N-Carbamoyl-L-Glutamic Acid: A Deep Dive
Historical Pathways: How N-Carbamoyl-L-Glutamic Acid Emerged
Between classic biochemistry labs and modern industry, N-Carbamoyl-L-Glutamic Acid found its role through a mix of curiosity and problem-solving. Researchers in the twentieth century saw potential in manipulating amino acids for practical benefits, especially in animal nutrition and rare metabolic disorders. Early experiments focused on upping arginine synthesis, a key function for urea cycle management in mammals. Over time, its application in feed additives and support for metabolic therapy grew, driven by gaps left by standard treatments for inherited disorders like N-acetylglutamate synthase deficiency.
A Closer Look at What N-Carbamoyl-L-Glutamic Acid Is
Most chemists look at N-Carbamoyl-L-Glutamic Acid and see a white, crystalline powder that brings more than stability to the table. This compound tweaks the biochemistry of the animals and humans who use it, especially when arginine production can't keep up with needs. In comparison to pure glutamic acid, the carbamoyl group makes it more than a dietary building block; it actively participates in metabolic rescue. Products end up tightly regulated, carrying detailed labeling about content, composition, and intended use, especially given the compound's place in the worlds of medicine and animal agriculture.
Physical Nature and Chemical Behavior
The compound typically delivers itself as a solid, melting only at higher temperatures which matters for storage and transport. It dissolves best in water, favoring use in pharmaceutical and feed mixtures. Structurally, the carbamoyl and glutamic acid parts work together to form a molecule both robust and ready for metabolic kickstarting. Handling it, one notices how its slight odor contrasts with the neutral appearance, a trait shared by many amino acid derivatives. On the chemical side, its formula C6H10N2O5 and molar mass around 190.16 g/mol frame every analysis you’re likely to run, whether it’s purity testing or formulation.
Technical Specs, Labeling, and Compliance
Manufacturers detail purity, particle size, and moisture content on spec sheets. From my time seeing regulatory reviews, I know that deviations mean batches face rejection. Labels don’t just list the name; they must spell out the concentration, source, and precise use instructions. Customer-facing documentation references international standards, since regulators like the FDA or EFSA analyze each claim. Certificates of analysis are another staple, lining up with batch numbers and guaranteeing that the promised quality matches what’s inside each container.
How It’s Made: The Preparation Process
Industrial synthesis favors methods that scale well without sacrificing purity. Most production lines kick off with L-glutamic acid and use a carbamoylating agent under watery, often alkaline, conditions. Temperature and pH stay tightly controlled, or you’ll end up with unwanted byproducts. Filtration, washing, and drying round out the steps before assessing the finished powder for contaminants. Workers in production suites know how even tiny pH swings can tilt yields, so there’s no room for improvisation. Recrystallization frequently steps in to push purity past ninety-nine percent, crucial for pharmaceutical markets.
Chemical Reactions and How Scientists Tweak the Molecule
N-Carbamoyl-L-Glutamic Acid doesn’t just sit still; it transforms in labs and factories. Exposure to strong acids or bases can hydrolyze the carbamoyl group, curbing its activity. Under controlled lab settings, those same reactions let chemists modify or tag the compound for research. Sometimes derivatives or analogues improve absorption, as researchers look for ways to extend benefits or limit side effects. Enzymatic reactions, especially those mimicking animal metabolism, keep revealing how this molecule interacts in real biological contexts, not just isolated test tubes.
Different Names and Marketplace Synonyms
Databases and product catalogs attach a few names to N-Carbamoyl-L-Glutamic Acid. You’ll encounter abbreviations like NCGA, or synonyms such as Carbamoylglutamate and Carbaglumic Acid, the latter showing up most often in pharmaceutical discussions. Trade names stick to straightforward translations, since regulatory frameworks leave little room for creative branding in this segment. In scientific papers, a blend of systematic and popular names often crops up, making careful reading crucial when combing through literature or regulatory filings.
Following Safety and Operation Guidelines
Factories set their own strict protocols when handling N-Carbamoyl-L-Glutamic Acid. Material safety data sheets stress gloves, masks, and dust masks because powders can sneak into the air. The risk profile looks mild compared to some chemicals, but the push for safety never takes a break. Operators train for proper spill management, waste disposal, and emergency steps. International organizations like the World Health Organization and domestic regulators keep updating standards, asking for fresh toxicity data before issuing safety endorsements. In labs, locked cabinets and clear labeling keep research samples out of unauthorized hands—something you learn to appreciate after a night audit.
Where It Matters: Key Application Areas
By far, animal nutrition and rare human metabolic disorders lead the application charts. Feed manufacturers boost pig and poultry diets to improve growth and prevent urea cycle disruptions. Pediatric hospitals rely on it for enzyme deficiencies where arginine won’t do the job. The compound supports kidney health and, in some pilot studies, helps with heart function under stress. Beyond medical and agricultural boundaries, scientists experiment with using it in fermentation and biotechnology, pushing new boundaries on how basic biochemistry supports complex systems.
Pushing Science Further: Ongoing Research and Innovation
Research never stops in the world of amino acid analogues. Academic teams and corporate engineers track how N-Carbamoyl-L-Glutamic Acid interacts with other nutrients, which enzymes speed or slow its metabolism, and how those reactions translate into better outcomes. Genome editing, precision nutrition, and targeted therapies rely on robust data, and recent clinical trials collect patient responses in ever more detail. Animal studies keep revealing connections between this molecule and gut health, immunity, and stress response in livestock. Researchers share findings at conferences where disagreements spark progress and help pin down how much, and for whom, supplementation truly makes a difference.
Unpacking Toxicity Research: Staying on the Safe Side
Toxicological studies back up every product release. Human and animal testing track doses, looking for metabolic disruptions, allergic reactions, and long-term effects. Even with favorable safety profiles, oversight committees notice every adverse event, no matter how rare. Detailed follow-ups and clinical monitoring feed into public databases, giving manufacturers and end users a clearer risk picture. Research teams also probe potential interactions with pharmaceuticals, vitamins, and minerals since metabolic pathways overlap and surprises hide in the details. Real-world monitoring, especially in children and livestock, keeps tightening guidelines for safe upper limits.
Looking to the Horizon: What the Future Holds
The evolution of N-Carbamoyl-L-Glutamic Acid is far from finished. New formulation techniques aim for improved absorption and lower dosing. Marketplace trends favor custom blends for species, breeds, or individual patients, reflecting a move toward precision nutrition and medicine. As regulatory frameworks face new data and applications, experts expect expanded use in diseases currently considered untreatable with standard amino acid therapies. Sustainability pressures drive research into greener production pathways, including biotechnological synthesis. Deep learning and genomics unlock more precise markers for use, while clinical and field data continue to fine-tune who benefits most and how to measure real impact.
What is N-Carbamoyl-L-Glutamic Acid used for?
A Look at a Little-Known Feed Additive
In livestock farming, animal diets often miss some of the critical building blocks needed for proper growth or production. One such building block is L-arginine, an amino acid that helps animals grow, reproduce, and deal with metabolic stress. Here’s where N-Carbamoyl-L-Glutamic Acid steps in.
How Feeding Efficiency Gets a Boost
N-Carbamoyl-L-Glutamic Acid, sometimes called NCG, isn’t a household name. In large livestock operations—think pigs, poultry, cattle—NCG is used as a feed additive. It doesn’t work as a direct nutrient for animals, but rather helps make L-arginine in the animal’s body. In newborn piglets or ruminants under stress, their own natural production of L-arginine doesn’t keep up with what their body asks for. Farmers, veterinarians, and animal nutritionists have found that feeding diets with NCG helps these animals build more protein, use nitrogen better, and gain weight more reliably.
Feed isn’t cheap. Anything that improves how much body mass an animal puts on per kilo of feed matters to the bottom line. I’ve talked with farmers who have seen improved litter size in sows and stronger early growth in calves simply from balanced arginine nutrition. NCG fits here by gently stepping up the animals’ arginine supply, without big swings in diet cost or other complicated changes.
Science Behind the Practice
A growing body of research backs up these field results. Studies published in journals such as Animal Nutrition and the Journal of Dairy Science have demonstrated that NCG improves nitrogen retention, which reduces the waste excreted into the environment. Piglets have shown higher daily weight gain and lower rates of diarrhea. In dairy cows, milk yield and reproductive health have both shown positive responses after feed formulas included NCG during challenging periods, like early lactation.
Animal scientists have focused on the environmental impact as well. NCG helps reduce the flow of unused nitrogen out of barns and into the watershed. For areas where nitrate pollution is a threat, small steps like adjusting feed can add up to major changes for local waterways. Less nitrogen in manure means fewer nitrates reaching rivers and groundwater—a win for communities living near farms.
Cutting Down Medicinal Use in Livestock
Veterinarians sometimes use NCG to support animals with inborn errors in metabolism—rare cases, such as certain urea cycle disorders, where an animal’s body can’t handle ammonia properly. Administering NCG helps keep ammonia at safe levels, which prevents crises in these animals. It replaces or reduces the need for expensive, hard-to-source drugs.
Better Outcomes, Fewer Chemicals
From what I’ve learned talking with producers, the appeal of NCG lies in its ability to improve animal health and growth naturally, without antibiotics or hormones. In many countries, farm managers feel pressure to cut down on unnecessary medications and focus on nutrition and management instead. NCG fits this movement—it works quietly but effectively, fitting alongside vitamins and minerals as part of a modern approach to animal care.
Finding the Right Balance
Animal diets need to adapt as science advances. If NCG supplements allow for healthier animals, better growth, and less strain on the environment, then it makes sense to consider them more widely. It’s always important to work with evidence, trial results, and veterinary guidance before changing feed plans. With continued research and transparent use, tools like NCG can play a valuable role in shaping the future of food production for both animals and people.
Is N-Carbamoyl-L-Glutamic Acid safe for human consumption?
Understanding What’s on Your Plate
Plenty of food ingredients have names that wouldn’t look out of place on a lab report. N-Carbamoyl-L-glutamic acid is one of them. Before it shows up in food products, people deserve a chance to know what it does and how it behaves in the body. Transparency matters, not just because it builds trust, but because it has real health consequences. When you walk down a grocery aisle, peace of mind is easier when you know the science lines up with safety.
What Is N-Carbamoyl-L-Glutamic Acid?
This compound is related to glutamic acid—a familiar amino acid found in meats, cheese, and even tomatoes. N-Carbamoyl-L-glutamic acid plays an important role in the urea cycle, a critical pathway in human biology that helps the body process and eliminate excess nitrogen. In medicine, doctors use it under the name carglumic acid to treat rare disorders where ammonia builds up in the blood. In other contexts, food chemists see its potential as a dietary supplement or additive to tweak flavor or nutrition profiles.
Evidence for Human Safety
The leap from clinical use to food ingredient gets complicated. Regulatory agencies—including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency—have looked at carglumic acid for prescription use. These reviews focus on rare metabolic disorders, so doses are much higher and given with a doctor’s supervision. Reports on side effects show reactions like gastrointestinal upset or changes in liver enzymes at these medical doses. In the hands of a doctor, side effects can be monitored and addressed.
No large-scale studies track the long-term effects of N-Carbamoyl-L-glutamic acid in healthy people. That’s a gap worth thinking about. The compound’s lineage as a non-protein amino acid closely related to substances already in a typical diet points to a low risk of trouble in small amounts, especially compared to industrial chemicals or heavy metals. Its degradation in the body also gives confidence: the pathways involved have been mapped out and don’t build up unwanted byproducts when functioning normally. But absence of harm at pharmaceutical doses doesn't always mean complete safety at population scale.
Why It Matters
Food safety isn't just about ‘does it poison you in a single dose?’ People have to consider allergies, unknown sensitivities, and the potential impact of consistent low-level exposure over years. Too often, novel food additives make their way into products before exhaustive reviews. Real-world experience tells me to check sources, trace the trail of approvals, and keep an eye out for new data. Most food products have a regulatory lag, so what’s permitted now might get reassessed down the line.
Regulators, scientists, and public watchdogs all play a role in filling research gaps. The best approach builds in follow-up studies, encourages transparency on ingredient labels, and urges independent review by third parties. If you’re worried about unknowns, supporting labeling laws and advocating for public access to safety data moves the needle more than waiting on public agencies alone.
Looking Forward
Right now, N-Carbamoyl-L-glutamic acid looks fairly low-risk in controlled doses, especially compared to plenty of unregulated supplements. Still, long-term data from the general population stays thin. Before calling it completely safe for everyday consumption, demands for more studies and open disclosure give everyone a chance to make informed choices at the dinner table. Food should be about both enjoyment and trust—and trust grows best when science stays out in the open.
What are the storage requirements for N-Carbamoyl-L-Glutamic Acid?
Getting Storage Right for Lab Safety and Consistency
The world of amino acid derivatives can seem technical, but keeping things real matters most when safety and results depend on careful storage. N-Carbamoyl-L-Glutamic Acid is one of those compounds that show up in lab protocols and manufacturing processes, and mistakenly treating it like off-the-shelf table salt can end up ruining samples or risking safety. Over the years, I’ve learned how even minor mishandling chips away at reliability in research or production.
Keep It Cool and Dry, Always
Heat never treats this compound kindly. I’ve seen a whole batch degrade after a summer power outage left a storeroom warm for just a day. The material straight-up discolored and failed the next lot test. Official documentation from chemical suppliers and research journals almost always calls for temperatures at or under 25°C. Think of a cool cupboard or climate-controlled storage area, not a bench in direct sunlight or anywhere the air-conditioning kicks off overnight.
Humidity plays just as big a role. This compound pulls water from the air, which often leads to clumping and, even worse, chemical breakdown. I always stash it with desiccants, double-sealed, in a moisture-tight container. There’s science behind this, too. Moisture speeds up the formation of degradation products, wiping out purity levels. Labs with tight quality standards never store it open, not even for an afternoon. We’ve lost precious reagents to stale air far more quickly than you’d expect.
Protect from Light and Air
Repeated exposure to sunlight or strong artificial lights can drive slow changes in some amino acid derivatives, including N-Carbamoyl-L-Glutamic Acid. I learned early in my career that transparent glass bottles don’t cut it. Dark, amber vials add that layer of protection. This isn't about being paranoid — it’s about cutting out unnecessary risks. Oxygen in the air gradually impacts shelf life, so I make sure the cap seals tight every time. For longer storage, we use vacuum-sealed or nitrogen-flushed vials, especially if the supply line looks uncertain.
Label Thoroughly and Track Dates
No matter how careful the storage, mistakes happen when labels fade or rub off. Every container gets the date received, date opened, and the supplier’s batch number. I wish I’d followed this rule every time — once, I watched a team scramble to trace a bad product lot because the key date wiped off from condensation. Tracking shelf-life isn’t about bureaucracy; it actually saves money and protects projects. Pharmaceutical standards recommend routine checks on stored reagents, and this habit fits right into that.
Don’t Mix Up Food or Drug-Use Grades
Sometimes people ask if bulk-purchased, industrial-grade material suits clinical research or food production. There’s a huge difference in storage and handling to avoid cross-contamination. Using food-grade containers, segregated spaces, and strict cross-checks guards against mistakes that can trigger audits or, worse, recalls. From my own experience, blending grades can lead to heat- and moisture-related loss, and regulators notice. Trust me — don’t take shortcuts here.
Why Good Storage Spares Headaches
Years spent in research and quality settings taught me — stable storage isn’t a luxury, it’s baked into making good science and safe products. Seeing the cost of failed batches and the stress of troubleshooting pushes teams to tighten routines. Chilling in the right space, staying dry and dark, and labeling with care prove that the little details turn out to be the big ones in the end.
How should N-Carbamoyl-L-Glutamic Acid be administered or dosed?
Understanding the Need for Proper Dosing
Giving N-Carbamoyl-L-Glutamic Acid (NCG) in the right way matters a lot in both clinical and veterinary settings. This molecule mimics a natural compound—N-acetylglutamate—that helps the body handle ammonia in rare metabolic disorders. The stakes get high for anyone dealing with errors of the urea cycle. Mistakes in dosing or timing can mean the difference between keeping a patient stable or landing them back in the hospital.
Practical Administration in Medicine
Most health providers start oral or intravenous dosing based on a person’s weight or clinical status. In pediatric patients struggling with hyperammonemia, doctors usually kick off with a loading dose of about 100 to 250 mg/kg, taken by mouth or through a feeding tube. The maintenance dose over the next day follows at about 25 to 62.5 mg/kg every six to eight hours. For adults, the calculations shift upward, but the method stays personal. Physicians keep a close eye on ammonia levels and tweak the amount as the hours or days go on.
Injectable forms come in handy for folks who can't swallow or who need a quick response. It’s never one-size-fits-all—too much can stir up side effects like stomach upset, and too little means ammonia sneaks up. I’ve seen how tricky this can be during hospital shifts. To catch silent problems, labs run frequent blood checks to track ammonia and other related lab values, and adjust accordingly. Certified pharmacists play a big role here, stopping dosing errors before they happen.
NCG in Animal Health
Not all the action happens in hospitals. In animal sciences, especially livestock care, NCG finds its way into feed to improve protein use and limit ammonia buildup. Large-scale farms stick closer to lower maintenance levels—about 0.05% of dry matter in feed—spread throughout the day. Jumping higher can backfire, so trained nutritionists adjust dose and timing on a per-herd basis, often tied to the season or feed composition.
People working with animals need to check not just the dose, but also overall herd health, since changes in food, stress, and environment mess with absorption. My time with rural veterinarians taught me to never trust theory without watching real results over weeks or months. Spot-checking urine and blood ammonia can catch trouble before yield drops or cattle get ill.
The Role of Accurate Information and Monitoring
So much relies on clear communication. Patients and caregivers need instructions they can follow. Too often, the biggest risks come from overlooked steps or confusing instructions on the bottle. Health teams that educate patients—showing how to measure dose at home, or translating milligrams into teaspoons—get better results.
The science keeps changing. Data published by rare disease organizations, hospital guidelines, or from the Food and Drug Administration shape how doctors and nutritionists recommend doses. The methods that worked a decade back sometimes shift with emerging evidence.
What Could Improve Safety and Results
Clinical experience shows the need for more accessible point-of-care ammonia measurements. Test strips or bedside meters could let families or farmers check levels and dose on the fly, catching spikes before real harm sets in. More practical training—whether for rural clinics or home caregivers—demystifies the process and makes the benefits of NCG safer for all.
Clearer product labeling would make a big difference in both hospital and farm stockrooms. Providing liquid formulations with marked syringes or ready-to-mix powders boosts success. Stakeholders from clinicians to agriculture experts should keep updating protocols as research grows, bridging experience, and evidence for the best dosing outcomes.
Are there any side effects or interactions with N-Carbamoyl-L-Glutamic Acid?
A Hands-On Look at N-Carbamoyl-L-Glutamic Acid
Most people outside medical labs or rare disease clinics have probably never heard of N-Carbamoyl-L-Glutamic Acid (NCG). In my experience talking to folks in metabolic medicine, this molecule means a lifeline for certain patients, mostly those with enzyme deficiencies like N-acetylglutamate synthase deficiency. It acts as a substitute for a broken part in the body’s ammonia-cleanup machinery. Using a treatment like this doesn’t come without concerns, though. Anyone interested in or prescribed NCG wants to understand what could go wrong and how it might play with other medications or supplements.
Understanding Side Effects with Real-World Impact
With any therapy, side effects often raise anxiety—patients and families deserve honest, fact-based information. After combing through clinical trials, rare case reports, and the stories shared by people who’ve actually tried NCG, one thing stands out: most side effects show up at the digestive level. Stomach upset, mild nausea, and loose stools lead the list. These don’t seem to knock people out of daily life, but they can worry parents of young kids using NCG for inborn errors of metabolism.
A handful of users mention headaches or tiredness, though the numbers stay low. Sometimes, lab results include shifts in electrolytes or mild changes in blood urea. Medical teams use regular bloodwork to track those, so any shift can be handled before it turns serious. I’ve talked to clinicians who say they counsel families about what to watch for: signs of dehydration, mood changes, or trouble sticking with daily dosing routines.
Medication Interactions: An Understudied Arena
Drug interactions matter, especially when someone takes other ammonia-lowering drugs, protein supplements, or seizure medicines. Researchers haven’t uncovered strong evidence of dangerous drug-to-drug issues. Still, no one pretends to have all the answers. NCG hasn’t clocked up the clinical trial miles of aspirin or insulin. Patients with rare metabolic conditions usually take several therapies at once, so real-life experience matters just as much as textbook knowledge.
Some doctors pay extra attention if someone’s on high-dose steroids or certain antibiotics, given how these can stress the liver and kidneys. The science behind drug metabolism keeps evolving. Until larger and longer real-world studies fill in the gaps, doctors and pharmacists take a cautious, personalized approach. Keeping an open line between patient, prescriber, and pharmacist makes a real difference in catching any strange reactions early.
Making Informed Choices and Embracing Vigilance
I’ve seen that what scares most people about new medications is the unknown. With a compound like NCG, used mainly in rare, high-risk situations, the benefits often outweigh the minor stomach upset or tired days. That doesn’t mean people facing these conditions should just assume safety. Clear communication, regular monitoring, and honest acknowledgment of what we do and don’t know matter.
Getting trustworthy information straight from the doctor or a registered pharmacist leads to confidence. Checking lab values, reporting new symptoms quickly, and using the medication exactly as prescribed make a positive difference. Nobody should feel brushed off—every question deserves attention, and anyone starting NCG or helping a loved one with it plays an important part in creating a safer world for rare disease treatment.
| Names | |
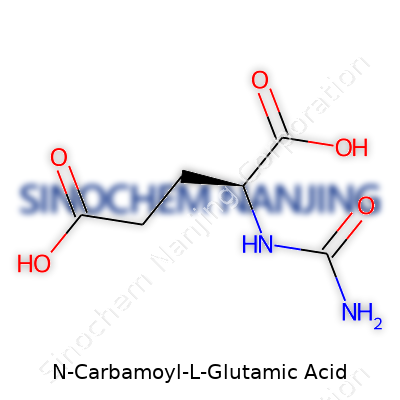
| Preferred IUPAC name | (2S)-2-[(Carbamoylamino)carbonyl]pentanoic acid |
| Other names |
Carbamylglutamate Carbamyl-L-glutamate N-Carbamylglutamic acid NCG N-Carbamoylglutamate |
| Pronunciation | /ɛn kɑːrˈbæmɔɪl ɛl ɡluːˈtæmɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | www-298-94-2 |
| Beilstein Reference | 120911 |
| ChEBI | CHEBI:27853 |
| ChEMBL | CHEMBL1205 |
| ChemSpider | 21581794 |
| DrugBank | DB00659 |
| ECHA InfoCard | 08d139e3-3ef4-4982-b765-776964e9b540 |
| EC Number | 3.5.1.2 |
| Gmelin Reference | 6178 |
| KEGG | C01879 |
| MeSH | D018080 |
| PubChem CID | 440013 |
| RTECS number | MW5086000 |
| UNII | 4R6Z587QDU |
| UN number | Not regulated |
| Properties | |
| Chemical formula | C6H10N2O5 |
| Molar mass | 216.17 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.642 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -2.41 |
| Vapor pressure | 3.3E-12 mmHg at 25°C |
| Acidity (pKa) | 2.1 |
| Basicity (pKb) | 7.96 |
| Magnetic susceptibility (χ) | -6.2e-6 |
| Dipole moment | 5.9206 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 198.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -924.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1401.7 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | A16AA06 |
| Hazards | |
| Main hazards | Causes serious eye irritation. |
| GHS labelling | GHS07, Warning, H319 |
| Pictograms | GHS07 |
| Hazard statements | Hazard statements: Not a hazardous substance or mixture according to Regulation (EC) No. 1272/2008. |
| NFPA 704 (fire diamond) | Health: 1, Flammability: 0, Instability: 0, Special: - |
| Lethal dose or concentration | LD50 > 2000 mg/kg (oral, rat) |
| LD50 (median dose) | LD50 (median dose): >5000 mg/kg (rat, oral) |
| NIOSH | KMH7H89E4F |
| REL (Recommended) | 4 mg/kg bw |
| IDLH (Immediate danger) | Not listed |
| Related compounds | |
| Related compounds |
Glutamic acid Carbamoyl phosphate N-Acetylglutamic acid L-Glutamine L-Arginine |

