Isopropyl Nitrate: Unpacking a High-Energy Chemical
Historical Development
Isopropyl Nitrate appeared on the scene during the 20th century, discovered as part of a broader search for versatile fuel additives and explosives. Its earliest documented uses tie closely to military and industrial pursuits, where demand rose for materials delivering substantial energy quickly. Over time, research spread from government labs into public sector technical reports, tracking the ways this nitrate could boost combustion or serve as a reliable propellant. This history shaped how the product is viewed even today, often pushing scientists to dig into both benefits and risks. Its invention and uptake run parallel with expanded knowledge about nitric esters in general, which changed how chemists design both peacetime and defense applications.
Product Overview
Isopropyl Nitrate falls under the family of organic nitrates, packing a strong punch in energy output. Users mostly look to it for its high reactivity and clean-burning properties. Its most common form comes as a clear, colorless liquid with a smell that hints at alcohol. In small-scale settings, hobbyists and engineers alike might experiment with it for model engines or specialty fuels, but regulations restrict access due to safety. Industry guidelines classify it in line with other nitrate esters, meaning product labeling stays strict about content and storage.
Physical & Chemical Properties
The molecule's structure, C3H7NO3, combines an isopropyl group linked to a nitrate functional group. It boils around 101°C and freezes well below standard room temperature, making it a liquid in most environments. Density sits near 1.03 g/cm³. It mixes well with other organic solvents, showing low solubility in water. The compound packs oxygen into its structure, supporting rapid combustion or even detonation in the right setting. This oxygen balance not only gives it power in engine applications but also presents major hazards; even a small spark can trigger aggressive burning, which demands respect and careful handling.
Technical Specifications & Labeling
Retail and bulk shipments require detailed information: purity (usually above 98%), water content, and identifiers such as CAS number 1712-64-7. Regulatory agencies require GHS-compliant hazard statements, warning about explosive potential and acute toxicity. Modern chemical suppliers offer certificates of analysis, making batch tracking easier and helping users pin down any impurities. This transparency matters for safe use, given the risks of unintended side reactions or contamination that can turn a useful compound into a liability. Labeling also covers recommended storage—cool, dry, and far from ignition sources.
Preparation Method
Isopropyl Nitrate usually gets made by reacting isopropanol with concentrated nitric acid, sometimes under chilled, controlled conditions to slow down the reaction and reduce unwanted byproducts. Concentrated sulfuric acid often acts as a drying or catalytic agent, stripping out water and driving the process forward. Chemists need to watch temperature and acidity closely; uncontrolled runs can get dangerously hot or create side-products that complicate purification. After synthesis, washing with cold water and neutralizing agents helps protect against residual acidity, then distillation purifies the main product.
Chemical Reactions & Modifications
This nitrate responds energetically to heat, shock, and light, decomposing into isopropanol, nitrogen dioxide, and oxygen, among other gases. In lab setups, adding a reducing agent can break it down in a controlled fashion, often capturing the organic residue for reuse or analysis. Under strong alkaline conditions, the ester bond cleaves to yield isopropanol and nitrate salts, a handy pathway for disposal. Some research teams explore blending it with other nitrate esters, hoping to tune detonation speed or slow down its flash. Such modifications carry risks, though, because mixing multiple energetic materials amplifies unpredictability.
Synonyms & Product Names
Isopropyl Nitrate goes by several names: IPN, 2-Propanol nitrate, and Nitric acid isopropyl ester rank among the most recognized in chemical literature. In older military documents, "PN" sometimes appears, causing confusion with other propellants. Global chemical suppliers now standardize on "Isopropyl Nitrate" for compliance and clarity, cutting down on mix-ups with similar esters. Product names may differ by brand, but the underlying formulas stay consistent—critical for both safe use and international shipping.
Safety & Operational Standards
Anyone handling Isopropyl Nitrate must respect its unpredictable nature. Improper storage or accidental mixing with trace metals or incompatible chemicals may touch off fires or rapid decomposition. Use requires chemically resistant gloves, face shields, and lab coats, plus access to proper ventilation—those nitrated vapors burn more than just eyes and throats. The chemical appears on multiple international watchlists, restricting use to licensed facilities. Safety data sheets stress emergency protocols, including foam or dry powder suppression, never water, which can accelerate reaction with spilled concentrates. Long-term exposure or careless disposal contaminates both air and water, making responsible management a priority for public health.
Application Area
The main draw for Isopropyl Nitrate comes from its value as a diesel engine initiator—used in rapid-start cartridges for older locomotives, construction machines, and the like. Its ability to ignite at low temperatures made it suddenly essential decades ago, especially in harsh winter conditions. Outside engines, research teams and military units investigated its potential as a rocket propellant or as part of small explosive charges. Civilian markets today use very little, given the safety restrictions and advances in cleaner, more stable alternatives. Still, textbook examples in chemical engineering courses keep IPN relevant in the academic world, where understanding its behavior illuminates bigger trends in energetic materials.
Research & Development
Efforts to unlock safer or greener nitrates mean Isopropyl Nitrate ends up in plenty of research pipelines. Labs measure everything from energy density to toxicity breakdown, using high-speed cameras and chromatographs to map detonation paths and residue. New studies look for additives or stabilizers that slow down unwanted reactions, especially in transport or storage. Increasingly, attention shifts toward environmental fate—how sunlight, water, and bioactivity transform leftover spills or emissions. Research continually weighs the efficiency of old methods against new, hoping to match performance with better safety profiles.
Toxicity Research
Studies confirm inhalation or skin contact with Isopropyl Nitrate can harm the nervous system, disrupt oxygen flow in blood, or irritate soft tissue. Blue baby syndrome (methemoglobinemia) remains a pressing concern for communities facing nitrate contamination in drinking water; while Isopropyl Nitrate itself may not persist, its breakdown products often linger. Regulatory agencies rely on animal testing and cellular assays to chart safe exposure levels, but complexities in metabolic breakdown mean labs must act cautiously. Chronic low-level exposure links to organ impairment, headaches, and increased risk of cancer. Proper disposal and diligent spill cleanup become both an ethical and scientific duty.
Future Prospects
Whether Isopropyl Nitrate finds a new purpose depends on how next-generation chemistry tackles safety, environmental challenge, and raw material sourcing. Demand for old-style diesel aids and explosives has slowed, but niche uses persist where nothing else matches its cold-weather reliability. If researchers succeed in designing stabilizers or green synthesis routes, it could make a comeback in specialty fuels, laboratory work, or even emergency rescue gear in freezing climates. Policy continues to shape its journey, as lawmakers respond to environmental concerns and public safety. Persistent curiosity from chemists ensures its story keeps unfolding—from risky molecule to carefully-managed tool, revisited by each new wave of scientists, engineers, and industry leaders.
What is Isopropyl Nitrate used for?
What Gets People Interested in Isopropyl Nitrate?
Some chemicals just spark people’s curiosity—and sometimes controversy. Isopropyl nitrate falls squarely into that group. If you ever walked through a hardware store or read about World War II history, you might have caught a mention of this liquid. It’s not exactly a household name. Still, its uses and risks come up more often than you’d think, especially in circles talking about automotive performance, pyrotechnics, or even dangerous recreational trends.
How Do People Use Isopropyl Nitrate?
Car enthusiasts started using isopropyl nitrate mainly as an additive in diesel fuel. The idea: jumpstart cold engines in rough weather and bump up performance. The fuel system gets a shot of extra oxygen, which makes diesel engines ignite more easily. Studies show additives like this can shave seconds off cold starts, which mattered when older engines struggled in the cold. Isopropyl nitrate also pops up in model rocketry. Model makers often look for anything that can push their engines higher and faster, and some turned to this chemical for an edge, despite the obvious risks.
Military and emergency sectors tried it, too. The U.S. Army once used isopropyl nitrate as a sort of single-use liquid propellant, like in the “start cartridges” that kicked big engines or generators into action with a flash. Aviation mechanics tell stories of cartridge starts—they’d bang, smoke, and sometimes, if you weren’t careful, leave scorch marks on the tarmac. It solved a real problem, but most have moved on thanks to safer, more reliable technology.
Why Do Safety Agencies Raise Red Flags?
Personal experience: I once worked at a place storing industrial chemicals. Isopropyl nitrate always demanded gloves, goggles, and extra ventilation. It evaporates quickly, smells sharply, and if a stray spark comes near, it can ignite in a flash—almost explosively. Exposure can also irritate your lungs and skin. Firefighters train specifically for chemical exposures like this one. The health risks led several countries to restrict sales to the public.
It’s not just the fire hazard—I’ve read credible reports linking inhalation to short-term euphoria followed by sudden headaches, nausea, and even fainting. Some outlets even market it for recreational inhalation, despite clear warnings from medical professionals. A 2020 CDC briefing detailed how misuse of volatile nitrates can damage the central nervous system, trigger irregular heartbeats, and cause fatal drops in blood pressure. Too often, someone looking for a quick thrill ends up in the emergency room.
Solving the Risks Around Isopropyl Nitrate
Greater awareness stands out as the first step. Regular people don’t usually realize products found online or in old workshop cabinets could give off toxic fumes or catch fire in seconds. Clearer labelling and better public health messaging would help. I’ve seen shops hide chemicals behind counters after too many close calls—retailers need to train staff to warn customers about risks and look for alternatives.
Innovation created substitutes that offer most benefits without the same danger. In the automotive world, chemicals like diethyl ether now start engines safely because they catch fire at lower temperatures with less explosive force. Pyrotechnics groups teach safe fuel handling and encourage people to stick to fuels with established safety records. Regulation steps in when needed—governments move certain chemicals out of public hands and into locked cabinets where only trained professionals access them.
Responsible Choices Matter
Whether it’s tinkering in a garage, running an old farm generator, or just browsing oddities online, every one of us relies on trustworthy sources to inform us about what we’re handling. Isopropyl nitrate isn’t some boring lab chemical—it’s a compound that shapes real-world outcomes, for better or for worse. The more we respect its power, the safer our communities become.
Is Isopropyl Nitrate safe to handle?
What Isopropyl Nitrate Brings to the Table
You’ll find isopropyl nitrate showing up in some pretty specific corners of industry—and also in conversations about party drugs. This clear, oily liquid with a sharp odor isn’t just another lab chemical. It’s a powerful oxidizer, which means it boosts combustion and gets things moving quickly in engines or other tech that needs a big, fast release of energy. It’s also been misused in recreational settings because of the rush it can cause when inhaled.
The Hazards People Tend to Overlook
If you’ve handled chemicals in a lab or worksite, you spot the usual suspects: splashes, fumes, spill cleanup. With isopropyl nitrate, the top risk comes from its volatility. It catches fire fast, even from a small spark or static. Those old videos of fires ripping through garages or workshops? Sometimes, it’s a bottle of this stuff at the center. Inhaling its vapors—whether accidental or on purpose—stresses the body in some ugly ways. Headaches, dizziness, and a drop in blood pressure hit almost right away. Some users black out. Enough exposure or misuse can injure lungs or the nervous system.
Mistakes aren’t rare. Even quick contact with skin causes burns. Vapors travel far and build up in closed rooms, so even a tiny leak turns dangerous. All it takes is a batch of forgotten rags or a cracked cap for a disaster to start slowly, then flare suddenly.
What Real Life Experience Shows
I’ve watched seasoned mechanics double-check every step while topping off fuel or cleaning parts, but still look uncomfortable opening a container of isopropyl nitrate. That’s respect earned by bitter experience: stories of fires that started with just a whiff of vapor escaping, safety gear that melted in seconds, and carefully planned days derailed by careless storage.
Across labs, proper training matters just as much as high-tech equipment. Seeing someone glove up with chemical-resistant material—not just flimsy latex—drives home how unforgiving isopropyl nitrate can be. My own lab days included a few emergency showers and burnt gloves—reminders that the best written rules only go so far against a splash or a slip.
Pushing Toward Safer Handling
Clear labeling does more good than any locked cabinet when people listen. Explaining what’s at stake in plain language—not just hazard codes—makes new workers pause before cutting corners. Stories make an impact: “Don’t leave this near anything remotely flammable,” carries more weight when you’ve seen burn marks on a table or heard a coworker talk about a close call.
Modern ventilation setups help, filtering vapor and keeping levels below the line where danger spikes. Regular training, safety drills, and real walkthroughs hold everyone accountable, making sure policies move beyond the binder and into daily routines.
For recreational use, public warnings must come across clearly. Calling something a “nitrate” might hide the fact it can stop a heart or cause lasting injuries. Honest conversations, led by both doctors and people with lived experience, bring the facts out from the fine print.
Bottom Line for Anyone Working With or Near It
Isopropyl nitrate demands respect—never casual handling. It’s not about being paranoid, but about learning from the people and places that have faced the fallout of mistakes. Chemical expertise, decent equipment, and steady habits are the only protection that counts. Every day spent working around it safely means every step of the process matters.
What are the storage requirements for Isopropyl Nitrate?
Handling an Unforgiving Compound
Anyone who has spent time with energetic chemicals knows the caution demanded by isopropyl nitrate. Unlike household solvents, this liquid brings an explosive temperament. Even folks who’ve handled fireworks or model rocket fuels will find it a different beast. Strong smells and quick evaporation hint at its volatility, but the real danger sits in its readiness to ignite or decompose without much provocation. One whiff of its sharp scent, and it’s clear nobody wants to see a bottle on the shelf next to regular lab alcohols.
Storing Isopropyl Nitrate: No Room for Complacency
Long before heading to regulations, it only makes sense to store isopropyl nitrate well away from heat, sunlight, or open flames. Static sparks sometimes lurk in ungrounded storage cabinets, and a single slip could offer enough energy to spark disaster. Warm environments—think cramped summer chemical closets—bring real risks for pressure build-up inside containers. A good rule from the field: keep the room as close to 20°C as possible and never let the stuff get anywhere near 30°C.
Moisture doesn’t play nicely with isopropyl nitrate. Rusty containers or leaky lids can trigger dangerous reactions. I always appreciate regulators insisting on tightly sealed, corrosion-resistant bottles—usually glass, but sometimes certain plastics do the trick. Every experienced chemist I know checks containers for surface flaws and only buys from reputable suppliers who know what they’re doing.
Strict Regulatory Compliance Isn’t Optional
Industrial facilities treat isopropyl nitrate as a hazardous material under OSHA and EPA frameworks. Labs and storage sites need proper hazardous material labeling—NFPA diamonds, GHS pictograms, and all identification kept visible. Emergency eye wash stations belong a few quick strides away. I’ve witnessed how ignoring one small detail, like missing sprinkler heads or poor signage, can give regulators a reason to shut down work on the spot.
Key regulations from the Department of Transportation and local fire marshals strictly control how much can be stored before extra permits or engineering controls step in. For most places, a cool, dedicated chemical storage room with explosion-proof ventilation and monitored temperature systems proves the safest setup. Portable fire extinguishers tagged for Class B and C fires never gather dust—everyone in the room knows where to find and how to use them.
Training and Best Practices: Protecting People
Anyone storing or moving isopropyl nitrate should go through specialty safety training. I have watched new chemists fumble, believing nitrates behave much like solvents or acids until a mentor steps in. PPE rules—chemical-resistant gloves, goggles, lab coats—aren’t optional. Nobody wants permanent scars or lung damage from casual mistakes.
Spill kits designed for flammable liquids keep incidents contained. Any leak or spill demands thorough response. I always advise against improvising with rags or household products. Disposal follows hazardous waste rules, relying on access to approved incineration or chemical disposal services; dumping it down a drain or into regular trash earns steep fines and puts the public at risk.
Making Safety Routine, Not Special
Labs, facilities, and occasional hobbyists owe their safety to consistency. Relying on documented protocols, regular inspections, and a culture where nobody skips steps reflects not just best practice but genuine respect for the risks. Attention to every detail, from light switches to inventory logs, sends everyone home whole at the end of the day.
What are the potential health hazards of Isopropyl Nitrate?
Understanding What We’re Dealing With
Walk into many workplaces or listen in on certain circles, and you’ll hear a few chemicals getting tossed around by nickname. “Poppers” is one of those names, usually pointing to a group that includes isopropyl nitrate. Some folks chase its rush for recreational highs, others handle it at work, like in laboratories or for cleaning electronics. Before anyone picks up a vial or sniffs the stuff at a party, it’s worth knowing what isopropyl nitrate can do to a body—both right away and down the line.
Immediate Dangers: It Really Packs a Punch
Inhaling isopropyl nitrate tends to hit fast. That high people get is a signal: The body isn’t happy. Dizziness and headaches show up quickly, sometimes alongside a pounding heart and chest tightness. Some users faint or get nausea bad enough to keep them off their feet for hours. This isn’t just discomfort—those symptoms mean oxygen in the blood isn’t keeping up, which stresses every organ.
I’ve worked around industrial solvents and other volatile stuff before; safety trainers go on and on about well-ventilated spaces. If you ever stood in a badly ventilated room after a chemical spill, this all makes sense. Breathing gets rough, you get cloudy in the head, and it can sneak up on you just how badly you’re affected—until you can’t stand up straight.
Blood-Related Hazards
One problem with chemicals like isopropyl nitrate: They push your red blood cells into trouble. Nitrites can cause something called methemoglobinemia, where cells carry less oxygen. Skin and lips look blue, people gasp for air, and if the dose runs high enough, the consequences get deadly. Emergency rooms sometimes see this after a “fun night out” gets out of hand.
Longer-Term Effects and Other Risks
No one plans to make a habit of using chemicals, but habits form anyway. Chronic exposure wrecks the immune system and can slowly gnaw away at the body’s defenses. Animal studies point to possible organ damage over weeks and months—liver and kidney mostly—though people rarely pay close attention unless the symptoms kick in hard.
Another overlooked angle: mixing isopropyl nitrate with other drugs, like erectile dysfunction meds, punches blood pressure way down. The combination drops people to the floor, fast. News stories show emergency crews arriving after someone’s heart gives out on the spot. There’s real danger in treating it like a party drug.
Hidden Dangers for At-Risk Groups
Kids, the elderly, and people with heart conditions don’t fare well when exposed. Even a whiff can push their bodies over the edge. Communities living in older buildings—often with poor ventilation—end up facing higher risks, especially if people try to clean with niche solvents that contain nitrates.
What Makes a Difference?
Education does some heavy lifting. Workplaces need plain-language instructions and real stories about what happens—not just signs on the wall. In recreation circles, honesty among friends about each other’s limits can be life-saving. Groups that support at-risk youth or vulnerable populations can talk openly about these chemicals without judgment, recognizing that harm reduction goes further than blanket bans.
Lawmakers and health agencies could focus more on packaging and clear labeling, way beyond fine print. Emergency rooms benefit when staff regularly drill on chemical emergencies. And it never hurts keeping a kit for oxygen and methemoglobinemia antidotes handy in places where exposure risk runs high.
Is Isopropyl Nitrate legal to purchase and possess?
Tracing the Legal Maze
Isopropyl nitrate, sometimes known among car enthusiasts as a fuel additive and among others for less scrupulous recreational use, stands at the center of a cloudy legal picture in many countries, including the United States. Laws change fast—faster yet since safety and public health keep landing in the headlines. At the local hardware store, nobody hands you a bottle of isopropyl nitrate over the counter. In fact, it sits in a gray area thanks to its history and its dual uses, both legitimate and illicit.
Why People Care So Much
This chemical made a reputation as a "booster" in racing scenes because it can spike engine performance. Others know it from so-called "poppers", chemical inhalants often misused for a quick, risky high. Most users hear conflicting stories about what is legal and what ends up getting people in trouble. For me, covering stories about chemical safety goes hand-in-hand with some tough conversations about drug misuse and how new products slip through regulatory cracks.
Federal law in the U.S. does not always draw a bright line around isopropyl nitrate. The Chemical Diversion and Trafficking Act targets chemicals used to make illegal drugs or explosives. Isopropyl nitrate does not show up by name in that act, but states set their own rules, and authorities look at purpose of use. Over the years, the Food and Drug Administration has cracked down on the sale of nitrate inhalants that claim to be “room deodorizers” or similar products, especially if the intention really looks like personal use as a drug. Retailers caught selling such products under false pretenses have faced hefty fines.
The Role of Safety and Public Health
Chemicals like isopropyl nitrate often harm more people than they help. Beyond addiction, medical experts link them to nerve damage, breathing problems, and sometimes even sudden death. Simple curiosity pushes buyers to try new things, rarely thinking about outcomes. I’ve reported on emergency rooms seeing young people come in with dangerous side effects after inhaling products that started as “household” chemicals. Doctors spend nights untangling chemical exposures and explaining to parents what went wrong—often something as basic as a bottle bought online with no clear warnings.
In the automotive world, some older manuals mention isopropyl nitrate as a starting fluid or performance enhancer. In many cases, safer and less volatile additives now fill its place. Modern fuel treatments and engine cleaners get better oversight, so manufacturers spell out ingredients and risks more clearly.
What Actually Helps
More clear-cut information helps everyone—buyers, retailers, and anyone stuck with the mystery of what they can or cannot legally own. If you plan to use any strong chemical, research local and federal laws, and talk to someone at a licensed shop. If questions remain, reach out to a poison control center, state health department, or regulatory agency. Too many people end up facing consequences because they simply missed a rule buried someplace online.
Stronger enforcement on mislabeled products on shelves—both virtual and physical—shields people from hidden dangers. Schools and workplaces also teach chemical safety as part of their own best practices, which keeps some risks at bay. Trustworthy product sources, detailed labeling, and transparency around risks play bigger roles than ever. We push for clearer rules and public messaging, especially online, so people spend less time guessing and more time making real decisions about what they bring into their garages and homes.
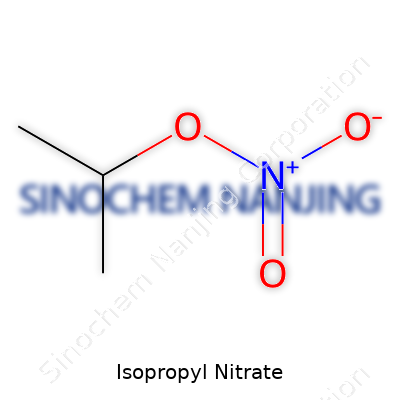
| Names | |
| Preferred IUPAC name | Propan-2-yl nitrate |
| Other names |
IPN 1-Methylethyl nitrate Propan-2-yl nitrate Isopropyl nitrite |
| Pronunciation | /ˌaɪsəˈproʊpəl ˈnaɪtreɪt/ |
| Identifiers | |
| CAS Number | 1712-64-7 |
| Beilstein Reference | 1091226 |
| ChEBI | CHEBI:51156 |
| ChEMBL | CHEMBL572907 |
| ChemSpider | 11016 |
| DrugBank | DB14076 |
| ECHA InfoCard | ECHA InfoCard: 100.006.180 |
| EC Number | 208-759-1 |
| Gmelin Reference | 163193 |
| KEGG | C19719 |
| MeSH | D009897 |
| PubChem CID | 10945 |
| RTECS number | TZ9620000 |
| UNII | E8V2X36Q3D |
| UN number | UN1222 |
| CompTox Dashboard (EPA) | `DTXSID6020824` |
| Properties | |
| Chemical formula | C3H7NO3 |
| Molar mass | 135.13 g/mol |
| Appearance | Clear, colorless liquid |
| Odor | fruity odor |
| Density | 1.065 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 0.61 |
| Vapor pressure | 40 mmHg (20°C) |
| Acidity (pKa) | 13.5 |
| Basicity (pKb) | 10.67 |
| Magnetic susceptibility (χ) | Diamagnetic |
| Refractive index (nD) | 1.401 |
| Viscosity | 0.695 cP (20°C) |
| Dipole moment | 3.54 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 183.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −199 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -1977 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H225, H301, H311, H331, H370, H373, H335, H319, H315 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P271, P280, P301+P310, P304+P340, P308+P311, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 3-3-1-OX |
| Flash point | The flash point of Isopropyl Nitrate is **-28°C**. |
| Autoignition temperature | 428 °C |
| Explosive limits | 0.9–40% |
| Lethal dose or concentration | LD50 (oral, rat): 170 mg/kg |
| LD50 (median dose) | LD50 (median dose), oral (rat): 160 mg/kg |
| NIOSH | PSA1340 |
| PEL (Permissible) | PEL: 6 ppm |
| REL (Recommended) | 200 ppm |
| IDLH (Immediate danger) | 200 ppm |
| Related compounds | |
| Related compounds |
Methyl nitrate Ethyl nitrate n-Propyl nitrate Isopropyl nitrite Isopropyl nitrate Nitroglycerin Amyl nitrate |

