Lanthanum Nitrate: An In-Depth Look at a Rare Earth Compound
Historical Development
The journey of lanthanum nitrate began with the wider exploration of rare earth elements in the nineteenth century, a time that fueled discoveries reshaping chemistry labs and tech industries alike. Early chemists like Carl Gustaf Mosander pulled lanthanum out of the mineral cerite and noticed its unique properties. Scientists worked with rudimentary isolation techniques for decades, but the material drew serious attention as advances in separation methods improved. By the mid-1900s, refined lanthanum nitrate became available beyond simple research labs. Demand for pure, reliable lanthanum nitrate grew as electronics, optics, and catalysis fields matured.
Product Overview
Lanthanum nitrate steps forward as a white crystalline solid, easily dissolving in water and known for delivering high lanthanum ion content in dissolved form. Manufacturers deliver it in hydrated form, usually hexahydrate. In the hands of researchers and makers of specialty glass, this compound offers a convenient way to introduce lanthanum into reactions or formulations. Its use spans polishing powders, phosphors in lighting, and catalysts in fuel conversion, connecting the old days of painstaking extraction to today's fast-paced technology development.
Physical & Chemical Properties
Solid lanthanum nitrate appears as colorless or faintly white crystals, taking up water from the air. Its molecular formula, La(NO3)3·6H2O, spells out the story: lanthanum binds with three nitrate groups and usually six water molecules. The crystals melt at around 50 °C, then lose water and eventually decompose, releasing toxic nitrogen oxides. In water, it breaks apart into lanthanum and nitrate ions, giving it high solubility and reactivity. The compound’s density sits just over 2.5 g/cm3, marking it as heavier than many common salts. This high solubility and strong oxidizing character bring both benefits and hazards to industrial and lab work.
Technical Specifications & Labeling
Industrial and research buyers want detailed labels, and for good reason. Purity rates push above 99% for high-end applications, sometimes labeled as 99.99%, signaling trace impurity controls. Labels routinely list hydrate form, batch number, assay percentage, lead and iron content, and the manufacturer’s name. Proper storage gets a nod, too, with warnings about moisture pickup and the need for sealed containers. For transport, lanthanum nitrate picks up hazardous material identifiers, mostly for its oxidizing power, and may ship under UN number 1477 or similar global codes, depending on jurisdiction.
Preparation Method
Producers commonly make lanthanum nitrate by reacting lanthanum oxide or carbonate with nitric acid. The process releases water and carbon dioxide (if starting from carbonate). Mixing the ingredients under controlled temperatures results in a clear solution, which cools to form hydrated crystals. Recrystallization steps follow for higher purity, where solid crystals form, get filtered, and dry in temperature-monitored rooms. Most manufacturers rely on closed systems to trap fumes and recycle gases, paying attention to environmental rules and worker safety since both acid and nitrate fumes corrode equipment and hurt lungs if containment fails.
Chemical Reactions & Modifications
Lanthanum nitrate acts as an entry point for other lanthanum compounds. Reacting it with sodium carbonate precipitates lanthanum carbonate, while exposure to phosphoric acid leads to lanthanum phosphate, prized for catalysts and ceramics. Mixing in an alkaline medium, it flips back to lanthanum hydroxide, part of more downstream chemistries. As a strong oxidizer, lanthanum nitrate helps break down organic compounds under certain lab protocols. Its responsive chemistry underlies many uses, but also demands respect, as improper mixing may cause release of toxic nitrogen oxides or rapid heat generation.
Synonyms & Product Names
Many catalogues and safety sheets list this material under names like lanthanum(III) nitrate, lanthanum trinitrate, or simply La(NO3)3. Some older texts call it lanthanum nitrate hexahydrate, matching its most common shipped form. Trade labels sometimes append “purified” or “high purity,” and language differences might spell out similar names, but confusion stays limited since rare earth chemistry draws a technical, well-informed crowd.
Safety & Operational Standards
Handling lanthanum nitrate means following strict safety priorities. The compound’s oxidizing power can turn combustible materials into fire risks if spilled or improperly stored. In halogenated labs, rulebooks set storage apart from organic solvents and keep the material in dry, cool places. Gloves, goggles, and masks block dust or fumes from reaching skin and lungs. Facilities rely on chemical fume hoods for weighing and mixing, reducing accident risk. Workers need access to clear safety data sheets, and emergency showers or eyewash stations cut response times after spills. First-hand, poorly ventilated prep rooms spell trouble fast, so routine air monitoring and staff drills crop up as standard practice.
Application Area
This compound leaves fingerprints in quite a few high-value industries. Makers of specialty and optical glass use lanthanum nitrate to boost refractive index and cut unwanted light transmission, which has become vital in smartphone cameras and telescope lenses. In petroleum refining, companies use its catalysts to upgrade fuel—maximizing output from crude. Lanthanum nitrate’s phosphor production role powers energy-efficient light bulbs and display screens, where color purity and brightness edge out rivals. Academic and government labs value it for developing separation science and studying rare earth behaviors. Its reach even spills into emerging areas, like water treatment, where modified products snatch phosphate from waste streams, helping slow the march of water pollution.
Research & Development
For me, nothing matches the thrill of experimenting with a substance central to current materials innovation. Modern research dives deep into new uses and purer grades, since both market pressure and environmental goals keep rising. Teams trial nano-scale versions for electronics and hybrid materials, combining lanthanum nitrate with doped crystals or polymer films. Graduate students chase improvements in LED phosphors, superconducting ceramics, or even medical imaging agents. The challenge remains high: every year, someone tweaks preparation, blending, or application and unearths data that push boundaries for greener, smarter manufacturing.
Toxicity Research
Lanthanum’s health effects stay a top concern. Acute exposure to nitrates, whether through inhalation or skin contact, triggers nausea, headache, or irritation, and lanthanum in its ionic form may show mild toxicity toward aquatic life and plants. Lab rats and aquatic tests show that excessive dosing disrupts cell function, although evidence for big-picture environmental or chronic health harm stays limited outside industrial settings. Researchers call for careful monitoring any time lanthanum nitrate enters wastewater or spills. Modern toxicology emphasizes worker controls, proper effluent treatment, and careful end-use evaluation—especially as lanthanum applications spread.
Future Prospects
As demand for clean energy, advanced optics, and smarter electronics climbs, the spotlight on lanthanum nitrate only gets brighter. Startups and global giants both eye new purification and recycling steps, hoping to lower extraction impacts and close waste loops. Data suggests that cleaner, denser phosphors and lighter-weight glass alloys will soon rely even more on high-quality lanthanum sources. Spectroscopy, battery tech, and water purification poised for takeoff could reshape the market, pulling in more research dollars. The call rings out for safer production, greener chemistry, and robust regulations to match the pace of innovation. Anyone in the field who has seen routines shift with one regulatory memo or supply disruption knows: innovation around lanthanum nitrate won’t slow down any time soon.
What is Lanthanum Nitrate used for?
The Role of Lanthanum Nitrate in Everyday Science
Lanthanum nitrate shows up in the background of a lot of products and research efforts, although few people outside laboratories talk about it. I remember seeing its name for the first time back in college chemistry class. My mentor waved a glass vial filled with a white powder and talked about rare earth elements that shape how we power devices and clean water—even if most of us never see this stuff up close.
Lanthanum Nitrate in Optical Glass and Ceramics
In the optics industry, manufacturers use lanthanum nitrate as a source of lanthanum for high-refractive index glass. This special kind of glass improves camera lenses, microscope objectives, and eyeglasses. It’s not about making the glass look fancy—it makes images sharper and reduces the blur at the edges. Tech companies take this edge seriously because clear images sell well in a world obsessed with megapixels. Many modern smartphones, armed with high-performance lenses, owe part of that clarity to rare earth ingredients, including lanthanum.
Lanthanum nitrate also makes its way into advanced ceramics. These heat-resistant ceramics end up insulating parts inside jet engines, protecting components from the punishing heat generated by combustion. As a result, engines run longer and more efficiently, which means airlines can run operations with fewer delays and lower fuel costs.
Lanthanum Nitrate in Catalysts and Environmental Applications
Catalytic converters clean exhaust fumes by breaking down pollutants before they escape from a car’s tailpipe. Lanthanum nitrate provides lanthanum for these catalysts, helping turn carbon monoxide or nitrogen oxides into gases that don’t poison our lungs. Major cities have seen marked improvements in air quality since these technologies became standard on cars.
Beyond automotive exhaust, water treatment engineers have found lanthanum compounds useful for trapping phosphates. Removing excess phosphates helps curb the spread of toxic algae blooms in lakes or reservoirs. I’ve spoken to local government officials who watch water quality numbers every season. For them, the difference between clean and green-sludge lakes often hinges on finding the right treatment chemicals. Lanthanum nitrate’s role in making clean water safer may not make the news headlines, but it keeps swimmers and fish safer every summer.
Lanthanum Nitrate in Research and Medicine
Lanthanum nitrate acts as a chemical reagent in labs to create other useful materials or to test for certain ions. Analytical chemists use it to hunt for traces of elements in samples where precision counts for everything. The stakes go beyond curiosity—mining companies evaluate ore samples for profitability, and environmental agencies monitor soil contamination.
In medicine, lanthanum compounds have been tested for use in radiography and as treatments for phosphate control in patients with kidney disease. These medical uses rely on the unique ways lanthanum compounds behave inside the body. Doctors and researchers run trials to weigh the safety and benefits, a process that sometimes takes years just to reach market approval.
Looking Forward
Lanthanum nitrate may not draw much attention, but its presence keeps research moving and technology advancing. These are the kinds of ingredients that quietly enable change, especially in fields pushing for cleaner energy, safer water, and stronger materials. When I think about what gives an edge to a lens or pulls toxins from water, it’s often unseen elements working behind the scenes. Lanthanum nitrate, in all its plainness, carries real weight in today’s push for better living.
Is Lanthanum Nitrate hazardous or toxic?
Understanding Lanthanum Nitrate
Lanthanum nitrate enters labs and industrial settings in the form of a white crystalline solid. It dissolves in water with ease and finds its way into catalysts, ceramics, water treatment, and research. People who handle lanthanum compounds usually pay extra attention to their material safety data sheets, but not everyone gets a clear sense of which risks demand respect and which ones get blown out of proportion.
Health Risks: Where They Come From
Breathing in lanthanum nitrate dust or fumes turns out to be the biggest worry. The compound can irritate the respiratory tract, eyes, and skin. Inhaling too much can bring coughing, shortness of breath, and in some rare cases, more severe lung trouble. The science behind chronic health effects from low-level work exposure isn’t fully settled. A paper in the journal Chemosphere published in 2021 looked at lanthanum exposure in rare earth mining. It pointed to possible connections between long-term contact and kidney stress, though researchers call for more studies on workers in high-exposure industries.
Swallowing lanthanum nitrate by accident isn’t common outside the lab. If it happens, symptoms may run from nausea to abdominal pain. There’s not enough real-world data to prove lasting organ harm just from brief dietary exposure, but regular ingestion could threaten the kidneys and liver. The LD50 in rats clocks in at around 3,800 mg/kg, according to Sigma-Aldrich, placing lanthanum nitrate below the threshold of acutely toxic chemicals. That doesn’t mean it’s safe to get careless. The science suggests effects add up over time, especially for people with kidney problems.
Environmental Impact
Accumulation in aquatic environments poses trouble for fish and plants. Lanthanum nitrate doesn’t break down in water, so it can move through the food chain once it enters lakes and rivers. Scientists have tracked bioaccumulation in freshwater snails and algae, even at levels that seem low. Some studies out of Sweden and China show lanthanum builds in fish tissues and disrupts enzyme systems. Not enough is known about exactly how much lands in the wild from factories or water treatment, but the signals are strong enough to warrant tighter spill controls.
Handling and Prevention in Workplaces
Too many people downplay their risk because lanthanum nitrate lacks the drama of classic hazards like cyanides or mercury. From my own lab days, I saw people wear gloves for handling acids but shrug off white powders like this one. Every few years, someone in research will flush this spent solution down the sink, thinking dilution solves everything. It doesn’t — municipal treatment plants aren’t built to pull lanthanides from the water. Many universities and responsible companies now insist on chemical waste bins just for rare earths and train staff to avoid skin contact. The value of safety goggles, lab coats, and working in a fume hood feels obvious after your first accidental splash or whiff.
Reducing the Risk Together
Clear communication beats warnings that get buried in paperwork. Printed hazard labels, real training with spill kits, and shared lessons from near-misses keep everyone honest about the risks. Efforts should focus on both acute exposure and the risk of cumulative buildup — especially in settings that handle large volumes or run processes at high temperatures. By collecting and disposing of lanthanum-containing waste properly, workplaces protect water quality and keep regulatory headaches away. Facilities that give environmental impact real weight tend to draw the line at pouring anything questionable down the drain.
Every compound brings its own story — lanthanum nitrate deserves respect but doesn’t mean panic. With common-sense handling, good housekeeping, and attention to research, people can manage its hazards while taking advantage of its unique chemistry.
What is the chemical formula of Lanthanum Nitrate?
The Bare Facts: La(NO3)3
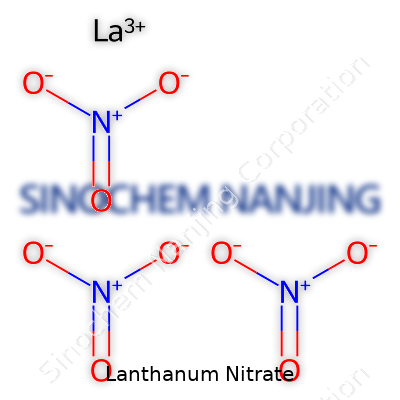
Lanthanum nitrate, known in the lab by the formula La(NO3)3, has a structure that spells out something both simple and intriguing. This compound falls under the group of rare earth metal nitrates. Each molecule consists of one lanthanum atom and three nitrate groups. It shows up as a white, highly soluble salt, which means it dissolves easily in water. Scientists and educators look at the formula and see the unmistakable presence of the lanthanide series, not just as a curiosity but as an ingredient powering a thin line between basic chemistry and advanced technology.
Why It Matters in Real Life
As a writer who’s spent some long nights wrangling with chemistry articles and industrial case studies, the real world sometimes throws surprises that textbooks skip. Lanthanum nitrate doesn’t draw crowds like gold or silicon, but industry and research labs rely heavily on it. It works well in producing specialty optical glasses and ceramics. Some of those shining optics in a camera, laser, or scientific instrument? They probably have roots in materials that once relied on this compound. Chemists use it as a starting point to prepare lanthanum oxide, another key material found in electronics and catalysis. This salt also pops up in the making of catalysts for petroleum refining—where it helps turn crude oil into more useful fuels.
Handling Hazards and Promoting Safe Practices
Every chemical, beneficial or not, comes with a tradeoff. La(NO3)3 deserves respect for its power. It’s an oxidizer, which means it can cause or intensify fires if mixed with the wrong substances. Inhaling or ingesting any nitrate—lanthanum's included—isn’t something to take lightly. Working in a lab, I’ve learned you can’t let your guard down with white powders. Even simple spills need to be cleaned up right away using gloves, goggles, and a steady hand. Regulations call for clear storage instructions and disposal plans. Waste shouldn’t go down the drain, as it poses environmental risks to waterways and aquatic life. Labs separate it in special containers, followed by disposal through licensed chemical waste handlers. These steps follow both common sense and government rules, not just to tick boxes but to protect real workers and students every day.
Strengthening Awareness and Education
Beyond the chemical formula and safety datasheets, there’s a problem of awareness. Not every lab or classroom has equal access to up-to-date safety protocols or training. Sometimes I’ve walked into teaching labs and seen students unsure about what to do with unused chemicals at the day’s end. It’s not enough to label bottles; everyone working with chemicals should know precisely what’s inside them and how to act in a spill or emergency. Universities, companies, and schools can boost safety by running regular training, keeping datasheets handy, and encouraging questions.
Seeking Sustainable Alternatives
Growing demand for electronic devices keeps rare earth minerals like lanthanum in the spotlight, and that brings up sustainability challenges. Mining, refining, and transporting rare earths comes with an environmental footprint that stretches well beyond the lab. Research labs and manufacturers are exploring ways to recover and recycle materials, reduce waste, and use less hazardous chemicals where possible. Staying ahead in science means not only knowing your formulas but thinking about the long game—the impact on people, the environment, and future innovation.
How should Lanthanum Nitrate be stored?
Understanding the Real Hazards
Lanthanum nitrate stands out among the rare earth salts because of its water solubility and its tendency to react in unpredictable ways if not handled with care. Many people see its pale crystals and underestimate the risks. My time in research labs taught me a different lesson—chemicals often surprise those who skip the small print on storage and safety data sheets. Left exposed or left out on a benchtop, lanthanum nitrate will absorb moisture from the air, clump up, and lose accuracy for any scientific work. Breathing dust can kick up bouts of coughing, sometimes more serious trouble if allergies come into play. Nobody wants to explain that to the safety officer.
Ideal Storage Conditions
A dry, cool location keeps lanthanum nitrate reliable. Many storage rooms end up too humid or warm, especially in older labs without air conditioning. I learned to stick each bottle in a dedicated desiccator filled with fresh silica gel or anhydrous calcium chloride. Night or day, that setup blocks moisture and helps avoid caking. Containers should always close with airtight screw caps, not crumbling parafilm, since the compound will pull water vapor through even tiny gaps.
Direct sunlight or heat will push the nitrate ions to break down. At my last job, sunlight from a window led to yellowing bottles after a single summer. That changed fast—everything moved into shielded cabinets. Flammable solvents might share a lab, but lanthanum nitrate doesn’t catch fire easily on its own. Still, it reacts with organics under the wrong conditions or if spilled. Segregating it from anything flammable or reactive gives some peace of mind—no one wants to create a violent mess just because a shelf tilts the wrong way.
Spill Control and Label Awareness
I once watched a careless spill go from a minor inconvenience to a safety meeting in ten minutes. The white powder spreads quickly, contaminating gloves, counters, and sometimes—without enough attention—food left on the bench. To prevent this, containers must always carry a clear label with hazard warnings and a visible date of purchase. That’s critical if the lab stores more than one nitrate salt or rare earth compound side by side. Different lots age in different ways, sometimes becoming useless for high-precision measurements. Rotate stock, check expiry dates, and never trust an open-top jar from who-knows-where.
Disposal and Handling Practices
The urge to pour small leftovers down the drain tempts many students, but environmental rules couldn’t be clearer. Lanthanum nitrate harms aquatic life. Proper chemical waste bins, lined and sealed, keep locals safe. During daily handling, disposable gloves and eye protection block exposure; training new users never stops. Sharing experience goes a long way—one person’s oversight can soon become everyone’s problem. Stubborn powder on a glove means a desk and a keyboard get contaminated. Gentle vacuuming beats sweeping, which only creates airborne dust.
Practical Solutions for Safe Storage
Even in busy workspaces, simple changes make a big difference. A dedicated lanthanum nitrate shelf, with posted handling rules, improves habits and cuts accidents. Regular audits ensure no bottles sit open or unlabeled. Ventilated, lockable cabinets offer both security and environmental protection. Colleagues appreciate checklists and ongoing safety reminders. It only takes one mistake to lose a week of research time or worse, so keeping the workspace tidy and organized keeps both people and data safe.
At the end of the day, safe storage and handling protects not just individuals, but the integrity of the whole lab.
What is the purity specification of your Lanthanum Nitrate product?
Real Value in Knowing Purity
Anybody who’s worked in a chemistry lab or dealt with rare earth materials knows it’s no small thing to ask about purity. For Lanthanum Nitrate, purity basically tells you how much of the bottle is really your requested compound — not leftovers, fillers, or sneaky trace metals. In research or manufacturing, this information separates predictable results from wasted work. Pure inputs make the difference between meaningful analysis and head-scratching anomalies. Personally, I’ve seen plenty of projects get knocked off track by one hidden impurity in a bottle that nobody double-checked.
Usual Specs and Why They Matter
On the market, high-quality Lanthanum Nitrate often shows up with a minimum purity of 99.9%. This means for every thousand grams, only one gram could be something else. I’ve seen some labs and suppliers push that even further — you’ll see 99.99% for especially sensitive jobs. The main enemies in low-purity samples aren’t just dirt; look for traces of other rare earth metals like cerium or neodymium, and the classic industrial headaches: iron, calcium, and copper. Even fractions of a percent can end up spoiling experiments, particularly in analytical chemistry, water treatment, or electronic material synthesis.
How Purity Impacts Real Work
It’s easy to shrug off numbers after the decimal. But one lesson I learned: if you’re trying to make a catalyst, or prepping a calibration standard for testing, that last bit of impurity shows up as noise — or worse, produces fake positives. For example, in atomic absorption spectroscopy, a contaminant can mess with the accuracy of the readings, bathing expensive equipment in errors and burning scientist time. Not once or twice, but over dozens of runs, impurities can empty procurement budgets without anybody noticing until the bad data lands on someone’s desk.
Assessing Supplier Claims
Most reliable companies list full trace impurity profiles alongside their certificate of analysis. It’s not enough to read “99.9%” and feel safe. Ask to see their actual breakdown and look for documentation on analytical methods used: Inductively Coupled Plasma (ICP) methods usually lend stronger confidence than colorimetric tests. Cross-check for the elements you know you can’t have — if you're doing glass manufacturing, iron impurities mean off-color glass; in energy storage, sodium or potassium sneaking in can foul ionic conductivity. Always match their data to your tolerance and don’t settle for vague answers.
Finding Solutions to Common Pitfalls
If purity falls short of what's needed, don’t just hope for the best. Labs with basic skills can sometimes purify using recrystallization, but that only suits small batches and relatively friendly contaminants. For industry-scale work, the only safe bet lies in opening an honest conversation with suppliers or switching vendors. Saving time by compromising on purity often ends up costing more — in materials, in trouble tickets, in rework. The answer seems simple, but I’ve seen even the smartest chemists learn this the hard way: always demand paperwork and don’t assume all “La(NO3)3” is created equal.
Conclusion
Choosing Lanthanum Nitrate with the right purity isn’t just paperwork for compliance. It sets the foundation for safe, repeatable work that supports science and industry moving forward. If the bottle isn’t labeled 99.9% or above, and you don’t see a breakdown of elements, move on. There’s nothing quite as disruptive as chasing ghosts from one small impurity nobody bothered to check.
| Names | |
| Preferred IUPAC name | lanthanum(3+) nitrate |
| Other names |
Lanthanum trinitrate Lanthanum(III) nitrate |
| Pronunciation | /ˈlænθə.nəm ˈnaɪ.treɪt/ |
| Identifiers | |
| CAS Number | 10099-58-8 |
| Beilstein Reference | 3839614 |
| ChEBI | CHEBI:60951 |
| ChEMBL | CHEMBL1201603 |
| ChemSpider | 83917 |
| DrugBank | DB11085 |
| ECHA InfoCard | 100.029.254 |
| EC Number | 233-238-0 |
| Gmelin Reference | 81478 |
| KEGG | C18668 |
| MeSH | D008383 |
| PubChem CID | 24845 |
| RTECS number | OX7530000 |
| UNII | JX8458I22P |
| UN number | UN1469 |
| Properties | |
| Chemical formula | La(NO3)3 |
| Molar mass | 432.91 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 2.37 g/cm³ |
| Solubility in water | Highly soluble |
| log P | -2.48 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 7.0 |
| Basicity (pKb) | 9.37 |
| Magnetic susceptibility (χ) | +94.0e-6 cm³/mol |
| Refractive index (nD) | 1.60 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 427.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1920 kJ/mol |
| Pharmacology | |
| ATC code | V07AB |
| Hazards | |
| Main hazards | Oxidizing, harmful if swallowed, causes serious eye irritation |
| GHS labelling | GHS05, GHS07, GHS08 |
| Pictograms | GHS03,GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H272, H315, H319, H335 |
| Precautionary statements | P264, P280, P301+P312, P305+P351+P338, P337+P313, P501 |
| NFPA 704 (fire diamond) | 1-0-0-OX |
| Explosive limits | Explosive limits: Non-explosive |
| Lethal dose or concentration | LD₅₀ Oral Rat: 4200 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50: 4050 mg/kg |
| NIOSH | SN12200 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 1 mg/m³ |
| IDLH (Immediate danger) | IDLH: Not established |
| Related compounds | |
| Related compounds |
Lanthanum chloride Lanthanum oxide Cerium(III) nitrate Praseodymium(III) nitrate |

