L-Malic Acid—More Than a Sour Note in Modern Industry
Historical Development
Few ingredients move quietly from orchard to lab with as much adaptability as L-malic acid. Its discovery traces back to apples and rhubarb—a telltale sign is its tartness—which French chemist Carl Wilhelm Scheele separated in the late 1700s. From natural extraction in the early days to today’s fermentation-based manufacturing, the journey of L-malic acid reflects the progress of the food and pharmaceutical industries over generations. Research on the acid advanced quickly in the twentieth century, spanning biochemistry, agricultural engineering, and medicine. With modern-scale fermentation using specifically bred microbial strains, makers capably boost yields, lower costs, and reduce contaminants. Historic shifts from fruit-based sources to bioengineered processes open discussions about sustainability and food authenticity. In my years as a food technologist, whenever someone mentions L-malic acid, I picture that balance—nature modified by science, but still built on a core of tart simplicity.
Product Overview
L-malic acid often appears as a crystalline white powder, sometimes in granular or microgranular form, depending on its intended use. Brands might call it E296 in Europe, or simply Malic Acid in ingredient panels, offering a familiar handle for those reading product labels. Supermarkets, pharmacies, and agriculture suppliers all recognize its presence. This consistency makes it a go-to additive across numerous goods, including beverages, candy, baked goods, animal feed, and health supplements. The purity requirements shift by sector, with the food and beverage market usually seeking higher grades than technical applications. The additive lines up with consumer trends for recognizable, “from nature” acids, even when produced with advanced fermentation.
Physical & Chemical Properties
On paper, L-malic acid’s structure seems simple: it holds a slightly higher melting point than citric acid, dissolves easily in water, and plays well with most sugars and sweeteners. In my lab days, technicians would note its sharp, persistent tartness in sensory tests. That long, sour finish matters more than pure sourness—bakers and winemakers both recognize how it stands up through processing and storage. L-malic acid’s chirality—its L- and D- forms—further separates natural from synthetic, as only L-malic exists in apples and most fruit, while D- and DL- forms usually signal petrochemical or synthetic origins. This distinction matters for natural labeling and dietary requirements.
Technical Specifications & Labeling
How regulators and brands label L-malic acid depends on purity, country of origin, and the application. High grade batches often exceed 99% purity on dry basis, tested using titration, HPLC, or spectroscopic analysis. Product labeling reflects the form—monohydrate or anhydrous—and regulatory codes, such as E296 in the EU or INS 296 in the Codex Alimentarius. In my experience, the debates at food safety symposia rarely center on malic acid itself; the details that spark concern usually come from residual impurities, allergens from fermentation feedstocks, or country-of-origin issues. For dietary supplements, proper designation of source and isomer matters more than regulatory code.
Preparation Method
Most of the world’s supply comes from the aerobic fermentation of glucose using strains of Aspergillus or Rhizopus, often genetically tweaked for higher output and enantiomeric purity. Old extraction methods—squeezing apple juice, then concentrating and precipitating—no longer meet commercial demand. The modern bioprocess starts with corn or sugar beet feedstocks, channeled into large-scale fermenters, under tight pH and oxygen controls. After fermentation, a suite of filtration and crystallization steps cleans and concentrates the product. Each stage impacts environmental footprint, cost, and local sourcing. Some producers still rely on chemical synthesis, which involves double hydration of maleic anhydride, but the food market gravitates to microbial or fruit-derived supplies for “clean label” needs.
Chemical Reactions & Modifications
L-malic acid’s chemical action punches above its molecular weight, serving as both an acidulant and a chelating agent. As winemakers know, malic acid in grape must can turn to lactic acid in fermentation, thanks to malolactic bacteria—dropping harsh acids and smoothing flavor. Its reactivity suits pH adjustment, buffering, and sequestration of metal ions in food, cosmetic, and cleaning formulas alike. In pharmaceutical work, L-malic acid’s salt-forming capacity helps modify solubility and absorption profiles of various actives. Under high heat, it degrades to fumaric acid or succinic acid, reshuffling its carbon backbone—a feature harnessed in some bioprocessing and food flavor development projects.
Synonyms & Product Names
Most chemists and food operators call it L-malic acid, but paperwork also lists synonyms like apple acid, hydroxybutanedioic acid, or simply E296. Ingredient lists sometimes use these terms interchangeably, but the L- prefix signals the desired isomer. Beverage makers smile when spotting it on sour candy wrappers, while animal feed formulators recognize both the acid and its salts in pellet labels.
Safety & Operational Standards
Global food safety authorities tightly regulate L-malic acid, with maximum usage levels for specific product categories. The Joint FAO/WHO Expert Committee on Food Additives lists it as a generally recognized as safe (GRAS) substance. Manufacturers adopt hazard analysis and risk management plans, with allergen reviews for fermentation feedstocks and cross-contamination checks along the way. Handling guidelines mirror those of other food acids—wear gloves and eye protection, keep storage sealed against moisture, and avoid prolonged inhalation of dust. The acid’s relatively low toxicity profile reassures operators, though those with chemical sensitivities or unique metabolic needs might report rare reactions.
Application Area
Food scientists rely on L-malic acid for the punch it adds to beverages and sweets—the “green apple” note and enduring acidity that make some drinks memorable. Bakers use it to balance dough fermentation, control yeast growth, and boost shelf life by dropping pH naturally. Nutraceutical brands tap its background in cellular energy pathways, using it alongside magnesium or in sports supplements. In oral care, mouthwashes and toothpaste include malic acid to brighten flavor and encourage saliva flow. On the farm, animal nutritionists blend it into piglet feed for palatability and to fight pathogens. The acid’s chelating skills show up in cleaning products, cosmetics, and even industrial water treatment.
Research & Development
Scientists continue to push L-malic acid production into greener territory: studies focus on using waste streams for fermentation feedstock, genetically optimizing microbes, and designing water-lean purification steps. In product formulation, research looks to combine malic acid with other organic acids for better antimicrobial action, improved taste curves, and lower cost. Pharmaceutical developers study new salt forms for enhanced solubility and stability. Some biochemists hope to boost yields with CRISPR-driven fermentation strains, trimming both energy use and process contaminants.
Toxicity Research
L-malic acid has a solid safety profile under standard usage guidelines. Toxicity studies in animals and humans over decades haven’t flagged significant health issues at dietary levels. The acid passes quickly through the citric acid cycle in human metabolism, ending up as CO₂ and water. That said, concentrated forms can cause mouth and stomach irritation, and excessive consumption—as seen with high-sour candies and drinks—can trigger digestive discomfort. Research continues into long-term exposure and any impacts for vulnerable populations, but modern risk assessments reaffirm the low hazard rating.
Future Prospects
L-malic acid finds itself at the center of several converging trends—natural flavor demand, “clean label” expectations, and the search for sustainable bioprocesses. Consumers continue to drive requests for recognizable, nature-based ingredients, and that old apple-tart acid fits the bill better than many of its synthetic cousins. The biotech push, with CRISPR and next-gen microbial factories, aims to drop environmental impact and open up resource cycles, possibly putting L-malic front and center as a model of circular chemistry. Food preservation, functional supplements, and specialty cleaning chemistries also represent growth sectors. In the years I spent connecting ingredient suppliers to product developers, it always struck me how a centuries-old compound can open new doors as technologies shift and consumer tastes evolve.
What is L-Malic Acid and what is it used for?
What L-Malic Acid Means for Food and Health
L-Malic acid shows up in almost every tart fruit you’ve tasted, especially green apples and cherries. Nature puts this compound right in the heart of ripening, helping shape the sharp, clean flavor in so many fresh foods. It doesn’t stop at orchard fruit, either. L-Malic acid gives a backbone to taste in tomato sauces, pickled vegetables, and drinks that really pop.
In the food world, L-malic acid turns plain into punchy. I remember the difference the first time I compared my homemade apple jam with and without it: one was flat, the other had a spark that lingered. Its ability to make flavors sing is real, and it keeps sour candies and “just-pressed” juices tasting pure. Sodas and electrolyte drinks also rely on it—not only for taste but for balancing out sweeteners and masking aftertastes. People can tell when it’s missing; there’s no zing, and the overall flavor turns muddy or just plain boring.
L-Malic Acid in Our Bodies
L-Malic acid isn’t just a flavor boost. Our cells use it every day. On a molecular level, this acid helps produce energy in our muscles. The “creak” after climbing a hill or finishing a long run partly comes from lactic acid’s build-up, but malic acid helps your muscles shift out of that state and keep going. That’s not just academic theory; athletes and people recovering from fatigue talk about supplements with malic acid for exactly this reason.
Doctors have looked at it for years. Some research suggests people with fibromyalgia may feel a difference combining malic acid with magnesium supplements. The science still needs a lot more evidence before you’ll hear any official medical endorsement, but the interest exists for a reason. Our body naturally produces malic acid, but in some circumstances, extra intake could nudge the system in a good direction.
Everyday Uses Beyond Your Plate
Grape harvests offer a unique view into how L-malic acid shapes things beyond the grocery aisle. Winemakers rely on it to judge when to bring in their fruit. If a wine tastes sharp, it’s likely because the malic acid hasn’t mellowed yet. Some winemakers use specific bacteria to convert it into lactic acid, softening flavors and adding complexity. The same goes for craft cider and even kombucha—people experiment with malic acid levels to reach a perfect balance between tartness and drinkability.
Household cleaners often include malic acid, too. It dissolves mineral deposits and breaks down stains that most soaps just skim over. Its mildness works well for sensitive skin in personal care products, from shampoos to toothpaste, bridging the gap between effectiveness and comfort.
Pushing for Safer, Smarter Choices
None of this works without careful oversight. Regulatory bodies like the FDA and EFSA regularly check safety data before letting L-malic acid near food or supplements. The global market runs on trust—so it really matters that sourcing stays ethical and manufacturing stays honest. Some brands lean on plant-derived malic acid, reassuring folks who want natural ingredients, while industrial versions allow for stable pricing and broad accessibility.
With more shoppers turning every bottle of juice or jar of jam around to read the label, understanding what goes in—and why—matters. L-malic acid isn’t a scary chemical with a name nobody can pronounce. It’s a molecule that helps both food and bodies work better, one tart flavor or energy boost at a time.
Is L-Malic Acid safe for consumption?
Understanding L-Malic Acid
L-Malic acid shows up in the ingredients list of lots of foods. Most apples, cherries, and even grapes contain it naturally. Food manufacturers add it to sour candies, juice, and other snacks to boost tartness and flavor. It’s easy to see why some folks wonder if this mysterious “acid” deserves a spot in your regular diet.
Safety Backed by Science
I remember once double-checking the ingredients in a sour candy my kid loved. L-Malic acid stood out, sending me into research mode. I learned that it’s more than a lab ingredient. Our bodies actually make malic acid every day as part of natural energy production.
The United States Food and Drug Administration added L-Malic acid to its list of safe food additives, and Europe also approves it as E296. Scientists reviewed studies where people and animals consumed reasonable levels, reporting very few adverse effects.
Doses in Food Stay Well Below Harmful Levels
Looking close at commercially processed products, typical doses reach just enough to enhance taste. Take a look at some sour candies, for instance — maybe 2 or 3 grams per package. Even eating those occasionally keeps total intake low.
Consuming huge amounts of any food additive – including L-Malic acid – could cause digestive issues like stomach cramps or diarrhea. Our bodies can only process so much. But those effects show up only with much higher doses than found in normal servings.
Digestive Process Mirrors Fruit Consumption
Every time you bite into a Granny Smith apple, you’re tasting L-Malic acid — and your body breaks it down using the same pathways as the version added to food. That connection between natural sources and food-grade additives helps clear up worries about weird chemicals. In moderate amounts, your liver takes care of the extra acid by converting it into energy or flushing it out.
People with Health Conditions Should Take Extra Care
People living with kidney disorders, or those who need to follow strict dietary restrictions, probably know to double-check anything new in their diet. Talking to a medical professional before adding food with higher acid levels is wise, simply because their bodies handle waste in a different way.
For most healthy adults and kids, the science and years of safe use point toward minimal risk from eating foods with L-Malic acid. Experts on nutrition keep their eye on new studies, and so should anyone who needs to avoid acids or has allergies.
Better Transparency in Food Labeling Helps
Folks deserve to know what goes into the food on their table. Good labeling helps people make smart choices. Food companies could provide more detail: explain whether malic acid comes from natural fermentation or synthetic sources, and specify actual amounts. The more we know, the easier it gets to decide what fits in a healthy diet.
Finding Balance in Eating Habits
Seeing “L-Malic acid” on a food label shouldn’t cause panic. Stressing about every additive can easily become overwhelming. Using facts from trusted health organizations and talking to nutritionists or doctors makes the path clearer. Focusing on variety — more fresh fruits and vegetables, less processed candy — puts worries about naturally occurring food acids in perspective.
What are the health benefits of L-Malic Acid?
Getting to Know L-Malic Acid
L-Malic acid shows up naturally in fruits like apples and grapes. It’s the stuff that gives you that tart bite in your mouth. Not much attention gets paid to it outside food science, but people miss out on what it brings to the table for daily health. I grew up with a backyard full of apple trees and didn’t appreciate at the time that I was eating a compound scientists say supports core functions in the body.
Energy That Lasts Through the Day
People often ask where steady energy can come from—real energy, not caffeine jitters. L-Malic acid takes part in the Krebs cycle, which powers up every cell. It helps the body turn carbs, fats, and proteins into ATP, the main fuel source for cells. Having enough of this acid in your food can set you up for less midday dragging and that all-too-common crash. Research published by the journal “Nutrients” points out how L-Malic acid gives mitochondrial function a boost, which promotes stamina and fewer aches during workouts.
Muscle Recovery: Not Just for Athletes
I used to dismiss cramps and soreness as part of getting old. I learned that athletes often add L-Malic acid to their routine because it helps reduce muscle fatigue from lactic acid build-up. A clinical study from Spain showed that supplementing with it can lower perceptions of tiredness during resistance training. This carries over to anyone physically active, including gardeners or warehouse workers. Pairing it with magnesium should bring more noticeable relief, according to research in the “Journal of Pain Research.”
Liver Health and Detox Support
The liver works overtime dealing with toxins and heavy food. L-Malic acid steps in as a natural chelator, helping remove heavy metals and other waste. Some reports describe how healthcare practitioners use apple juice before procedures like gallbladder flushes for its high L-Malic acid content. Even if someone’s not diving into detox trends, getting more of this nutrient may lighten the burden on the liver and kidneys, especially for those exposed to pollution and processed foods.
Oral and Skin Benefits
Acids get a bad rap for dental erosion, but L-Malic acid tells a different story. It spurs saliva production, which keeps the mouth clean and breaks up plaque. Dental journals highlight how chewing gum with this acid can help those with dry mouth and reduce bad breath. On the skin, manufacturers use it in creams and exfoliants for gentler turnover of old skin cells. That’s the science behind “fruit acid facials.” The acid’s mild nature makes it safer for sensitive types compared to harsher exfoliants.
Natural Sources Offer Real Advantages
Anyone can grab a supplement bottle, but whole foods carry the benefits with extra nutrients. Biting into a crisp apple or reaching for cherries delivers L-Malic acid, along with fiber, vitamin C, and other antioxidants. The tartness signals that health boost, especially for folks who want improvements in energy or muscle recovery without harsh additives. Individual needs vary, so checking with a healthcare provider ensures no conflict with current medications or health conditions.
Moving Toward Better Health Choices
People crave simple steps toward feeling better, not just cures for illness. Including more L-Malic acid-rich foods and learning about its role lets people get ahead of problems like fatigue, soreness, or sluggish digestion. Nutrition doesn’t have to be mysterious, and sometimes eating what grows on a tree outside does more than any supplement aisle promise. Eating with purpose and understanding helps every cell work better, so the impact shows up in daily life, not just lab reports.
How should L-Malic Acid be stored?
Looking After a Reliable Ingredient
L-Malic acid shows up everywhere, from tart apples and tangy fruit drinks to granola bars and sports supplements. Every box, bag, or barrel of this sour ingredient looks pretty simple on the outside, but proper storage tells the difference between a potent product and a sticky lump of disappointment. My experience in food labs and ingredient warehouses taught me that careless storage can ruin an entire lot—a waste both financially and in lost production time.
Real Dangers of Moisture and Clumping
L-Malic acid draws water like a sponge. Open a bag and leave it on a shelf, and soon you’ll face a mess. Moisture doesn’t just turn a bag of powder into rock-hard chunks; it sets off chemical changes, especially when acid granules get sticky or damp. The sour bite can fade, and the off-taste creeps in. Reputable suppliers flag this ingredient as hygroscopic, meaning it grabs water from the air at every chance. Sealing the package after use really does matter, and tossing in a silica gel packet helps a lot—an old but reliable trick from chemical storerooms everywhere.
Cool Temperatures Keep It Potent
Heat speeds up the breakdown of L-malic acid, even before water gets involved. Warehouses without air conditioning or even racks that sit next to a sun-baked window can gradually cause the product to break down. Keeping it in a dry, shaded place at room temperature or lower protects both taste and shelf life. In manufacturing, I saw pallets set too close to radiators or heat vents; that decision often ruined texture and flavor. Once, a summer heatwave cut the shelf life of three months’ supply, leading to a headache of recalls and a revised, tighter protocol for every shipment we handled after that.
Keeping It Away From Metals and Strong Odors
Pure acids, even food-grade, play rough with certain metals. Leaky containers touching iron shelves or exposed to metal shavings usually show sign of rust or a strange taste months down the line. High-quality storage uses plastic bins or polyethylene-lined drums. Don’t store L-malic acid next to solvents, paint, or pungent chemicals—acids absorb odors easily, and bad smells transfer to the finished food or supplement. I once received grape powder that reeked faintly of cleaning solution, traced back to a shared shelf in a tiny warehouse.
Safe Labeling and Stock Rotation Reduce Loss
Foods and supplements always face recalls because someone misread the storage date or pulled the wrong lot. Label every container clearly with both arrival and expiration dates. Using the “first in, first out” method sounds basic, but slapping a bright sticker or mark on every shipment that comes through keeps mistakes from happening. This approach cuts costs and reduces waste. It’s satisfying seeing a storeroom where nothing lingers forgotten in the back: just a clean row of containers, all legibly marked, easy to access.
Final Tips from the Field
Experience always trumps theory in ingredient storage. Zip the inner liners closed tight after each use. Use the acid quickly after opening rather than letting it sit. Train staff to spot powder that’s turned clumpy or smells off. Set up storage areas free from high heat, moisture, and bad air. Treating L-malic acid with respect keeps every batch of food or drink tasting right and safe for everyone down the line.
Is L-Malic Acid suitable for vegetarians and vegans?
Looking Deeper into L-Malic Acid’s Origin
L-Malic acid pops up everywhere—juices, candy, bakery treats, that sour tang in apples and cherries. As an additive, it helps food taste fresher and last longer. The real question is whether it fits into vegetarian and vegan lifestyles. I remember standing in the grocery aisle with a friend who checks each label meticulously, always asking about ingredients like these. It’s not paranoia, just wanting clear answers about what lands on the plate.
How L-Malic Acid Gets Made
This acid starts its life in fruit and veggies by nature’s design. Commercial food production takes a shortcut, creating it through synthetic processes. One common method involves fermenting sugars with select bacteria or fungi. Another uses chemical synthesis from petrochemical feedstocks. In most cases—based on food manufacturing data—no animal products play a role. This means synthetic L-malic acid, which you’re most likely to encounter, comes from plants or pure chemistry rather than meat, milk, or eggs.
Animal Byproducts: Rare, but Always a Possibility
It’s easy to assume every “plant-derived” additive is safe ground, though sometimes processes can sneak animal residues into the mix. Some old fermentation methods once used animal-based nutrients to grow bacteria or fungi, but these have faded as companies lean toward allergen-free and cruelty-free labels. Today, producers choose corn, sugar beets, or even apple pulp—ingredients vegetarians and vegans have always enjoyed.
Still, food laws in different countries aren’t uniform. In a few rare cases, fermentation might run on nutrients from milk or gelatin. Certification matters here. Labels like “vegan” or a green “V” badge often signal the manufacturer checked every step for animal involvement. For vegans chasing purity, brands that show their process and ingredient sources stay ahead of the pack.
How to Check If L-Malic Acid Respects Dietary Choices
Reading food labels isn’t just a habit, it’s a skill. I always tell folks: look for signs such as “suitable for vegans” or recognized vegan certifications. If a package stays quiet, it helps to contact the maker directly. Reputable brands won’t shy away from sharing ingredient sources. Even food scientists urge customers to reach out, since transparency builds long-term trust.
Some supplement makers include L-malic acid in capsules or powders. It’s smart to check not only the acid but also the capsule shell or any “natural flavors” attached. Gelatin capsules are a no-go for plant-based diets. Thankfully, many companies have shifted to cellulose-based alternatives. It’s not just what’s inside the jar, but what the jar itself is made of.
Cutting Through the Confusion
I’ve met parents, athletes, and home cooks—everyone tries to line up their food choices with their ethics. L-malic acid rarely causes problems for vegetarians and vegans in most modern markets. Still, honest labeling and reliable sourcing make a world of difference. The industry has the tools to keep animal products out of the mix. Public demand keeps that process moving. Knowledge, a quick look at the label, and a direct line to the manufacturer keep dietary slip-ups to a minimum.
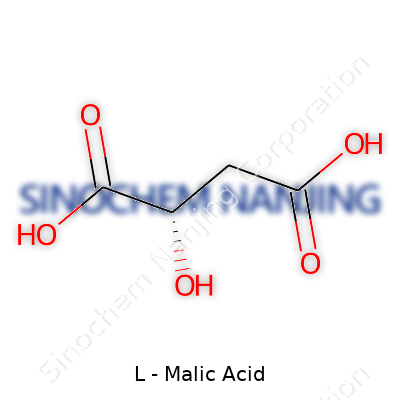
| Names | |
| Preferred IUPAC name | (2S)-2-hydroxybutanedioic acid |
| Other names |
apple acid levo-malic acid hydroxybutanedioic acid malic acid (l-form) |
| Pronunciation | /ˈɛl ˈmælɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 97-67-6 |
| Beilstein Reference | 1721812 |
| ChEBI | CHEBI:17896 |
| ChEMBL | CHEMBL1231462 |
| ChemSpider | 887 |
| DrugBank | DB01308 |
| ECHA InfoCard | 03bb9c3c-cce5-402b-9ea0-3b6da5e4fd01 |
| EC Number | EC 200-293-6 |
| Gmelin Reference | 82137 |
| KEGG | C00149 |
| MeSH | D008287 |
| PubChem CID | 525 |
| RTECS number | OM5250000 |
| UNII | 817L1N4CKP |
| UN number | UN1789 |
| Properties | |
| Chemical formula | C4H6O5 |
| Molar mass | 134.09 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.601 g/cm³ |
| Solubility in water | Miscible |
| log P | -1.26 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 3.40 |
| Basicity (pKb) | pKb: 12.47 |
| Magnetic susceptibility (χ) | -48.2·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.554 |
| Dipole moment | 2.70 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 157.4 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -991.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1343.5 kJ/mol |
| Pharmacology | |
| ATC code | A16AA02 |
| Hazards | |
| Main hazards | Irritating to eyes, respiratory system, and skin |
| GHS labelling | GHS07, Exclamation mark |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H319: Causes serious eye irritation. |
| Precautionary statements | P264, P270, P280, P301+P312, P330, P501 |
| Flash point | 130°C |
| Autoignition temperature | 220°C |
| Lethal dose or concentration | LD50 Oral Rat 1600 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50: 3200 mg/kg |
| NIOSH | SN4325000 |
| PEL (Permissible) | 50 mg/m³ |
| REL (Recommended) | 400 mg |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Fumaric acid Succinic acid L-Aspartic acid Tartaric acid Maleic acid |

