L-Mandelic Acid: The Evolution of a Key Chemical Player
Historical Development
Interest in L-Mandelic acid stretches back to the 19th century, when chemists isolated it during research on bitter almonds. Using crude extraction and crystallization, early researchers could only obtain small amounts. This alpha-hydroxy acid's chiral nature grabbed the attention of the pioneers in stereochemistry and organic synthesis. Later, the pharmaceutical industry adopted it as synthetic techniques advanced, transforming L-Mandelic acid from a rare chemical curiosity into a substance found in many laboratories and manufacturing plants around the world. This evolution followed the demand for effective antibacterial agents, safer chemical peels, and versatile intermediates in drug production. The decades since have seen steady innovation, marked by the rise of enantioselective catalysis and microbial fermentation methods that push both production efficiency and purity to new heights.
Product Overview
L-Mandelic acid commonly appears as a white, crystalline powder, dissolving well in water and ethanol. Those handling the material find a powder with no strong odor, easy to store under standard conditions. In commercial settings, purity often tops 98 percent, which proves adequate for research, cosmetic formulations, or pharmaceutical synthesis. Because of its natural chiral purity, industries value L-Mandelic acid over racemic forms when results depend on stereochemistry, especially in certain drug syntheses or in dermatology, where side reactions can prove unpredictable. Its source can be fully synthetic or produced from renewable feedstocks by microbial or enzymatic routes, with the method chosen based on cost structure and downstream requirements.
Physical & Chemical Properties
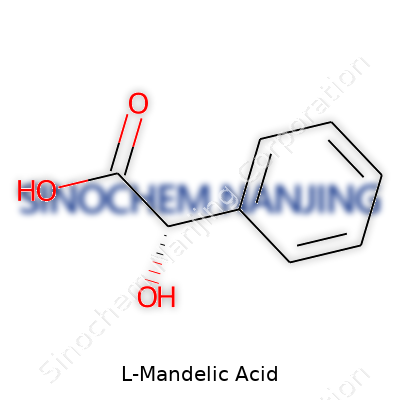
L-Mandelic acid’s chemical formula is C8H8O3, revealing a core of a phenyl group attached to a hydroxyacetic acid side chain. The melting point hovers around 119°C to 122°C. As a weak acid, its pKa measures near 3.85, letting it serve as a gentle yet effective exfoliant in skincare products, since it slowly releases protons compared to stronger acids. As for solubility, natural acids often disappoint, but L-Mandelic acid dissolves at many concentrations, opening up possibilities for both aqueous and alcoholic solutions. Hydrophilic tendencies let formulators use it in liquids, gels, or even crystalline solids without constraints usually seen with other aromatic hydroxy acids.
Technical Specifications & Labeling
Every lot of L-Mandelic acid sold to industry and research labs typically comes with a certificate of analysis. Such documentation reports purity—usually over 98 percent—and lists trace impurity levels, heavy metals, optical rotation, and moisture content. Labels display not just basic identity, but also batch numbers, shelf-life data, CAS number (611-72-3), and safety guidelines that highlight its classification as a mild irritant. Supply chains rely on this rigorous reporting, both for regulatory reasons and because product consistency matters in fine chemistry and pharmaceutical synthesis. Some suppliers also provide certifications for compliance with REACH or other chemical safety frameworks, depending on the end destination.
Preparation Method
Manufacturers started with chemical synthesis using benzaldehyde. These processes once used cyanide intermediates, raising numerous safety and environmental challenges. Developments in biocatalysis and enzyme engineering have given producers cleaner, more sustainable routes, often based on engineered microbial strains or isolated enzymes that convert precursors with impressive selectivity toward the L-isomer. Yields often approach 90 percent or more, with reduced byproduct formation. While the chemistry sounds abstract on paper, operators in production facilities deal with real-world challenges—reaction times, pH stability, purification through crystallization and filtration, waste treatment, and maintaining the right reaction temperature to avoid oxidation. Downstream, quality control teams use high-performance liquid chromatography (HPLC) and chiral capillary electrophoresis to rule out contamination or unwanted stereoisomers.
Chemical Reactions & Modifications
L-Mandelic acid offers a range of chemical transformations thanks to its phenyl ring and reactive alpha-hydroxy carboxylic acid group. In pharmaceutical chemistry, teams use it as a resolving agent to separate racemic amines. This compound reacts with bases to make salts valuable in chiral separations or as chemical building blocks. As a substrate, it undergoes esterification, amidation, and can even serve as a precursor in the total synthesis of more complex molecules. For cosmetic R&D, its gentle exfoliating properties stand out, and simple pH adjustments tailor it for skin formulations. Researchers also use it to probe enzyme specificity or screen for new catalysts; its chirality makes it a standard in studies about enantioselective synthesis or asymmetric catalysis.
Synonyms & Product Names
The technical literature refers to L-Mandelic acid by several names, including (S)-2-Hydroxy-2-phenylacetic acid, (S)-Mandelic acid, and Mandelic acid (L-form). Product catalogs might feature code designations like Mandelate or its enantiomeric identifier. Synonyms serve a practical purpose across supply chains, where detailed paperwork uses systematic nomenclature, but researchers and formulators might pick a shorthand. Consistency in naming throughout ordering, handling, and application reduces mistakes and keeps projects running smoothly.
Safety & Operational Standards
In any facility that moves kilogram quantities of L-Mandelic acid, staff use gloves and safety goggles. Inhalation hazards run low, but the dust can still cause mild irritation. Good laboratory practice means working with adequate ventilation and clean workspaces. In cosmetics, concentrations stay well below levels that risk systemic toxicity, but restricted use in sensitive applications like injection reflects a cautious approach. Material safety data sheets review both acute and chronic risks, most of which stem from the acid’s mild corrosiveness and skin/eye irritation potential. Tight control over storage—dry, cool, airtight containers—means product quality stays high for months. Periodic staff training supports recognition of spill risks or off-spec materials, and disposal protocols favor environmentally safe routes consistent with regional regulations.
Application Area
L-Mandelic acid appears in a surprising number of industries for a compound with such a simple structure. Dermatology values it for its gentle exfoliation, helping treat acne and brighten skin without triggering the sharp irritation associated with glycolic or lactic acids. In pharmaceuticals, its ability to resolve racemic mixtures opens access to pure drug enantiomers. Some clinics combine it with antibiotics, using its antimicrobial properties in topical formulations. Research scientists treat it as a benchmark for studies on enzyme specificity, chiral catalysis, and biochemical pathways. Water treatment companies and specialty chemical suppliers also tap its mild chelating effect. Each field tailors its use to the acid's specific strengths—low irritation, strong chiral selectivity, and broad solubility.
Research & Development
R&D labs keep pushing L-Mandelic acid to new uses, from nanoparticle synthesis through to investigating new delivery systems for skincare. Several research groups target more efficient bioprocesses that use renewable carbon sources, cutting costs and the environmental footprint at the same time. Scientists keep testing structural analogs or changing protective groups to tune reactivity and solubility further, hoping to unlock new drug candidates or more stable formulations. The acid's unique place in studying chirality also makes it a fixture in chemical education and foundational research. Most work shares the same goal—finding ways to improve efficiency, push purity, or broaden the range of practical applications in medicine, industry, and environmental science.
Toxicity Research
Toxicologists have tested L-Mandelic acid both in vitro and in animal models. Most data points to low acute toxicity, with rats showing high LD50 figures when administered orally. Skin patch tests reveal only mild irritation even at concentrations higher than typically used in skincare products. Long-term exposure studies raise no red flags; reports of sensitization remain rare. That said, regulatory agencies treat all alpha-hydroxy acids with care because of observed photosensitivity and irritation in human trials. As a result, approved usage concentrations in consumer products stay below thresholds known to trigger reactions, safeguarding vulnerable populations.
Future Prospects
L-Mandelic acid holds strong promise as green chemistry shifts to the mainstream. Biocatalysis keeps advancing, which means coming years could greatly increase the availability of high-purity, renewable L-isomer. In the medical field, advances in drug delivery and enantioselective synthesis will keep L-Mandelic acid relevant for specialty drug pipelines. The cosmetic sector shows rising demand for mild, effective ingredients that deliver real results without harsh side effects, and L-Mandelic acid's profile lines up perfectly. Public awareness around safe skincare and reduced irritation keeps pushing formulators to revisit established ingredients. Academic groups keep the acid under the spotlight as a tool for teaching chemistry fundamentals, asymmetric synthesis, and the practical challenges of industrial scale-up. If future developments stay on trend, L-Mandelic acid should keep attracting R&D investment and commercial innovation across a growing range of technical and consumer-focused industries.
What are the benefits of using L-Mandelic Acid?
A Gentle Touch With Serious Results
L-Mandelic acid has picked up plenty of fans in the skincare community, but its benefits don’t just float around on hype. Dermatologists and skin therapists have taken to recommending it for both oily and sensitive skin types. One of the biggest reasons is how its larger molecule size gives a slower, more even penetration. That means less irritation than you get with many other acids. Having tried various acids on my own sensitive skin—everything from glycolic to lactic—L-mandelic acid didn’t give me that burning, itchy feeling that kicks in with harsher formulas. It’s one of the few exfoliants I can use regularly, even during winter, without worrying about flakiness or redness.
Acne and Hyperpigmentation: Two Birds, One Acid
People struggling with breakouts and dark spots often land in the dermatologist’s office with a mix of frustration and skepticism. Here’s where science meets visible change: published research has shown that mandelic acid helps break down the “glue” holding dead skin cells together. By keeping pores clear, it reduces congestion that leads to acne. I’ve seen improvements using L-mandelic acid-based serums, especially in stubborn areas around the nose and chin, which tend to flare up most often.
Also, L-mandelic acid goes deeper than daily surface cleansing. Its exfoliating action targets uneven tone and dark marks left after breakouts. Melanin (skin pigment) doesn’t move out of the skin quickly on its own, especially for folks with medium or deep complexions. Mandelic acid helps fade these spots in a steady, visible way. You won’t get overnight miracles, but after a month of using it a few times a week, the improvement can change how you feel about your skin.
Fighting the Signs of Aging Without Wreaking Havoc
Most anti-aging products promise smoother skin, but then leave you red. My biggest frustration with retinol and some of the stronger acids has been that they bring on peeling and sore skin. L-mandelic acid boosts collagen and skin cell turnover at a steady pace. You get fewer fine lines and a brighter look, and you’re far less likely to end up with aggravated or peeling skin. People with rosacea-prone or reactive skin don’t always have safe anti-aging options. Mandelic acid levels the playing field.
Safe for a Range of Skin Tones and Types
Skin experts recommend patch testing for everything, but mandelic acid stands out for its track record on darker skin. Some acids (especially glycolic) can trigger postinflammatory hyperpigmentation. Mandelic acid’s slower absorption lowers that risk. This acid lets more people see exfoliation’s benefits without worrying about new marks.
Easy to Layer, Easy to Use
Using multiple skincare products at once can feel like a chemistry project. L-mandelic blends well with simple routines because it doesn’t tend to clash with hydrators or niacinamide. I’ll use a light, leave-on mandelic serum in the evening, followed by a moisturizer. The next day, my skin still feels smooth, even if I only rinsed with water in the morning. Unlike some exfoliants, you don’t have to choose between clear pores and comfort.
Practicality and Cautions
Start gently. Using too much too often can still lead to dryness. If you avoid any step, make sure it isn’t daily sun protection—any exfoliation can make skin more sun-sensitive, and that’s where mistakes turn into damage. Look for formulas that make their concentration clear, and remember: your skin knows when it’s being pushed. Stick to two or three times weekly unless a dermatologist tells you otherwise. With some patience and consistency, L-mandelic acid can become a mainstay in keeping skin clear, strong, and even-toned.
How do I use L-Mandelic Acid in my skincare routine?
The Place for L-Mandelic Acid
L-Mandelic acid isn’t just another bottle on the bathroom shelf. With roots in both acne treatments and gentle chemical exfoliation, it stands apart from stronger acids like glycolic or salicylic. Its molecule is bigger and moves a bit slower on the skin, helping to prevent irritation. This point matters plenty for people with sensitive skin or for those who’ve been burned – sometimes literally – by overzealous use of other exfoliants.
Why People Use L-Mandelic Acid
This alpha hydroxy acid has become popular among dermatology circles as a more forgiving choice for fading dark spots, evening out skin tone, and reducing acne breakouts. There’s solid research showing it fights hyperpigmentation and gently loosens dead skin. Plus, because it has mild antibacterial properties, it works well in routines tackling both aging and acne.
I remember testing retinol too young, hoping for magazine-gloss skin, only to wind up stinging and peeling for days. L-Mandelic doesn’t torch your face like that; with thoughtful use, the skin feels smoother and looks brighter without the drama.
Building It Into Your Routine
Consistency and patience make or break chemical exfoliants. I add L-Mandelic acid in the evening. I wash with a gentle cleanser, dry thoroughly, and then swipe on a few drops or a pea-sized amount, depending on the formula. I wait around 10 minutes; this gives the acid a real shot at loosening bond-hardened dead skin. No fancy tools, just clean hands and a habit of reading the label twice.
I avoid overdoing it. A few times per week is plenty for most people. Signs of overuse—flaky patches, burning, tightness—tell you to slow down. Mixing it with other strong acids or retinoids can make trouble, so I keep those on opposite nights. For anyone layering vitamin C in the morning, sticking to basics at night becomes more important.
People Who Benefit Most
Those dealing with hormonal breakouts, mild rosacea, or ongoing pigmentation see real value from L-Mandelic when other acids fall short or sting too much. People with darker skin often turn to it too, since the risk of post-inflammatory marks runs lower than with some harsher exfoliants.
Skin isn’t a science experiment or a place for shortcuts. Dermatologists like Dr. Elyse Love recommend always following up with moisturizer and broad-spectrum sunscreen, day in and day out. Moisture locks in the benefits without stripping, and sunscreen prevents dark marks from getting worse. In my circle, using both takes discipline—but on tough skin days, you notice the payoff.
Pitfalls and Solutions
Tingling usually happens at first. If it sticks around or burns, it’s time to dial down or stop. It makes sense to patch-test every new acid on a clean patch of arm skin before going all in. For those unsure, a quick call or visit to a board-certified dermatologist saves a lot of guesswork.
Drugstore aisles and online shops now carry L-Mandelic in serums, toners, and cleansers. Reliable brands publicize pH and percentage, which matters for results. Look for concentrations between 5% and 10% for regular at-home use. Higher strengths belong in clinics.
Getting Results Without Damage
Success with L-Mandelic acid comes from respect—respect for your skin’s limits and for the science behind slow, consistent exfoliation. The goal isn’t starting over; it’s helping healthy skin along, night by night. I’ve learned that clearer, calmer skin rarely shows up overnight, but it does come with tools like this one, used wisely.
Is L-Mandelic Acid suitable for sensitive skin?
Personal Experience with Sensitive Skin
Growing up, I always looked forward to trying a new skincare product, only to end up with redness, itching, or dry patches. My skin reacted to everything—retinol, glycolic acid, and even gentle cleansers. Frustration became the norm, and I stuck with basic moisturizer for years out of fear that chemical exfoliants would cause more harm than good. For anyone living with skin that gets angry at the slightest shift in routine, curiosity about so-called “gentle” acids is completely understandable.
What Sets L-Mandelic Acid Apart
L-Mandelic acid belongs to the alpha hydroxy acid family—like glycolic and lactic acid—but features a larger molecular size. Dermatology research points to this as a core reason why it penetrates the skin more slowly. Because of this slower absorption, many users notice less stinging and irritation compared to glycolic acid. It helps break down dead skin and brightens overall tone, and it’s often recommended for those with acne-prone skin as well as sensitivity.
The American Academy of Dermatology highlights that slower-absorbing AHAs can sidestep the harshness some experience with other exfoliants. Clinical studies back up the claim: in one study published by the Journal of Cosmetic Dermatology, participants using mandelic acid reported reduced redness and flakiness after four weeks, compared to those on glycolic acid. Doctors see it as a safer bet for anyone worried about burning or excessive dryness.
Why Sensitive Skin Is Tricky
Someone with sensitive skin doesn’t just have thin or dry skin. Genetics, allergies, compromised barriers, or past overuse of harsh products are all pieces of the puzzle. Stripping away oil, even with something meant to help, leaves skin exposed and more reactive. I’ve learned that no product—no matter what the label says—works as a miracle fix for everyone.
Fragrance, high alcohol content, and essential oils in products still pose risks. Even mandelic acid, promoted as milder, can cause discomfort if the formulation includes irritating extras. A patch test on a small area, waiting 24 hours, and then tracking changes closely gives better protection from breakouts or hypersensitivity than any promise on a package ever could.
Looking at Real Solutions
Dermatologists I’ve spoken with recommend taking it slow. Instead of jumping all in, they suggest starting with a lower concentration—around 5%—and using it a couple of times per week. They also point to layering serum after cleansing at night, and always following up with a simple moisturizer that skips fragrance and dyes.
Checking ingredient lists remains key. Some formulas combine L-mandelic acid with calming agents like niacinamide or squalane. These ingredients can further soothe and support the skin barrier. Avoiding combinations with other exfoliating acids, retinols, or harsh cleansers in the same routine prevents overload, a lesson I had to learn the hard way.
Responsible Use Makes a Difference
Sensitive skin doesn’t mean zero exfoliation forever. With awareness and a healthy dose of skepticism about “miracle” claims, even those of us who react to everything can add something new. L-mandelic acid sits alongside lactic acid as a gentler option, but patience and consistent observation make the biggest impact. Listening to how skin responds—tightness, stinging, smoothness, or sudden redness—teaches more than any quick fix ever can.
What are the possible side effects of L-Mandelic Acid?
Skin Care’s New Darling, But Not Always Gentle
L-Mandelic acid’s been making the rounds as a gentle exfoliant for folks with sensitive or acne-prone skin. It feels good to have something that promises smoother, brighter skin without the sting some acids bring. But like anything you put on your face, L-mandelic acid comes with its own set of risks. Understanding possible side effects means fewer surprises and better skin health down the road.
Common Side Effects: No Acid is Truly “Mild” for Everyone
L-mandelic acid is derived from almonds. The molecules are larger than those found in glycolic or lactic acids, so they tend to sink in more slowly. That slow and steady approach helps people with sensitive skin, but it doesn’t mean irritation never happens. Redness, dryness, mild stinging—these aren’t rare. My first time using a mandelic acid serum, my cheeks flushed and tingled uncomfortably, even though that product had “for sensitive skin” on the label.
Some see peeling or flakiness within days. Skin may look a bit parched or feel tight. Dry patches can get worse for those already dealing with eczema or rosacea. Sun sensitivity is another biggie. Acid use leaves skin vulnerable. Without regular sunscreen, there’s a bigger risk for redness, sunburn, and long-term pigment changes.
Rare But Serious Reactions
Most folks will only see mild effects. On the rare side, some develop hives, swelling, or painful inflamed areas. Allergic reactions are possible, especially for people with a known nut allergy. Though L-mandelic acid gets made in a lab and contains only the isolated acid, cross-contamination or individual immune responses can kick in. Anyone who swells or itches after use should wipe off the product and seek advice from a healthcare provider.
Overuse: The Downside of Chasing Smooth Skin
The temptation to layer chemical exfoliants can catch up with you. Using mandelic acid with other alpha hydroxy acids, retinoids, or scrubs leads to over-exfoliation. In my years working alongside skincare specialists, I’ve often seen folks strip away their skin’s barrier this way. Signs include raw patches, cracking, burning, or breakouts that look like a rash. At this point, skin loses its ability to protect against germs and environmental stress.
Prevention and Care: Start Low, Go Slow
A little patience pays off. Starting with low percentages (many products begin at 5–10%), using only on clean, dry skin, and waiting before adding moisturizers can cut down the risk of side effects. For folks brand new to chemical exfoliation, it often helps to apply once or twice a week before ramping up. Hydration is key—think fragrance-free moisturizers that restore what acids take away. Sunscreen ends up being non-negotiable. Even on cloudy days, that extra barrier helps prevent discoloration and all sorts of lingering damage.
Watch for Hidden Irritants
Ingredient lists matter. Some products sneak in alcohol or fragrance that stings. Check for simple formulas: L-mandelic acid, water, and gentle supports like sodium hyaluronate or glycerin do the job well without added risk. If you’re taking any prescription acne treatments, or using other acids, give your skin time off between applications. Dermatologists can help build a safe routine for tricky skin types or anyone not sure where to start.
The Bottom Line: Stay Informed, Listen to Your Skin
Research and common sense help dodge unnecessary skin troubles. Public sources like the American Academy of Dermatology and peer-reviewed journals give solid, evidence-backed advice. Listening to your own body matters just as much. Tingling, redness, or even that feeling that your usual moisturizer burns are all signs to pause and rethink your routine. Healthy skin reflects daily choices—slow progress, plenty of protection, and willingness to adjust when things don’t feel right.
How often should I apply L-Mandelic Acid?
Getting Real About L-Mandelic Acid and Your Skin
L-mandelic acid stands out as an alpha hydroxy acid (AHA) with a mild touch. It pulls in people who want smoother, clearer skin but feel wary of irritation. Those who deal with acne, hyperpigmentation, or uneven texture see this as a gentle alternative to classic exfoliants. So many want a perfect calendar: “Is every day too much? Once a week not enough?” The answer sits somewhere in between, and daily reality plays a huge part.
Why Frequency Matters
Every face tells a different story. I’ve tried a dozen exfoliating acids. Some stung or brought on a peel, while others seemed to do nothing at all. Mandelic acid joined my routine because of its larger molecule size, meaning it soaks in more slowly and doesn’t rush inflammation. Science backs that up: research from dermatology clinics shows people tolerate mandelic acid better than glycolic or lactic for sensitive or darker skin tones, often with less peeling and burning. Even so, gentle doesn’t mean harmless. Overdoing it can strip the skin’s natural barrier, for anyone.
Setting Up Your Routine
If you’re new to L-mandelic acid, start small. Most dermatologists suggest starting at two to three times a week. Plenty of consumers feel the urge to push for faster results by layering on more, but patience keeps the skin in top form for the long haul. After a few weeks without redness or dryness, you can go to every other day. For some, building up to daily use feels fine. But stop if your skin looks red, dry, or tight. From personal experience—using mandelic acid nightly right away led to weeks of flaking and stinging. Slowing down made a world of difference.
Look at other products in your cabinet. Mixing too many exfoliants (like retinoids or other AHAs) can spark irritation. If you’re using prescription acne meds, chat with your doctor before starting any acid at all. Keeping exfoliating acids on different nights protects the skin barrier and keeps things calm.
Supporting Healthy Use
No polish can fix skin overnight. Mandelic acid supports gradual improvement. Apply at night, after gentle cleansing and before moisturizer. Using a sunscreen every day matters even more. Alpha hydroxy acids like mandelic leave skin prone to sun damage for up to a week after use. I learned that lesson quickly: skipping sunscreen after an exfoliating routine left me with dark spots that lasted months. It's far easier to prevent that damage than to fix it later.
What To Watch For
If you're dealing with active breakouts or trying to fade dark marks, consistency wins over intensity. Using L-mandelic acid regularly, even at lower frequency, often brings better results than harsh treatments that leave skin raw. Listen to the signals your skin gives you—tightness, stinging or flaking means scale back. For questions on combining products, especially if you’re seeing a dermatologist for acne, ask for guidance tailored to your situation.
Quality matters, too. Choose serums or toners with clearly marked percentages (many products land between 5% and 10%) and brands that share their ingredient lists openly. Trusted companies like The Ordinary, Face Reality, or Allies of Skin have made mandelic acid accessible, which helps consumers make safe, informed picks.
Future Paths
Developing healthy skin isn't about finding the most potent acid or trending serum—it’s about building a routine you can keep up with. L-mandelic acid gives a softer route toward healthy, glowing skin. Take all signals seriously, go slow, and respect your skin’s unique story.
| Names | |
| Preferred IUPAC name | (2R)-2-hydroxy-2-phenylacetic acid |
| Other names |
(R)-(+)-Mandelic acid L-(+)-Mandelic acid L-2-Hydroxy-2-phenylacetic acid L-α-Hydroxyphenylacetic acid |
| Pronunciation | /ɛl mænˈdɛlɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 611-72-3 |
| Beilstein Reference | 1365074 |
| ChEBI | CHEBI:36538 |
| ChEMBL | CHEMBL50455 |
| ChemSpider | 65153 |
| DrugBank | DB04284 |
| ECHA InfoCard | 100.013.861 |
| EC Number | EC 200-193-6 |
| Gmelin Reference | 71562 |
| KEGG | C00811 |
| MeSH | D009351 |
| PubChem CID | 69951 |
| RTECS number | PB7610000 |
| UNII | Q81N96F97T |
| UN number | UN2811 |
| CompTox Dashboard (EPA) | DTXSID7034368 |
| Properties | |
| Chemical formula | C8H8O3 |
| Molar mass | 152.15 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.30 g/cm³ |
| Solubility in water | slightly soluble |
| log P | -0.62 |
| Vapor pressure | 0.000108 mmHg at 25°C |
| Acidity (pKa) | 3.85 |
| Basicity (pKb) | pKb: 13.18 |
| Magnetic susceptibility (χ) | -8.0e-6 cm³/mol |
| Refractive index (nD) | 1.541 |
| Viscosity | 33 mPa·s (25 °C) |
| Dipole moment | 2.72 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 186.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -841.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -843.5 kJ/mol |
| Pharmacology | |
| ATC code | D01AE22 |
| Hazards | |
| Main hazards | Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS07, GHS05 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H315: Causes skin irritation. H319: Causes serious eye irritation. |
| Precautionary statements | Precautionary statements: "P264, P280, P305+P351+P338, P337+P313 |
| Flash point | 110 °C |
| Autoignition temperature | 185 °C |
| Lethal dose or concentration | LD50 Oral - rat - 4,000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral Rat 3400 mg/kg |
| NIOSH | SNK7300000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 4% |
| Related compounds | |
| Related compounds |
Benzoic acid Mandelonitrile Phenylglyoxylic acid Phenylacetic acid Lactic acid |

