Isopropyl Propionate: A Closer Look at a Quiet Workhorse in Modern Chemistry
Historical Development
The rise of isopropyl propionate in the global chemical market connects to decades of refining organic ester production. Chemists in the early 20th century noticed a growing need for solvents and intermediates combining good volatility with mild odor and safety. From my perspective, the shift from more hazardous solvents, like diethyl ether or chlorinated hydrocarbons, drove research into esters such as isopropyl propionate. Industrial chemists realized its value especially in the post-war years, when paint, coating, and cosmetic industries were hungry for cleaner, less pungent solvents. As demand for versatile, safe-to-handle solvents grew, manufacturers adjusted their facilities to favor esters, and isopropyl propionate started driving both research and sales.
Product Overview
Anyone working in a paint, fragrance, or flavor laboratory probably spotted isopropyl propionate at some point. This colorless liquid ester forms from isopropyl alcohol and propionic acid. The result: a compound balancing pleasant scent, rapid evaporation, and easy compatibility with all kinds of organic materials. Its modest boiling point and lack of strong odor make it a helpful agent for blending fragrances or formulating specialized coatings. Speaking with colleagues in formulation, this ester comes up as an effective fixative in perfumes, or a mild solvent in sun care products, usually outshining alternatives whenever a lighter hand is needed.
Physical & Chemical Properties
Isopropyl propionate stands out because it manages to dissolve many organic substances without creating a harsh working environment. It usually appears as a clear, mobile liquid with a mild, fruity odor, boiling somewhere above 100°C depending on purity. Its density hovers slightly below that of water, and it doesn’t mix with water but mixes well with most common organic liquids. Its low surface tension, easy spreadability, and limited reactivity help in applications ranging from coatings to cosmetics. I’ve seen lab techs appreciate the fact that it evaporates quickly but doesn’t linger in the air, unlike some more aggressive solvents. This characteristic keeps workspaces comfortable—a little thing that matters over long hours.
Technical Specifications & Labeling
When shopping for or formulating with isopropyl propionate, technicians expect a specification sheet listing its minimum purity, water content, and acceptable acidity and color. Packaging typically lists the chemical name, any hazard warnings (mainly flammable liquid and vapor), and recommended storage. It’s sold in steel drums or HDPE containers, sealed to prevent moisture uptake or evaporation. Barcode labeling, batch numbers, and country of origin have become standard, partly to satisfy traceability demands from regulators. Walking into any material storage area, one can spot these pale-blue or silver drums marked for safe storage away from heating elements.
Preparation Method
Industrial production leans on esterification, a reaction of isopropyl alcohol with propionic acid, typically performed under acidic conditions. Large reactors combine these inputs at moderate temperatures, using acid catalysts like sulfuric acid. Manufacturers monitor water content throughout the process to drive the reaction toward completion, then separate the crude product using distillation. This routine draws from the classic approach chemists use in the lab—mix, heat, eliminate water, distill—but scaled for drums, not glassware. Over the years, producers experimented with continuous processes and water removal techniques to boost yields and lower costs, reflecting a broader industry shift toward greener, more energy-efficient operations.
Chemical Reactions & Modifications
Most labs treat isopropyl propionate as fairly inert. Under typical storage and use conditions, it resists reacting, making it a stable option for production lines or bench chemists. Exposure to strong acids or bases can break it back down into isopropyl alcohol and propionic acid. In some niche syntheses, researchers alter it further, especially if they’re seeking to introduce new functional groups or chains into the molecule. Yet compared to more reactive esters or ethers, it mostly serves as a carrier—not a star player in complex reaction networks.
Synonyms & Product Names
The chemical often appears under alternate names such as isopropyl propanoate, propionic acid isopropyl ester, or 1-methylethyl propionate. Catalogs from suppliers list these alongside their CAS numbers, letting buyers match the material to required standards and applications. For those new to the compound, the variety of names can confuse, but anyone working in formulation soon recognizes them as one and the same.
Safety & Operational Standards
Every practical chemist pays close attention to solvent safety. Isopropyl propionate, while not especially toxic, deserves basic handling precautions: avoiding open flames, providing solid ventilation, and wearing splash protection. Storage follows the same logic as most organic solvents—cool, dry, well-ventilated rooms with tightly sealed drums. Over the last decade, I’ve seen more labs move to explosion-proof storage lockers and limit stock to prevent fire hazards. Operating guidelines suggest working with small amounts under fume hoods, especially in schools or smaller-scale companies, where expertise and equipment may be limited.
Application Area
This ester’s real strengths show in its daily uses. In personal experience, formulators return to it for perfumes, room sprays, and paints, as it can carry volatile oils and dissolve resins without heavy scent or lingering residue. The food industry uses it in trace quantities for flavor production, relying on both regulatory approvals and strict batch controls to guarantee safety. Nail polish, sunscreen, and inks often feature it for its ability to dissolve and spread organic components smoothly. Its mildness suits skin-contact products, making it more appealing than rougher solvents like acetone or toluene. Professional artists and craftsmen also value it because, unlike fast-evaporating solvents, it gives them just enough working time before drying.
Research & Development
Some researchers continue to push boundaries, exploring tweaks to the esterification process or testing new catalysts aimed at lower waste and higher purity output. In academic settings, chemists look for ways to optimize yield, energy demands, or even to produce isopropyl propionate from renewable feedstocks such as corn-based isopropanol. R&D efforts also test compatibility with next-generation resins or explore novel uses—in my network, people in biotechnology have run pilot studies on ester-based extractants or specialized delivery systems for pharmaceuticals. The quest often circles back to balancing safety, cost, and performance for industrial customers.
Toxicity Research
Compared to many industrial solvents, isopropyl propionate enjoys a decent safety record. Acute toxicity studies suggest low hazard from skin or inhalation exposure at the concentrations and durations encountered during typical factory or laboratory work. Regulatory agencies keep tabs on environmental persistence, too, as improper disposal could threaten waterways. Teams working on safety testing focus on chronic exposure scenarios, with animal model data showing only mild effects at relatively high doses. Safety data sheets mention eye and skin irritation in concentrated form, so gloves and goggles remain standard kit. As health standards rise worldwide, firms now re-examine legacy solvents and keep looking for even gentler alternatives in the ester family.
Future Prospects
Isopropyl propionate stands at an interesting crossroads. Changing consumer expectations push for both gentler ingredients and greener production. As groups seek nontoxic, rapidly biodegradable solvents, demand for simple esters should keep rising. The ongoing refinement of renewable chemical processes holds promise for biobased isopropyl propionate, which could help makers label products as sustainable. Regulatory limits on solvent emissions will challenge the industry to reduce waste and innovate at every step. My bet is that we will see isopropyl propionate hold its ground, especially where human contact or environmental worries rule out older, harsher chemicals. Upcoming research may even open new roles in drug delivery, fine chemical synthesis, or advanced coatings, keeping this quiet workhorse relevant for years to come.
What is Isopropyl Propionate used for?
The Workhorse in Beauty Formulations
People don't spend much time wondering why lotions spread smoothly or why some creams feel silky rather than sticky. Isopropyl propionate plays a big role here. In the beauty industry, this chemical acts as a key emollient and solvent. What does that mean for the average person? Creams and serums don’t drip or clump, and makeup applies without streaks. A smooth texture helps consumers feel like they’re getting quality, not just empty promises.
My experience in skincare circles taught me to check labels and trace back common ingredients. Isopropyl propionate keeps popping up, not just for how it mixes with other compounds, but for the quick glide it gives. A moisturizer absorbs quickly without a heavy feel, and that’s partly down to this compound. It brings comfort, especially for folks dealing with humidity or oily skin—nobody enjoys waiting for lotion to dry.
Improving Fragrance and Deodorants
Fragrances cling to the skin better with help from certain solvents. Isopropyl propionate brings out those base and mid notes in a perfume, letting a scent last longer. The same science applies to roll-on deodorants, which dry fast without leaving sticky patches. Making perfume and antiperspirant without this ingredient would end up with subpar results—quick fading or patchy application—and customers catch on fast.
Mistakes in formulating these daily essentials lead to complaints online and loss of trust. It’s why producers lean on proven ingredients like isopropyl propionate. In my own trial and error with making soaps for community fairs, I learned quickly that not all solvents behave alike. This one gives better fragrance delivery and less residue on the countertop.
A Helper in Pharmaceuticals and Medical Supplies
Ointments, topical prescription creams, and even some wound cleaning products list isopropyl propionate as part of their formula. The compound aids with delivering medication through the skin, and also ensures that a product doesn’t feel greasy. For anyone dealing with chronic skin conditions, texture matters as much as medicine. Sticky, unpleasant ointments discourage people from following through on care plans.
Solutions and What the Industry Should Watch Out For
There’s a push in recent years to scrutinize every ingredient, not only for results but for health and environmental impact. Isopropyl propionate hasn’t shown evidence of major issues when used as intended. Regulatory bodies like the US Food and Drug Administration review safety data, and industry experts publish new research to keep practices smart. For most people using lotions or perfumes or skin products, worry doesn’t rank high unless allergies exist.
Producers need honesty when listing ingredients. Open labeling gives consumers a voice and builds trust. Too many products crowd store shelves with mystery chemicals, and that turns off smart shoppers. If concerns ever come up—like persistence in water or air—industry leaders ought to invest in green chemistry, searching for alternatives without performance drops.
Finding balance matters. Companies should use isopropyl propionate where it adds real value and keep an eye on research and safety standards. Staying informed, both as consumers and as producers, gives everyone a better shot at safe, comfortable products that actually work.
Is Isopropyl Propionate safe for skin?
Understanding Isopropyl Propionate in Daily Products
Scan the label on almost any bottle of lotion, foundation, sunscreen, or shave cream and there’s a good chance you’ll spot isopropyl propionate. In personal care, this chemical works as an emollient and a solvent. It helps make creams glide across skin, gives that light, almost silky finish, and lets everything blend without feeling greasy. That might sound like a win for the senses, but skin safety deserves a closer look.
My Experience & Why Ingredient Scrutiny Matters
After years of wrestling with sensitive skin, I’ve learned to watch ingredients, especially anything synthetic. It’s easy to overlook substances like isopropyl propionate since so many mainstream brands use them without any warning signals. I don’t believe in scare tactics, but I do trust a practical approach—learn about what touches your skin and weigh the risks with the benefits.
What Science Tells Us
Researchers and safety panels have looked at isopropyl propionate because it shows up so often in cosmetics. The Cosmetic Ingredient Review (CIR) Expert Panel reviewed studies and came to the conclusion that isopropyl propionate counts as safe for use on skin in current concentrations. They found no evidence of harmful buildup, genetic problems, or cancer risk. Toxicologists have even checked long-term use, and nothing in the data signaled major red flags for healthy adults.
That doesn’t mean everyone gets the same result. Dermatologists say it can cause irritation or breakouts in some people, especially those prone to acne or with very sensitive skin. Oils and esters like this sometimes clog pores, even if just a little bit, so folks with blemish-prone faces face some risk. The FDA and European safety authorities haven’t banned it, but they watch data and update rules if new concerns pop up.
Who Should Think Twice?
Anyone with a history of acne or stubborn “mystery” breakouts probably wants to patch-test products using isopropyl propionate. If you’ve noticed skin congestion or irritation with certain lotions, there’s a chance this ingredient plays a part. It isn’t as comedogenic as some other esters, but it doesn’t fall on the “non-pore-clogging” side, either.
People with allergies or major chemical sensitivities should start slow with any new skincare formula. A dermatologist can patch-test for specific reactions and offer tailored advice. It helps to keep a product journal—write down if and how your skin changes after using anything new.
Smart Skincare: Questions and Solutions
Ask yourself some straightforward questions before adding any new product: Does my skin react to most synthetic ingredients? Do I trust this brand’s full ingredient list? How much of this product do I expect to use every day? Sometimes the solution isn’t swapping a product but adjusting how often you use it or mixing it with more soothing, gentle options. For those with persistent reactions, fragrance-free and fewer-ingredient routines work better.
Brands can help by sharing concentration percentages, conducting more trials on reactive skin types, and clearly labeling products for acne-prone folks. The industry also benefits from honest consumer feedback—if a formula clogs pores or triggers irritation, those stories drive better safety research and new blends.
Balancing Science and Skin Sensitivity
For most people, isopropyl propionate slips under the radar and causes no harm. For those with tricky, reactive skin, learning ingredient labels and sticking to simple routines makes a difference. Honest conversations with dermatologists, relying on peer-reviewed safety studies, and tracking your own skin’s reactions give you control over your choices—without hype or fear.
What are the potential side effects of Isopropyl Propionate?
Everyday Exposure, Everyday Choices
Isopropyl propionate probably isn’t a name that comes up at the dinner table. It shows up on labels for skincare, haircare, and cosmetics mostly because of two things: it helps products spread smoothly and gives a soft feel. It doesn’t feel right to ignore what’s actually in everyday lotion, so digging into ingredient safety becomes part of taking care of yourself.
Common Side Effects and Who Gets Them
Redness, itchiness, and a tingling or burning feeling often end up as the most noticed side effects, and these usually show up on people with sensitive or damaged skin. I learned that after using a moisturizer packed with isopropyl propionate after a mild sunburn. I didn’t think twice, but the stinging started within minutes. A lot of people with eczema or allergies tell similar stories—sometimes it’s a minor rash, other times the reaction calls for a dermatologist and a switch of brands.
Studies from the Environmental Working Group pointed out that allergic reactions don’t happen very often, but when they do, they can leave people feeling wary of anything unfamiliar on a label. It’s not about fear, it’s just being careful. Folks with acne also may see more breakouts with heavy use, as isopropyl propionate can act like an emollient, sometimes clogging pores when used on oily or acne-prone skin.
What Science Says about Behind-the-Scenes Risks
Doctors and toxicologists look at isopropyl propionate as a low-to-moderate risk chemical when used externally. The real trouble starts only at high concentrations or with repeated, long-term exposure. Acute toxicity only comes up with swallowing or breathing in large amounts. The FDA puts isopropyl esters in the “generally recognized as safe” group when used within prescribed limits. But “safe” still means paying attention to how skin reacts, since every person is wired differently.
A 2021 review in the journal Contact Dermatitis linked isopropyl esters with a handful of contact allergy cases, especially among professional hair stylists and people who use personal care products daily. Vitamin E-based moisturizers and serums sometimes rely on similar lightweight esters and produce similar reactions in folks with sensitive skin.
Pushing for Transparency and Safer Choices
People can’t always tell what will trigger a reaction without full ingredient lists or more education from brands. Dermatologists stress patch testing for new creams or cleansers, especially with sensitive or allergy-prone skin. In my own experience, a quick patch test behind the ear made all the difference in avoiding an uncomfortable week of itching. Sharing this advice with family members made them more confident choosing safer products too.
Industry groups and advocacy organizations have been pushing brands to disclose full lists of ingredients, not just “key actives.” This helps consumers spot potential triggers before buying. Tools like the EWG’s Skin Deep database let people check safety profiles for virtually every ingredient, including isopropyl propionate, which can help guide smarter shopping. Sometimes clean beauty brands step ahead with detailed ingredient breakdowns and offer fragrance-free or low-residue blends for sensitive skin.
What People Can Do
Even without a deep science background, regular folks can keep their skin calm and healthy by:
- Using a patch test with new skincare products
- Looking up unfamiliar ingredients
- Choosing fragrance-free or dermatologist-tested items when sensitive skin is an issue
- Checking in with a doctor if any reaction happens
Ingredient transparency means people spend less time guessing what went wrong and lets them choose what’s best for their skin, comfort, and peace of mind.
Is Isopropyl Propionate the same as isopropyl alcohol?
Clearing Up a Common Mix-Up
A lot of folks see the words “isopropyl” and immediately picture the classic bottle of rubbing alcohol in their medicine cabinet. Isopropyl alcohol has built up a reputation and trust over decades, especially as a household cleaning item and disinfectant. Isopropyl propionate, on the other hand, rarely gets a shoutout in daily life. Many people wonder: aren’t these two just the same thing in different packaging? They aren’t. In fact, they have different structures, uses, and even risks.
Chemical Identity Shapes Everything
Isopropyl alcohol, which some call 2-propanol, comes off as a solid pick for quick disinfection and even as a solvent in home and industrial settings. The smell, the cooling sensation on the skin, and the near-instant evaporation make it familiar to everyone from parents to paramedics. Its molecular formula, C3H8O, tells a story of a small, volatile compound.
Isopropyl propionate, with the formula C6H12O2, stands in a different league. It forms when propionic acid and isopropyl alcohol react together. It looks more like a neat trick in a chemist’s toolkit. Isopropyl propionate is much less common on grocery shelves. This compound finds its place in cosmetics and perfumes because it doesn’t linger with a strong smell and carries oils and fragrances smoothly on the skin.
Health and Safety Matter
Many people imagine both liquid chemicals have about the same risks. Isopropyl alcohol dries out skin and burns a bit on cuts, but it generally washes away with a bit of soap and water. Swallowing it can land you in the hospital, so parents lock it high up. It has a long medical track record and gets used in everything from sanitizing thermometers to prepping skin before an injection.
Isopropyl propionate doesn’t see the same direct use. It’s more common as an ingredient you don’t notice: a carrier in makeup, sunscreen, or shaving cream. Regulatory bodies, such as the FDA and the European Chemicals Agency, keep a close eye on its concentration in cosmetics. Skin irritation or allergies can occur, so manufacturers often patch-test. The safety profile comes from a long paper trail of studies, not from decades of household experience.
Usage Should Drive Choices
Mixing up these two can cause trouble. If someone tried to sanitize a kitchen counter with isopropyl propionate, they wouldn’t get the bacteria-fighting punch isopropyl alcohol gives. The same goes the other way: isopropyl alcohol works for cleaning but doesn’t belong in face creams.
The distinction between these products underlines an important point: names that sound alike might lead to wrong decisions if you rely on guesswork. I once chatted with a neighbor who accidentally bought a specialty solvent for his woodworking, thinking it was safe on skin. He ended up with a rash. Reading those small-print labels and knowing the purpose changes everything.
Smarter Decisions Need Trusted Information
Between online shopping and crowded ingredient lists, the opportunity for confusion keeps growing. Trustworthy, science-backed sources become more important each year. Doctors, pharmacists, and product safety organizations put in the work so people don’t have to play chemist in their bathrooms.
Taking Action for Clarity and Safety
Manufacturers can help by making labels bolder and clearer and spreading knowledge through education campaigns. Retailers, too, play a part: their staff often fields questions from customers who just want to grab the right bottle for the task. If you’re not sure, ask someone trained to help or check agencies like the CDC or the FDA. Health and safety begin with the right information, not with guessing at similarities between two chemicals that are worlds apart in use.
Can Isopropyl Propionate be used in cosmetics and personal care products?
A Closer Look at Isopropyl Propionate
Walking down the aisles of any drugstore, rows of lotions and creams catch the eye. Many labels list a heap of ingredients people barely recognize. Isopropyl propionate usually pops up there, tucked inside the fine print. This chemical, made from propionic acid and isopropanol, acts as an emollient and solvent. It helps spread oils and other good stuff smoothly onto the skin. That slick, pleasant feel in creams and some hair products owes a lot to it.
Why Formulators Turn to It
Product developers want items to glide easily and soak in fast. Isopropyl propionate has a lightweight nature and blends seamlessly with oils. It gives formulas a slipperier feel without leaving a greasy film on skin. Beauty brands aiming for quick-drying makeup removers and light facial serums often turn to it. I have seen how richer creams benefit from its texture-enhancing abilities, making heavy lotions feel less sticky.
What Does Science Say?
Safety always stirs up concern in beauty. Most dermatologists and toxicologists agree: in current usage concentrations, isopropyl propionate shows a low chance of skin irritation or allergies. The Cosmetic Ingredient Review (CIR) panel, after combing through studies, said this ingredient remains safe to use in rinse-off and leave-on products. Authorities in the European Union also cleared it for cosmetic use after their assessments. As with any substance, rare sensitivities exist, but so far, large-scale reactions remain almost unheard of.
Keeping Transparency on the Table
Consumers grow warier of complex chemicals these days. People want clarity about what goes on their bodies. Ingredient transparency takes top priority for brands that want to keep trust. A quick search pulls up scientific literature showing the minimal toxicity of isopropyl propionate. Full ingredient lists, clear labeling, and fact-backed explanations help both brands and users feel confident about daily choices.
Walking the Line with Sensitive Skin
Some skin types react to everything. Even a gentle ingredient might cause a flare-up under certain conditions. People with known sensitivities or allergies might want to patch test products with isopropyl propionate or consult a dermatologist. Based on data from published patch tests, the risk seems low, but a personal approach always protects best.
Sustainability and Sourcing
As sustainability takes center stage, cosmetic chemists keep a close eye on how ingredients get made. Isopropyl propionate comes from synthetic routes, mostly from petrochemicals. Companies now aim to find greener routes and lower their environmental footprint. The push for clean alternatives drives research for renewable versions of familiar emollients like this one. I believe consumer demand for sustainability means the industry will continue to innovate in this area.
Moving Toward Smarter Choices
Choosing products involves more than flipping bottles and reading claims. Relying on evidence, science-backed studies, and regulations brings real confidence. Years of use, clear regulatory oversight, and research support isopropyl propionate’s role in personal care. People deserve honest answers about what touches their skin; trustworthy information, clear communication, and a commitment to safe sourcing pave the way forward in cosmetics.
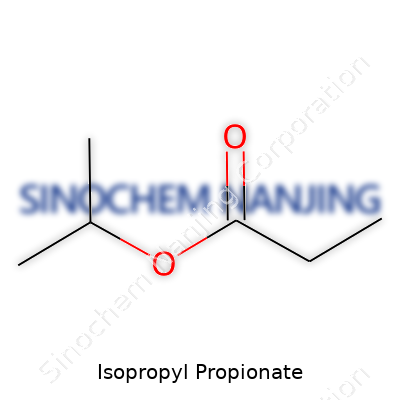
| Names | |
| Preferred IUPAC name | Propan-2-yl propanoate |
| Other names |
Propionic acid, isopropyl ester Isopropyl n-propionate Isopropyl propanoate 1-Methylethyl propionate Propanoic acid isopropyl ester |
| Pronunciation | /ˌaɪ.səˈprəʊ.pɪl prəˈpɒn.i.eɪt/ |
| Identifiers | |
| CAS Number | 637-78-9 |
| 3D model (JSmol) | `JSmol` string for **Isopropyl Propionate** (C6H12O2): ``` CC(C)COC(=O)CC ``` This is its **SMILES** string representation, which is used in JSmol and other 3D viewers to render the chemical structure. |
| Beilstein Reference | 803836 |
| ChEBI | CHEBI:31210 |
| ChEMBL | CHEMBL3184852 |
| ChemSpider | 10788 |
| DrugBank | DB14096 |
| ECHA InfoCard | 100.032.805 |
| EC Number | 203-561-1 |
| Gmelin Reference | 119348 |
| KEGG | C19607 |
| MeSH | D011808 |
| PubChem CID | 8016 |
| RTECS number | UJ8750000 |
| UNII | 6M7QXD6EK1 |
| UN number | UN3276 |
| CompTox Dashboard (EPA) | DTXSID8020292 |
| Properties | |
| Chemical formula | C6H12O2 |
| Molar mass | **130.187 g/mol** |
| Appearance | Colorless transparent liquid |
| Odor | fruity |
| Density | 0.858 g/cm3 |
| Solubility in water | Slightly soluble |
| log P | 1.84 |
| Vapor pressure | 0.6 kPa (20 °C) |
| Acidity (pKa) | pKa ≈ 25 |
| Magnetic susceptibility (χ) | -7.72×10⁻⁶ |
| Refractive index (nD) | 1.387 |
| Viscosity | 4.1 mPa·s (25°C) |
| Dipole moment | 2.05 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 377.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -428.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3487.7 kJ/mol |
| Pharmacology | |
| ATC code | D02AX10 |
| Hazards | |
| GHS labelling | `GHS02, GHS07` |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H319, H336 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P273, P280, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P370+P378, P403+P235, P403+P233, P501 |
| Flash point | 54 °C |
| Autoignition temperature | 400 °C |
| Explosive limits | 1.1% - 8.0% |
| Lethal dose or concentration | LD50 oral rat 3440 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 14,700 mg/kg |
| NIOSH | RN 05-034-00 |
| PEL (Permissible) | PEL: Isopropyl Propionate: 100 ppm (525 mg/m³) |
| REL (Recommended) | Exempt (JP), IECIC (CN), FDA Generally Recognized as Safe (GRAS), EU. |
| IDLH (Immediate danger) | 800 ppm |
| Related compounds | |
| Related compounds |
Isopropyl acetate Propyl propionate Isopropyl butyrate Methyl propionate Ethyl propionate |

