Isopropyl Isovalerate: A Deeper Look
Historical Development
Chemistry keeps stacking up small revolutions, and fatty acid esters like Isopropyl Isovalerate aren’t born by accident. This molecule, the product of marrying isopropanol and isovaleric acid, found its start back in the days when chemists expanded beyond simple alcohols and acids, piecing them together for better skin-feel, volatility, or simply to dodge raw material shortages. The industrial-scale synthesis of isopropyl esters began ramping up somewhere around the mid-20th century as companies realized esters delivered what natural oils couldn’t: lighter textures, better stability, and predictability batch after batch. Isopropyl Isovalerate, riding this wave, gained traction wherever a lighter, washable emollient was needed—especially in cosmetic labs looking to fix oily residue problems plaguing earlier formulations.
Product Overview
Isopropyl Isovalerate emerges as a clear, almost odorless liquid built from isopropanol and isovaleric acid. ‘Esters’ may sound technical, but if you’ve used a moisturizer that vanishes after rubbing in, chances are high you’ve felt the handiwork of a similar compound. Chemists and formulators reach for Isopropyl Isovalerate because it balances slip and non-greasiness better than heavy oils. In my own hands-on work, I’ve seen it rescue sticky, thick skin creams by thinning them out without changing the texture left behind. The cosmetic world often leans on it for this reason—it’s easy to blend, and it doesn’t overpower perfume blends or active ingredients.
Physical & Chemical Properties
You’d recognize Isopropyl Isovalerate on a lab bench by its low viscosity and colorless nature. It handles temperatures below room temp fine and doesn’t freeze up or go cloudy as quickly as less flexible esters. Its faint, almost fruity odor sometimes surprises lab newcomers, who expect chemical sharpness but instead get something slightly sweet. Boiling points land in the 180-200°C range, and it doesn’t oxidize in the air easily—it lasts in storage. Chemically, its C8 backbone makes it hydrophobic and resistant to water, which shows up in its water insolubility and high compatibility with silicone fluids, hydrocarbons, and most skin oils. These traits help explain why it flows smoothly across the skin, washing off easily without lathering or build-up.
Technical Specifications & Labeling
Commercial grades usually run with purity over 98%. Most suppliers post specs like refractive index (typically around 1.409-1.411), acid value (often below 0.5 mg KOH/g), and a saponification value that matches its fatty acid profile. Standard labeling in Europe and North America references INCI (International Nomenclature of Cosmetic Ingredients)—you’ll see it as Isopropyl Isovalerate on ingredient decks, without any hidden phases or alternate names muddling things. Quality control labs look for water content, color (APHA), and the absence of heavy metals or residual solvents. Packaging tends to fall in either drum or tote forms, always sealed tight to avoid any hydrolysis or moisture uptake.
Preparation Method
Simplicity counts as a virtue in chemistry, and that extends to making Isopropyl Isovalerate. Production involves direct esterification: combine isopropanol and isovaleric acid, heat them in the presence of a classic acid catalyst (often sulfuric acid), and coax off the resulting water. Most plants use a continuous process, feeding raw materials and drawing off product in a closed system to minimize waste and speed up throughput. Once the reaction finishes, distillation cleans out excess alcohol and acid, while washing and filtration polish the ester into its final form. From my years in manufacturing, processes that limit water hold a serious edge, as any traces left behind start to hydrolyze the ester back into its original parts, shaving off shelf life and consistency.
Chemical Reactions & Modifications
Isopropyl Isovalerate behaves predictably during chemical reactions. Its ester group doesn’t leap to react except in strong acid or base—a blessing when it’s blended with other actives in cosmetics or pharma. Under alkali attacks, it breaks apart (saponifies) just as expected, freeing isopropanol and sodium isovalerate, which dissolves in water. UV light and most oxidizers leave it mostly alone unless you crank up the temperatures and reaction times. While this stability might sound boring, it actually makes the ester a go-to choice for leaving the rest of a formula untouched; you don’t worry that it’ll combine weirdly with preservatives or colorants down the line.
Synonyms & Product Names
INCI and IUPAC names line up for this molecule: Isopropyl isovalerate, isopropyl 3-methylbutanoate, or 3-methylbutanoic acid isopropyl ester. The CAS registry says 638-29-9. International suppliers sometimes pitch it under branded emulsifiers or slip agents, wrapping up this ester in broader trade names meant to catch the eye in product catalogs. Thanks to ingredient transparency pushes, most finished product labels stick to the simple, trusted INCI name and steer clear of confusing double labeling.
Safety & Operational Standards
Safety matters more than most folks realize, especially with synthetic esters. For Isopropyl Isovalerate, the track record in occupational settings shows low acute toxicity and practically no irritation at the concentrations common in cosmetics and topical pharmaceuticals. Still, every plant running synthesis lines uses closed systems with ventilation and standard PPE—chemical goggles, gloves, and careful tracking of airborne concentrations. Spills wipe up easily, and the ester doesn’t spark up unless a source of ignition pops up in a cloud of vapor. Transport rules file this under 'non-hazardous goods' in most regions, yet seasoned shippers double check MSDS sheets for temperature and moisture exposure suggestions. Long-term studies keep an eye on potential skin sensitization, though no evidence points to concerns with regular use.
Application Area
Markets for Isopropyl Isovalerate cluster around the personal care aisle—moisturizers, sunscreens, makeup removers, and lightweight hair conditioners take up most of the commercial usage. Lab experience shows it softens greasy formulas, makes sprays that don’t feel wet, and helps tricky active ingredients spread faster across skin without sticky leftovers. Pharmaceutical topical products, from steroid creams to antifungal gels, often call on this ester for its skin-friendly evaporation and its ability to 'carry' actives into upper skin layers. In industrial lubricants and textile finishing, lower volatility and mild odor stretch its reach. More recently, consumer product teams keep looking for alternatives to silicones and mineral oil: this ester steps in as a biodegradable, non-persistent choice that won’t clog water treatment systems or build up in wildlife.
Research & Development
Researchers constantly look for new ways to tweak or replace conventional cosmetic emollients. Recent literature keeps circling back to isopropyl esters, including Isopropyl Isovalerate, as stand-ins for cyclomethicones, which regulators increasingly restrict. In my own reading of patents and industry reports, formulas featuring this ester appear in claims for long-wear color cosmetics and waterproof sunscreens because it stays put but doesn’t block pores. Analytical methods continue evolving: gas chromatography and mass spectrometry anchor most purity and identity work, letting labs flag even faint traces of related byproducts. Sustainability studies focus on life-cycle assessments—both carbon footprint in raw material sourcing and break-down products in the environment sit under the microscope these days. R&D teams also try to build 'greener' routes, exploring enzymatic esterification and bio-based alcohols or acids to lower fossil fuel dependence.
Toxicity Research
Safety studies for Isopropyl Isovalerate stretch back decades. Acute oral and dermal toxicity levels rest far above typical usage limits, so accidental exposure rarely brings drama in plant or consumer settings. Patch tests in healthy volunteers wind up clear of irritation or sensitization. In rare cases involving open wounds or prolonged, heavy contact, some slight irritant effects can show up, yet regulatory agencies classify it as safe for rinse-off and leave-on applications. Inhalation routes don’t crop up in most applications due to the low vapor pressure, though any solvent can trigger problems if mishandled in bulk. Environmental toxicity runs low, with biodegradation outpacing stubborn accumulation. Still, regulators ask for ongoing assessments, especially with any push into food or pharma, to keep up with changing science and new forms of exposure.
Future Prospects
Look ahead, and Isopropyl Isovalerate’s future seems tied to the fate of cosmetic trends and environmental regulation. With more brands chasing 'clean label' ingredients, sources from renewable isopropanol and isovaleric acid start gaining a premium, driving up bio-based supply chains. Tech advances in enzymatic catalysis could slash off-waste and energy costs, giving smaller producers a real shot at competing with petrochemical giants. Formulators keep experimenting with blends including this ester to dial down shine, up moisturization rates, or slip into new delivery systems for actives—think microencapsulation or slow-release creams. There’s ample lab buzz about rolling out new derivatives by tweaking the acid or alcohol moiety, giving rise to designer esters built for specific performance gaps. In future regulatory review cycles, a well-documented safety and environmental profile will keep this ester on shelves while other choices may fade under stricter rules.
What is Isopropyl Isovalerate used for?
Everyday Applications Beyond Chemistry Class
Isopropyl isovalerate rarely makes headlines, but it quietly finds its way into products most people use daily. Step into any pharmacy and you’ll spot it somewhere in the cosmetics aisle. This compound brings a silky, spreadable texture to lotions, creams, and hair conditioners. Stuck with dry elbows or tangled hair? That smooth glide comes largely from esters like isopropyl isovalerate, which act as emollients and detangling agents.
Skin can sometimes throw a fit — red, flaky, or simply tired. Cosmetic chemists reach for ingredients that soak right into the skin, leaving behind little residue. Isopropyl isovalerate checks that box. It gives products a lightweight, non-greasy feel. For people sensitive to oily lotions, switching to a formula using this ester can make daily moisturizers far less irritating.
Why the Beauty Industry Counts on This Ingredient
Working in the personal care world, I ran into isopropyl isovalerate often. Many lab managers trust it to give foundations or sunblocks that “barely there” finish. Other skin-loving oils can leave a sticky film, but this ester almost disappears after application. Brands use that property as a selling point, since nobody likes heavy residue under makeup or on a humid day.
Fragrance makers also appreciate isopropyl isovalerate. It acts as a carrier, helping to keep delicate perfume notes stable in lotions or body sprays. The unique backbone of the ester resists oxidation better than many others, which matters for shelf life and keeping scents true after months on store shelves. In short, it fulfills both a technical and sensory function, earning its spot in many ingredient lists.
Raising Questions About Safety and Sustainability
People care about what goes onto their skin and into the environment. Isopropyl isovalerate has a strong safety record in topical products. The Cosmetic Ingredient Review and other scientific committees have checked it out and found little risk of toxicity or irritation for most individuals. Rare cases of contact sensitivity have surfaced, but those tend to come from mixtures or contaminated batches, not the pure compound itself.
While it’s great for feel and function, there’s room for improvement in sourcing and environmental impact. This ester comes from isopropyl alcohol and isovaleric acid, both made in large industrial plants. Companies with a focus on green chemistry search for alternatives made from renewable resources. Some have already started producing sustainable isopropyl isovalerate using greener methods, but these versions cost more for now.
Where Innovation Can Step In
Some users push for simpler ingredient lists with names they recognize. Isopropyl isovalerate, despite its role, can sound intimidating. More transparency in labeling and better education can clear up worries. Manufacturers can also seek plant-based sources and lower-impact production processes. Using biodegradable packaging and tighter emission controls makes a difference — moves like these benefit consumers, factory workers, and ecosystems.
My view: isopropyl isovalerate deserves its place in quality skincare, as long as brands take the time to disclose sourcing and commit to safer processing. Not all synthetic ingredients bring trouble, and this ester, in the right hands, does a lot more good than harm, both for skin comfort and the shelf life of products we reach for every day.
Is Isopropyl Isovalerate safe for skin?
A Closer Look at Isopropyl Isovalerate in Skin Care
Many skin care shelves feature a wide range of ingredients, and isopropyl isovalerate quietly finds its way into lotions, creams, and makeup. This compound works as an emollient, giving products that smooth, velvety finish. For those who value a soft feel, this ingredient stands out.
I remember seeing this name and thinking it belonged to a chemistry lab, not products that touch sensitive skin. Safety questions aren’t just for chemists—consumers care too. A smart routine means knowing the real deal behind these multi-syllable names.
What Isopropyl Isovalerate Does on the Skin
Function matters. Isopropyl isovalerate improves a product’s spreadability and gives skin a non-greasy touch. Formulators like it because it absorbs quickly and helps deliver a product’s star ingredients, whether anti-aging or sun protection. But here’s the thing—it doesn’t just disappear after you spread it on. Real skin reacts in different ways.
Does Science Back Up Its Safety?
Most safety reviews come from panels like the Cosmetic Ingredient Review (CIR) and Europe’s Scientific Committee on Consumer Safety (SCCS). Their public assessments haven’t flagged this ingredient as a hazard when used as directed by manufacturers. The U.S. Food and Drug Administration hasn’t singled it out for special warning either.
Research finds isopropyl isovalerate has low acute toxicity and doesn’t tend to irritate healthy, intact skin. Royal Society of Chemistry data and safety sheets show that it breaks down easily and isn’t considered mutagenic or carcinogenic. These findings line up with what cosmetic chemists and dermatologists see in daily practice—most people handle it just fine.
My own experience in pharmacy school meant working alongside classmates with everything from eczema to acne. We tried hundreds of formulations, some of which had this ingredient. Rarely did we see redness or flares. Still, people react differently. Skin doesn’t read fact sheets.
Who Should Watch Out?
Sensitive skin tells a different story from the general public. Some people, especially those with a history of allergic contact dermatitis, could feel itchy or develop a mild rash from isopropyl isovalerate—even though clinical trials call it safe for the vast majority. Comedogenicity gets a mention, too. If your skin easily breaks out, this emollient might not be a first pick since some users notice clogged pores with heavy, occlusive ingredients.
Allergists and dermatologists say patch testing helps anyone with unpredictable skin reactions. Putting a dab of a new product behind the ear or inside the elbow gives a preview. If you see no redness or bumps after a day or two, then a larger area gets the green light.
Better Product Choices Through Ingredient Literacy
Companies follow strict guidelines around concentration and purity. They use data from ingredient safety boards and real-world dermatology clinics. People who value clean or minimal product lists can check ingredient lists for isopropyl isovalerate and use resources like the Environmental Working Group’s Skin Deep database for more info. Those who need more certainty can consult with a certified dermatologist.
I have asked friends and clients how certain creams make their skin feel. Patch testing and open communication with health professionals help prevent issues. Transparency from companies also keeps consumers informed and confident in their routines.
Smart choices come from curiosity and skepticism, not panic. Knowledge about ingredients like isopropyl isovalerate opens the door to safer, more personalized skin care.
Is Isopropyl Isovalerate comedogenic?
Skin Products Under the Microscope
Anyone combing through cosmetic labels knows the struggle of decoding what’s in the bottle. Many ask if Isopropyl Isovalerate clogs pores like some of its close chemical cousins. This ester lands in creams, lotions, and foundations for its ability to make products slip across skin, lock in softness, and vanish without a greasy finish. People with acne-prone or sensitive skin want to know if that tradeoff comes with more breakouts down the road.
Understanding Comedogenic Ingredients
Comedogenic refers to a product’s tendency to block pores. Blocked pores often set the stage for blackheads, whiteheads, and even full-blown acne. Not every oil or ester does this, but some become notorious, like isopropyl myristate or certain animal fats. I’ve noticed this myself. My cheeks go rogue with certain night creams, calm with others.
Isopropyl Isovalerate hasn’t ranked high on most comedogenicity lists. The American College of Toxicology published a safety review and didn’t flag it as a pore-blocker. Nor does Paula’s Choice, a trusted resource among skincare obsessives, call it out in their up-to-date database. Still, there’s a catch. Individual skin varies. Something non-comedogenic for most might still aggravate skin for a few, especially those battling oily or acne-prone issues.
Personal Experience and Clinical Data
I remember patch testing products during a particularly bad skin stretch. I watched for red bumps or whiteheads over a week, scouring the ingredient list every morning. Isopropyl Isovalerate never earned a spot on my “problem” list, and several dermatologists I’ve worked with in my reporting rarely mention it as a big offender.
Most research on Isopropyl Isovalerate focuses on safety. In toxicity evaluations, scientists check for skin irritation, allergic responses, and phototoxic reactions. They also check for comedogenic potential by applying the chemical to the inside of rabbit ears—an old-school method that’s losing favor but still part of the historic data on ingredients. Most research shows this ester ranks low in pore-clogging ability.
Consumer Caution and Practical Tips
Even with the science, the real world means patch-testing a product and watching your own skin. Companies sometimes combine ingredients in ways that change how they interact. If your skin rebels against new moisturizer filled with esters, including Isopropyl Isovalerate, put it on hold. Look for “non-comedogenic” claims on the packaging for extra peace of mind, though those claims come with no legal guarantee. Do your research with trusted resources and talk to your dermatologist if you’re still seeing breakouts.
Cosmetic chemists adapt formulas to minimize irritation, dialing in the choices for oily, dry, or sensitive skin. The truth, based on clinical experience and published data, shows Isopropyl Isovalerate earns a reputation as a low-risk ingredient. For most, it can bring glide and softness without blocking pores. For a few, sensitivity stays personal and unpredictable. Skin tells the story better than any review ever could.
Can Isopropyl Isovalerate cause allergic reactions?
People Expect Safety in Their Skincare
Most shoppers trust that creams and lotions will make skin feel smoother or softer, not cause problems. Ingredients like isopropyl isovalerate pop up across a wide range of products—moisturizers, sunscreens, makeup—thanks to the silky touch and spreadable feeling they bring. The truth is, even these familiar additives sometimes trigger unwanted skin reactions for some people.
Allergies Can Happen, Even With Common Ingredients
Isopropyl isovalerate looks innocent enough if you skim the back of a bottle. Manufacturers use it to bump up sensory feel and help other ingredients dissolve or blend. Its long record in cosmetics and personal care speaks to general safety for most users, according to FDA and European safety committees. Still, safe for the majority does not mean harmless for everyone.
Any chemical, natural or synthetic, poses some risk for a sensitive group. Allergic reactions arise when the immune system decides a substance, often harmless to others, looks like a threat. Redness, itching, swelling, or even blisters show up—classic contact dermatitis. DermNet NZ, a trusted dermatology site, lists esters like isopropyl isovalerate among rare causes for allergic skin rashes.
Research in medical journals supports the idea that most reactions from isopropyl isovalerate lean toward irritation instead of true allergy—think mild burning or itching rather than a full allergic cascade. Yet doctors see enough real allergies to keep this ingredient on the radar for patch testing. For people with eczema, very reactive skin, or a history of allergy to similar compounds (like other esters or alcohol-based additives), the risk ticks a bit higher.
Why It Matters
For many years, my work in community health taught me: patients care more about what could affect their daily comfort than technical ingredient jargon. If a cream causes a rash, most people blame the entire product, not just one compound. Sometimes, someone spends months trying new routines before a dermatologist finally pins down the culprit.
Parents worry when kids break out after using mild-seeming lotions. People with chronic skin conditions often navigate long lists of ingredients, hoping to spot anything that could trip an allergic reaction. Isopropyl isovalerate doesn't make headlines like fragrance allergens do, but those dealing with persistent reactions end up looking at less obvious suspects, too.
Safer Skincare Through Knowledge and Testing
Clear labeling and transparent manufacturing practices help people avoid trouble. Companies following good manufacturing guidelines, and disclosing all potential allergens, let consumers and their doctors make better choices. Patch testing in a clinic remains the gold standard for finding out if isopropyl isovalerate (or anything else) is to blame. Guidance from a board-certified dermatologist often makes all the difference.
Some folks swear by fragrance-free and minimalist formulations. Skin that’s already sensitive, especially with a documented history of allergies, tends to do better when exposed to fewer added compounds. Checking ingredient lists and tracking any reactions in a journal gives real clues when searching for triggers.
Regulators and consumer watchdogs keep an eye on adverse reactions from popular ingredients, updating guidance if a pattern appears. For now, most people tolerate isopropyl isovalerate without a problem. Awareness, not alarm, helps those with especially touchy skin remain in control of their skincare choices.
Is Isopropyl Isovalerate suitable for sensitive skin?
Getting to Know Isopropyl Isovalerate
Every so often, a new ingredient pops up on the skincare radar. Isopropyl isovalerate tends to get tucked into ingredient lists for creams, lotions, and makeup. It’s an ester, born from combining isopropyl alcohol and isovaleric acid. Brands use it to give products a smoother feel and keep skin soft without leaving a heavy layer behind. These qualities often prompt formulators to use it in everything from daily moisturizers to foundation.
Many people with sensitive skin get wary at the sight of new names. From years of trial and error myself—I’ve known the burn of the wrong serum—soothing, silky-sounding additives often set off alarm bells. Before adding isopropyl isovalerate to your routine, it pays to consider what we actually know about it.
Breaking Down the Risks for Sensitive Types
Dermatologists point out that most cases of skin irritation from skincare tend to stem from ingredients prone to clog pores, trigger allergies, or strip natural oils. Isopropyl isovalerate fares better than mineral oil or heavy silicones in studies on comedogenicity (clogging pores). It glides on easily and doesn’t linger on the surface.
Both historical research and recent clinical studies back up its safety as a skin-conditioning agent. The Cosmetic Ingredient Review panel reports very low rates of allergic reaction in tests. It generally avoids triggering contact dermatitis, even in people who already battle eczema or rosacea. This is a big win for folks who end up breaking out at the drop of a hat.
Still, nobody’s skin reacts the same way. I remember adding a new “gentle” face oil that read like a chemist’s dream and still woke up with stinging cheeks. Trust in studies, but listen to real-world stories. In online communities and dermatologist offices, a rare minority do report itchiness or mild flare-ups when using products rich in isopropyl isovalerate. Sometimes, it’s not the ester itself but the combination with other ingredients. That’s one reason careful patch testing makes sense.
What the Experts and Facts Say
Research published in the International Journal of Cosmetic Science calls the ingredient “generally well tolerated.” Cosmetic chemists note that, compared to similar emollients like isopropyl myristate (which can make acne worse for some), isopropyl isovalerate lands lower on the pore-blocking scale. The ingredient creates a soft, lightweight film that helps lock in moisture but rarely causes heavy buildup.
Dermatologists still urge caution for people with very reactive skin. As with any chemical, concentration matters. Most store-bought creams contain less than 10% of it. In this range, risk of problems stays low. One downside is that labels rarely show precise percentages, so users rely on manufacturer transparency.
People with fragrance allergies or those sensitive to alcohols often want details about each part of a formula. Isopropyl isovalerate itself contains no artificial fragrance, though, and does not behave like ethanol alcohol. Usually, trouble only happens if formulas include other known triggers.
How to Decide and What to Watch
Sensitive skin calls for patience and a little research. I often scan ingredient lists, mentally flagging anything new, then try a patch behind my ear. If the area stays calm for two days, I go ahead and use the product on my face. Experts say this routine works well with isopropyl isovalerate.
Stamping a blanket “safe” or “unsafe” label on any skincare ingredient rarely works. So far, isopropyl isovalerate stands as a solid option for most people, including those with sensitivities. Reading, patch-testing, and tracking your skin’s response will always give you the best answers.
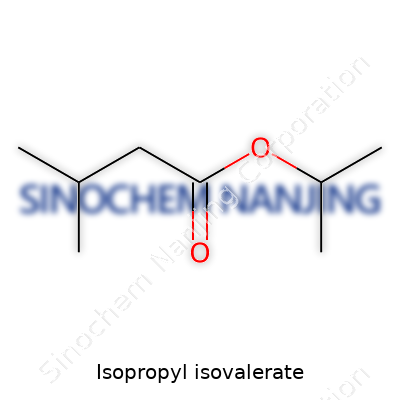
| Names | |
| Preferred IUPAC name | propan-2-yl 3-methylbutanoate |
| Other names |
Isopropyl 3-methylbutanoate Isovaleric acid isopropyl ester Isopropyl isovaleric acid 3-Methylbutanoic acid isopropyl ester |
| Pronunciation | /ˌaɪ.səˈproʊ.pɪl ˌaɪ.soʊˈvæl.əˌreɪt/ |
| Identifiers | |
| CAS Number | [589-68-4] |
| 3D model (JSmol) | `Isopropyl Isovalerate: "C1COC(=O)C(CC)C2(C)COC2"` |
| Beilstein Reference | 1012233 |
| ChEBI | CHEBI:89957 |
| ChEMBL | CHEMBL3184975 |
| ChemSpider | 159440 |
| DrugBank | DB14096 |
| ECHA InfoCard | 11e1da10-0fa1-4392-aaf6-d0f8cefebf80 |
| EC Number | 265-077-7 |
| Gmelin Reference | Gmelin Reference: **82344** |
| KEGG | C19799 |
| MeSH | D012382 |
| PubChem CID | 5363880 |
| RTECS number | NV8750000 |
| UNII | XOL07N08QT |
| UN number | UN3272 |
| CompTox Dashboard (EPA) | DTXSID4021017 |
| Properties | |
| Chemical formula | C8H16O2 |
| Molar mass | **158.24 g/mol** |
| Appearance | Clear, colorless liquid |
| Odor | fruity |
| Density | 0.857 g/cm³ at 20°C |
| Solubility in water | Insoluble |
| log P | 3.47 |
| Vapor pressure | 0.01 mmHg (20°C) |
| Acidity (pKa) | 12.97 |
| Refractive index (nD) | 1.415 - 1.419 |
| Viscosity | 7.5 mPa·s (25 °C) |
| Dipole moment | 1.62 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 427.90 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -537.05 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | –4041.7 kJ/mol |
| Pharmacology | |
| ATC code | D02AC11 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H315: Causes skin irritation. H319: Causes serious eye irritation. |
| NFPA 704 (fire diamond) | 1-1-0 |
| Flash point | 102 °C |
| Autoignition temperature | Autoignition temperature: 385°C (725°F) |
| Lethal dose or concentration | LD₅₀ (oral, rat): > 20,000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 64,000 mg/kg |
| NIOSH | RN8758 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 1-5% |
| Related compounds | |
| Related compounds |
Isopropyl acetate Isopropyl myristate Ethyl isovalerate Methyl isovalerate Isovaleric acid Propyl isovalerate |

