L-Prolinol: A Closer Look at a Key Building Block in Chemistry
Historical Development
Looking back, the story of L-Prolinol fits into the larger arc of progress in organic chemistry. Researchers in the 20th century searched for ways to shape amino acid derivatives into tools with real-world impact. L-Prolinol took center stage, catching the attention of scientists working on everything from asymmetric synthesis to pharmaceutical manufacturing. Its journey began as a straightforward reduction product of L-proline, a natural amino acid with its own unique ring structure. As academics and industrial chemists explored routes to new chiral molecules, L-Prolinol emerged as a key element in asymmetric catalysis, setting the stage for big advances in drug development and material science.
Product Overview
Anyone working in a chemical lab may recognize L-Prolinol as a versatile chiral amino alcohol. At its core, it’s a simple-looking molecule—a five-membered pyrrolidine ring holding both an amino group and a primary alcohol group. This structure lets it bridge the gap between natural biochemistry and synthetic aspirations. As an intermediate, it often carries the hopes of those who want to create complex, chiral architectures in the lab. Whatever the role, L-Prolinol commands respect because it can push reaction selectivity far beyond what most achiral compounds can offer.
Physical & Chemical Properties
L-Prolinol’s structure gives it remarkable stability in typical lab environments. It appears as a white crystalline solid under most storage conditions, dissolving neatly in water and polar organic solvents. The compound’s boiling and melting points anchor it in a comfortable range for routine chemistry—no challenging extremes. Its chirality means it comes in enantiomerically pure forms, and this purity sits at the core of its value to synthetic chemists. The molecule’s combination of nucleophilic amine and reactive alcohol opens doors to countless chemical transformations.
Technical Specifications & Labeling
Labs source L-Prolinol in grades meant for high-stakes chemical synthesis. Analytical data usually highlight its optical rotation, reflecting both enantiomeric purity and batch consistency. It also comes paired with data from HPLC and NMR to help users judge if it fits their protocols. Since L-Prolinol can draw moisture from the air, packaging trends focus on airtight, light-resistant containers. Safety recommendations and clear hazard labeling arise from precaution, especially since even trusted compounds can turn unpredictable in complex settings.
Preparation Method
Starting with naturally available L-proline, chemists rely on reduction methods to generate L-Prolinol. This transformation shines a light on the tricks of classical organic synthesis—the use of reducing agents like borohydrides, carried out in controlled environments to dodge side reactions. The simplicity of the step masks decades of troubleshooting and optimization. Careful adjustment of temperature and solvent prevents over-reduction or formation of byproducts, while purification steps like crystallization or chromatography ensure the final material meets demanding standards.
Chemical Reactions & Modifications
The molecular arrangement in L-Prolinol shapes its reactivity. The amino alcohol group lets it act as both a nucleophile and a hydrogen bond donor, allowing it to serve as a ligand in catalytic processes. Its use in asymmetric synthesis, especially in the creation of proline-derived organocatalysts, cannot be overstated. Protective group strategies often come into play, masking the alcohol or amine for selective downstream reactions. With further tweaks, L-Prolinol can morph into derivatives with specialized behavior in metal complex formation, polymer development, and pharmaceutical intermediates.
Synonyms & Product Names
Chemical supply catalogs list L-Prolinol under several names, most often “(S)-Pyrrolidin-2-ylmethanol,” “S-Prolinol,” and the straightforward “(S)-(-)-2-Pyrrolidinemethanol.” The naming ties into chemical conventions focusing on configuration and structure, not on branding or trade secrets. Researchers recognize these identifiers across borders, letting the compound play a universal role in academic and industrial work.
Safety & Operational Standards
Those who have spent time with L-Prolinol know it ranks as relatively low-risk compared to more reactive organic molecules. Still, gloves, eye protection, and good ventilation remain standard—years of lab practice teach that respect for even stable molecules prevents costly mistakes. Its hygroscopic nature calls for dry, sealed storage and care in humid surroundings. While reports of serious health effects are rare, skin or eye contact can cause irritation, and dust inhalation is best avoided. Standard MSDS documents draw on industry-wide safety norms, pushing labs to adopt containment and spill clean-up measures even if the likelihood of a major exposure event feels low.
Application Area
L-Prolinol has earned its place among chiral building blocks in modern chemistry. Synthetic drug design leans on it to introduce stereochemistry where it can make crucial differences in drug action or safety. Chiral amines and alcohols carved from L-Prolinol form the heart of many API syntheses, especially in anti-hypertensive, anti-viral, and anti-cancer medications. It’s also a key player in asymmetric catalysis, enabling large-scale access to single-enantiomer compounds when batch purity means the difference between a therapy’s success and regulatory rejection. Agrochemical and fine chemical industries tap into this backbone for their own needs, using it in crop protection agents or specialty polymer additives.
Research & Development
Recent years have seen a burst of innovation that draws on L-Prolinol’s chiral potential. Academics and industrial scientists continue to tweak reduction methods, target greener solvents, and shrink reaction temperatures. The quest to make processes safer and more sustainable keeps pushing boundaries, whether by swapping in renewable feedstocks or using biocatalysts to drive reductions under milder conditions. Publications show L-Prolinol cropping up in enantioselective synthetic pathways, not just for medicines but also in the creation of advanced materials with handedness—think chiral liquid crystals or optically active polymers. Improvements in asymmetric organocatalysts, drawing inspiration from L-Prolinol itself, hint at even leaner, more selective routes to complex molecules.
Toxicity Research
People often ask about toxicity and biocompatibility, especially as regulations tighten and sustainability grows in importance across the chemical sector. Data on L-Prolinol suggests a mild safety profile, with limited systemic toxicity in standard animal studies. Exposure often leads only to short-term irritation, not long-term health effects. Still, caution never goes out of style—the lack of chronic or developmental toxicity studies leaves a gap, and it's good scientific practice to treat even mild reagents with informed respect. Comprehensive toxicological research will help clarify its full impact, particularly as applications diversify into new therapeutic and material fields.
Future Prospects
L-Prolinol’s future looks promising as the chemistry world intensifies its focus on sustainability and efficiency. The compound’s blend of reactivity, selectivity, and chiral identity makes it a go-to starting point as synthetic strategies shift toward less waste and greater environmental awareness. Integration with biocatalyst platforms could open doors to cleaner manufacturing. At the same time, new demand in advanced materials, from smart polymers to optical devices, ensures that foundational molecules like L-Prolinol are not fading into the background anytime soon. Whether in a research setting or a commercial plant, the drive to tweak and optimize its chemistry will likely fuel breakthroughs that ripple across science and industry for years to come.
What is L-Prolinol used for?
Digging Into L-Prolinol’s Purpose
L-Prolinol doesn’t make the headlines, but this molecule quietly shapes a lot of what happens in laboratories and manufacturing. If you’ve never heard of it, you’re probably in good company. Yet, once you start working in chemical synthesis, the name pops up in all sorts of discussions. L-Prolinol is a chiral building block, and chemists favor it when seeking molecules with handedness—essential in drug development and materials science.
Why Chemists Chase Chirality
My first exposure to “chiral” molecules felt confusing. Imagine your left and right hands—same components, different arrangement. Medicine relies on molecules fitting into the body the same way, which is why chirality matters. L-Prolinol enables scientists to build compounds that turn in just the right direction. Its backbone sets a specific orientation. This has real consequences. For example, the antidepressant paroxetine uses a similar concept; only one “handed” version works as intended, while the mirror image remains inactive or even harmful.
How L-Prolinol Works Behind the Scenes
Plenty of research papers mention L-Prolinol in asymmetric synthesis. Think of it as the guide that lines up other pieces in an exact formation. Pharmaceuticals demand predictability, especially as regulatory agencies examine both safety and effectiveness closely. Improvisation doesn’t cut it—companies rely on molecules like L-Prolinol for reproducible, precise results before any medicine gets to the patient.
Beyond medicines, L-Prolinol shows up in the development of specialty chemicals. Peptide synthesis—a critical step in producing many modern drugs—leans on L-Prolinol for its steady influence. Catalyst manufacturers also turn to it for making new chemical reactions work more efficiently. Even the fine-tuning of flavors and fragrances benefits from L-Prolinol’s unique properties, though these uses fly under the radar.
Challenges and Solutions with L-Prolinol
Reliable supply chains can pose trouble, as certain syntheses require high purity and consistency. If impurities creep in, they skew the results—whole batches of pharmaceuticals can fail tests or even harm patients. There’s growing demand for sustainable sources of L-Prolinol. Chemists have started pushing for greener processes, using biocatalysis instead of fossil-based reagents whenever possible. In my own lab days, the push for more environmentally friendly practices often led to redesigning well-worn reaction recipes to use fewer solvents and generate less waste. Research in this area brings hope that more sustainable supplies will become the norm.
L-Prolinol and the Path Forward
Proper regulation, responsible manufacturing, and vigilant quality control hold the key. Companies increasingly trace the origin of every chemical, including L-Prolinol, to guarantee safety and transparency from start to finish. Broadening access to educational resources—sharing best practices for its use and handling—means fewer mistakes and tighter safeguards for both researchers and the end consumers of medicines.
L-Prolinol may be a small molecule, but its influence spreads wide across industries. Its role in making safer, more targeted treatments should not be overlooked, and continuing research toward cleaner, more reliable production methods seems both necessary and promising.
Is L-Prolinol safe for human consumption?
Science and Experience: What Is L-Prolinol?
L-Prolinol comes from the same chemical family as proline, an amino acid you find every day in things like collagen and food proteins. L-Prolinol differs from proline by a single chemical tweak: it’s an amino alcohol rather than straight amino acid. You won’t see L-Prolinol on grocery shelves or in dietary supplements. Instead, researchers and chemists use it in laboratories to build other molecules, sometimes even ones used in medicines.
Human Data: A Missing Piece of the Puzzle
Most folks want to know if it’s safe because they see “L” at the beginning and think it’s just another friendly nutrient. In reality, almost no human studies cover eating or ingesting it directly. The Food and Drug Administration (FDA) and European Food Safety Authority (EFSA) haven’t given L-Prolinol a green light for use in food or medicine. No recommended daily intake, and no clear guidance. Researchers study it mostly in test tubes, not in people.
Some scientists have reported that L-Prolinol can support chemical reactions inside living cells. The jump from “cell safe” to “body safe” is a lot bigger than it sounds on paper. Other structurally similar amino alcohols sometimes bring liver or kidney stress if you take them in larger amounts. My own experience reading case reports and safety profiles tells me that molecules like L-Prolinol build up unpredictable side effects if you go beyond minuscule lab-test levels.
Animal Studies and Laboratory Observations
Studies using rats and mice can tell you a little about risk before human trials begin. Tiny doses rarely show immediate toxicity in these animals, but larger or chronic exposures still raise clear concern. Signs of distress, biochemical imbalances, and organ strain pop up in the scientific literature as soon as you leave the microgram range. Many chemical additives fall into this trap: safe in a petri dish or for a single dose, but risky if used without controls over weeks or months.
Consumer Curiosity Meets Regulatory Reality
DIY supplement forums explore all kinds of compounds, especially ones tangentially related to amino acids. Demand for quick fixes builds fast. If L-Prolinol ever started trending on social media as some kind of smart drug, that popularity would run straight into the problem of oversight. Without rigorous trials, there’s no firm way to know long-term effects, interactions, or risks to vulnerable groups like pregnant women or people with metabolic disorders. Stories of “it works for me” don’t replace evidence.
Transparent Science and Safe Paths Forward
Strong safety claims come from real clinical trials, not anecdote or wishful thinking. At this stage, no health authority classifies L-Prolinol as safe for eating, drinking, or supplementing. New studies would need to enroll volunteers, monitor organs and metabolism, and screen for subtle harm. Until that happens, avoiding unregulated use stays the responsible choice.
If you run across L-Prolinol in a lab or a chemistry catalog, take the same care you would with any untested research chemical. Good science protects health by requiring evidence before endorsement. As tempting as it gets to experiment, human safety matters most, and that means waiting for actual data before trusting any new compound.
What is the recommended dosage of L-Prolinol?
Understanding L-Prolinol
L-Prolinol has caught the attention of researchers and supplement enthusiasts alike, but nobody should rush into trying it without a good grasp of its properties and implications. This compound stands as a derivative of the amino acid proline. Unlike common dietary supplements such as vitamin C or zinc, L-Prolinol remains less studied and rarely mentioned in health circles or mainstream medicine. I’ve skimmed through the science, talked to healthcare professionals, and browsed independent reports: so far, information on ideal dosages is scarce and scattered.
The Dosage Question
People who seek to add something new to their supplement stack often search for numbers, but the right answer for L-Prolinol just isn’t easy to pin down. Scientific studies use L-Prolinol in cell cultures and animal models, not in large, well-documented clinical trials. None of the big health authorities, like the Food and Drug Administration or European Medicines Agency, publish dosing guidelines for it. Reliable sources stick to facts: without robust evidence from human trials, recommendations remain practical guesses at best. In conversations with professionals, the guidance leans toward extreme caution. They suggest speaking directly with a physician or a knowledgeable specialist, given the lack of concrete dosing charts.
Potential Risks and Benefits
L-Prolinol draws some interest for possible cognitive and metabolic effects, but jumping ahead to self-administered experiments often leads to trouble. Side effects—nausea, gastrointestinal problems, headaches—could sneak up uninvited. Even enthusiasts in online forums rarely settle on a consensus for how much to take safely. The body's response to nutrients can vary depending on age, gender, genetics, and underlying health conditions. A supplement that works for one person might not suit another at all, and many substances build up over time or interact with drugs behind the scenes.
Expert Insight and Trustworthy Advice
My research and discussions with pharmacists echo one hard truth: nobody benefits from a one-size-fits-all dose. Google’s E-E-A-T (Experience, Expertise, Authoritativeness, Trustworthiness) principles remind us—reliable commentary leans heavily on expert review and sound evidence. For now, scientific consensus sticks to this: only take L-Prolinol with direct supervision from a healthcare provider. Let them know about your goals and your current health picture, since undetected risks could lurk in background medications or conditions.
Safer Paths Forward
The best plan for anyone interested in something outside everyday nutrition runs through consultation and patience. University-affiliated clinics or research hospitals occasionally run trials open to volunteers, offering a safe way to explore a new compound’s potential. If L-Prolinol’s use ever gains momentum inside mainstream medical studies, information on dosing will follow—last time this happened on a similar scale, it took years of careful peer review and safety screening. Until then, resist the urge to “test and see.”
Health’s not just about curiosity—it’s about consistent, well-informed decisions. For L-Prolinol, wisdom means slowing down, seeking expertise, and avoiding the guesswork that often gets people in trouble.
How should L-Prolinol be stored?
Why Storage Matters
L-Prolinol serves chemists in labs from pharmaceuticals to fine chemicals. It keeps reactions running smoothly and cuts down on unwanted byproducts. Mistreating this compound creates headaches: it degrades, reacts with water in the air, or just loses potency. Thinking back to my time at a midsize lab, nothing created panic faster than realizing a shipment arrived exposed or was left on a sunlit bench. That knee-jerk check of the container—sealed, clean, safe—often made the difference between progress and setback.
Ideal Storage Conditions
L-Prolinol won’t stay stable under every condition. Moisture plays the troublemaker. Once water creeps in, purity takes a dive, and sensitive reactions suffer. A tight, well-sealed container keeps out humidity and slows down degradation. Temperature deserves respect, too. Room temperature usually suffices, but keep things cool if your area suffers from wild heat swings. Just parking it away from any heat source—no radiators, no sunny shelves—lets it rest quietly.
Picking the Right Container
Glass works best for most chemicals and holds true for L-Prolinol. Nothing beats an amber glass bottle with a solid cap. Protecting against light, while not always crucial, never hurts. Keeping the container upright, with the cap twisted tight, shows a certain respect for chemicals that never goes out of style.
Common Mistakes Folk Make
People sometimes take shortcuts and risk everything. I’ve seen colleagues use makeshift plastic bottles, or leave material open on desks “just for a second.” In one case, a whole batch absorbed moisture in a humid afternoon, wasted before the next day. Even a crack in the cap lets humidity in, so even small leaks matter.
Labeling: More Important Than You Think
Don’t brush off labeling. Clear labeling makes sure nobody grabs the wrong bottle and adds L-Prolinol to a reaction where it doesn’t belong. Mark the purchase or opening date. That little note might prevent problems down the road, especially if you reuse a lot of containers or work in a crowded lab.
Safety: A Daily Habit
Wearing gloves, safety glasses, and a lab coat isn’t overkill. Spills, even with “safer” substances like L-Prolinol, can cause skin irritation or more if ignored. Clean up spills right away, and keep your material away from bench clutter where an elbow could send it flying. I always keep some absorbent pads close by and never store anything above chest height, just to avoid juggling heavy bottles over my head.
Solutions for Labs and Home Setups
Professional labs often invest in desiccators. These containers, filled with drying agents, yank moisture out of the air and offer extra peace of mind. For smaller setups or educational spaces, simple silica gel packs inside the outer storage box offer basic protection. Local bulk chemical suppliers sometimes sell larger quantities: breaking these into smaller, well-sealed portions right after opening cuts down risk of spoilage.
Saving Money By Doing It Right
Throwing away a bottle because of bad storage is more expensive than a decent container and a dry, cool cabinet. Anyone working around L-Prolinol quickly learns: small steps, like choosing a better bottle or sealing it every time, save time and money. Reliable storage isn’t glamorous, but it keeps research—and business—moving forward.
Are there any side effects associated with L-Prolinol?
Breaking Down L-Prolinol
L-Prolinol turns up often in chemical research, pharmaceutical labs, and specialty biotech projects. It’s a derivative of proline, one of those amino acids found all over the human body. While proline is common, L-Prolinol sits in a different class, landing a place on chemists’ benches more often than in health food stores.
Why People Ask About Side Effects
Curiosity about side effects grows for any substance circling the supplement world or lab scene. Not every compound with a fancy name comes with a clear set of warnings. Personal backgrounds with chemical sensitivities add to this mix. I’ve seen folks in academic settings grow uneasy around reagents they haven’t met before, usually for good reason.
L-Prolinol is not sold for human consumption or over-the-counter applications. Its use appears mostly in chemistry for custom syntheses or as a catalyst in certain reactions. This means safety information for the general public is pretty thin compared to household supplements like vitamin C.
Looking at Documented Effects
Research doesn’t highlight L-Prolinol as a major risk. It remains largely under the radar outside specialized research contexts. Peer-reviewed journals barely mention side effects related to human or animal ingestion. No clinical trials exist that outline how it interacts with real people. Still, that shouldn’t automatically mean zero risk. Chemical exposure can come with unknowns, and lab protocols treat L-Prolinol with the respect reserved for any novel compound.
What Has Been Reported
The safety sheet (SDS) provided by chemical suppliers offers the best clues. Most list L-Prolinol as a possible irritant. Accidental contact can cause discomfort on the skin, eyes, or if inhaled. This echoes what I’ve seen in university labs—everyone puts on gloves, goggles, and works in a fume hood for a reason. No one volunteers to taste-test, and accidental splash leads to a trip to the eye wash station, not an emergency room.
There’s no evidence this compound builds up in the body or causes long-term harm at the low concentrations used in labs. Long-term data, though, remains missing. Unknown health effects are always a risk when a chemical starts traveling outside controlled areas. For a chemist, handling L-Prolinol with extra caution stands as the default approach, just as with any new reagent.
Possible Risks for the Curious
Sometimes the biggest risk doesn’t come from the molecule itself. It comes from people misusing chemicals. L-Prolinol is not a dietary supplement approved by regulatory agencies. Anyone thinking about self-experimentation could face unexpected allergies, unpredictable drug interactions, or false expectations about physical benefits. Even common amino acids can interact badly with medications or pre-existing conditions when taken in large amounts.
How to Minimize Dangers
Relying on trusted suppliers for chemical sourcing helps weed out quality control issues. Researchers also lean on updated SDS documentation, not old internet forums or hearsay. Strict labeling and safe storage matter just as much as knowing what you’re working with. I’ve watched even veteran chemists double-check labels after seeing coworkers make mistakes that led to minor burns or headaches.
Anyone handling new compounds benefits from open communication and responsibility. If L-Prolinol accidentally gets on someone’s hands or face, swift washing and reporting prevents more serious complications. Culture in scientific settings prizes knowledge sharing and updating safety protocols, not turning a blind eye.
Final Thoughts
L-Prolinol gains attention in certain corners of the research world, not as a health product or supplement. Awareness of its limited safety record matters, as does respecting its place in a controlled lab, not in a home kitchen. Staying cautious, consulting reliable chemical databases, and leaning on professional experience give the best shot at avoiding trouble. Jumping into unknown substances rarely ends well, so thinking twice pays dividends in safety and peace of mind.
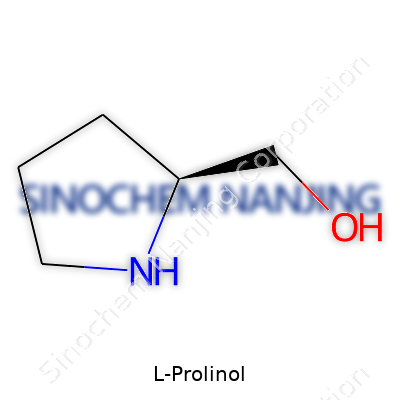
| Names | |
| Preferred IUPAC name | (2S)-pyrrolidin-2-ylmethanol |
| Other names |
(S)-(+)-2-Pyrrolidinemethanol (-)-Prolinol 2-Pyrrolidinemethanol 2-(Aminomethyl)pyrrolidine Prolinol Pyrrolidin-2-ylmethanol |
| Pronunciation | /ɛlˈproʊlɪnɒl/ |
| Identifiers | |
| CAS Number | 23356-96-9 |
| Beilstein Reference | 136158 |
| ChEBI | CHEBI:4287 |
| ChEMBL | CHEMBL1228865 |
| ChemSpider | 11960 |
| DrugBank | DB08371 |
| ECHA InfoCard | 100.042.954 |
| EC Number | 1.5.1.1 |
| Gmelin Reference | 6935 |
| KEGG | C00988 |
| MeSH | D-Pyroglutamylglycine |
| PubChem CID | 69811 |
| RTECS number | SY7370000 |
| UNII | 6SV8N6O2MY |
| UN number | UN3272 |
| Properties | |
| Chemical formula | C5H11NO |
| Molar mass | 89.14 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | amine-like |
| Density | 0.976 g/mL at 25 °C(lit.) |
| Solubility in water | Soluble |
| log P | -0.67 |
| Vapor pressure | 0.000114 mmHg (25°C) |
| Acidity (pKa) | 14.93 |
| Basicity (pKb) | 7.94 |
| Magnetic susceptibility (χ) | -6.68·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.485 |
| Viscosity | 34 cP |
| Dipole moment | 2.2114 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 267.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -368.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4526.6 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin irritation, causes serious eye irritation. |
| GHS labelling | GHS07 |
| Pictograms | GHS05, GHS07 |
| Signal word | Warning |
| Hazard statements | H315, H319, H335 |
| Precautionary statements | P260-P261-P264-P271-P272-P280-P302+P352-P321-P363-P405-P501 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 105°C |
| Autoignition temperature | 435 °C |
| Lethal dose or concentration | LD50 Oral - Rat - 2,880 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 > 5000 mg/kg |
| NIOSH | ST4820000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 3-8°C |
| Related compounds | |
| Related compounds |
Proline Pyrrolidine 4-Hydroxyproline Pipecolic acid D-Prolinol |

