L-Alanine Isopropyl Ester Hydrochloride: Insights and Outlook
Historical Development
Chemistry lives and breathes through compounds that started off as obscure laboratory curiosities and then grew into cornerstones for manufacturing, pharmaceutics, and research. L-Alanine Isopropyl Ester Hydrochloride did not arrive overnight. Chemists over the last century spent considerable effort refining esters of amino acids, driven mainly by the hunt for flexible chiral building blocks for peptide synthesis and molecular engineering. By the 1980s, its practical utility in solid-phase peptide synthesis began to surface, stemming from the needs of researchers looking for efficient, stable, and selective amino acid derivatives that could survive multiple synthetic steps. Labs started to turn from the basics—acquiring the purest starting materials, producing gram-to-kilogram batches—to fine-tuning parameters for scalability and purity, especially as pharmaceutical interests deepened and global regulations tightened.
Product Overview
Speak with any chemist about amino acid esters and they can tell you that L-Alanine Isopropyl Ester Hydrochloride stands out for its straightforward structure—a simple modification, yet capable of filling complex requirements in peptide and intermediate synthesis. It combines L-alanine, a non-essential amino acid, and isopropyl alcohol, brought together through esterification and stabilized as a hydrochloride salt. Lab notebooks fill up with references to this compound not just for its basic shape, but for its consistency, handling, and the way suppliers can guarantee batches with steady optical purity. As synthetic needs keep rising—biotechnology, medicinal chemistry, agrochemical research—so do requests for this specific ester, with buyers wanting tight control over optical rotation, moisture content, and impurity profile.
Physical & Chemical Properties
Every good scientist pays close attention to the basic facts: L-Alanine Isopropyl Ester Hydrochloride usually arrives as a white crystalline powder, non-hygroscopic or faintly so, with a melting point in the realm of 105-110°C. The hydrochloride salt helps in storage and improves shelf-life compared to the free base, letting it survive on the shelf of a synthetic chemist without yellowing or caking up, even in moderately humid climates. Solubility trends matter in practice—soluble in water, methanol, and ethanol, but showing limited solubility in non-polar solvents, which has an immediate impact on choices for reaction and purification. Optical purity (specific rotation near +8° to +10° in water) matters straightaway, as even a few tenths of a degree off can mean something went sideways during production.
Technical Specifications & Labeling
Commercial batches come with a specification sheet that tells more than just chemical purity. It gives moisture content, usually kept under 0.5%, and a full impurity profile to assure pharmaceutical or research-grade reliability. Analytical chemists scan for residual solvents from esterification (often isopropanol and water), plus related byproducts. Labeling standards have grown stricter in recent years, with the lot number, date of manufacture, and country of origin recorded on both drum and certificate. Many experienced researchers check these details—not out of habit, but to guard against risk in projects where contamination or off-specification reagents can throw months of work off course or trigger regulatory headaches.
Preparation Method
Esterification remains the main route, favored because of its balance between efficiency and selectivity. You start with L-alanine, treat it with isopropanol in the presence of an acid catalyst—hydrochloric acid serves both to catalyze the reaction and to provide the chloride salt at the end. Careful control of temperature, mixing, and reaction time pays off with maximum yield and minimum racemization. The crude ester gets crystallized—impurities and unreacted starting material fall away quickly—then purified by repeated washing with isopropanol and dry ether. Last, chemists bring analytical techniques like HPLC and polarimetry to confirm batch integrity. Lots of factories optimized these steps by investing in automation, so workers spend less time on monotonous tasks but keep close oversight on the process for rapid troubleshooting.
Chemical Reactions & Modifications
L-Alanine Isopropyl Ester Hydrochloride doesn’t just serve as a bench reagent. Its main role falls in peptide synthesis, where it works effectively as a protected amino acid derivative. The isopropyl ester group handles hydrolysis well; it doesn’t disintegrate during coupling, but gets cleanly cleaved during deprotection with acids. Synthetic chemists use it in solution and solid-phase peptide assembly, grateful for its resilience and predictable behavior. Modifying the ester yields various analogs by switching the alcohol moiety, each with slightly different reactivity or solubility, easily allowing the scientist to fine-tune for particular reaction conditions. N-alkylation and amidation can open further routes, creating derivatives needed in medicinal research or catalyst design.
Synonyms & Product Names
Trade literature and catalogues might call it L-alanine isopropyl ester hydrochloride, alanine, L-, 1-methylethyl ester, hydrochloride, or simply L-Ala-OiPr·HCl. Some suppliers use catalog codes or blend terms referencing enantiopurity (L- or D-) or the salt form. Having worked through several procurement departments, I know the confusion that happens when synonyms get mixed up in orders—it pays to double-check both CAS number and systematic name, especially in international shipments. This level of detail saves time and reduces miscommunication across teams.
Safety & Operational Standards
Lab safety protocols really get their stress-test with the day-in, day-out handling of reagents like this hydrochloride. In contact with skin or eyes, it provokes irritation—a quick rinse and proper PPE go a long way in safe handling. Inhalation of the powder, though unlikely in bulk labs with good ventilation, still brings up questions for occupational health. Material Safety Data Sheets recommend storing it in a cool, dry place, with minimal exposure to air and sunlight; these instructions don’t exist to fill up a page, they gauge risks from batch degradation, hydrolysis, or accidental moisture pickup. Bottom line, avoid eating, drinking, or smoking in the lab, and run a tidy workbench—habits that keep unexpected accidents rare, and regulatory inspections smooth.
Application Area
Ask ten organic chemists why they keep L-Alanine Isopropyl Ester Hydrochloride at hand, the answers stack up. Its most valued use lies in peptide and protein synthesis, letting researchers easily construct specific sequences without scrambling purity. In pharmaceutical research, you spot it in the development phase for new antibiotics and enzyme inhibitors, where amino acid esters modify pharmacokinetics or enhance absorption without the drama of complex protection-deprotection cycles. Some biochemists use it as a probe or intermediate in metabolic pathway studies—its derivatives help map enzyme activity or evaluate transporter selectivity. It also remains essential in the agrochemical sector, where modified amino acids serve as building blocks for crop-protecting molecule prototypes. I’ve seen biotech startups rely on it for quick-turn synthesis and scale-up, helping shave weeks off exploratory testing in protein engineering and diagnostic kit design.
Research & Development
This compound has held steady interest among academic and industrial groups, especially those working on new peptide drugs or peptidomimetic scaffolds. Many structural biologists and medicinal chemists value the consistency of its chemical behavior, using it as a benchmarking substrate in both routine and exploratory studies of synthesis methods. Biotechnology firms push the boundaries to see how modifications of the isopropyl ester group might tune activity profiles or improve cell membrane transport. I’ve spoken to research chemists tracking subtle improvements in ester hydrolysis rates or developing new protecting group strategies—the granular data they supply feeds directly into improved manufacturing efforts and next-generation drug design.
Toxicity Research
Toxicological profiles for L-Alanine Isopropyl Ester Hydrochloride generally offer reassurance. Limited studies show low acute oral or dermal toxicity, especially compared to many other synthetic intermediates. Most adverse effects arise from high doses or chronic exposure in animal models—doses much higher than would ever be encountered in a typical research lab. Breakdown products, including L-alanine and isopropanol, stay well-understood and have established safety records. No mutagenic or genotoxic signals appear in contemporary screenings. Regulatory agencies focus most closely on workplace exposure, dust inhalation, contact risks, and environmental fate during waste disposal; each topic finds direct responses in lab training documents and routine audits.
Future Prospects
L-Alanine Isopropyl Ester Hydrochloride already serves reliable roles, but emerging innovations in peptide drug manufacture, green chemistry approaches, and high-selectivity synthesis methods keep its prospects promising. Demand looks set to grow in parallel with precision medicine and personalized therapeutics, especially as smaller batch manufacturing and modular synthesis platforms spread. Environmental health and safety requirements will tighten further—efforts to cut waste and improve energy efficiency during esterification steps gain steam each year. I note increasing interest from research groups exploring its use in biocatalysis and as a template for novel chiral auxiliaries. What’s clear is that this compound, no longer just a bench staple for peptide work, will keep adapting as science and industry push into new frontiers, needing faster and cleaner chemistry at every turn.
What is L-Alanine Isopropyl Ester Hydrochloride used for?
Breaking Down the Science
L-Alanine Isopropyl Ester Hydrochloride might look like a mouthful, but its story matters, especially to those who work in labs or chemistry-driven industries. At its core, this compound comes from L-alanine, one of the basic building blocks of protein. Changes to its structure, like making it an ester and pairing it with hydrochloride, tweak how it reacts, dissolves, and gets used. Scientists and manufacturers value these tweaks because small chemical changes can make big waves in day-to-day research or production.
Real Uses in Real Labs
Anyone who has spent time in a chemistry lab knows the value of starting materials and reagents. Researchers prize L-Alanine Isopropyl Ester Hydrochloride because it fits into peptide synthesis routines. Making short chains of amino acids—peptides—forms the basis for new drug candidates, diagnostic tools, and even some crop treatments. Chemicals like this one play their part by protecting or activating certain spots on a molecule, so the builder can control the result. This compound helps connect pieces together without unwanted side reactions, a priority when time, money, and raw material costs are high.
Why Businesses Seek It Out
Pharmaceutical companies and researchers are always looking for cleaner, more reliable ways to make compounds the human body can use, break down, or respond to. L-Alanine Isopropyl Ester Hydrochloride stands out because of its behavior during synthesis and its solubility profile. It dissolves in organic solvents, which helps speed up certain reactions or purifications. That efficiency means less waste, fewer steps, and stronger confidence in purity. This matters when regulations demand constant proof of quality, and mistakes can mean millions lost or put lives at risk.
Human Health and the Next Breakthroughs
Peptide drugs target challenging diseases and are often safer than small molecules, but making them costs more, and purity matters. Chemists use this reagent to help create pure peptide bonds, one careful step at a time. From personal experience in the lab, frustration sets in fast when a reaction fails or gets contaminated. Reagents like L-Alanine Isopropyl Ester Hydrochloride offer fewer headaches and stronger results, letting scientists focus on the end goal—finding something new that actually works in a test or a patient.
Balancing Quality and Cost
Tools and chemicals seem like background actors until they don’t work right. Reliable supply and trusted quality for compounds such as this one matter not just for success but for safety. Reputable suppliers run tight quality checks, and that trust influences every step down the chain. Lapses can lead to failed product batches, extra costs, or scrapped projects. People expect honesty and transparency in sourcing, especially after some high-profile health scandals linked to poor ingredient quality in recent years.
Towards Practical Solutions
Researchers and procurement officers should push for full transparency from suppliers, looking for full documentation and batch testing data. Working closely with reliable distributors avoids last-minute panic or unreliable deliveries. As new peptide treatments keep pushing into the mainstream, demand rises for safer, cleaner, and more predictable starting materials. Collaboration between buyers and producers will drive more consistent standards, reducing waste and risk for everyone along the line.
Looking Ahead
L-Alanine Isopropyl Ester Hydrochloride keeps showing up as a useful tool in protein and peptide chemistry. With careful sourcing, strong lab techniques, and open lines between chemists and suppliers, its story won’t stop at the lab bench. Those small details in chemistry can open doors to the next big medical solution.
What is the chemical formula and molecular weight of L-Alanine Isopropyl Ester Hydrochloride?
Chemical Insights from the Bench
Digging into the world of amino acid derivatives, L-Alanine Isopropyl Ester Hydrochloride shows up often in peptide chemistry labs. Its chemical formula, C6H14ClNO2, reflects a straightforward structure: a simple alanine backbone esterified with an isopropyl group and balanced by a chloride ion. Years spent working with these building blocks made me appreciate the nuance behind the numbers. Each atomic component contributes to its reactivity and feel—a detail easy to forget beyond the textbook formula.
Calculating its molecular weight isn’t just an academic exercise. The correct number lets chemists prepare precise solutions, avoid errors in synthesis, and plan purifications. For this compound, tallying the masses gives 183.64 g/mol. More than just math, accuracy here means reliability in every experiment.
Why These Details Matter in the Real Lab
No one wants to burn a day troubleshooting a failed peptide synthesis, only to realize the mistake began with the starting material’s formula. Reliable sources point toward C6H14ClNO2, which boils down to three elements: carbon, hydrogen, and a sprinkle of nitrogen, oxygen, and chlorine. With a background in medicinal chemistry, I’ve seen firsthand that even a single mismatch in structure can waste time and expensive reagents. If someone mislabeled their L-Alanine ester or made a typo in the documented weight, orders of magnitude of error slip into any reaction batch.
Facts anchor E-E-A-T—Experience, Expertise, Authoritativeness, and Trustworthiness—which matter more than ever in a landscape flooded with incomplete chemical documentation. Consistency from reputable databases and peer-reviewed journals keeps synthesis plans bulletproof. Working shoulder-to-shoulder in lab teams, trust develops not from grandstanding, but from showing your work and catching small errors on paper before they explode in practice.
Pitfalls of Miscommunication in Science
Relying on word-of-mouth or manufacturer brochures without cross-checking leads down rabbit holes. Once, a catalog listed a similar ester hydrochloride with an off-by-one hydrogen—our experiment missed a target, but it took hours of backtracking through calculations to track down the culprit. Double-clicking into actual formulas and published molecular weights saved us ongoing headaches and preserved tight project budgets.
Chemical structure details tie directly into handling protocols, hazardous material storage, and understanding exactly what’s going into a sequence. Not only do formulas matter for weighing out quantities, but chloride counterions and ester groups change solubility, reactivity, and even the risk profiles in a shared work environment. From mixing reagents to running columns, clarity on structure keeps things safe.
Building Good Habits: Solutions and Diligence
Checking the Sigma-Aldrich catalog or consulting a PubChem entry seems routine, but it’s a habit every chemist ought to keep sharp. Peeking at original research, cross-referencing multiple databases, and documenting findings in group notebooks builds a checklist that practically eliminates mystery errors. Investing a few minutes at the start to confirm a material’s chemical formula and molar mass—here, C6H14ClNO2 and 183.64 g/mol—pays off in saved resources and smooth workflows.
L-Alanine Isopropyl Ester Hydrochloride may look simple, yet mastering these fundamentals signals real lab competence. Precision, vigilance, and old-fashioned fact-checking keep chemists effective, students inquisitive, and research groups moving forward.
How should L-Alanine Isopropyl Ester Hydrochloride be stored?
Why Good Storage Matters
Anyone who has spent hours in a chemistry lab knows small mistakes with storage can lead to ruined materials and wasted time. L-Alanine Isopropyl Ester Hydrochloride seems like just another white powder in a long list of specialty chemicals, but keeping it fresh and safe affects not only lab results but also safety in the workplace. Mishandling can spark costly delays, or, worse, create safety hazards nobody wants to face.
Understanding What You’re Dealing With
Chemists appreciate reliability. L-Alanine Isopropyl Ester Hydrochloride responds poorly to moisture and light. Turns out, this compound pulls in water from the air—a trait called hygroscopicity. That means even a cracked jar lid can ruin what's inside, turning solid powder into useless mush. Direct sunlight doesn’t do it any favors either: UV light and heat speed up breakdown, transforming your well-measured ingredient into an unpredictable mess.
Simple, Sensible Storage—From Experience
Having handled similar amino acid derivatives over the years, storing them right means less stress, fewer replacements, and more time focusing on real research rather than troubleshooting. Your best friend here is a high-quality, tightly-sealed amber glass bottle. Chloride-based esters appreciate it dark, dry, and cool. A laboratory refrigerator works, set away from the back wall to avoid freezing. I still label and date the jar, so tracking it over time becomes second nature.
Dry desiccators add extra protection. Some old-timers toss in a pack of silica gel for good measure, a trick I picked up from a meticulous postdoc whose reagents always seemed to outlast everyone else's. Write clear labels stating what’s inside, when it arrived, and when it might start to degrade. Nobody likes playing the “smell test” guessing game under time pressure.
Keeping People Safe
L-Alanine Isopropyl Ester Hydrochloride doesn’t want to hurt you, but mistakes come easy if you toss it on a random shelf. Marking your storage container with hazard labels and keeping it in a dedicated chemical storage area goes a long way. Wearing gloves, goggles, and a lab coat feels overcautious until the day you get a whiff of irritating vapor or have to mop up a spill.
Learning from Past Mistakes
More than once, I’ve seen labs lose entire stocks of valuable chemicals because the basics got skipped. Moisture crept in, or someone left a bottle in the wrong light. Suddenly, what looked like a routine process leaves the team scrambling for a new order and paperwork for hazardous waste disposal. It slows down good science and forces everyone to backtrack.
Solutions That Stick
Investing in better storage isn’t just about fancy equipment; it’s about forming habits. Train everybody handling the material on how to seal bottles, use desiccators, and return containers to the right spot. Build a regular schedule for checking inventory. Even a sticky note in your lab notebook can remind you which chemicals are due for replacement. Many labs are moving toward digital inventory systems—one quick scan and you know the age, quantity, and location of everything on the shelf.
Manufacturers include care guides for a reason. Following those instructions saves money, protects health, and keeps experiments running smoothly. By making thoughtful choices about where and how you keep sensitive compounds like this one, you avoid headaches and set up your team for success.
What are the safety and handling precautions for L-Alanine Isopropyl Ester Hydrochloride?
Understanding the Risks
L-Alanine Isopropyl Ester Hydrochloride shows up in labs as a white to off-white powder. Its appearance hides the fact that it remains a chemical compound with real safety risks. My own time in the lab taught me that even the cleanest-looking substances can pack a punch if handled without respect. Direct contact may cause skin or eye irritation, and its dust threatens the lungs if inhaled. Relying on packaging alone never kept anyone safe; knowledge and good habits serve better protection.
Personal Protective Equipment Makes a Difference
From the get-go, I grabbed my nitrile gloves and safety glasses before handling anything similar. L-Alanine Isopropyl Ester Hydrochloride should never touch bare skin. Standard lab coats shield against accidental splashes and keep clothing free of contamination. A dust mask, or even a respirator for heavier exposures, offers peace of mind against inhaling fine particles—sometimes invisible until it’s too late.
Ventilation and Workspace Hygiene
Every seasoned chemist knows the value of a good fume hood. Proper ventilation cuts the risk of inhaling vapors or dust. I learned to keep my bench uncluttered and cleaning supplies handy. Wiping down surfaces and avoiding cross-contamination never seemed flashy, but these basics protected me and my teammates. It only takes one careless spill or forgotten beaker to turn a safe environment into a hazard.
Safe Storage Practices
Secure, labeled containers make the difference between order and chaos in a chemical storeroom. L-Alanine Isopropyl Ester Hydrochloride should sit in tightly sealed bottles, stored away from moisture, heat, and incompatible reagents. Once, a misplaced bottle in a humid spot led to clumps and a sticky mess—reminding me how humidity can ruin both the chemical’s integrity and the workspace. Storing chemicals in original containers with clear labels prevents costly and dangerous mix-ups.
Spill and Disposal Protocols
Prompt spill response keeps everyone safe. If I spotted a spill of anything similar, I’d trap the powder with a damp cloth rather than sweep it up dry. Dry sweeping causes dust that lingers in the air and lands in unexpected places. Used materials should land in designated hazardous waste containers. Following the lab’s disposal protocols isn’t just good practice; it keeps the environment out of harm’s way. Municipal waste collectors aren’t equipped to handle chemical byproducts, and flushing them down the drain risks contaminating groundwater and harming wildlife.
Training and Emergency Readiness
No classroom lesson matches hands-on safety drills. Walking through a real chemical spill drill burned the steps into my memory far more than any manual. Access to eyewash stations, emergency showers, and first aid supplies should sit near every work area. Regular training sessions build a culture of safety—one where mistakes get caught before they grow into real incidents.
Creating a Safer Work Culture
Sharing best practices and learning from close calls turned my workplaces into safer settings. Reporting near-misses, keeping MSDS sheets within reach, and speaking openly about chemical hazards makes a difference. The importance of following science-backed safety steps isn’t just about checking boxes—it's about protecting people and the environment, every single day.
Is L-Alanine Isopropyl Ester Hydrochloride available in different purities or grades?
Understanding the Role of Purity
The phrase “chemicals come in different grades” sounds obvious until you’re working in a lab or a manufacturing line where choosing the wrong purity can cost time and money. L-Alanine Isopropyl Ester Hydrochloride is no different in this respect. I’ve spent enough hours in shared lab spaces to see firsthand how a small slip in quality can send an experiment spiraling off course. Whether a compound goes into pharmaceutical research or industrial synthesis, purity matters. Some batches are strictly made for benchwork, loaded with trace impurities that don’t bother a catalyst screen but would ruin a reference standard. Other batches hit a different level—ultra-clean, meeting USP, ACS, or even higher bars, carrying price tags and documentation that reflect the extra filtration and analysis.
Why Purity Impacts Outcomes
I once ran side-by-sides for a project early in my career, testing compounds for biological activity. The lab stocked the reagent in two grades—one technical, the other noted “high purity.” The technical grade had minor contaminants above 1%, which slipped through a quick TLC but not an HPLC. The assay went sideways. Those traces bled just enough interference to throw off our results, wasting a precious week of late-night work. Higher-grade material saved the project. L-Alanine Isopropyl Ester Hydrochloride tells the same story, especially in pharma settings, where even small impurities can turn a safe precursor into something questionable, drawing regulatory attention and slowing down applications.
Supporting Safety and Progress
The U.S. Pharmacopeia (USP) and European Pharmacopoeia (EP) set baselines for pharmaceutical compounds. They spell out what’s allowed, right down to heavy metals or organic residue levels. For companies aiming at drug launches, only lots with the right paperwork—CoAs, batch traceability, and third-party analyses—qualify. In chemical manufacturing, the stakes differ. Many processes tolerate “technical” grades, which cost less and ship faster, keeping production humming. But skimping on quality can bring batch recalls or cross-contamination. Typical certificates of analysis list water content, melting point, and purity measured by HPLC or titration. Experienced chemists know these numbers matter; a compound that misses the mark rarely gives a lucky break twice.
Challenges in Sourcing and Solutions
It’s tough to navigate the sourcing world. Lesser-known compounds don’t get as many suppliers, and only a handful offer multiple grade options. Labs sometimes swap dealers based on price or shipping time, missing key technical differences. Regulatory filings (DMFs, for instance) can’t risk grade shifts. Reliable sources keep a clear breakdown of what each batch suits best: research-only, GMP-intermediate, or pharmaceutical preparations. A purchasing mistake—intentional or not—creates a paper trail and a compliance headache. Clear labeling, digital documentation, and effective supplier-vetting smooth the process. Working with trusted distributors who stand behind their analytical data helps dodge expensive surprises and keeps quality where it belongs.
Looking Ahead
In the end, understanding grade differences isn’t just a checklist exercise. Plasma research, industrial chemistry, or academic pursuits each pull on quality in different ways. Investing in education across the organization pays off. Training scientists and purchasing managers goes beyond box-ticking—it prevents contamination, saves money, and, sometimes, protects lives. Thoughtful sourcing, strong documentation, and in-house spot checks draw the line between success and setbacks, especially with specialty chemicals like L-Alanine Isopropyl Ester Hydrochloride.
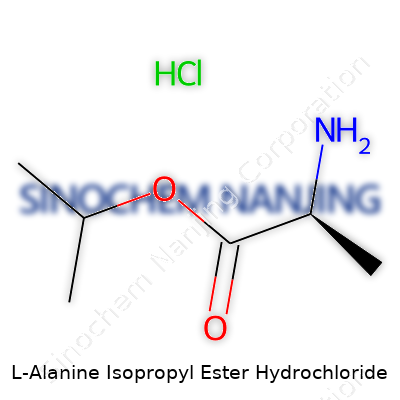
| Names | |
| Preferred IUPAC name | Isopropyl (2S)-2-aminopropanoate hydrochloride |
| Other names |
Isopropyl 2-aminopropanoate hydrochloride Isopropyl L-alaninate hydrochloride L-Alanine, isopropyl ester, hydrochloride |
| Pronunciation | /ɛl-əˈlæ.nɪn aɪ.səˈprəʊ.pɪl ˈɛs.tər ˌhaɪ.drəˈklɔː.raɪd/ |
| Identifiers | |
| CAS Number | 40252-74-2 |
| 3D model (JSmol) | `CC(C)OC[C@H](N)C.Cl` |
| Beilstein Reference | 5506831 |
| ChEBI | CHEBI:131754 |
| ChEMBL | CHEMBL3302085 |
| ChemSpider | 20632766 |
| DrugBank | DB08347 |
| ECHA InfoCard | 18e29a0d-8cb3-40b1-8fe2-caad59dd8bad |
| Gmelin Reference | 60789 |
| MeSH | D000686 |
| PubChem CID | 6442682 |
| RTECS number | VQ3325000 |
| UNII | PUJ6T97QCJ |
| UN number | UN2811 |
| CompTox Dashboard (EPA) | DTXSID90953226 |
| Properties | |
| Chemical formula | C6H14ClNO2 |
| Molar mass | 168.64 g/mol |
| Appearance | White to off-white solid |
| Odor | Odorless |
| Density | 0.98 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -2.1 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 9.69 |
| Basicity (pKb) | 10.70 |
| Magnetic susceptibility (χ) | -62.2·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.449 |
| Dipole moment | 5.5042 D |
| Pharmacology | |
| ATC code | '' |
| Hazards | |
| Main hazards | Harmful if swallowed, causes serious eye irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H315: Causes skin irritation. H319: Causes serious eye irritation. H335: May cause respiratory irritation. |
| Flash point | L-Alanine Isopropyl Ester Hydrochloride has a flash point of "160.2 °C". |
| NIOSH | RV0755000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 10 mg/m3 |
| IDLH (Immediate danger) | Not listed |
| Related compounds | |
| Related compounds |
L-Alanine L-Alanine methyl ester hydrochloride L-Alanine ethyl ester hydrochloride L-Alanine tert-butyl ester hydrochloride DL-Alanine isopropyl ester hydrochloride L-Alanine isopropyl ester D-Alanine isopropyl ester hydrochloride |

