Lead Arsenate: A Closer Look Through Science, History, and Human Health
Historical Development
Lead arsenate sits in a unique place in agricultural history, stepping onto the scene in the late 19th century just as apple growers were desperate to fight the relentless codling moth. Chemists designed lead arsenate as a compound able to deliver the lethality of arsenic with some control, aiming to avoid killing the tree along with the insect. By the early 1900s, entire orchards in North America saw regular dustings of this compound. Major horticultural bulletins from universities and government agencies openly promoted the use of lead arsenate, touting its higher effectiveness and lower “burn” compared to Paris green or straight arsenic trioxide. Factories scaled up production to meet demand as the farming sector raced to keep yields high. This lead-heavy pesticide stood front and center in rural life until public health sparked a rethink after decades of buildup in soil, crops, and even the food chain.
Product Overview
Lead arsenate splits into two basic forms: acid lead arsenate and basic lead arsenate, each suited to different crop conditions. Both forms combine lead, a soft dense metal, with arsenic, a notorious element known for its danger. In commercial settings, the product appeared as a white or greyish powder or as a wettable paste. Suppliers packaged it in drums or sacks, labeled for agricultural use and sometimes for lumber treatment and anti-fouling paints. Local suppliers grew familiar with the dangers of handling it, as word spread about those who got careless. Even children knew to give lead arsenate containers a wide berth in farm sheds.
Physical & Chemical Properties
Acid lead arsenate shows up as a heavy, white crystalline powder, and it doesn’t dissolve well in water, so it hangs on leaves after rain. Chemical formulas set acid lead arsenate as PbHAsO4, while basic lead arsenate carries more lead ions, often written as Pb4(AsO4)2·Pb(OH)2. Melting points exceed 600°C, and the product resists breakdown by sunlight or typical weather. Mixed in water with lime, it forms a fine suspension, clinging better to trees than older dusts. The lead content hits up to 63% by weight in some mixtures, meaning even a spoonful goes much farther than softer baits. The chemical stability that made it effective as a pesticide also drove its risks, building up in soils for generations.
Technical Specifications & Labeling
Manufacturers supplied detailed product labels, listing active ingredients and providing minimum lead and arsenic percentages. The packaging included hazard pictograms well before modern GHS labeling, and instructions strictly warned not to inhale or swallow the dust or let it contact skin. Labels cautioned users to wear gloves, goggles, and masks—a tall order before proper safety gear reached rural towns. The technical documentation that shipped with commercial drums described recommended dilutions, suggested sprayer calibrations, and intervals to harvest, but historical records show many farmers ignored those instructions whether out of haste, ignorance, or habit. Official recommendations specified safe application rates and mixing instructions to limit crop and worker exposure, but the line between enough and too much often blurred.
Preparation Method
Large-scale producers synthesized lead arsenate by slowly reacting solutions of lead acetate or lead nitrate with sodium arsenate, precipitating a solid sublimate of the product. The process required precise control of temperature and pH to prevent runaway reactions. Backyard recipes circulated as well—mixing lead oxide (red lead) with arsenic acid over heat, stirring until the mixture thickened before washing and drying the powder. Factories installed scrubbers and waste ponds, since even minor leaks or mishandling left workers exposed to fumes and dust. The final product got sifted and milled before packing, since clumping reduced the evenness of field spray applications.
Chemical Reactions & Modifications
Lead arsenate barely reacts with most household chemicals, but it breaks down under extremely acidic or alkaline conditions, liberating arsenic as soluble ions and forming lead salts that settle into sediments. This behavior led to slow but steady release of toxic species wherever the compound soaked into orchard soil. Research labs tested additives like calcium hydroxide or ferric chloride to tie up the lead, minimizing uptake by roots or water. Attempts to reduce runoff included blending the pesticide with oils or using stickier wetting agents, though the base product chemistry changed little until phased out. Heavy use also sparked research into soil-plant transfer and environmental persistence, as groundwater monitoring picked up traces decades after application.
Synonyms & Product Names
Chemists listed lead arsenate under names like lead hydrogen arsenate, acid lead arsenate, and the more chemical-sounding dihydrogen arsenate of lead. Commercial trade names included “Gypsy Moth Arsenate,” “Orchard Lead Dust,” and “Arseno Plomb,” depending on the manufacturer and local regulations. Early 20th-century advertisements painted the product as a miracle for orchardists, sometimes with the simple branding “Guaranteed Codling Moth Killer” in bold script. These names reflected both the compound’s direct application and the plain talk of the farming community, who cared more about yield than about Latin root words.
Safety & Operational Standards
Occupational safety around lead arsenate lagged until mounting cases of chronic poisoning in orchardists brought change. Regular users described symptoms like headaches, stomach pain, and numb legs, decades before the science caught up. Over time, legislative bodies in the United States and Europe tightened rules: mandatory gloves, respiratory masks, and no eating or smoking while handling the substances. Companies rolled out education campaigns after industrial accidents, urging everyone from managers to field hands to shower, wash work clothes separately, and record daily exposure in logbooks. Standard operating procedures called for secure storage, locked cabinets, and rigid workplace hygiene—a far cry from the sacks stored beside livestock feed at the turn of the 20th century.
Application Area
Lead arsenate found its widest use in orchards, especially for apples, pears, cherries, and even blueberries where surface pests threatened entire crops. Forestry departments deployed it in aerial spraying runs against gypsy moths and tent caterpillars, leaving visible residue in forest canopies and ponds below. Some city governments even dusted public parks, and railroads mixed the powder into timber to ward off wood-eating insects. Commercial reach extended into paints for ship hulls and irrigation pipes, thanks to its resistance to microbial action. By the 1950s, widespread contamination forced public health agencies to re-examine sites: kids’ playgrounds, baseball fields, and yard gardens all turned up high lead and arsenic levels where no one expected them.
Research & Development
Early research treated lead arsenate as a necessary evil, documenting effective doses and crop tolerances in seasonal extension handbooks. By the mid-20th century, labs tracked how the compound bound to various soils, registering how long it sat near roots and how much leached to wells. Over time, epidemiologists picked up troubling patterns in communities with heavy spray histories, driving new rounds of government-funded research. Testing focused on chelation—the process of binding and removing heavy metals from contaminated soils—and on identifying replacement compounds with lower toxicity. Regulatory bodies pushed for field trials using synthetic organic pesticides, but nothing matched the cost-effectiveness of lead arsenate until DDT entered the market after World War II. By the 1970s, most research aimed at remediation: can plants extract the arsenic, or can bacteria break it down in washed soils? Growing public demands for organic agriculture have sparked renewed interest in soil legacy and cleanup technology.
Toxicity Research
Lead arsenate ranks high on the list of environmental toxicants, with dual burdens from both the lead and arsenic it contains. Acute exposure cases often started with nausea, vomiting, and cramps, and deaths occurred at lower doses than many pesticides. Chronic exposure presented more subtle dangers: learning disabilities in children, anemia, immune suppression, and neurological deterioration observed among farm families decades after the spraying stopped. Soil tests from long-standing orchards still show high residuals. Research linked lead arsenate to waterway poisoning, as arsenic runoff killed aquatic invertebrates and tainted wells. Both the European Union and the United States now list the compound as a banned substance, recognizing risks too great for continued use. Cleanup remains costly, and the real lesson comes from years of slow, silent exposure.
Future Prospects
Nobody puts lead arsenate front-and-center in pest control plans anymore, yet its legacy remains in thousands of acres of contaminated land. Future prospects depend on a commitment to environmental remediation and the search for gentler chemistry that won’t haunt future generations. Current research follows two paths: cleaning up the orphaned soils, and finding markets for safe, effective pesticides that break down quickly. Cities and farmers now collaborate with bioremediation firms and government labs, using plants and microbes to extract or degrade the lingering arsenate and lead. Regulatory frameworks continue to evolve, spurred by lessons learned from the past, aiming for a future where public health, crop protection, and environmental safety hold equal weight. Until every last orchard has been decontaminated, the story of lead arsenate keeps pressing on, reminding us how easily powerful solutions can slip beyond their intended boundaries.
What is lead arsenate and what is it used for?
What Lead Arsenate Is
Lead arsenate sits on the shelf of history as one of the most prominent pesticides from the early 1900s. Chemists combined lead and arsenic to make a compound lethal to insects, mainly targeting the codling moth in apple orchards. Farmers all over the United States dusted or sprayed it onto their crops, hoping to save the harvest. This mix promises effectiveness against pests, but anybody who spent time in an orchard decades ago will remember the chalky residue it left behind. As a kid, walking through those groves, I never realized how much harm these practices set in motion.
Why People Used Lead Arsenate
The codling moth drove growers to desperation. Apple crops, their mainstay income, suffered from infestations with no real alternative in sight. Paris Green—a previous poison—lost its edge due to insect resistance. Lead arsenate became a hero for a while, drastically reducing crop losses. In its heyday, you couldn’t find a commercial apple orchard in North America that didn’t know the powdery taste and smell of lead arsenate. This sense of a silver bullet came at an enormous health cost, but farmers acted on what seemed to be the best science at the time.
The Downside Nobody Wanted
Moving through old orchard country, people today still stumble across the true cost of lead arsenate. Lead and arsenic don’t just wash away—they stick around. They get into the soil, settle deeper each year, and stay poisonous for generations. My neighbor once bought a charming farmhouse with an abandoned orchard, then found his well water full of arsenic. Playgrounds on old orchards in suburban areas, like some I knew growing up in New Jersey, draw news crews for toxic soil tests. This isn’t just a rural problem—it ends up in urban gardens and tree-lined backyards where parents want their kids to dig in the dirt.
Health Risks and Lasting Impact
Exposure to lead can stunt brain development, lower IQ, and hurt kids most of all. Arsenic brings cancer risk, gut issues, and skin diseases. Studies, like those from the US Geological Survey and Cornell University, show that locations once doused with lead arsenate often hold persistent danger. Most states now keep registries of where orchards once stood. Buyer beware doesn’t quite cover it; the contamination outlasts the people who spread it.
What Can Be Done Now?
Testing soil stands as the best place to start. Homeowners near former orchards need to send soil to labs for lead and arsenic checks. If the numbers run high, covering soil with grass, mulch, or even pavement keeps exposure low. Excavating and hauling away the topsoil costs a fortune, making prevention and education all the more important. Many local health departments offer discounted tests or community workshops, which help fill knowledge gaps.
Switching the focus to safe gardening methods, raised beds with clean soil, and avoiding deep digging helps a lot. Communities need open records on former pesticide use, so new residents know what’s underground. My experience has shown that people want to do the right thing, but only if they know the risks they face.
The tough lesson in the story of lead arsenate is about short-term fixes leaving long-term scars. By paying attention to the land's history, we do better for the next generation, and maybe avoid repeating old mistakes with the new technologies we knock on today’s door.
Is lead arsenate dangerous to humans or the environment?
What Happens When Lead Arsenate Gets Into Our Lives
Growing up near apple orchards, I remember the smell of chemicals after big sprays in early summer. People didn't always like talking about what those sprays included, but lead arsenate used to stand out. Farmers turned to this chemical, hoping it would fight off pests eating fruit. Tough as it is to hear, that decision didn't fade with the fruit season – the impact stuck around.
Lead arsenate is no longer sprayed on crops in most countries, but the soil is still holding onto it. Research from Cornell University shows that lead and arsenic levels remain in former orchard soils even fifty years after spraying. Local kids playing in the dirt, gardeners planting vegetables, neighbors working on new housing – all can kick up old dust, breathing in or carrying home contaminated soil.
Why People Worry: Health and the Environment
Lead doesn’t leave the body easily. Even small exposures in children can damage brain development, sparking lifelong trouble with focusing or learning. Centers for Disease Control and Prevention (CDC) studies highlight no safe level of lead in blood, and lead arsenate gives you two poisons in one hit. Most people know about lead paint in old houses, but poisoned soil flies under the radar until a test picks up surprising results.
Arsenic adds its punch. Long-term exposure links to heart disease and certain types of cancer, according to studies in Environmental Health Perspectives. Drinking water across the globe faces arsenic worries, but leftover sprays increase the danger in yard dust, local wells, and homegrown food. Birds, insects, earthworms – all pay the price too, and poison moves up the food chain.
Cleaning Up a Mess Left Behind
The mess left by lead arsenate isn’t simple to fix. Digging up and removing soil costs a bundle. Most families and communities living on former orchards cannot afford that. Even cities struggle with the price tag. Covering the most contaminated spots with fresh soil, planting deep-rooted grasses, or building raised beds for gardening – these are real-world ways people have tried to limit exposure.
Testing matters. Many states offer soil screening for lead and arsenic at little or no cost. If levels show up high, planting fruit trees or vegetables in raised containers, washing hands after digging, and swapping out shoes before coming indoors can help cut risk for everyone at home. Public health groups suggest simple routines: wet-mopping floors, dusting with damp rags, and keeping soil off playgrounds.
Looking Forward: Less Use, More Awareness
A lot of people want stronger rules to prevent new poisons from being used this way. Bans made a difference but didn’t erase the residue left behind. Education remains the strongest tool we have: teaching families, gardeners, and developers to test and protect their lots. Federally-backed cleanup programs, and research into new plant-based soil treatments, look promising. None of this goes fast, but people rooted in these communities know too well that action beats letting kids play in silence where invisible danger lives.
How do you safely handle and dispose of lead arsenate?
Understanding the Risk of Lead Arsenate
Lead arsenate holds a nasty reputation as a historic insecticide, especially for apples and other fruit crops. Years ago, orchards leaned on it for pest control because it worked well, and no one gave much thought to what stuck around in the soil. Over time, health experts uncovered serious dangers. Both lead and arsenic damage the nervous system and organs, sometimes long after exposure. Breathing dust, getting it on your hands, or eating food grown in contaminated ground slowly chips away at your health, especially for kids. Once in the body, both metals stick around for a long time, building up over the years.
Personal Responsibility and Protective Measures
If you come across old lead arsenate, maybe from farm work or home renovation, don’t treat it like just another bag of fertilizer. Start with gloves, goggles, and a dust mask—skipping even one of these ramps up the chance of breathing in or absorbing these poisons through your skin. Do the work outdoors on calm days. Never wash work clothes with your regular laundry. Wash hands and exposed skin right after handling contaminated stuff. It’s tempting to power-wash tools afterward, but that just spreads the mess. Scrape off visible dirt, and seal any rubbish in sturdy containers.
Disposal—Where Mistakes Get Costly
Tossing leftovers or dirt laced with lead arsenate in the backyard means trouble for pets, kids, and ground water. Local laws put the responsibility on individuals not to dump hazardous waste. Most communities point to a household hazardous waste collection site, and these places know how to keep dangerous metals from leaking into the ecosystem. I’ve hauled a bucket of orchard soil to a county event once, and it surprised me how little paperwork and hassle they needed—just transparency about what I brought along.
Long-Term Solutions and Remediation
Whole neighborhoods in old orchard country still fight with these residues. Lead and arsenic stay put near the top few inches of soil for decades. Some landowners fence off hot spots, lay thick mulch, or cover old orchard beds with landscape fabric. These steps may not pull the metals out, but help block dust and direct contact. Universities and extension offices often run soil tests at low cost, making it easier for families to check garden plots before planting food crops. Removing soil and trucking in clean fill works, though the price stops most people. For community parks or schoolyards, federal and state help sometimes funds these large fixes.
Building Awareness and Prevention
People shouldn’t have to guess whether their yard or garden is safe to use. Towns can work together to map old spraying locations and share the news. Real estate agents and property records ought to flag properties with known contamination history, so each new owner isn’t left in the dark. Local workshops and school science lessons can show how tiny traces of metal make a big difference for health, planting the idea early to check soil before growing food.
Bringing Science into Action
Doctors, public health workers, and agriculture pros all bring good evidence to the table about lead and arsenic. They know which foods pick up more metals from dirt, and which building lots match historic orchard maps. Community action rooted in science and local knowledge helps keep these poisons out of kitchens and playgrounds. People who take a little time to learn the risks, suit up, and follow rules for disposal save themselves and their neighbors from decades of trouble.
What crops or pests was lead arsenate commonly used on?
The Role of Lead Arsenate in Farming
Farmers looking for ways to keep their crops from being ruined by pests once leaned heavily on lead arsenate. This chemical blended arsenic with lead and became a go-to weapon in the hands of growers, especially before World War II. In my own work cataloging the trends in agriculture, I keep seeing references to how widespread this stuff got, particularly on fruit trees. Apple and pear orchards in places like New York and Washington ended up depending on it year after year. If you can picture standing among those trees, you’d understand the challenge: each fruit susceptible to codling moths boring their way in. These moths would wreck entire crops and leave families who depended on a harvest with little to show for their labor.
Battling the Codling Moth on Fruit Trees
Growers weren’t just fighting a minor annoyance. The codling moth made apples and pears nearly unmarketable. Lead arsenate dust got sprayed straight onto the leaves and roots in a fine white mist, blanketing orchards. Farmers felt relief once the damage slowed, and for decades nobody paid much mind to what happened in the soil or what washed away in heavy rains. Strawberries and cherries also saw doses of this pesticide, mainly out West, where large acreages could be threatened all at once.
Expanding Use to Other Crops and Pests
Lead arsenate proved to be versatile, at least in those days. In the South, it helped cotton farmers battle the boll weevil, a pest that demolished fields. Cotton represented a means of survival for entire communities, so folks did whatever they thought necessary. In California, grape growers used it against grape leaf folders. Even vegetables grown for local markets received treatments, with leafy greens at risk from armyworms and cabbage worms. Gardeners and truck farmers once followed suit, trusting what seemed to be good science and what the local ag agent endorsed.
Health, Soil, and Water Impact
Few people realized the long-term problems tied to lead arsenate. Over years of spraying, lead and arsenic built up in orchard soil. No rainstorm or winter freeze seemed to wash this legacy away. I’ve walked in some of those old orchards—decades since the trees last saw a pesticide sprayer—and growers still test soil before planting anything new. The tools used for old problems left a mark; the lead and arsenic won’t break down or disappear. Even backyard gardeners in neighborhoods built on these sites worry about planting tomatoes or letting children play in the dirt.I’ve seen the anxiety that can take hold when groundwater tests come back showing arsenic traces. Even at low levels, long-term exposure carries big risks, especially for children. Not every community has access to frequent soil or water testing, which can mean years of exposure before anyone realizes something is wrong.
Balancing Crop Protection With Health
Today, science offers safer options for managing pests. Integrated Pest Management (IPM), for example, combines monitoring, biological enemies, careful targeting of chemicals, and smarter crop choices. Teaching farmers and homeowners about these choices gets more important with every story of lingering contamination. It’s not about blaming past generations—they worked with what they were given—but making sure fields today support crops and families safely, both above and below ground.
Moving Forward: Testing and Remediation
Ongoing sampling of soil and water in former orchard areas stays critical. Certain plants, like sunflowers, can help remove some lead from soil. Sometimes more drastic steps—like excavating contaminated earth—need to happen around homes and old orchards. Policymakers listening to folks in affected areas build trust when they offer funding for cleanups or support for healthier farming techniques.
Lessons for Crop Protection
The story of lead arsenate stands as a warning and a lesson. Innovations need real vetting, but ongoing care matters too. Research keeps things honest, but community involvement and regular testing will always add a layer of safety that chemistry alone can’t guarantee.
Are there alternatives to using lead arsenate for pest control?
Lead arsenate once played a big role in farming. For decades, growers across the world sprayed their fields and orchards with it to keep crops from being devoured by insects. The results looked good at the time, but the health risks grew more obvious with every season. Cancer, developmental problems in children, and poisoned soil and water—these are not small matters. Today, many countries outlaw or tightly regulate lead arsenate, but some places still struggle with its legacy. If people keep using dangerous chemicals like this, the toll on public health and food safety keeps going up.
Alternatives With Less Long-Term Risk
Farmers and gardeners find lots of options if they want to leave lead arsenate behind. Biological pesticides, such as those based on Bacillus thuringiensis (Bt), do solid work against many insects but rarely hurt people or wildlife. Bt’s proteins target certain bugs, leaving pollinators and other helpful creatures unharmed. That’s a real step forward. For tree fruit and vegetable crops, insecticidal soaps and horticultural oils break down faster and carry less risk for water and soil. These products, often approved for organic use, have years of research behind them showing low residue and fast breakdown.
Integrated Pest Management (IPM) changes the game even more. Instead of putting down chemicals by the calendar, IPM uses careful scouting and traps. Growers hit pests only after numbers hit a certain level, so fewer sprays go out. Crop rotation and resistant varieties slow down or block pests before chemicals become necessary. Many universities back this approach, showing growers how to mix physical, biological, and chemical methods.
Community Knowledge Shapes Solutions
Modern farming relies on sharing experience. In my own small orchard, I watched neighbors use pheromone sprays that disrupt insect mating. These products confuse the pests long enough to drop their numbers without heavy-duty sprays. I tried it myself on my apples last year. Less fruit got bitten, and pollinators like bees stuck around in higher numbers. Local extension agents run workshops where farmers learn about this and swap stories. Their firsthand tips beat leaflets or company ads.
Switching away from old-school chemicals isn’t always quick. New methods often cost more up front, and learning a new routine can frustrate people set in their ways. But resistance to old sprays like lead arsenate keeps increasing among pests, leading to more sprays for less effect. The cycle drives up costs and risk. There’s real evidence that communities investing in organic and low-impact approaches see bigger savings and higher land values over time. Health benefits, too, add up for both workers and neighbors. Data from the U.S. EPA and World Health Organization link long-term pesticide exposure with higher rates of cancer and neurological disorders in farm families and children. By taking steps toward less-toxic tools, farmers protect their own kids and land as much as their harvests.
Moving Toward Safer Farming
Farmers, gardeners, and local officials come together to pick smarter pest control options. Access to reliable information makes a difference. Public funding for research into natural enemies—like parasitic wasps or beneficial nematodes—gives growers more paths away from harsh chemicals. Policies can push for buffer zones around homes, schools, and water sources and reward farms that protect soil and water. In my own experience, even small changes—switching one chemical for another or tweaking the spray schedule—add up to cleaner air, healthier soil, and a safer harvest for everyone.
| Names | |
| Preferred IUPAC name | dilead(II) arsenate |
| Other names |
Acid lead arsenate Basic lead arsenate Lead hydrogen arsenate Arsenic acid, lead(2+) salt (2:3) Ceresan Gypsine |
| Pronunciation | /ˈliːd ɑːˈsɛneɪt/ |
| Identifiers | |
| CAS Number | 7784-40-9 |
| Beilstein Reference | 35367 |
| ChEBI | CHEBI:33155 |
| ChEMBL | CHEMBL42961 |
| ChemSpider | 15921 |
| DrugBank | DB06778 |
| ECHA InfoCard | 100.004.756 |
| EC Number | 232-099-9 |
| Gmelin Reference | 7891 |
| KEGG | C18622 |
| MeSH | D007864 |
| PubChem CID | 3032646 |
| RTECS number | CG3325000 |
| UNII | 2P299V78G6 |
| UN number | UN1616 |
| Properties | |
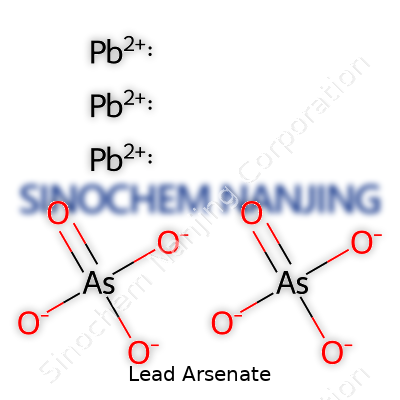
| Chemical formula | Pb₃(AsO₄)₂ |
| Molar mass | 807.13 g/mol |
| Appearance | White powder |
| Odor | Odorless |
| Density | 6.86 g/cm³ |
| Solubility in water | Insoluble |
| log P | -2.48 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 6.3 |
| Magnetic susceptibility (χ) | Diamagnetic |
| Refractive index (nD) | 1.687 |
| Dipole moment | 0.00 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 626.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1767 kJ/mol |
| Pharmacology | |
| ATC code | S01BA06 |
| Hazards | |
| Main hazards | May be fatal if swallowed, inhaled, or absorbed through skin; causes damage to organs; suspected of causing cancer; very toxic to aquatic life. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08,GHS09 |
| Signal word | Danger |
| Hazard statements | H300 + H330: Fatal if swallowed or if inhaled. H350: May cause cancer. H360: May damage fertility or the unborn child. H372: Causes damage to organs through prolonged or repeated exposure. H410: Very toxic to aquatic life with long lasting effects. |
| Precautionary statements | P201, P202, P260, P264, P270, P273, P280, P308+P313, P314, P391, P405, P501 |
| NFPA 704 (fire diamond) | 4-2-3-Pb |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD₅₀ (oral, rat): 100 mg/kg |
| LD50 (median dose) | LD50 (median dose): 105 mg/kg (oral, rat) |
| NIOSH | SN4540000 |
| PEL (Permissible) | 0.15 mg/m³ |
| REL (Recommended) | 0.01 |
| IDLH (Immediate danger) | 100 mg/m3 |
| Related compounds | |
| Related compounds |
Lead(II) acetate Lead(II) carbonate Arsenic trioxide Paris green |

