L-Aspartic Acid Diethyl Ester Hydrochloride: Insight and Analysis
Historical Development
Learning about L-Aspartic Acid Diethyl Ester Hydrochloride’s journey gives a good sense of how chemical compounds evolve from ideas into crucial lab tools. Organic chemists started modifying amino acids seriously back in the early twentieth century, and aspartic acid quickly grabbed attention for its workable structure. Tweaking its ester forms, including the diethyl version, let researchers test new reactions and syntheses easily. The hydrochloride salt came after, locking the molecule into a stable, usable format that held up better both on the shelf and in water-based reactions. That legacy of careful development echoes in today’s catalog listings and published protocols, where this compound’s name pops up regularly in amino acid chemistry discussions and method sections.
Product Overview
Anyone who’s spent time in a chemical storeroom knows L-Aspartic Acid Diethyl Ester Hydrochloride’s familiar pale crystalline look. Its full chemical name signals its makeup: it’s the hydrochloride salt of an ethyl-esterified aspartic acid. Suppliers usually offer the compound as a lab reagent, making life easier for organic chemists who need a predictable starting point for peptide modification or building complex molecules. Beyond just raw materials, this compound can serve as a building block for everything from pharmaceuticals to specialty polymers. Some companies list it under synonyms like “Diethyl L-aspartate HCl” or “Aspartic acid, diethyl ester hydrochloride,” but the core structure stays the same.
Physical & Chemical Properties
In the weighing station, L-Aspartic Acid Diethyl Ester Hydrochloride appears as a fine, white powder, usually odorless, clumping only slightly if the air gets too humid. Typical molar mass hovers around 243 g/mol. The compound dissolves fairly easily in water and ethanol, far more so in acidified solutions than in strictly basic ones. The melting point often lands near 155-160°C, with good thermal stability for short reaction runs but some risk of decomposition—especially if moisture creeps in or storage conditions drop below best-practices. The hydrochloride definitely boosts shelf life, and also offers more consistent solubility than the free ester. One big plus: the lack of offensive odor means easier bench work when prepping buffers or synthesizing peptides.
Technical Specifications & Labeling
Supplier data sheets almost always promise high purity, usually at 98% or more, and show crucial data: melting point, optical rotation, and sometimes a thin-layer chromatography Rf. Labels spell out not just the IUPAC name but also the main CAS number (often 7488-99-5), storage conditions (cool, dry place, tightly sealed container), and hazard notes. Labels with GHS pictograms make safety guidance clearer, giving even seasoned bench hands a needed nudge towards eye protection and good ventilation. Barcode tracking and batch records matter too, especially for those in regulated labs or GLP settings. Good labeling doesn’t just check a box: it saves time and prevents hard-to-fix mistakes.
Preparation Method
Making L-Aspartic Acid Diethyl Ester Hydrochloride takes careful stepwise synthesis. The classic approach starts with L-aspartic acid, which reacts with ethanol in the presence of an acid catalyst—often HCl—leading to the diethyl ester. Once that reaction has gone to completion (usually by refluxing for several hours), the crude ester forms and the hydrochloride moiety attaches by bubbling hydrogen chloride gas or adding concentrated aqueous HCl. Afterward, careful solvent removal and crystallization pull out the pure hydrochloride salt. Washing with cold ethanol helps strip away side-products, and a controlled drying step removes moisture that could lower purity or cause clumping. It’s the kind of preparation that rewards close attention, with final crystallinity and color acting as visual guides to quality.
Chemical Reactions & Modifications
L-Aspartic Acid Diethyl Ester Hydrochloride’s value in the lab shines when it becomes a substrate for peptide synthesis or further derivatization. Its ester groups activate for mild hydrolysis or transesterification, unlocking routes to more complicated amino acid derivatives. Direct amidation, alkylation, and even cyclization come up in branch-chain peptide studies or polymer chemistry. I’ve seen teams tweak this molecule into specialty monomers for biodegradable plastics or tag it with functional groups to probe enzyme selectivity. Even simple substitution reactions can expand the molecule’s chemical space—practically every organic chemist runs across an aspartic acid ester variant at some phase in their project, and the HCl salt version usually wins out for ease of handling.
Synonyms & Product Names
L-Aspartic Acid Diethyl Ester Hydrochloride circles lab catalogs under a few different names. “Diethyl L-aspartate hydrochloride” and “Aspartic acid diethyl ester HCl” fill out most order forms. I’ve seen one supplier list “(S)-2-Aminobutanedioic acid diethyl ester hydrochloride,” which clarifies the stereochemistry but hardly rolls off the tongue. CAS 7488-99-5 tracks down the compound fast, regardless of vendor, and helps dodge mix-ups with similar-sounding acid esters. Some companies paste their own brand or code on the label, but those cross-references help trace inventory and check quality.
Safety & Operational Standards
Lab veterans treat all amino acid esters with respect, and L-Aspartic Acid Diethyl Ester Hydrochloride gets the standard protocols: gloves, goggles, and fume hood work for any weighing or transfer. GHS classification usually calls it an irritant to eyes and skin. I always remind newcomers to crack open the MSDS before opening a fresh bottle—it lists not just spill protocols but also exposure symptoms (eye redness, mild respiratory discomfort). Storage away from acids, bases, and strong oxidizers improves stability. Many operations add a layer of secondary containment, especially if moisture in the storeroom means bottles can cake up or labels smear. Each batch runs through quality controls to confirm both purity and absence of byproducts, trimming risk on both user and environmental fronts.
Application Area
The compound’s strongest pull comes in peptide synthesis and pharmaceutical research. Diethyl aspartate forms stand up well under coupling reactions, letting chemists string together peptide chains with precision. Formulating drugs often involves tweaking the molecule for solubility or bioavailability, and this ester finds a home as a starting point in both generic and specialty syntheses. Outside pharma, researchers test L-Aspartic Acid Diethyl Ester Hydrochloride in agricultural chemistry (novel plant-growth regulators), biodegradable plastics, and even specialty coatings. Each field values its reliable shelf life and friendly handling, and as one who has run purification columns with this compound, I know it holds up well in prep work, making it a backbone of diverse R&D efforts.
Research & Development
Academic teams and private labs both use L-Aspartic Acid Diethyl Ester Hydrochloride in different lines of research. In peptide engineering, it helps test new coupling reagents or explore backbone conformations, and lab books fill up with variants created by modifying its ester groups or chirality. Polymer research leans on diethyl aspartate salts to generate new chains with tunable properties, and some groups probe its use as a chiral auxiliary in asymmetric synthesis. Digital databases show citations growing year by year, as more projects pick this compound to build proof-of-concept molecules or explore biological applications. It’s that workhorse status, showing up in reaction trials and yield tables, that sets this ester apart in a crowded field of amino acid derivatives.
Toxicity Research
Toxicity studies—though less frequent than for pharmaceutical products—still tag along with any new application of L-Aspartic Acid Diethyl Ester Hydrochloride. Lab handling data puts toxicity as relatively low for small-scale exposure, mainly limited to local irritation and rare allergic reactions. Animal testing for regulatory review checks acute and chronic toxicity, with results generally indicating low systemic risk at research dosages. Environmental fate draws the eye in larger-scale use, so some groups assess what breakdown products appear after hydrolysis or incineration. Waste management plans reflect those findings, emphasizing thorough dilution and minimizing drain disposal. Each new use case brings a fresh round of toxicology checks, especially with novel modifications or delivery methods, as regulatory frameworks demand ongoing vigilance.
Future Prospects
Looking past today’s uses, this compound has plenty of future promise. The steady global need for new peptide therapies and “greener” polymers means L-Aspartic Acid Diethyl Ester Hydrochloride will stay on the shopping list for drug and material developers. Advances in green chemistry push for milder, solvent-free esterification methods, which could shrink the environmental footprint of large-scale production. Synthetic biologists keep hunting for new enzyme-based routes to this and similar esters, hinting at a push from batch chemistry toward biocatalysis. At smaller scales, its role as a precursor for chiral building blocks may only grow, especially as more startups chase niche pharmaceuticals and specialty polymers. Well-designed safety data and robust technical support will help users stay confident handling the compound, and as research deepens into its unexpected roles, L-Aspartic Acid Diethyl Ester Hydrochloride’s reputation as a ‘go-to’ amino acid derivative won’t fade any time soon.
What is L-Aspartic Acid Diethyl Ester Hydrochloride used for?
What L-Aspartic Acid Diethyl Ester Hydrochloride Really Does
L-Aspartic acid diethyl ester hydrochloride might sound like a mouthful, but for researchers and folks working in labs, this compound brings some value to the table. Chemically, it builds on the foundation of aspartic acid—a well-known amino acid—but in the form of an ester, making it useful for things regular aspartic acid just can’t do. What stands out for me about this molecule isn’t just its complexity; it’s how it steps up as a building block for making bigger, more interesting molecules. In many cases, it serves as a stepping stone in the synthesis of peptides and pharmaceuticals.
Applications in Peptide Synthesis
Building peptides forms a huge part of modern drug development and biochemical research. You run into the issue of unwanted reactions or breakdowns during the hybrid assembly of complex molecules. By turning aspartic acid into the diethyl ester hydrochloride form, researchers get extra stability and solubility. It handles conditions that break down less protected molecules. This means better yields and more reliable results. Speaking from experience in a research setting, the frustration of unwanted side reactions eats up time and money. Working with protected amino acids like this one helps clean up those experiments, letting teams cycle through ideas much quicker.
Value in Chemical Research and Development
Lab workers use this compound not only because it fits into peptide syntheses but also since it can mask certain chemical groups. If you leave the carboxyl groups in their original form, they easily react with things you might wish they wouldn’t. Converting those to esters changes the game. Suddenly, you can target another spot on the molecule or add other chemicals one at a time, almost like following a recipe. That level of control sits at the core of designing safer, more effective drugs in the long run.
Contributions to Pharmaceuticals
Drug design benefits from molecules like L-aspartic acid diethyl ester hydrochloride. Medicinal chemists often test long chains of modified amino acids to find just the right combination for a new treatment—whether that means better pain relief, reduced inflammation, or stronger antibiotics. By using esters, researchers can guide those chains to react only when and where they want during the manufacturing process. Consider how closely regulated the pharmaceutical industry remains—both for safety and quality. Streamlining synthesis with building blocks that behave as expected helps deliver consistent results, keeps production costs in check, and reduces waste.
Concerns and Ways Forward
Like many specialty chemicals, this compound does bring up some safety concerns. Exposure guidelines and proper handling need attention. Teams working with it rely on clear labeling, up-to-date safety sheets, and protective gear. What matters even more is education. Each time I started using a new compound in my own research, I spent time reading, asking for advice, and double-checking procedures. It keeps mistakes at bay and protects not only health but also the reliability of the results coming from the lab.
Looking for Balance in Innovation
The push for new medicines keeps driving demand for specialty reagents like L-aspartic acid diethyl ester hydrochloride. Labs look for purer chemicals, greener synthesis methods, and safer byproducts. As researchers and chemical suppliers continue raising the bar, the hope is that the next steps in medicinal chemistry involve not just novel science but practical solutions that weigh health, safety, and progress all at once.
What is the chemical formula and molecular weight of L-Aspartic Acid Diethyl Ester Hydrochloride?
The Formula in Black and White
L-Aspartic acid diethyl ester hydrochloride doesn’t just sound like a mouthful—it has a chemical backbone that actually means something. The chemical formula is C8H16ClNO4. For scientists and anyone trying to get a grip on pharmaceutical ingredients or lab reagents, the formula gives you the recipe to its molecular makeup. The molecular weight sits at 225.67 g/mol, a number as dry as it seems, but one with practical consequences for those mixing, measuring, and creating both in university labs and commercial operations.
Why Formula and Weight Aren’t Just for Fancy Calculations
People work with chemicals like L-aspartic acid derivatives because they form the backbone of bigger, more useful molecules. I’ve seen research teams get tripped up over mismeasured ingredients, wasting days or blowing the budget because a decimal point was off. In the lab, knowing you’re looking at C8H16ClNO4 and its molecular weight tells you exactly how much to use for your experiment or synthesis. Mistakes stick out fast; one gram off, and your reaction might stall or produce something totally useless.
L-Aspartic Acid Diethyl Ester Hydrochloride in the Real World
Talking with chemists, it always comes back to reliability. Nobody wants to redo a run because the reagent didn’t behave as expected. If you need to convert amino acids into something more reactive for peptide synthesis, this compound saves steps. Its tweaked ester pairs play nice in organic synthesis, improving yields and letting you avoid tricky purification steps. Researchers using this ester as a building block produce specialty polymers, intermediates, and even go for specialty pharmaceuticals, riding on the consistency this molecule offers.
Purity and Integrity—Don’t Skimp
Letting quality slide with L-aspartic acid derivatives stirs up problems. Impurities lead to batch failures. The hydrochloride salt keeps the basic structure stable and soluble, cutting down on the gunk that shows up if you cheap out on suppliers. If a manufacturer can’t provide clear details on molecular formula, assay, or polishes off the certificate of analysis with questions unanswered, most chemists roll their eyes and walk away. You can’t chase reproducibility with mystery material.
Challenges and Smarter Solutions
Labs run into headaches sourcing this ester in high purity, and it’s no small fix. Sticking with reputable suppliers who document traceability can help. There’s a push in the research community for better testing protocols. Instead of trusting the label, some groups run NMR and mass spec to double-check that the C8H16ClNO4 on the jar is what’s actually inside. That costs money and time, but saves even more on headaches down the road.
It wouldn’t hurt for more suppliers to get transparency up front—keep clear batch records, answer chemistry questions, and skip vague marketing. If a mistake in formula or weight ruins even a single run, that’s hours lost and budgets stretched. Reliable chemical data isn’t just science trivia; it’s the difference between discoveries and dead ends.
Is L-Aspartic Acid Diethyl Ester Hydrochloride safe to handle and what are its storage requirements?
Safety Takes More Than Gloves
I have worked in labs that hustle through all kinds of chemicals, simple or exotic. L-Aspartic Acid Diethyl Ester Hydrochloride might sound niche, but its risks sit right alongside more familiar reagents. The powder gives off little smell, and it doesn’t sizzle on contact, so many new hands might gloss over real hazards. No, it’s not the worst thing you’ll find in a chemical storeroom. Still, you wouldn't trust a kitchen cleaner under your nose without knowing what’s inside.
A chemical this pure can irritate the skin or eyes fast. Stir it up in the air, and suddenly the headache doesn’t feel so distant. OSHA and Sigma-Aldrich flag it for causing irritation. No, it’s not likely to burn through your gloves after splash, but basic safety means more than hoping for the best. Nitrile gloves and a face shield reduce stupid accidents. Open bottles under a fume hood, and you avoid breathing stray dust. Once, a colleague laughed off safety glasses—until a speckled lens taught him not to. The reminder left a sharper memory than any checklist.
What Storage Demands
L-Aspartic Acid Diethyl Ester Hydrochloride doesn’t do well with moisture. Think of how flour absorbs damp—this powder clumps just as fast, and loses its chemical punch before you notice. A broken seal, or a jar set on a warm shelf, spoils the whole batch. Best practice says: keep it dry, cool, and shut tight. Store it in tightly sealed containers. Keep those away from sunlight and any room where the thermometer creeps up. Walk through any pharma or university lab, and you’ll find this sitting with other esters, tucked away in low-light cabinets.
Each time someone leaves a jar cracked open, the risk piles up. Hydrochloride salts in moist air pull water like a sponge, turning powder gummy. Doing regular checks of stock keeps folks honest—nobody wants to spend the afternoon tracing why a synthesis ran flat.
Reducing Accidents Means Routine
Big safety improvements come from small habits. L-Aspartic Acid Diethyl Ester Hydrochloride won’t explode from dropping a tube, but treating it like a reckless cleaner tempts fate. Anyone assigned to handle these stocks needs repeated, real-world training—a thick binder on GHS labeling doesn’t replace direct, on-the-job experience.
Every lab should post clear signage reminding people of risks. Easy-to-read instructions give new hires a fighting chance. A good habit and routine supplies like spill blankets, eyewash stations, and smart inventory management cut emergencies before they start. Old-timers working through recipes usually know: a neat workspace beats crisis calls.
Toward Better Handling Practices
Solutions don’t cost an arm and leg. Change gloves before moving between projects. Label every bottle by date and batch. Review safety data sheets every time a new container comes in—not just once a year. Encourage staff to speak up about worn seals or strange smells, no matter their role on the team. Building a culture of accountability doesn’t just protect individuals. It lifts your whole lab’s reliability, protecting reputations and, in the long run, budgets.
No one has to become a chemist to handle L-Aspartic Acid Diethyl Ester Hydrochloride safely. With respect, routine checks, and the right training, this chemical stops being a source of worry and stays a science tool—just as intended.
What is the purity level of the L-Aspartic Acid Diethyl Ester Hydrochloride product?
Purity Shapes the Story
It feels odd to talk about chemistry and purity without getting bogged down in numbers and technicalities, but the purity of L-Aspartic Acid Diethyl Ester Hydrochloride is not some abstract box to check. It shapes what researchers, pharmaceutical manufacturers, and even supplement formulators can trust about their end result. The most common standard found on the market is a purity level above 98%, often reaching beyond 99%. That number is more than a bragging point—it directly ties to safety, reliability, and even legality.
The Impact on Research and Production
Purity carries weight because contaminants, even in tiny amounts, can distort research findings or introduce risks. Anyone who's run synthesis in the lab knows frustration when impurities muddy outcomes. I’ve seen high-purity chemicals help research avoid those re-runs and head-scratching moments. In pharmaceutical settings, even minor contamination can slow regulatory approval, delay release, or in worst cases, prompt recalls. Trust in a product starts with this number.
Industry Demands Real Clarity
Groups responsible for large-scale production expect suppliers to document analytical data—things like HPLC and NMR spectra to confirm claimed purity. I’ve had conversations with procurement managers who won’t even consider a bid unless a certificate of analysis accompanies every batch, mostly because that provides traceability. In my experience, batch-to-batch consistency trumps a few tenths of a percent on the COA, so suppliers working in the pharmaceutical field usually go out of their way to document controls.
Facts Back Up the Hype
This is not just about nitpicking. Publications from sources like the European Pharmacopoeia or USP typically demand greater than 98% purity for L-Aspartic Acid derivatives, including the diethyl ester hydrochloride. Analytical testing using chromatography, titration, and spectroscopy backs up these claims. Manufacturer fact sheets reveal that reputable suppliers not only meet these numbers but also publish data on potential residual solvents or heavy metals. I have checked several supplier sheets during spec evaluations; most top suppliers easily hit or surpass the 98% mark.
Possible Solutions for Purity Issues
Problems sometimes come up—occasional impurities by synthesis shortcuts, inadequate drying, shipping conditions—so quality control is more than a rubber stamp. Companies can invest in better purification steps, sometimes recrystallization or advanced chromatography. Third-party audits keep suppliers honest. In some research labs, we have re-tested purchased lots to make sure no corners were cut, particularly when scaling up.
Why Not Go Lower?
Cutting the purity to save cost is a false economy. Lower grades might work for some industrial applications, but for anything involving biological or pharmaceutical work, the risk isn't worth it. Any impurity could either block reactions, cause toxicity, or ruin months of experiments. That’s no small sum when you consider the cost of wasted time.
Sticking to Science and Transparency
Lots of places can supply this compound, but the recurring message from every credible pharmacist, researcher, and chemical supplier I’ve met: verify purity, ask for the documentation, and don’t fall for vague promises. Whether the project is small scale or an industrial run, the level of scrutiny on purity tells you how seriously the supplier—and the end user—take their science.
How should L-Aspartic Acid Diethyl Ester Hydrochloride be dissolved and prepared for experiments?
Getting the Basics Right
If you’ve ever stood at the bench eyeing a bottle labeled “L-Aspartic Acid Diethyl Ester Hydrochloride,” chances are you’ve wondered what’s the straightforward way to get this compound into solution, clear and ready for your next protocol. From experience, things rarely go smoothly if you rush ahead. Once, I added a new compound like this straight into cold water—clumps everywhere, cloudy with undissolved chunks, and a wasted hour repeating the prep. Taking time to understand a compound pays off.
Choosing a Solvent
This hydrochloride salt dissolves best in polar solvents. For most experiments, distilled water or saline works well, provided you don’t go overboard with the concentration. Going higher than 10-20 mg/mL often leads to stubborn precipitates floating at the top. If water alone doesn’t do the trick, tweaking with a little ethanol helps, but keep it to a minimum to avoid unwanted effects on cell cultures or bioassays. Reading the compound’s SDS recommends water as a primary go-to, and PubChem notes solubility in both water and alcohol.
Weighing and Handling
Some mistakes stick. Years ago, I measured out a compound with an open spatula while the lab fans were running. The powder scattered. L-Aspartic Acid Diethyl Ester Hydrochloride is a fine powder, quick to leap in a draft. Always close the bottle fast and weigh the sample near the balance, away from busy traffic. For accurate results, the powder should go directly into a clean glass beaker or flask, not onto random surfaces that can trap residue.
Mixing and Dissolving
Patience with stirring beats brute force. Gentle mixing at room temperature or in a warm water bath (no higher than body temp) does more than hitting it with a vortex for ten minutes. Too much agitation can introduce bubbles or throw off pH, especially if you mix in a buffer. If the solution stays cloudy, a bit of gentle heating (up to 37°C) breaks stubborn crystals, but don’t boil. Heat can change chemical structure if it gets too aggressive.
pH Matters More Than You’d Think
L-Aspartic Acid derivatives are touchy about pH. The hydrochloride raises the acidity, and downstream reactions or cell work often demand near-neutral conditions. Always measure pH after dissolving and adjust slowly with sodium hydroxide or hydrochloric acid as needed. I’ve lost valuable work to pH drift—cells behave differently, enzymes lose pep, and results suddenly look strange, all because I skipped this check.
Filtration and Storage
Particularly for experiments with cells or proteins, microfiltration (0.22 μm filter) takes out dust and undissolved bits that can mess with sensitive assays. Solutions stay most stable in amber bottles at 4°C, away from light. In my own routine, clear labeling of the date and concentration stops confusion down the line. Chemical breakdown creeps up over weeks. Using freshly prepared solution always gives cleaner, more reliable results.
Staying Safe and Consistent
Lab safety and good documentation never go out of style. Gloves, eye protection, and clear notes in your lab book may seem basic, yet those steps head off headaches from spills, exposure, or forgotten details about pH or concentration. L-Aspartic Acid Diethyl Ester Hydrochloride isn’t as risky as some corrosive acids or toxic chemicals, but it deserves respect and smart habits—especially since your data depends on that first prep going right.
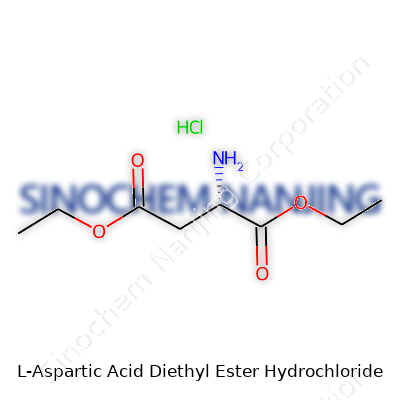
| Names | |
| Preferred IUPAC name | diethyl (2S)-2-aminobutanedioate hydrochloride |
| Other names |
Diethyl L-aspartate hydrochloride L-Aspartic acid diethyl ester HCl L-Aspartic acid, diethyl ester, hydrochloride L-Aspartic acid diethyl ester monohydrochloride |
| Pronunciation | /ɛl æˈspɑːrtɪk ˈæsɪd daɪˈɛθɪl ˈɛstər ˌhaɪdroʊˈklɔːraɪd/ |
| Identifiers | |
| CAS Number | [128941-15-1] |
| Beilstein Reference | 1720946 |
| ChEBI | CHEBI:131266 |
| ChEMBL | CHEMBL559872 |
| ChemSpider | 167351 |
| DrugBank | DB08343 |
| ECHA InfoCard | 100.143.033 |
| EC Number | 6342-14-7 |
| Gmelin Reference | 126640 |
| KEGG | C06160 |
| MeSH | D020155 |
| PubChem CID | 61360 |
| RTECS number | AR8400000 |
| UNII | V2H4G36E5D |
| UN number | 2811 |
| CompTox Dashboard (EPA) | DJRXHEGKBWWFDU-UHFFFAOYSA-N |
| Properties | |
| Chemical formula | C8H16ClNO4 |
| Molar mass | 209.67 g/mol |
| Appearance | White to off-white solid |
| Odor | Odorless |
| Density | 1.01 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -2.08 |
| Vapor pressure | 0.0 mmHg at 25°C |
| Acidity (pKa) | 3.9 |
| Basicity (pKb) | 12.82 |
| Magnetic susceptibility (χ) | -51.2×10^-6 cm³/mol |
| Refractive index (nD) | 1.4390 |
| Dipole moment | 10.06 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 387.9 J/mol·K |
| Std enthalpy of combustion (ΔcH⦵298) | -1933 kJ/mol |
| Pharmacology | |
| ATC code | A16AA08 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. Causes skin irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | No Hazard Statements |
| Precautionary statements | Precautionary statements: P261, P305+P351+P338 |
| Flash point | 102.6 °C |
| LD50 (median dose) | LD50: 5000 mg/kg (Rat, Oral) |
| NIOSH | RN:67642-97-3 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 1 gram |
| IDLH (Immediate danger) | Not listed |
| Related compounds | |
| Related compounds |
L-Aspartic acid L-Aspartic acid dimethyl ester L-Glutamic acid diethyl ester hydrochloride DL-Aspartic acid diethyl ester hydrochloride L-Aspartic acid beta-methyl ester hydrochloride |

