L-Cysteine Hydrochloride Monohydrate: A Deep Dive into Past, Present, and Future
Historical Development
People have paid attention to amino acids for over a century, but the journey of L-Cysteine Hydrochloride Monohydrate started in earnest in the early twentieth century. Chemists first drew it out from protein hydrolysates, using time-intensive techniques that produced only modest yields. For decades, most manufacturers relied on animal hair and feathers as starting materials, simply because extraction from other sources didn’t pay off. By the 1980s, new fermentation methods turned up — harnessed by teams in Japan and Germany who recognized the need for more ethical and cleaner supply chains. Through all these shifts, the underlying drive stayed the same: industry needed a building block, and health professionals needed a reliable reagent.
Product Overview
L-Cysteine Hydrochloride Monohydrate shows up as a crystalline white powder, dissolving quickly in water and giving off a slightly sulfuric scent. It falls under a class of amino acids known for their functional thiol groups, which brings broad utility in both synthesis and food production. You’ll find it in labs and factory floors across the world, everywhere from bakeries to biopharmaceutical clean rooms. This amino acid salt doesn’t just act as a supplement; it changes the way flour behaves, unlocks protein cross-linking, and even scavenges oxygen in certain formulations.
Physical & Chemical Properties
L-Cysteine Hydrochloride Monohydrate offers a molecular formula of C3H7NO2S·HCl·H2O with a molecular weight of about 175.63 g/mol. In practice, the powder flows well, stays stable under most dry conditions, and resists clumping. Its solubility in water marks one of its most helpful features — for bakers, this means it can be mixed right into doughs, and for scientists, it gets dissolved easily for reaction setups. Its melting point rests around 175°C (decomposes), and at its core, the thiol (-SH) group drives chemical reactivity, making it a strong nucleophile and a valued reducing agent.
Technical Specifications & Labeling
Every bag or bottle rolling out of production sports labels that detail batch numbers, storage conditions, and the percentages of key components. Pharmacopeias such as USP, EP, and JP set standards for purity and heavy metal content. Real-life uses often demand a purity of 99% or higher, checked through HPLC and titration. Labels warn of the sulfurous odor, potential for skin sensitization, and proper storage — cool, dry places with solid ventilation. Users in pharma, food, or research environments look for documentation with a clear certificate of analysis (COA) and a transparent supply chain.
Preparation Method
Industrial methods for turning raw materials into usable L-Cysteine Hydrochloride Monohydrate rely on microbial fermentation. Sugar, spit out by genetically tweaked Escherichia coli, converts into L-cysteine in huge fermenters. This biotechnological leap nearly wiped out reliance on animal-derived sources. After fermentation finishes, staff separate out solids and perform multiple crystallizations. The hydrochloride salt forms last, an essential step so the final powder is easy to use and stable. The monohydrate comes from adding precise amounts of water during crystallization, skipping old pitfalls like inconsistent product and higher costs.
Chemical Reactions & Modifications
Chemists value L-cysteine’s -SH group for more than just its role in redox reactions. It acts as a protecting group in peptide synthesis, blocks unwanted side reactions, and stabilizes certain drug molecules. Under oxidative conditions, the thiol can form disulfide bonds, creating cystine — a process harnessed in protein refolding and hair treatments. The amino group also serves as a nucleophile in N-acylation reactions, opening more avenues for tailored compounds. In the lab, L-cysteine often helps regenerate enzymatically active forms of proteins or breaks disulfide bridges in denatured samples.
Synonyms & Product Names
Over the years, this compound has taken many names: L-Cysteine HCl Monohydrate, 2-Amino-3-mercaptopropanoic acid hydrochloride monohydrate, and even E920 in the bakery industry. Each synonym signals context — E920 for food regulators, L-Cysteine Hydrochloride when sourcing for pharma. The distinction helps buyers cut through the market jargon and get exactly what their workflow demands.
Safety & Operational Standards
Production and transportation bring hazards—the sulfur smell’s a hint, but there’s more. Powders float in the air and irritate lungs, so operators wear masks and gloves. MSDS sheets warn against splashing it in the eyes and call for washing with water if contact happens. Regulatory checkpoints, especially in Europe and North America, tightly monitor heavy metal residues, microbial content, and packaging integrity. Factories invest in air handling, clean rooms, and traceability measures, not just to satisfy auditors but to keep workers out of harm’s way and reassure end users.
Application Area
Bakeries use this amino acid salt to make dough less tough and sliceable, which keeps loaves consistent across gigantic production runs. In pharmaceuticals, it stabilizes active ingredients, feeds cell cultures, and serves as a reference standard in nutrition assays. Cosmetics companies include it in hair treatments, drawing on its relationship with keratin. Many labs rely on it as a reducing agent for preparing protein samples. Its use isn’t limited by geography — both east and west line up to secure reliable supply chains.
Research & Development
Scientists keep pushing to expand what this building block can do. Some experiment with new bacterial strains to boost yield in fermentation or reduce energy consumption. Recent work explores custom modifications — swapping side chains, tweaking solubility, or linking cysteine to other nutrients for better absorption. In proteomics, researchers seek to tag cysteine selectively for tracking in living systems, which could lead to new diagnostic tools. The field of green chemistry leans heavily on amino acids like this, hunting for alternatives to traditional petrochemical reagents.
Toxicity Research
Toxicologists have tested L-Cysteine HCl Monohydrate thoroughly. Oral doses far above recommended amounts bring nausea and diarrhea, but the quantities found in food or supplements barely nudge the danger scale. For those handling tonnage or working with powder daily, inhalation risks demand attention — dust control and ventilation go from nice-to-have to non-negotiable. Reports show rare allergic reactions, mainly among workers with prolonged skin exposure. Surveillance by agencies like the FDA keeps product recalls infrequent.
Future Prospects
Growing demand for clean label foods, ethical sourcing, and greener manufacturing keeps L-Cysteine Hydrochloride Monohydrate in the spotlight. More companies seek microbial production, ditching older animal-based sources for both ethical reasons and economic efficiency. Innovations in fermentation technology promise better yields and fewer byproducts. Functional foods and advanced drug delivery systems might pull demand in new directions, spurring fresh regulatory scrutiny and product development. I see education as a missing link — more transparency around sourcing, quality, and risks could calm lingering public doubts and speed up adoption of biotechnological breakthroughs.
What are the main uses of L-Cysteine Hydrochloride Monohydrate?
Bread Softening and Dough Conditioning
Most people probably don’t spend time thinking about what makes supermarket bread so soft and fluffy. L-Cysteine Hydrochloride Monohydrate shows up in bakery supply shops as a must-have for commercial bakers. This amino acid helps break down gluten bonds, so dough becomes easier to knead and shape. Batches become more reliable, and the final product tends to come out consistently airy. Fast food restaurants rely on this ingredient to deliver buns with the same squish each time, not the dense loaves you might get from a small bakery using traditional methods.
Improving Flavor and Shelf Life in Processed Foods
Large food companies don’t just want their products to look good right out of the oven—they want them to last. L-Cysteine Hydrochloride Monohydrate deals with this problem in a very practical way by reducing the effects of oxidation. Processed meats and convenience snacks, for example, stay fresher for longer because this additive helps fight off the chemical changes that make food go stale or taste “off.” This kind of additive has caught on around the globe. The Food and Agriculture Organization, together with the World Health Organization, has weighed the evidence and provided guidelines for how much makes sense in food.
Pharmaceutical Formulations and Intravenous Solutions
Anyone who has spent time studying pharmacy ingredients will notice L-Cysteine Hydrochloride Monohydrate listed in certain injectable medications. Its role here feels critical. Hospitals turn to it because it works as a precursor for glutathione, a powerful antioxidant in the human body. Conditions like paracetamol (acetaminophen) poisoning get treated with N-acetylcysteine, a close cousin. The monohydrate form shows up in parenteral nutrition formulas, especially for patients who can’t get nutrients through regular eating. The FDA includes L-cysteine on its list of safe food and medical additives, so there’s quite a bit of confidence behind its use.
Supplements and Hair Products
I know gym enthusiasts always look for ways to recover faster and build muscle better. Amino acid supplements feature L-Cysteine Hydrochloride Monohydrate on their labels for its reported roles in detoxification, immune health, and hair quality. My own barber once talked about how this same ingredient strengthens the bonds in keratin, the protein that makes up hair. Some shampoo and conditioner brands proudly list it to attract health-conscious buyers. Researchers continue to study its exact effects, but for now, sales pitch and science both seem to agree: there’s potential benefit.
Addressing Concerns and the Way Forward
L-Cysteine Hydrochloride Monohydrate sometimes leads to questions about origin, since some sources derive it from animal products, like poultry feathers. A growing number of manufacturers now produce it through fermentation, using microorganisms instead of animal sources. Demand for vegan or halal-certified products pushes this shift. As shoppers learn more about what’s in their bread, medicine, or hair products, I see more companies moving toward transparency. Labels say more than ever. Regulatory oversight keeps increasing. People have good reason to expect clear information, especially when it comes to what we eat or put on our bodies.
Practical Considerations for Industry and Everyday Life
Anyone who works in food manufacturing, pharmaceuticals, or even the beauty sector knows efficiency matters. L-Cysteine Hydrochloride Monohydrate acts as a reliable tool, whether the job involves producing pallets of hamburger rolls or compounding a specific IV medication. Safety limits rest on strong scientific reviews. Plenty of industry experts and nutritionists suggest keeping an eye out for new methods and consumer preferences. My own grocery store now stocks vegan-friendly bread and shampoo, a small but clear sign that the market keeps changing. The science supports its importance, but public trust rides on honesty and safer alternatives where possible.
Is L-Cysteine Hydrochloride Monohydrate safe for consumption?
The Purpose of L-Cysteine Hydrochloride Monohydrate in Food
I’ve worked in ingredient research for years and seen all kinds of chemical names on labels rattle consumers. L-Cysteine Hydrochloride Monohydrate often triggers questions, especially in the bakery aisle. In practice, this amino acid derivative breaks down gluten and helps make dough soft, pliable, and easy to handle. Breads, bagels, and some Asian noodles rely on this additive for that signature fluff most people expect.
How Regulatory Agencies View Its Safety
L-Cysteine Hydrochloride Monohydrate has gone through safety evaluations from agencies including the US Food and Drug Administration (FDA), the European Food Safety Authority (EFSA), and Health Canada. These folks look at long-term studies, toxicology data, and the practical use of the ingredient in daily diets. The FDA gives it GRAS (Generally Recognized As Safe) status. EFSA considers it safe in quantities used in food processing. Decades of bakery production back up this position with real-world data, not just lab tests.
The Source of L-Cysteine: A Real-World Concern
Many people stumble over where this amino acid comes from. Traditionally, manufacturers extracted it from animal sources like duck or hog feathers. In the last several years, fermentation methods using bacteria have become a leading source, especially for products marketed to vegetarians and vegans. I’ve spoken to bakers and manufacturers who say their suppliers switched over almost entirely to the fermentation route. If you want to know the source, it pays to check with a producer that shares ingredient origins on request. This matters most for vegetarians, vegans, and people following Halal or Kosher diets.
Potential Side Effects and Who Should Be Cautious
I've seen almost no reports of healthy people reacting to this ingredient. L-Cysteine naturally exists in many proteins folks eat each day. The amounts used in food are far lower than doses sometimes given for medical therapy. For people with rare metabolic conditions affecting amino acid processing, such as cystinuria, high intake might not be recommended. If you live with a chronic health condition, it makes sense to point out new ingredients or supplement intake to your doctor.
Transparency, Labeling, and Informed Choice
Trust grows when companies clearly label where their ingredients come from. Sometimes labels say “L-Cysteine,” other times you find the full chemical name. If it matters to you that it’s animal-free, or you need non-GMO, verified information from companies helps make confident choices. Some manufacturers use QR codes or website links to add extra detail beyond the label.
Ideas for Moving Forward
With rising consumer interest in ingredient origins, food makers need to keep lines open about the source and processing details behind the ingredients they add. Greater transparency not only addresses dietary restrictions but also boosts shopper knowledge. Investing in research on how these processing aids interact with other food components deepens understanding and could spark new, gentler baking methods down the road. For now, the science and day-to-day experience say L-Cysteine Hydrochloride Monohydrate is safe at the levels used in food.
What is the recommended dosage for L-Cysteine Hydrochloride Monohydrate?
Why People Use L-Cysteine Hydrochloride Monohydrate
L-Cysteine Hydrochloride Monohydrate shows up in the world of supplements, food processing, and even some pharmaceuticals. Many of us spot it on food labels—think bread and some hair treatments. But what’s the right amount to use? That’s been a bit of a puzzle, and plenty of people have questions around safety, effectiveness, and medical advice.
What Science Says About Dosage
The U.S. Food and Drug Administration gives L-cysteine a Generally Recognized As Safe (GRAS) status for its specific use in food within certain levels. The tolerable daily intake for L-cysteine—not the hydrochloride monohydrate form—is often pegged around 1 to 1.5 grams for adults, spread out during the day. Most products with it, like bakery bread or supplement capsules, keep the dose well under this mark.
Researchers have dug into the role of L-cysteine as an amino acid, noting it gets involved in glutathione production, an antioxidant important for cell protection. The recommended intake doesn’t just depend on what a label says. Health status, age, diet, liver health all play into it. Too much can lead to side effects. Upset stomach, nausea, and even increased oxidative stress in rare cases, especially when the body doesn’t need extra cysteine.
How I Approach Supplement Decisions
I read about supplements regularly, and over the years, I’ve watched friends try pretty much every new nutrient on the market. I always nudge folks to ask their doctor before jumping on any new supplement, even if it seems harmless or “natural.” Most people actually get enough cysteine from regular foods—think poultry, eggs, dairy, beans—so they don’t need to add more unless a health provider sees a real need.
Dosing without guidance can backfire. Elevated cysteine levels sometimes get linked with kidney stones or cardiovascular problems, though data is mixed. Medical professionals lean on studies and bloodwork to say how much, if any, extra cysteine helps. Chasing better health is important, but it works better with facts and oversight.
Label Reading and Smart Choices
Most supplement bottles list L-Cysteine Hydrochloride Monohydrate doses between 100 mg and 600 mg per serving. Multivitamins or hair/skin formulas may use 50 mg or less. Bread and food ingredients add much smaller amounts, just enough to help with texture or shelf life. The European Food Safety Authority, for example, sets a similar conservative limit for food uses.
People should always keep an eye on what they’re actually swallowing. It’s easy to double up with a supplement and a multivitamin and not notice. L-Cysteine works best in balance with other amino acids, not as a solo act. Nuanced nutrition matters, and context helps: what suits an athlete recovering from intense training might not serve someone with a chronic illness or a completely different diet.
Considering Alternatives and Solutions
Most of us do better investing time in a balanced diet instead of chasing after isolated amino acids. If someone’s worried about their intake, doctors can check dietary patterns and might recommend adding eggs, chicken, or legumes before reaching for a bottle of L-cysteine. Those with diagnosed deficiencies—like some folks with rare metabolic conditions—do require more, but that’s always under medical guidance, with regular monitoring.
In the fast-moving supplement world, it’s easy to get lost or caught up by marketing. Trusted health sources and transparent labeling support better decisions. Getting science-backed advice from dietitians or providers leads to fewer risks and more real benefits. It never hurts to ask smart questions.
Are there any side effects associated with L-Cysteine Hydrochloride Monohydrate?
What Goes Into the Food We Eat?
L-Cysteine Hydrochloride Monohydrate pops up on food labels more often than people realize. From sliced bread in the supermarket to some snack foods, it works as a dough conditioner, a way to help make products softer and easier to process. Some supplement shelves carry it too, with promises around hair, skin, and immunity. Awareness about what we eat matters. Nutrient additives and food technology can help, but questions about side effects deserve honesty.
What the Research Tells Us
People often ask if L-Cysteine Hydrochloride Monohydrate actually leaves any negative health marks. The story’s pretty straightforward in the scientific literature: for most healthy adults, L-cysteine in the amounts used in food or even many supplements rarely causes trouble. The US Food and Drug Administration classifies it as “Generally Recognized As Safe.” It’s a non-essential amino acid, which means healthy bodies make it on their own.
That said, eating too much of anything can make trouble, and L-cysteine stands no exception. Researchers have looked at side effects after people swallowed large amounts—far higher than anything found in a normal diet. Some reported nausea, vomiting, or digestive cramps. High doses may upset the stomach or bring headaches. It’s easy to underestimate how much ‘just a little extra’ turns into ‘way too much’ if someone adds supplements to a diet already packed with processed foods boosted with this ingredient.
Liver and kidney function play a part too. The liver processes amino acids and if someone’s organs aren’t working well, even modest doses become risky. There have been case reports of kidney stones after excess cysteine buildup. Anyone with cystinuria—a rare genetic disorder—should be cautious, as excess cysteine in the urine may trigger stone formation.
Food Industry Practices and Transparency
A challenge comes with food processing. Bakery chains use L-Cysteine Hydrochloride Monohydrate to make dough pliable, reducing waste and speeding up bake times. Sourcing can raise ethical eyebrows, since some manufacturers previously used hair or feathers, but more use synthetic or microbial fermentation now. Consumers deserve to know not just what’s inside, but how it ends up there.
Allergies to L-cysteine sit at the extreme end of rare, but those with sensitivities or special diets should always check food labels. Just because something’s in ‘everything’ doesn’t make it totally risk-free for everyone.
Practical Solutions and Mindful Choices
It helps to treat ingredient lists like road maps. If someone sees L-Cysteine Hydrochloride Monohydrate on a label and worries, limiting highly processed foods can offer peace of mind. Whole foods rarely contain such additives, and even for those who rely on packaged goods, moderation stands as the safest bet. Supplement seekers should talk to healthcare providers, especially if they take medications or manage chronic illnesses.
Simple food choices reflect a lot more than nutrition: trust, transparency, and respect for health all get wrapped up in how additives like L-Cysteine Hydrochloride Monohydrate appear on store shelves. Following up with questions, reading labels, and asking who makes what goes into food goes a long way toward protecting personal well-being.
Is L-Cysteine Hydrochloride Monohydrate derived from animal or plant sources?
Chemicals with a Backstory
Not every food additive comes with a clean, straightforward background. L-Cysteine Hydrochloride Monohydrate sits in that gray area where science meets industry shortcuts. You’ll find it in bread, in packaged baked goods, and even in pharmaceuticals. It gets added because it breaks down dough, softens texture, and sometimes, helps food last longer. Yet, very few stop to ask: what exactly goes into making it?
Behind the Name: Animal or Plant?
L-Cysteine isn’t just a mouthful to pronounce; it hides a story that many people who care about the source of their food might want to know. For a long run, most of the global supply came from human hair—a resource considered ‘waste’ in many salons across countries like China and India. The process uses acid hydrolysis to break down hair and extract the amino acid. This is an industrial process, and regulatory agencies in the U.S. and Europe know all about it.
People who follow vegan, kosher, or halal diets get less enthusiastic when they realize the origins. Human hair doesn’t fit the bill for meeting these food standards. Consumer pressure has forced some companies to reconsider. Similarly, duck feathers serve as another animal-based source. The chemical process is the same: break it down and isolate the L-Cysteine. There’s no secret about it, but the packaging never mentions it either.
Plant-Based Production: A Shift in Focus
Alternative sourcing did not show up because the industry suddenly found a moral compass; rising demand from those with dietary restrictions led to changes. Fermentation using genetically modified microbes is now picking up. Companies like CJ CheilJedang and Wacker Chemie produce L-Cysteine via fermentation with bacteria fed on plant sugars. It sidesteps animal products completely.
Fermentation requires investment and expertise, so costs rise. Many smaller manufacturers stick with animal or human sources to keep prices down for commercial bakeries and food service giants. Plant-based sources haven’t reached global dominance yet, but pressure keeps mounting.
Transparency—A Stubborn Problem
If you scan a food label, you’ll see “L-Cysteine Hydrochloride Monohydrate” but rarely see where it comes from. Regulators such as the FDA allow the label as long as the chemical makeup matches the approved additive, regardless of how it’s made. This loophole creates confusion for consumers. More people demand transparency, especially as plant-based diets gain followers.
Retailers, bakeries, and food manufacturers say they want to be transparent, but changing supply chains doesn’t happen overnight. Certifying plant-based supply starts with tracing every part of the production, plus regular audits. These steps bring reassurance to customers who care, but only if companies commit wholeheartedly.
Where Solutions Grow
If you want to avoid animal-sourced L-Cysteine, there’s only one safe bet—choose brands that clearly state plant-based or microbial fermentation sourcing, or reach out to their customer service. Advocacy groups push for tougher labeling laws, and some food retailers already use plant-sourced versions in their vegan lines. With sustained customer pressure, and as technology improves, wider plant-based sourcing will become the norm.
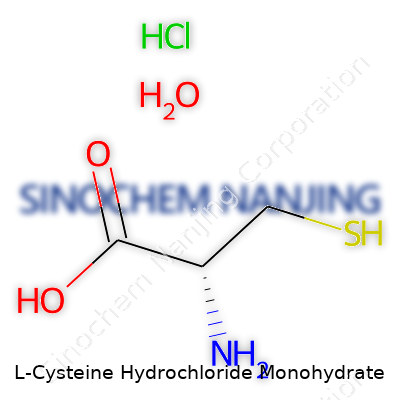
| Names | |
| Preferred IUPAC name | 2-amino-3-sulfanylpropanoic acid;hydrochloride;monohydrate |
| Other names |
L-Cysteine HCl Monohydrate L-Cysteine hydrochloride monohydrate Cysteine hydrochloride monohydrate L-Cysteine, monohydrochloride, monohydrate L-2-Amino-3-mercaptopropionic acid hydrochloride monohydrate |
| Pronunciation | /ˌɛlˈsɪstiːn haɪˈdrɒklaɪd ˌmɒnəˈhaɪdreɪt/ |
| Identifiers | |
| CAS Number | 7048-04-6 |
| 3D model (JSmol) | `3D model (JSmol) string for L-Cysteine Hydrochloride Monohydrate:` ``` CSCC(N)C(=O)O.Cl.O ``` *This is the SMILES string representation, compatible with JSmol for visualization.* |
| Beilstein Reference | 3569724 |
| ChEBI | CHEBI:61343 |
| ChEMBL | CHEMBL1201471 |
| ChemSpider | 25969 |
| DrugBank | **DB00141** |
| ECHA InfoCard | 03a21e8b-48d9-47b5-bfa6-3ff394c56b52 |
| EC Number | 3.1.1.75 |
| Gmelin Reference | 57481 |
| KEGG | C00097 |
| MeSH | D-Cysteine |
| PubChem CID | 61336 |
| RTECS number | MF4300000 |
| UNII | F6F0HK1URN |
| UN number | UN3077 |
| Properties | |
| Chemical formula | C3H7NO2S·HCl·H2O |
| Molar mass | 175.63 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 0.6 g/cm³ |
| Solubility in water | Freely soluble in water |
| log P | -7.0 |
| Acidity (pKa) | 8.33 |
| Basicity (pKb) | pKb ~ 7.64 |
| Magnetic susceptibility (χ) | -9.6 × 10⁻⁶ cm³/mol |
| Dipole moment | 1.66 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 221.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -947.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -163.2 kJ/mol |
| Pharmacology | |
| ATC code | A16AA17 |
| Hazards | |
| Main hazards | May cause respiratory, skin, and eye irritation. |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | Hazard statements: "Causes serious eye irritation. |
| Precautionary statements | P264, P270, P273, P301+P312, P330, P501 |
| NFPA 704 (fire diamond) | 1-0-0 |
| Lethal dose or concentration | LD50 Oral Rat >3980 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 1890 mg/kg |
| NIOSH | MI7700000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 30 mg/kg |
| IDLH (Immediate danger) | No IDLH established |
| Related compounds | |
| Related compounds |
Cysteine L-Cysteine L-Cysteine Hydrochloride N-Acetylcysteine DL-Cysteine Cystine Glutathione |

