Lead Arsenite: A Deep Dive into Its Past, Present, and Unfolding Future
Historical Development
In the nineteenth century, lead arsenite began its run as a pigment, especially prized for its vibrant green color. Back then, industrial painters and artists alike turned to it for applications that stretched from grand public murals to everyday wallpaper. Early manufacturing rarely asked questions about long-term health, so safety standards lagged behind. During the Victorian era, households with ornate, decorative interiors sometimes unwittingly brought significant health risks right into their living rooms. Much of the hindsight came too late, as reports of poisoning and mysterious weakening of people living in colorful homes started piling up. This pigment's history shows how industries grew comfortable with short-term benefits, even when the drawbacks loomed just beneath the surface. Industrial revolutions always seem to teach the same lesson—progress without respect for health exacts a steep price. I remember reading old paint catalogs as a student, struck by how common these toxic colors appeared in even the most ordinary catalogs.
Product Overview
Today, lead arsenite barely holds market share, having been crowded out by safer alternatives after modern regulations caught up with its hazards. At its core, the product contains lead and arsenic bound into a crystalline powder, often found with a green tint. For a long stretch, it helped manufacturers produce goods in construction, paints, fireworks, and even alloys. Modern chemical factories rarely encounter it directly anymore outside of specialized legacy processes, and its legacy has faded, but echoes remain in old sites and collections.
Physical & Chemical Properties
Lead arsenite shows up as a fine powder, usually white to pale green. Its molecular structure offers little in terms of solubility, refusing to break down easily in water but giving in somewhat to acids. That feature caused years of trouble in soils and water near old factories. The powder weighs heavily due to the lead content, feeling far denser than most pigment powders. It smells somewhat earthy, but having handled pigment jars myself in conservation labs, there's always a faint, unnerving metallic note. Stable at room temperature, it degrades under strong heat, emitting fumes that can become hazardous. The chemical formula PbHAsO3 maps out the balance between lead and arsenic, tying toxicity to the very features that once made it desirable.
Technical Specifications & Labeling
Regulations have tightened labeling across the world. Packages must declare both lead and arsenic clearly, and hazard pictograms cover every container. Even in research settings, batches must carry trackable batch numbers, manufacturer addresses, risk statements, and required GHS safety codes. Typical technical sheets outline particle size, lead and arsenic mass fractions (often above 60% each by dry weight), purity standards, and instructions for immediate action in case of spills or exposure. Any product with even trace content faces restrictions, prompting manufacturers to keep levels of both contaminants lower than ever before, out of pragmatic self-interest. The cost of ignoring these rules adds up fast, both legally and in bad press.
Preparation Method
The classic method mixes lead nitrate with sodium arsenite in solution, causing an immediate, visible precipitation of lead arsenite. Years ago, I watched chemists run this reaction for demonstration—thick, chalky clouds swirling in a beaker, a stark visual warning of danger. Filtering and drying the precipitate yielded the powder. In industry, large vats with mechanized stirring replaced laboratory glassware, but the heart of the process stayed the same. The operation never thrived without proper exhaust or containment, as impurities or loose dust could quickly compromise environments and leave residues. No surprise, most plants converted to alternative pigments as soon as viable options showed up.
Chemical Reactions & Modifications
Lead arsenite stands up well under neutral conditions but succumbs to acids, breaking apart and releasing both lead and arsenic ions—both of which pose health risks. Strong bases can also attack the crystal lattice. The pigment resists oxidation under normal conditions but burns with an acrid smoke when exposed to extreme heat; this became a real risk in fire-prone warehouses or old structures. While chemists occasionally explored adding stabilizers or changing particle size to reduce hazards, the workarounds never really fixed the core problems; they only changed the look or feel, rarely the underlying health risk. Even so, reactions with certain organic molecules provided clues for remediation, an area still under study today.
Synonyms & Product Names
Lead arsenite goes by several names in older literature: “Schweinfurth Green,” “Vienna Green,” even “Emerald Green” when blended with other copper compounds. Manufacturers gave it dozens of local names, especially during periods of high demand in European markets. Each name pointed to a slightly different blend but shared the key pair of lead and arsenic at their heart. Some commerce records muddled the distinctions, passing off synthetic mixtures as “true green” to cut costs or boost demand. Whether called by a technical chemical name or a marketing term, the dangers stuck around regardless.
Safety & Operational Standards
Modern labs treat any exposure to lead arsenite as a top-tier hazard. Air monitoring stays in place wherever the powder could appear, and workers must wear full protective equipment, including respirators and sealed suits. Waste streams from any site using it fall under hazardous regulation and must travel to approved disposal zones. Training covers everything from accidental ingestion risks to clean-up drills. OSHA and its global counterparts set occupational exposure limits far below what earlier generations ever imagined. I saw firsthand the look of old-timers, who remembered no gloves or masks, as they attended annual health checks now required by law. It’s a stark display of how risk perception grows sharper once science weighs in.
Application Area
Lead arsenite once filled a surprisingly wide range of uses: decorative paint, dye for textiles and wallpaper, and a pigment in printing inks. In pyrotechnics, its oxidizing power made certain flares and colored smoke devices possible. Some legacy batteries and glass ceramics tinkered with it for specialized properties, but even minor roles faded in the wake of widespread toxicity awareness. Occasionally, conservation work in museums still encounters it hiding under layers in historical artifacts, forcing curators to take special measures during restoration. Residual contamination appears in older buildings, driving remediation projects to dig beneath coats of paint and patch up contaminated sites.
Research & Development
Academic groups keep poking at lead arsenite, less out of product interest and more to answer questions about remediation, analysis, and long-term environmental fate. Teams run spectroscopic studies to model how the compound moves in soil or leaches into water under different pH values. New detectors, including portable XRF (X-ray fluorescence) tools, now help field teams scan objects and soils for traces of legacy pigment. Some research aims to find microbes or plants able to reduce bioavailability or sequester arsenic and lead safely—early work with certain ferns and bacteria shows promise, though scaling up these discoveries poses challenges. Most innovation has shifted to safer pigments and remediation tools, but scientific curiosity keeps the subject on university syllabi.
Toxicity Research
The danger of lead arsenite rests squarely with its constituent elements. Both lead and arsenic damage organs, disrupt brain development in children, and increase cancer risk. Animal studies confirm that even low, chronic exposures cause cumulative harm. After a round of exposure, lead accumulates in bones, slowly releasing over time and interfering with the central nervous system; arsenic often attacks cell replication and immune function. Epidemiological work in communities near factories historically using the compound traced lines from pigment dust to clusters of illness. Decades of shared stories back up the dry numbers. Today, advances in medical testing allow earlier detection, but a clean bill of health after exposure stays rare. Personal protective equipment and rigorous monitoring can keep current scientific staff safe, but clean-up workers in old industrial zones still face real danger.
Future Prospects
As safer pigments take center stage, lead arsenite’s days in new products look numbered. The push now centers on site clean-up, environmental monitoring, and historical analysis. Regulatory authorities continue to tighten restrictions and expand bans, while green chemistry research urges industry to learn from the past—prioritize safety at the earliest stages of material design. Scientists turning to bioremediation and advanced waste handling could help mend the damage in old sites. Young researchers revisit the pigment in museums or archives, more as a cautionary tale than a resource. Even with curiosity piqued by its striking color, respecting its toxic past builds stronger products and healthier workspaces going forward.
What is Lead Arsenite used for?
Lead Arsenite: A Glance at Its Role
Lead arsenite looks like just another chemical from the long list of industrial compounds, but it has carried a hefty load in history. For most people, hearing “lead arsenite” doesn’t stir any emotions. Say “poison,” and eyes widen. There’s a good reason for that. Used over a century ago, lead arsenite played a big role in the world of pigments, especially for brightening up paint. The compound became famous for its ability to provide a deep, vivid yellow—"Scheele’s Green" and “Paris Green” had cousins in lead arsenite pigments. It turned up in everything from children’s toys to wallpaper. Funny how harmless a dash of color on a toy can seem until science peels back the curtain.
What Made It Popular?
The pigment packed a punch. Artists, manufacturers, and even homeowners flocked to it for bold yellow shades that wouldn’t fade in the sun. Paints stuck with it for its durability and brilliance. Gardeners and orchard keepers mixed it into insecticides, where the combo of lead and arsenic took out bugs, fungus, and mold with gusto. As a young science enthusiast, I used to wander through antique stores with my father, marveling at old toys and painted boxes. Many of those colors came from toxic compounds like this one. Back then, nobody thought much about what lingered in the dust or where the runoff went.
What Are the Dangers?
The very qualities that made lead arsenite useful also made it dangerous. Breathing in or swallowing even small amounts does heavy damage. Lead goes after the brain and nervous system, especially in children. Arsenic attacks kidneys, skin, and sometimes turns up later as cancer. It travels fast, finds its way into dirt and water, and is tough to clean up. In the old houses I walked through, chipped paint and dust often meant silent risk to anyone inside.
Why We Moved On
It took too many sick workers and poisoned children for society to realize what they had invited home. The science got better and the stories turned tragic enough to change laws. Today, you won’t find lead arsenite in common paints or pesticides. Countries ban it—evidence overrules tradition. Better and safer choices stepped in: synthetic organic pigments color our walls and modern insecticides target pests without long-term fallout.
Better Approaches For Today
Banning a toxin is just half the solution. Many old homes, barns, and garden sheds still hide these threats in their walls, floors, and soil. I once joined a local group cleaning abandoned playgrounds, scraping up dust and safely removing paint chips, because no parent wants to gamble with a child’s health. Testing kits tell the truth, and certified cleanup crews do the hard work. Governments push for more research into safe alternatives and protocols for dealing with contamination.
Holding Companies Accountable
Looking at how companies sold lead arsenite without proper warning, it’s clear regulations matter. Today, product safety laws and honest labeling stand in place, but oversight needs to stay sharp, especially with imported goods. If something doesn’t meet safety standards, it shouldn't make it to store shelves—plain and simple.
The Big Picture
Every chemical tells a story, and lead arsenite’s tale is a warning. Chasing performance and profit without regard for health can haunt generations. Trust and safety come from learning, remembering, and doing better the next time. Nobody needs a history lesson in poison—just a push to make smarter choices moving forward.
Is Lead Arsenite hazardous to health?
Unpacking Lead Arsenite and Its Uses
Most folks have never heard of lead arsenite, but they’ve probably stumbled across something colored with it. Painters in the past loved its bright yellow and green pigments, letting walls, canvases, and toys glow with unnatural color. Tasks that called for stubborn insect control also relied on it, since the stuff worked well as a pesticide. Underneath those pretty hues and protective finishes sits a problem—lead arsenite packs a serious health punch.
Health Risks Come from More Than a Warning Label
Trouble with lead arsenite starts at the atomic level. Both lead and arsenic belong on lists nobody wants in their food, water, or air. Slicing through the noise, this compound isn’t just “potentially harmful”—it’s downright toxic. Old lead-based paints might look harmless on a sunny windowsill, but breathing in their dust or getting flakes near your mouth lets the poison enter your system. Children pay the steepest price. Their growing bodies suck up lead faster, causing learning problems, behavioral issues, lower IQ, and long-term neurological damage. Arsenic adds its own dangerous twist, increasing risks for skin lesions, heart disease, lung and bladder cancer, and developmental setbacks.
Crunching the numbers, the Centers for Disease Control and Prevention say there’s no “safe” level of lead for kids. Even relentless hand washing can’t keep dust from getting onto mouths and toys. The World Health Organization confirms arsenic lands among the most hazardous substances known to man. Prolonged exposure sets people up for decades of trouble, long after the fresh paint smell disappears.
Old Buildings, New Dangers: Where Lead Arsenite Hides
Communities find danger in unexpected places. Many homes built before the late 1970s hold paint that quietly chips away. Workers scraping old barn doors, sanding window sills, or tearing down playgrounds risk spreading tiny bits of lead arsenite everywhere. Water systems catch a share of the fallout if flakes or dust settle in soil and make their way to local taps. Cleanup gets tricky, expensive, and sometimes political. Low-income neighborhoods catch the worst ends of these risks since repairs lag and information doesn’t always reach every household.
Growing up in a city where peeling paint marked every corner, I remember warnings about which steps to avoid, where not to dig in the yard, and the importance of shutting windows during summer renovations. Friends whose families couldn’t afford prompt repairs wound up battling headaches, fatigue, and learning concerns teachers sometimes dismissed as laziness. Years later, doctors traced those issues back to nothing fancier than an old layer of lead arsenite-rich paint.
No Silver Bullet—But Plenty Can Be Done
Wide-scale solutions take grit, not just good intentions. Regular home inspections help spot risks before they land kids or workers in the hospital. Education, especially in high-exposure areas, lifts the curtain on invisible hazards. Banning the use of toxic pigments sets a clear line, but millions still deal with lead arsenite left behind. States can step up by funding safer housing programs and offering assistance for thorough paint removal. Simple habits—wet-cleaning dusty spots, filtering tap water, teaching kids to avoid chipping paint—buy families extra protection where bigger fixes haven’t arrived yet.
Doctors and nurses staying alert to subtle symptoms play a key part. With early testing, families learn their risk and take steps to knock exposure levels down. It’s tough to see, harder to erase entirely, but not impossible. Armed with education, practical changes, and firm support, communities stand a fighting chance against the threats hidden in something as ordinary as paint.
What are the precautions for handling Lead Arsenite?
Know What You’re Dealing With
Lead arsenite carries a toxic punch, combining two heavyweights—lead and arsenic. Old paint factories, pigment shops, and some agricultural setups may still come across traces of this compound. Anyone who’s spent time in older industrial buildings or worked near contaminated soils knows the worry grows real fast if you don’t respect the hazards.
Prepare Before You Even Open the Jar
Good preparation means less risk. If you’ve worked around chemicals longer than a few months, you’ve already heard supervisors stress having the right gear. Don’t just toss on generic gloves and call it a day. For lead arsenite, a full set of personal protective equipment—nitrile gloves, goggles, and a fitted respirator—turns a risky project into a manageable chore.
Check your workspace. A cluttered or cramped setup makes accidents and spills far more likely. I always clear off benches, keep only the tools I need close by, and line up a sealed disposal bin nearby. This habit, picked up from a meticulous lab manager, has saved a lot of headaches when dealing with anything toxic.
Work Methods Matter
Avoid producing dust at all costs. Don’t scrape, grind, or shake containers. When mixing or pouring, move slowly and pay attention to drafts or sudden gusts from open windows or AC units. Even tiny airborne particles carry enough poison to send someone to the doctor or worse. Wet processes and proper fume hoods make a huge difference.
Having practiced clean handling over the years, I’ve learned that small steps—washing hands before touching anything, never bringing food into the work zone, and always labeling everything clearly—do more than check boxes on a safety form. They actually save lives. Workers exposed to lead arsenite face higher risks of nerve damage, kidney failure, and cancer. Families suffer too when someone’s exposed and brings dust home on their clothes.
Disposal: Don’t Cut Corners
Throwing the remnants in a regular trash bin would seem quick, but it only spreads toxins down the line. Every region sets strict rules—labeled, sealed containers and certified pickup or drop-off. I’ve personally seen confiscated shipments after folks skipped proper procedures. Lead arsenite won’t degrade in a landfill; it leaches into groundwater, harming your neighbors as well as local wildlife.
Plan for Trouble
Spills do happen, even for veterans. A good spill kit, including absorbent pads and neutralizing agents, ought to sit close by. Colleagues once cleaned a minor spill without one, and the stress and cleanup costs soared. If clothing gets contaminated, strip and rinse off immediately. Every minute of hesitation makes the cleanup harder. Showers on-site and routine medical checkups for workers who handle branded toxins remain critical parts of the job.
Education Goes Further Than Warnings
Talking honestly about risks beats waving around pamphlets. Newcomers often underestimate how invisible some dangers become, especially with chemicals like lead arsenite. I’ve seen coworkers change their habits after hearing a real story rather than reading a warning sign. Whether holding regular training, inviting experts, or just sharing a personal close call, education cuts through complacency.
Better Substitutes Make Work Safer
Switching out lead arsenite for safer pigments or pesticides isn’t always easy or cheap, but it pays long-term. More companies now fund research into alternatives. As someone who’s seen friends develop health problems after years of exposure, I’d take any slower job or pricier option over a lifelong illness.
Staying ahead means caring enough to protect yourself and everyone around you. Respect the hazards, keep your procedures sharp, and push for safer materials where you can. That approach keeps work safe for today, and for everyone who comes next.
What is the chemical formula of Lead Arsenite?
The Basics: What Does Lead Arsenite Look Like on Paper?
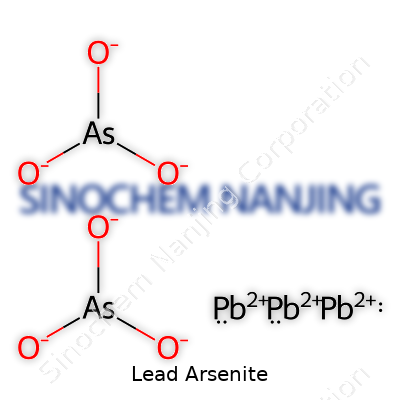
Anyone who has spent time in a chemistry lab or even flipped through an old textbook might spot compounds like lead arsenite. Its chemical formula reads Pb(AsO2)2. That formula reveals a story about the atoms tucked inside: one lead atom joined up with two arsenite groups. Arsenite means you’ve got arsenic and oxygen together, but not in the same way as in arsenate. For lead arsenite, its structure brings up unique challenges and risks.
Why Should This Compound Matter to You?
Flip back a few decades and lead arsenite found a place in household paints and pigments. Bright, durable, and dependable—on the surface at least. My grandfather’s old tool shed, painted bright green, probably had some of this stuff layered into the wood. He painted rooms without knowing the long-term health risks piled up in those glossy colors. Researchers later found both lead and arsenic carry heavy baggage: nervous system damage, developmental hazards for kids, and higher risk of chronic illness for adults. That's not just lab talk. The World Health Organization and the EPA both flag lead as a top-tier health threat, and arsenic traces in groundwater have driven public health campaigns across the globe.
Digging Deeper: The Chemistry Behind the Headlines
Pb(AsO2)2 isn’t some rare, barely-used compound. Its use in pigments stood out because it blended vivid colors with a chemical backbone tough enough to withstand sunlight and weather. Factories pumped out gallons of lead arsenite paints through the 19th and 20th centuries. As science caught up, so did the list of problems. Families living in homes coated with these products ended up with children testing positive for lead poisoning. My own childhood memories include city crews peeling old paint from window frames because of the threat to neighborhood kids.
Real-Life Impact: Health, Environment, and the Long Arm of Regulation
People didn’t always have the facts at their fingertips. Once the dangers became clear, regulations moved in. Paint standards tightened, and consumer groups pushed for full disclosure about product contents. Environmental clean-up crews started testing for lead in soil near old buildings and playgrounds, a lesson learned after seeing how quickly dust or chips could travel through a household. No parent wants to discover a child has been exposed to heavy metals just from playing in the yard.
Lead arsenite contamination lingers. Soil and water don’t heal instantly after exposure. Cleanup teams sometimes spend months restoring contaminated lots, using data from environmental toxicologists to guide their work. These steps cost money, but prevention saves much more down the line. According to the CDC, even small lead exposures can drop IQ scores and boost risks for attention problems. That’s a price too high for any community to pay.
A Safer Future: Practical Solutions For Homes and Communities
We can’t rewrite the past, but safer choices keep families out of harm’s way. Getting your home checked for lead-based paint protects children—especially in houses built before lead bans took effect. Dust wipes and approved sealants cut down on exposure if paint can’t be replaced right away. For folks living near older industrial sites, regular testing and prompt communication help build trust in local services. Schools and landlords taking these steps show care for the people who depend on their buildings.
Smarter Chemistry, Healthier Lives
It’s not just about memorizing chemical formulas. Pb(AsO2)2 tells a story about progress in science, regulation, and public action. Families, teachers, and policymakers all have a part to play in keeping dangerous legacy chemicals out of daily life. That’s one lesson I hope gets passed down along with those old paint cans—know what’s in your environment, and act early to protect what matters most.
How should Lead Arsenite be stored and disposed of?
Understanding the Hazards
Lead arsenite carries a long shadow of risk for human health and the environment. Breathing dust or touching contaminated items brings poisoning closer to home. Authorities like the EPA and CDC have warned for years about effects that range from nerve damage to cancer. There’s more at stake here than workplace safety files; a mishap sends shockwaves through families and communities. From the start, clear procedures matter more than haste or convenience.
Safe Storage: Cutting Corners Brings Trouble
My years working near labs have left memories of well-meaning shortcuts that, at best, triggered a messy clean-up. Locking lead arsenite in a labeled, airtight container seems simple, but careless seals or worn labels create confusion and risk. Metal drums or tough HDPE containers don’t just protect from leaks; they shield workers and keep dust from escaping.
Cool, dry, ventilated spaces offer the extra layer of defense. These substances react badly with moisture—sloppy shelving or pipe drips can mean a dangerous mess. Shelves need a lip so nothing slides off. Safety officers should always have eyes on expiration dates, because old, degraded containers bring fresh hazards. It’s never a one-and-done process; checking storage regularly turns up problems before disaster strikes.
PPE: More Than a Piece of Paper
Gloves, goggles, and sturdy overalls aren’t just a formality. I’ve seen burns and rashes from people who tried to “just handle it real quick.” Respirators rated for toxic dusts keep invisible harm out of your lungs. So does using a fume hood, even for simple repackaging chores. Changing out of work clothes and washing hands right away slashes the risk to kids and pets at home.
Disposal: No Room for DIY Solutions
Tossing lead arsenite in the regular trash or pouring it down the drain never solves the problem. Waste facilities can’t stop toxins from slipping into soil and groundwater. In my small town, a landfill spill took years to fix; wells stayed closed the entire summer.
Hazardous waste companies have trained teams for this kind of job. They seal up the material, tag every drum, move it using routes away from neighborhoods, and track everything. Documentation follows every step. The law often demands a paper trail from start to finish, partly because mishaps from sloppy handling wind up on the news. Emergency response plans can’t save the day if nobody knows what’s in the back corner of the shed.
The Human Part
No one wants health scares, EPA fines, or lawsuits. Storing and billing for proper disposal costs more up front, but the headache of clean-up after a spill turns that price tag into chump change. Honest conversations with everyone involved—janitors, transport workers, managers—help stop mistakes before they spread. If something feels off, speaking up beats hiding the problem until it bites.
Lead arsenite reminds us that diligence isn’t overkill; it’s just good sense. Approaching the task with care, not complacency, keeps families safe and water drinkable. The difference between disaster and normal life often comes down to a label, a checklist, or a quick question at the right time.
| Names | |
| Preferred IUPAC name | Trilead diarsenite |
| Other names |
Arsenous acid, lead(2+) salt C.I. 77585 Lead(II) arsenite |
| Pronunciation | /ˈliːd ˈɑː.səˌnaɪt/ |
| Identifiers | |
| CAS Number | 12006-76-3 |
| Beilstein Reference | 35361 |
| ChEBI | CHEBI:86344 |
| ChEMBL | CHEMBL510181 |
| ChemSpider | 25416 |
| DrugBank | DB11577 |
| ECHA InfoCard | 100.006.819 |
| EC Number | 206-104-4 |
| Gmelin Reference | 72350 |
| KEGG | C18946 |
| MeSH | D007857 |
| PubChem CID | 166829 |
| RTECS number | OS9275000 |
| UNII | N747C26R24 |
| UN number | UN1617 |
| Properties | |
| Chemical formula | PbHAsO3 |
| Molar mass | Molar mass of Lead Arsenite: 533.01 g/mol |
| Appearance | Pale yellow powder |
| Odor | Odorless |
| Density | 6.86 g/cm³ |
| Solubility in water | Insoluble |
| log P | -0.33 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 6.5 |
| Basicity (pKb) | 11.84 |
| Magnetic susceptibility (χ) | Diamagnetic |
| Refractive index (nD) | 2.0 |
| Viscosity | Viscous liquid |
| Dipole moment | 0 Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 207.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -668.1 kJ/mol |
| Pharmacology | |
| ATC code | V03BA04 |
| Hazards | |
| Main hazards | Toxic by inhalation, ingestion, and skin contact; carcinogenic; may cause genetic defects; environmental hazard. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08,GHS09 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. H350: May cause cancer. H360: May damage fertility or the unborn child. H400: Very toxic to aquatic life. |
| Precautionary statements | P201, P202, P260, P264, P270, P272, P273, P280, P301+P310, P302+P352, P308+P313, P314, P321, P330, P361+P364, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-3-⨁ |
| Lethal dose or concentration | LD50 oral rat 20 mg/kg |
| LD50 (median dose) | LD50 (median dose): 20 mg/kg (oral, rat) |
| NIOSH | NAK6225000 |
| PEL (Permissible) | 0.01 mg/m³ |
| REL (Recommended) | 0.01 mg As/m3 |
| IDLH (Immediate danger) | IDLH: 15 mg/m3 |
| Related compounds | |
| Related compounds |
Lead(II) arsenate Lead(II) acetate Lead(II) oxide Arsenic trioxide Lead antimonate |

