Isopropyl Mercaptan: Finding Value in a Sharp-Smelling Compound
Historical Development
The story behind isopropyl mercaptan isn’t just about sulfur and sharp odors. Long before engineers began piping natural gas through cities, chemists were already toying with sulfur compounds. In the late 1800s, early oil refineries noticed unpleasant scents in their processing lines, which led them to isolate compounds like isopropyl mercaptan. The real push came in the twentieth century after major gas leaks underlined the need for reliable leak detection. Natural gas, being odorless, needed a warning system. Mercaptans stepped up not just for their smell, but for their staying power and easy detection even at concentrations almost too small to measure. Refineries and lab benches across Germany and the United States became proving grounds for this family of compounds, and isopropyl mercaptan carved out a role for itself as a practical, effective tool—one shaped as much by industrial need as by its stubborn stench.
Product Overview
Isopropyl mercaptan often comes up in the context of odorant applications—especially in the energy sector. Suppliers deal with this colorless to pale yellow liquid, notorious for its sulfur-rich odor, and package it in drums or specialized containers to prevent leakage and evaporation. Major chemical producers manufacture it to rigorous standards, tracking every step from raw isopropanol and hydrogen sulfide input to end-product purity. Regulatory agencies focus on its role in safety systems, but it also appears in agricultural products, flavor chemistry, and even specialized lubricants. Despite its reputation, industries see isopropyl mercaptan as the right tool for jobs where a nose for danger or a pinch of sulfur chemistry brings vital results.
Physical & Chemical Properties
A bottle of isopropyl mercaptan doesn’t need a label for someone to notice—the distinct, skunky scent announces itself. Its boiling point lands around 59°C, and the liquid forms vapor easily, which can be daunting in the summer heat or inside a poorly ventilated lab. The low threshold for olfactory detection—on the order of parts per billion—gives it a unique commercial value. The chemical formula, C3H8S, puts two methyl groups on either side of a sulfur, making the substance both volatile and stubbornly persistent. It mixes well with organic solvents and dissolves only sparingly in water. Sulfur in the backbone of the molecule loves to react, especially with oxidants and certain metals, so chemists run processes with a careful eye on temperature and pressure to keep everything on track.
Technical Specifications & Labeling
Every shipment of isopropyl mercaptan carries a raft of technical paperwork. Product grades differ—some destined for gas odorization, others branded for laboratory or specialty chemical use. Purity typically surpasses 98%. Water content, acidity, and levels of heavy metal residues sit at the heart of certificate of analysis documents. Bulk containers bear hazard numbers and flammable liquid warnings regulated by agencies like OSHA and the Department of Transportation. Safety Data Sheets spell out flash point values (often around -18°C), recommended storage temperatures, and incompatibility with oxidizing agents.
Preparation Method
Manufacturers rely on alkylation methods, joining isopropanol with hydrogen sulfide under pressure and the guidance of catalysts such as aluminum oxide or zinc chloride. This process runs continuously in large-scale reactors, and operators work under strict protocols to contain both the toxicity of hydrogen sulfide and the corrosiveness of sulfur intermediates. After the main reaction, distillation under reduced pressure separates isopropyl mercaptan from byproducts—often methyl mercaptan, diisopropyl sulfide, or unreacted precursors. Purification doesn’t stop with distillation; it sometimes calls for washing the product with caustic or brine and keeping a close watch on anything that might affect odor or stability.
Chemical Reactions & Modifications
Working with isopropyl mercaptan means expecting reactions with halogens, acids, and even certain light metals. The thiol group (–SH) acts as a ready handle for building new molecules—chemists transform it into sulfoxides or disulfides, both of which find uses in synthesis and research. In contact with alkali metals or bases, isopropyl mercaptan forms salts and thiolate ions, opening doors to more complex organosulfur compounds. When oxidized, its volatility changes, and researchers tracking these transformations rely on gas chromatography for detailed analysis. Some processes convert isopropyl mercaptan to less odorous forms, as a way to clean up intermediate streams in chemical production or control storage risks.
Synonyms & Product Names
You might find isopropyl mercaptan under a dozen names depending on the supplier or country. The most common include 2-Propanethiol, isopropanethiol, and 2-methyl-1-propanethiol. Chemical catalogs might list it under CAS number 75-33-2. Some product lines refer to it as a “natural gas odorant” or mention trade names developed by specific industrial suppliers. Regulatory frameworks sometimes use older or alternate descriptions, so keeping a cross-reference at hand helps keep shipments running smoothly and paperwork accurate.
Safety & Operational Standards
Nothing about isopropyl mercaptan’s safety profile allows for shortcuts. The strong odor proves both a blessing and a curse; early detection of leaks saves lives, but even tiny spills require immediate cleanup to prevent nausea and workplace complaints. Workers need flame-resistant gloves, tight-sealing goggles, and chemical-resistant clothing, especially in areas with poor airflow. Ventilation goes beyond the basics: high-capacity scrubbers and careful monitoring of air concentrations cut down on lingering odors and accidental exposure. Storage rules demand tightly closed metal containers kept away from heat, sunlight, and oxidizing chemicals. Facilities working with isopropyl mercaptan train teams in spill control, evacuation procedures, and fire response. Regular drills and equipment checks sharpen response times and help spot problems before they scale up.
Application Area
Natural gas companies rely on isopropyl mercaptan to warn customers of leaks—a role rooted in hard experience with gas explosions and public outcry. Its powerful odor, detectable in minute amounts, lets maintenance crews and neighbors spot leaks before problems spiral. Other industries draw on isopropyl mercaptan’s chemistry for synthesis, using it to produce herbicides, pharmaceuticals, and flavoring agents where a burst of sulfur notes is needed. Laboratory-scale reactions benefit from its reactivity, especially in building blocks for novel sulfur-containing molecules. In the past, some electronics manufacturers used sulfur compounds like isopropyl mercaptan for specialized corrosion inhibitors, though tighter workplace limits dialed back usage. Companies making adhesives and rubber chemicals keep isopropyl mercaptan on hand for cross-linking and modification steps.
Research & Development
Research focuses on both safety and new uses for isopropyl mercaptan. Analytical chemists track air concentrations with portable sensors based on electrochemical or UV detection, hoping to improve workplace safety and reduce nuisance odors. Teams working on cleaner fuels have explored ways to break down mercaptans during refinery processes, aiming for lower emissions and easier regulatory compliance. Some researchers aim for modified odorants with similar detection properties but less persistent odor, trimming community complaints near storage sites. Others study how to recycle thiol-rich waste for upstream chemical use, turning what was once a disposal challenge into a source of feedstock.
Toxicity Research
Toxicologists found that isopropyl mercaptan, like other low-molecular thiols, delivers acute effects at surprisingly low concentrations. Inhalation at levels above 10 ppm can lead to headaches, dizziness, and irritation to eyes and breathing passages. Long-term animal studies show no clear evidence for cancer risk, but they point to possible central nervous system impacts after repeated high-dose exposure. Workplace limits in the United States (and echoed worldwide) fall under tight control—usually at 0.5 ppm for an 8-hour exposure. Research teams comb through case reports from refinery incidents and accidental releases, looking for patterns that can inform emergency response rules. Personal monitoring devices and real-time air quality sensors have become crucial tools, especially in older facilities.
Future Prospects
Chemistry doesn’t stand still, and prospects for isopropyl mercaptan reflect both ongoing needs and rising challenges. Urban populations grow, so reliable gas odorization stays crucial, but alternatives that blend sulfur notes with milder odors may appeal to expanding cities sensitive to air quality. Tighter environmental regulations have spurred research into recovery and recycling methods, turning waste into raw material and cutting down on emissions. Advances in analytical technology—like real-time odorant monitoring and rapid gas chromatography—support safer, more efficient operations. Researchers keep refining preparation routes to limit byproducts and energy use. Finding safer, less obtrusive warning compounds remains an active field, but the blend of proven effectiveness and industrial inertia means isopropyl mercaptan likely endures awhile yet, both in the pipes under our neighborhoods and in the hands of chemists shaping what’s next.
What is Isopropyl Mercaptan used for?
The Power of a Strong Scent
Stop by a natural gas plant and someone will mention the rotten egg smell that hangs in the air. That’s not the gas itself. It’s isopropyl mercaptan, a compound with one of the most memorable scents on the planet. Companies add it as a warning agent. Natural gas doesn’t have an odor, and that can be dangerously deceptive. Without a distinctive smell, leaks may go unnoticed, leading to explosions or poisoning. That’s where isopropyl mercaptan steps in. It wakes up the senses, demanding attention if trouble starts brewing.
Safety Through Science
Plenty of folks haven’t heard the term “odorizing agent,” but lives depend on it every day. Pipelines and storage tanks line communities, and technicians need a method to alert everyone near a leak before problems escalate. Isopropyl mercaptan brings an unmistakable signal, cutting through everything else in the air. This makes it one of the standard choices in the utility industry. Regulators back up its use, with rules in place across North America and Europe mandating odorization for public safety.
Other Places It Shows Up
My years around industrial sites taught me to recognize the smell in other settings. Chemical makers rely on isopropyl mercaptan during production processes. Within specialty chemistry, it acts as a building block—often in the creation of pesticides or pharmaceuticals. Workers use thick gloves and face protection because of its intensity. I’ve seen bottles in research labs, always sealed up tight. One drop outside the bottle, and the whole building starts to notice.
Problems and Health Risks
There’s no getting around it: the aroma comes with trade-offs. Extended exposure leads to headaches, nausea, or worse. Emergency crews train for spills, using high-powered fans and absorbents to soak up leaks as fast as possible. The Environmental Protection Agency and OSHA warn about safe handling. Most workplaces put strict guidelines in place. I’ve met plant operators who described bolting outdoors the moment they caught a whiff indoors—better safe than sorry.
The Path Ahead: Safer Use and Better Detection
Technology always looks for more precise options. Companies now experiment with remote sensors tied to smartphones and alarms, aiming for quicker response. Some work goes into training homeowners as well—recognize the smell, leave the building, and call for help. Efforts to cut down on accidents start with education, right alongside safer packaging and storage. The chemical’s usefulness persists, but only where responsibility sits front and center.
Why It Matters
Each time I pass a gas meter or notice those little yellow warning stakes on a neighbor’s lawn, I think about the unseen risks. Isopropyl mercaptan isn’t glamorous. Still, it saves lives every year, and smart handling ensures it only surprises us for the right reasons. By paying attention to where and how it gets used, people keep their families, coworkers, and communities out of harm’s way. That simple, sharp smell carries a message nobody should ever ignore.
What are the safety precautions when handling Isopropyl Mercaptan?
Understanding Isopropyl Mercaptan Risks
If you’ve ever stepped into a chemical storage room, you know the sharp, rotten odor that comes from a broken seal or a loose cap. That’s isopropyl mercaptan for you. I’ve crossed paths with it on more than one occasion inside busy plants, and it doesn’t take much to respect just how volatile and pungent this chemical gets.
The key thing here is that isopropyl mercaptan is flammable and toxic. Even though it often shows up in small quantities as a warning agent in gas, its fumes pack a punch. Exposure isn’t just unpleasant. Contact or inhalation can cause headaches, dizziness, and irritation. Higher concentrations can even knock someone unconscious, and skin exposure leaves rashes and burns behind.
Practical Precautions You Should Never Skip
Every day on the job, you pick up tips from those who’ve learned the hard way. There’s nothing flashy about slipping on a chemical-resistant pair of gloves—just peace of mind. Nitrile or neoprene hold up well against leaks. Goggles that hug the face tightly and splash-protective aprons give a reminder not to cut corners on protection, even if the job seems simple.
A proper respirator—NIOSH-approved, cartridge fitted—is essential when there’s potential for fumes. It’s tempting to believe a quick task doesn’t need a mask, but I’ve seen coworkers regret that mistake. Even a few breaths in an enclosed area can cause fits of coughing and nausea.
Ventilation and Storage Really Matter
Chemical fumes drift fast. An open window doesn’t cut it in most labs or maintenance bays; good exhaust systems and fume hoods change the game. Take the time to double-check the airflow before uncapping anything. I remember a time a vent failed in a small lab. The difference between a humming fan and quiet air made the room unbearable in seconds.
As for storage, most fire codes require flammable chemicals like isopropyl mercaptan to stay in tightly sealed containers, away from sparks or heat. I always keep it locked up in a flame-resistant cabinet, nowhere near acids or oxidizers. Stories float around about forgotten bottles left in sunny windows or next to motors—accidents just waiting for the right mix of heat and vapors.
Training, Labels, and Emergency Preparedness
Anyone handling chemicals should read Safety Data Sheets closely. I always check containers for clear hazard labels, and I help others brush up on what each color or signal word means. Training goes beyond paperwork. Drills on what to do after a spill or exposure save time that could make the difference between a scare and an emergency room trip.
The eyewash station and emergency shower should sit within a quick dash from the work area. Colleagues have told me there’s no shame in rehearsing the path to safety because in the panic of an actual splash, instincts can freeze up.
Sharing Responsibility
Workers, supervisors, and safety officers need to look out for one another. If something feels off—leak, smell, unlabeled jar—it pays to speak up. Companies must stick to standards and invest in routine inspections, but nothing beats the watchdogs working side by side. I’ve noticed that a team culture built around communication and vigilance often avoids trouble before it starts.
Better Outcomes Through Common Sense
Being around isopropyl mercaptan doesn’t have to be risky. Wearing protective gear, respecting ventilation rules, and storing everything properly come down to simple habits. Teams that talk openly about what could go wrong, and who drill the basics, make everyone safer. It’s consistent, everyday choices—the sort anyone can make—that form the real shield against harm.
What are the physical and chemical properties of Isopropyl Mercaptan?
Getting Familiar with Isopropyl Mercaptan
People in the chemical industry don’t forget the smell of isopropyl mercaptan. The moment a container opens, it fills the air with a strong, sulfur-like odor. Skunk spray, rotting cabbage, natural gas leaks — the comparison fits. This distinct smell isn’t just a quirk. Utility companies purposely add it to otherwise odorless natural gas to warn us about leaks.
On the molecular level, isopropyl mercaptan’s formula — C3H8S — reveals its true nature as a thiol, with a sulfur atom bonded to an isopropyl group. It shows up as a colorless, volatile liquid that boils around 59°C (138°F). Because of its low boiling point, isopropyl mercaptan evaporates readily at room temperature. This property makes it easy to detect its presence in the air but also brings safety issues, because inhaling high concentrations can irritate the nose, throat, or even the lungs. Anyone who’s poured it or used it in industrial settings will tell you — ventilate, ventilate, ventilate.
Physical and Chemical Behavior
Isopropyl mercaptan mixes with many organic solvents but doesn’t blend well with water. Its low density — less than that of water — means it floats and spreads out quickly if spilled. This spreading, plus the smell, is a double-edged sword. Spills in a closed lab or facility do not stay a secret for long. Keeping it contained in storage becomes practical as a matter of safety and comfort for everyone working nearby.
The chemical’s reactivity deserves attention. That sulfur-hydrogen bond at the heart of every mercaptan brings a tendency to react with oxidizing agents. In real-world terms, that means you won’t find isopropyl mercaptan stored near strong bleach or peroxides. Mixing them can produce heat, even fire, or dangerous sulfurous gases. Industrial users learn to respect these hazards, not just because safety laws require it, but because the risks in real terms include property damage, injuries, or worse.
Importance in Everyday Life
Keeping natural gas safe hasn’t always been easy. The addition of mercaptans, including isopropyl mercaptan, has probably saved countless lives. Before this practice, gas leaks often went undetected, causing explosions or poisoning. Even as a chemist in training, I learned early on that the tiniest drop would alert everyone in the building. While that often meant some groans or jokes in the lab, it taught me how attention to detail and respect for sensory warning signs matter far beyond textbooks.
Addressing Hazards and Solutions
Anyone handling isopropyl mercaptan faces two main challenges: controlling its strong odor and minimizing exposure. Working with this substance safely means using tight-sealing containers, proper ventilation, and personal protective equipment. Facility design matters too — chemical storage areas need air handling systems capable of diluting and removing fumes.
Training and procedures must go beyond theory. Spill drills, protective gear checks, and careful inventory control form the backbone of a safe work environment. Digital sensors and alarm systems, along with old-fashioned “nose tests,” help catch leaks or evaporation early. Solutions like activated carbon filtration help capture stray vapors before the smell spreads or concentrations become hazardous.
Between its role as a warning agent and the unique physical and chemical quirks, isopropyl mercaptan demands respect. Real-world experience, not just charts and numbers, shapes how its properties impact safety and daily life in labs, factories, and utility systems everywhere.
What are the storage requirements for Isopropyl Mercaptan?
Why Handling This Chemical Matters
Working with Isopropyl Mercaptan, you notice right away that even a tiny whiff tells you trouble’s nearby. This liquid’s harsh, sulfur-like smell is no joke and points to its volatility and ability to rapidly affect air quality. Folks in factories, labs, and storage facilities have to take that seriously. Nausea, headaches, and even more serious health hits can result from accidental exposure. The risks are not just for those handling the substance. Leaks impact neighbors, first responders, and even whole communities if managed poorly.
The Science and Storage Reality
Isopropyl Mercaptan lights up at low temperatures, somewhere around room heat, and can tear through seals if the material can’t stand up to organic sulfides. So corrosion quickly drains tanks, and vapors slide through loose lids or cheap gaskets. Stainless steel and high-grade plastics like HDPE hold up better after years of testing. The best storage always means containers not just rated for hazardous liquids, but more specifically for flammables with a reputation for chemical attacks on softer materials.
Ventilation: Life-Saving Simplicity
No matter how secure the storage vessel, you can’t skip good airflow. Closed spaces with vapors trapped can create a flash-fire hazard—one static spark spells disaster. Local exhaust hoods, spark-free fans, and sometimes vapor-detectors have to stand guard in the room where drums or tanks wait. A single broken pipe can force building-wide evacuations. Air-handling gear needs regular checks unless you want to wake up to alarms or, worse, a call from the fire brigade.
Rule Book and Real Experience
The regulations aren’t just paper exercises. OSHA and NFPA codes grew out of painful lessons from chemical fires and poisonings. From my own time walking plant floors and helping set up chemical supply rooms, it’s clear: any shortcuts add up to risk. Shops that kept drums off the ground on proper spill pallets and away from sunbaked windows rarely faced leaks or chemical burns. Combining those steps with strict no-smoking signs, a detailed map of exits, and quick access to firefighting equipment keeps surprises manageable.
Spill Control and Site Specifics
Spillage routines matter just as much as long-term storage. Granular absorbents, not rags, stay ready close by. Emergency showers show up on site maps. Working with Isopropyl Mercaptan often forces companies and labs to re-evaluate their safety routines. Double-checking for hidden ignition sources and keeping personal protective gear accessible can turn a crisis into a cleanup. Having written procedures speeds up response and limits property and health damage.
Moving Beyond Bare Minimums
Too often, the question ends at meeting codes instead of practical safety. Workers talk about actual incidents or near-misses, not just compliance. A locked cabinet in a cool, shaded area often beats forgetting a drum near a heat source or letting a relief vent point toward walkways. Routine training and regular walk-arounds catch problems before they balloon. Some folks set up remote monitoring for tank pressures and temperatures: investing early keeps big costs at bay later.
Solutions that Stick
Actively managing storage for Isopropyl Mercaptan, instead of hoping the gear holds up, pays off. Honest safety culture, the right equipment, and a willingness to fix small leaks before they grow keep people healthy and sites running. The right attitude isn’t just about following the rules, but learning from past failures and treating every drum like it could be the next headline if ignored.
What should I do in case of Isopropyl Mercaptan exposure or spill?
Why You Should Take Isopropyl Mercaptan Seriously
I’ve dealt with more than a few chemical incidents in workplace settings, and none leave a lasting impression quite like Isopropyl Mercaptan. Its rotten egg smell hits your nose hard and fast, making you think about the health risks before you even read a label. This stuff packs a punch—causing irritation to eyes, skin, and lungs. It’s easy to underestimate volatile compounds if you haven’t had a close call, but I’ve seen firsthand how even small slips can cause headaches, nausea, and an all-around bad time for everyone in the area.
Immediate Steps for Personal Safety
If you or your coworkers get exposed, don’t stick around hoping the smell clears up. Ventilation becomes key. Open nearby windows and doors. Turn on exhaust fans if you have them. Fresh air often helps people recover from breathing in low levels of mercaptans.
Skin contact? Head straight for a wash station. Soapy water washes away contamination much better than just a quick rinse, so don’t skimp on the scrub time. If you get chemical in your eyes, hold your eyelids open and flush with water for at least 15 minutes. Don’t take risks if someone has a lingering cough or dizziness—call a doctor and bring along the chemical safety data.
Containing Spills Without Panic
Having spill kits on hand turns chaos into a controlled clean-up. I used to work in maintenance, where speed and steady hands avoided bigger problems. Wear gloves and chemical-resistant goggles. Put on a respirator if the stink fills the whole room. Absorb the spill with inert material, like clay or commercial spill pillows. Avoid water on small spills—it can spread vapors and make the smell worse.
Gather up the soaked absorbents and put them in a sealed, labeled container. That part matters, since janitors and waste haulers rely on knowing what’s inside. Tossing stuff in the regular trash just passes the danger onto someone else down the line. Notify the right people—supervisor, safety officer, or building management—so nobody misses hidden hot spots days later.
Learning from Incidents and Staying Ready
A close call with mercaptans convinced me that reading MSDS sheets before starting a job isn’t overkill. If a company doesn’t provide clear signage, ventilation, and cleanup materials, that’s a sign to speak up or refuse to work.
Emergency drills weed out confusion. Running a mock spill means people know which exits stay clear, where the eyewash stations hide, and who takes charge if a room fills with fumes. It’s a lot easier to make mistakes in practice than when someone’s coughing, so don’t let drills become paperwork exercises that everyone ignores.
Getting Ahead of the Problem
Wearing the right personal protective equipment saves skin and lungs. As someone who’s ruined more than one favorite shirt on cleanup jobs, I can vouch for specialized gloves and aprons. If you work with mercaptans, store chemicals in tight, labeled containers, away from vents or break rooms. Make air monitors standard in spaces where vapors could build up—don’t rely on your nose as a warning system.
By speaking out for safety and sharing lessons learned, we help less experienced coworkers avoid nasty run-ins with this chemical. Relying on teamwork—rather than lone cleanup efforts—cuts down on risk and helps everyone breathe a little easier at the end of the day.
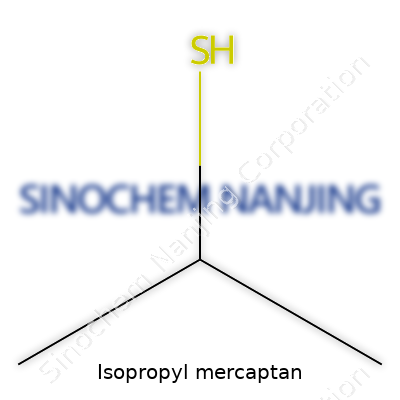
| Names | |
| Preferred IUPAC name | propan-2-thiol |
| Other names |
2-Propanethiol Isopropanethiol Propan-2-thiol 2-Mercaptopropane IPM |
| Pronunciation | /ˌaɪsəˈproʊpɪl mərˈkæptæn/ |
| Identifiers | |
| CAS Number | 75-33-2 |
| 3D model (JSmol) | `Isopropyl Mercaptan JSmol 3D model string`: ``` CS(C)C ``` |
| Beilstein Reference | 637505 |
| ChEBI | CHEBI:59416 |
| ChEMBL | CHEMBL137098 |
| ChemSpider | 10313 |
| DrugBank | DB14008 |
| ECHA InfoCard | 100.002.696 |
| EC Number | 200-453-6 |
| Gmelin Reference | 844 |
| KEGG | C14133 |
| MeSH | D008438 |
| PubChem CID | 7922 |
| RTECS number | NT5250000 |
| UNII | DJ8R97C43M |
| UN number | UN 2347 |
| CompTox Dashboard (EPA) | DTXSID3020226 |
| Properties | |
| Chemical formula | C3H8S |
| Molar mass | 76.16 g/mol |
| Appearance | Colorless transparent liquid with unpleasant odor |
| Odor | Unpleasant, skunk-like |
| Density | 0.794 g/mL at 25 °C |
| Solubility in water | Slightly soluble |
| log P | 1.96 |
| Vapor pressure | 40 mmHg (20°C) |
| Acidity (pKa) | 10.6 |
| Basicity (pKb) | Product has pKb of 3.77 |
| Magnetic susceptibility (χ) | -38.2×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.443 |
| Viscosity | 0.42 mPa·s (at 20°C) |
| Dipole moment | 1.59 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 199.0 J mol⁻¹ K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -117 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2286 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS07, GHS08, GHS09 |
| Pictograms | GHS02,GHS06 |
| Signal word | Danger |
| Hazard statements | H225, H301, H311, H331, H315, H319, H335, H410 |
| Precautionary statements | P210, P233, P260, P264, P271, P273, P301+P310, P302+P352, P304+P340, P305+P351+P338, P308+P311, P332+P313, P333+P313, P337+P313, P362+P364, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 3-4-2-W |
| Flash point | -38 °C (-36 °F) |
| Autoignition temperature | 399 °C (750 °F) |
| Explosive limits | 2%-9.2% |
| Lethal dose or concentration | Lethal dose or concentration (LD50/LC50) for Isopropyl Mercaptan: "LC50 (rat, inhalation): 1600 ppm/4H |
| LD50 (median dose) | LD50 (median dose): 179 mg/kg (oral, rat) |
| NIOSH | NIOSH: CN1400000 |
| PEL (Permissible) | PEL: 0.5 ppm (as an 8-hour TWA) |
| REL (Recommended) | 160 ppm |
| IDLH (Immediate danger) | IDLH: 500 ppm |
| Related compounds | |
| Related compounds |
Ethanethiol n-Propyl mercaptan tert-Butyl mercaptan |

