Look at Lead Bromate
Lead bromate, known by its chemical formula Pb(BrO3)2, isn’t something that pops up in most households. Still, it draws attention in chemistry classes and labs. Sometimes, folks wonder about its behavior in water. That’s a good question, especially for people who care about chemical safety or environmental impact.
The Science Behind Solubility
Solubility isn’t just a fancy word in the world of chemicals—it’s about whether a substance mixes well with water. Some salts, like sodium chloride, dissolve easily. Others, especially those containing heavier metals, resist and stay mostly solid. Lead bromate falls into this later category. It barely dissolves in water. Strong ionic bonds and the nature of lead ions play a part here. You’d need to work hard to get a decent amount of this compound to dissolve. Data shows the solubility of lead bromate at room temperature remains extremely low, often measured in fractions of grams per liter. The numbers don't lie: this compound hangs back rather than breaking apart in water, even with a bit of heat.
Why It Matters in Real Life
Water solubility tells much about possible risks. As someone who’s handled chemicals in labs, I see the value of knowing which substances float away in water and which stubbornly sink to the bottom of a beaker. Low solubility limits the spread in water. Yet, even a tiny bit of dissolved lead in water sparks worry. The Centers for Disease Control and Prevention (CDC) keeps a close eye on lead in drinking water. Lead exposure at low concentrations can cause serious health concerns, especially for kids. So despite poor solubility, lead bromate should never be dismissed as harmless.
Environmental and Health Concerns
Lead doesn’t belong in rivers, lakes, or drinking water. Bromates add another layer of trouble. In 2022, studies confirmed bromate ions can form unintendedly during certain water treatment processes. When a compound like lead bromate enters water, lead ions can affect nervous systems and other organs. Environmental agencies worldwide set super-low limits for lead in water—most public water systems in the U.S. face a limit of 15 parts per billion. Even if lead bromate doesn’t dissolve much, trace amounts could accumulate over time, hurting wildlife and leaking into food sources.
Balancing Usage, Disposal, and Safety
Taking steps to handle lead bromate properly stays key. In chemical storage rooms I've managed, lead compounds always go on a separate shelf, away from anything that might bump up risk. Heading off pollution means securing waste properly and following legal disposal rules. Never pour lead compounds down a drain. Labs and manufacturers need strict procedures. Investing in wastewater treatment upgrades, using cheaper alternatives, or favoring greener chemistry—these practices make a real difference. Ordinary people can contribute by recycling electronics properly and speaking out for clean water programs. Every little effort to reduce lead in the environment helps community health in the long run.
Final Thoughts on Solubility and Care
Lead bromate may not dissolve much, but that doesn’t cancel out its risk factors. Treat every bit of it with respect and always lean toward safer handling, both for people and the planet. Knowledge, careful storage, and thoughtful disposal join forces to protect future generations. Everyone who works around chemicals plays a part in this ongoing effort.
How should Lead Bromate be stored?
Understanding What’s on the Shelf
Lead bromate carries a long history of being treated with caution in both laboratories and industrial settings. Every chemist who’s lifted a jar of this stuff knows it isn’t just another dry powder. The compound comes with a double edge: toxicity from lead and reactivity from bromate. Leaving it in the wrong condition isn’t just a careless mistake; it means putting people and the wider environment at risk.
Awareness Before Action
Forget complicated jargon. Lead bromate doesn’t belong near food, water, or places where someone might lean over and get a whiff of dust. My years working alongside researchers in teaching labs showed me people sometimes develop a false sense of security after working with less hazardous reagents. Lead bromate wipes that out. Every seasoned technician remembers hearing about someone who cut corners and paid for it later.
Keeping It Secure
Want to protect yourself and others? Choose a dry, cool space. Heat and sunlight break down bromates and make lead dust easier to spread. Once I found a container stuck next to a radiator — disaster waiting to happen. Shelves made from metal or thick plastic work best. Anything wooden collects particles and holds onto spills, turning a simple task into a future cleanup headache.
Containers must be airtight and clearly labeled. Big black letters. I tell every new assistant: If you can’t read the label from two steps back, fix it. Never rely only on factory labels, especially after repackaging. Use sturdy materials, not thin plastic. Glass with a tight cap works, but never risk cracked containers. No one likes finding powder along the cracks later.
Don’t stash lead bromate with acids or organic solvents. Mixing mistakes can mean toxic fumes or fires. Certain labs use color-coded shelves—red for oxidizers like bromates, blue or white for everything else. Segregation keeps accidents away.
The Human Factor
Training makes all the difference. I learned to never skip orientation. Even seasoned chemists slip up if they’re in a hurry or distracted. Every storage cabinet should show clear rules for access and usage. Someone must track inventory, and it can’t sit forgotten for years. Regular checks catch leaks or broken seals before the stuff becomes everyone’s problem.
Think long-term. Store only the amount you plan to use within a reasonable timeframe. Old stock piles up risk and nobody remembers when it arrived. If it’s not needed, arrange for proper waste removal. I’ve seen more than a few back rooms packed with chemicals from past researchers—each bottle a mystery.
Protecting People and Place
Lead enters the bloodstream easily, especially as dust. Bromates damage lungs. This isn’t a risk worth taking. Always wash hands after handling, wear gloves, and use face protection if there’s a chance of a spill. Work in a ventilated space. Accidents don’t just hurt the careless—they threaten everyone working nearby.
Safe storage starts with respect. It’s not enough to have rules on paper. People remember stories, and the best labs talk openly about what can go wrong and how to stop it before it begins. If we keep learning from each other’s mistakes, more people make it home safe each night.
| Names | |
| Preferred IUPAC name | lead(II) bromate |
| Other names |
Plumbous bromate Lead(II) bromate |
| Pronunciation | /ˈliːd ˈbroʊ.meɪt/ |
| Identifiers | |
| CAS Number | 10031-23-9 |
| 3D model (JSmol) | `'JSmol: [Pb2+].[O-]Br(O-)=O'` |
| Beilstein Reference | 1721125 |
| ChEBI | CHEBI:86458 |
| ChEMBL | CHEMBL4580638 |
| ChemSpider | 22040973 |
| DrugBank | DB14555 |
| ECHA InfoCard | 100.236.623 |
| EC Number | 236-483-9 |
| Gmelin Reference | Gm. 21113 |
| KEGG | C18606 |
| MeSH | D007864 |
| PubChem CID | 24636 |
| RTECS number | EO9625000 |
| UNII | Z8K4C7G286 |
| UN number | UN1479 |
| Properties | |
| Chemical formula | Pb(BrO3)2 |
| Molar mass | 437.01 g/mol |
| Appearance | White powder |
| Odor | Odorless |
| Density | 6.662 g/cm³ |
| Solubility in water | slightly soluble |
| log P | -1.93 |
| Vapor pressure | Negligible |
| Magnetic susceptibility (χ) | −78.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.853 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 430.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -835 kJ mol⁻¹ |
| Pharmacology | |
| ATC code | V03AB56 |
| Hazards | |
| Main hazards | Toxic if swallowed, causes severe skin burns and eye damage, may cause genetic defects, may cause cancer, harmful to aquatic life with long lasting effects. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS05,GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H302 + H332: Harmful if swallowed or if inhaled. H360Df: May damage the unborn child. Suspected of damaging fertility. H373: May cause damage to organs through prolonged or repeated exposure. H410: Very toxic to aquatic life with long lasting effects. |
| Precautionary statements | P260, P261, P264, P270, P271, P272, P273, P280, P284, P301+P310, P302+P352, P304+P340, P305+P351+P338, P308+P313, P314, P320, P330, P361+P364, P391, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2 0 3 OX |
| Lethal dose or concentration | LD₅₀ (oral, rat): 340 mg/kg |
| LD50 (median dose) | LD50 (median dose) of Lead Bromate: 175 mg/kg (oral, rat) |
| NIOSH | WI2600000 |
| PEL (Permissible) | 0.05 mg/m3 |
| REL (Recommended) | 10 mg/m3 |
| IDLH (Immediate danger) | IDLH: 1 mg/m³ |
| Related compounds | |
| Related compounds |
Lead(II) nitrate Lead(II) chloride Lead(II) bromide Sodium bromate Potassium bromate |

