Isopropyl Isocyanate: Insightful Perspective on Its Role in Modern Chemistry
Historical Development
Chemists in the twentieth century pushed the boundaries of industrial organic synthesis, and Isopropyl Isocyanate surfaced in the wave of innovation that swept through the field. Tracing its development links back to the rise of isocyanate chemistry, fueled by a booming plastics and coatings industry. Companies and researchers worked to design new alkyl isocyanates for specialized applications, giving birth to isopropyl derivatives as demand for selectable reactivity and volatility increased. Over time, this compound gained recognition for distinct chemical behavior, earning a spot among key precursors for rigid polyurethane foams and specialty intermediates. Generations of researchers have navigated safety and production hurdles, each trade-off shaping the standards and methods recognized today.
Product Overview
Standing out as a specialty chemical, Isopropyl Isocyanate bridges laboratory theory and practical industrial application. It carries an unmistakable pungent odor and serves as a powerful alkylating agent. Most often, industry professionals reach for this chemical during the synthesis of urea and carbamate derivatives or in stepwise construction of pharmaceutical intermediates. Companies engaged in fine chemical synthesis store and ship this compound under controlled environments because batches can rapidly degrade without proper containment. Packaging sizes and handling recommendations reflect not just commercial scale, but attention to safety, given its reactive nature.
Physical & Chemical Properties
This organic isocyanate presents as a colorless to pale yellow liquid at room temperature. Volatility remains high, and even slight heating triggers the quick release of toxic vapor. With a molecular weight of about 99.13 g/mol and a boiling point near 71°C, professionals encounter a material demanding technique during handling. It dissolves in a variety of organic solvents, showing keen reactivity with water to release carbon dioxide along with heat. The isocyanate group, sitting opposite the isopropyl unit, gives the molecule its electrophilic punch—making it one to handle with care and respect in any lab setting.
Technical Specifications & Labeling
Industry technical sheets supply CAS numbers and molecular formulas to reduce the risk of mix-ups, especially since many isocyanates share similar physical characteristics. Clear hazard symbols warn of respiratory and dermal hazard, and labeling leans into regulations meant to protect both users and the environment. Detailed shelf-life and storage instructions underscore that temperature, humidity, and light exposure all affect the product. The material safety datasheet sets standards across sites so that no staff member runs blind when using or moving the compound.
Preparation Method
Synthesis usually starts with isopropylamine—an accessible feedstock—connected to the phosgenation process that revolutionized isocyanate preparation last century. Phosgene, toxic in its own right, reacts with isopropylamine via a tightly controlled gas-phase reaction, producing Isopropyl Isocyanate along with side products like hydrochloric acid. Macro-scale operations require closed systems to trap and neutralize both intermediate vapors and byproducts, reflecting years of engineering to keep emissions down and yields up. Some innovators experiment with safer carbonylation agents or milder oxidative routes, but industry often returns to the proven reliability of phosgene chemistry despite its demanding protocols.
Chemical Reactions & Modifications
Once in hand, chemists value Isopropyl Isocyanate for its rich reaction portfolio. It rapidly reacts with alcohols to give urethanes, one of the foundational linkages in the plastics sector. In another vein, it delivers stable urea derivatives in the presence of amines, creating structures used for agricultural and drug intermediates. Treating it with water yields carbon dioxide and isopropylamine—useful for purposeful deactivation or scrubbing. Cyclizations and other ring-forming steps sometimes use this isocyanate for inserting nitrogen and carbon units into larger frameworks. Rarely do professionals expose it to open air for long, recognizing its tendency to decompose or form unwanted polymers almost without warning.
Synonyms & Product Names
In the chemical trade, Isopropyl Isocyanate may appear as 2-Isopropyl Isocyanate, Propan-2-yl isocyanate, or its systematic name, 1-methylethyl isocyanate. Each label follows international standards to clear up any confusion on sourcing or documentation. Large chemical suppliers often attach their own catalog numbers, and distributors list common product codes on invoices to distinguish high-purity grades from technical or stabilized forms. Even seasoned buyers double-check synonyms before signing off on procurement to eliminate confusion with other isocyanates that might only differ by a methyl group.
Safety & Operational Standards
Acute toxicity stands front and center in any discussion on handling or storing Isopropyl Isocyanate. Teams working with this chemical don full-face respirators and protective gloves, knowing vapors irritate eyes, skin, and respiratory tracts almost immediately. Facilities require fume hoods and specialized ventilation, and emergency showers sit close to reaction spaces. Spills set off alarm bells because even small amounts can quickly vaporize, meaning cleanup teams must respond with absorbents and neutralizing solutions containing ammonia or similar chemicals. Local fire marshals and industrial insurers drill into facilities about tightly monitored storage temperatures—never exceeding 30°C—and ensure stock rotation prevents dangerous buildup of old, degraded material. Every lab tech and engineer gets trained on disposal and accident response, because experience shows unpreparedness leads to serious, sometimes irreversible injuries.
Application Area
Isopropyl Isocyanate carves a niche where reactive intermediates take center stage. In pharmaceutical synthesis, it slots into schemes for making selective carbamates, which find their way into medicines or protective coatings for sensitive molecules. Agrochemical labs pick it as a building block in pesticide development, tailored to disrupt specific insect metabolic pathways. The plastics industry pulls on this compound for specialized polyurethane elastomers and coatings, especially where resistance to certain solvents or abrasion is non-negotiable. Some electronics manufacturers reach for urethane-thin films, depending on precisely formulated isocyanates. Its short shelf life and aggressive reactivity mean it rarely gets stored in bulk outside of a few specialized sectors.
Research & Development
Academic interest in Isopropyl Isocyanate sits at the intersection of synthetic methodology and green chemistry. Chemists keep searching for alternatives to phosgene routes, mindful of both safety and sustainability. In some places, researchers try to design catalytic cycles relying on less hazardous reagents or renewable feedstocks, but have yet to report a broadly adopted breakthrough. Analytical experts keep deepening their understanding of isocyanate’s environmental fate, developing techniques to detect low levels of vapor in workplace air. Tools such as gas chromatography and NMR help trace the fate of both starting materials and decomposition products. For those in scale-up or production, constant tweaking of reactor conditions and material flows forms part of the ongoing R&D effort to drive down cost without sacrificing safety.
Toxicity Research
Every credible risk assessment highlights the hazards associated with exposure to Isopropyl Isocyanate. Acute inhalation provokes respiratory distress, sometimes requiring medical intervention for chemical pneumonitis or edema. Chronic exposure in poorly controlled environments sometimes links to sensitization—a risk mirrored across the isocyanate class. Government agencies in Europe and North America review new toxicology data every few years, updating workplace exposure limits or pushing for better monitoring and reporting of incidents. Industry partners work with public health researchers to refine first-aid recommendations, supply chain traceability, and the development of smart badges or sensors to give early warnings at the plant floor. While animal studies highlight some gaps in long-term carcinogenicity or reproductive toxicity, most regulatory action centers on acute risk. This attention drives companies to assign product stewardship leads so that every downstream user gets fair warning and advice on engineering controls.
Future Prospects
Looking down the road, Isopropyl Isocyanate stands as a bellwether for innovation in both chemistry and safety practices. Chemists face increasing regulation of phosgene use and growing expectations from downstream users to cut workplace hazard levels. Companies willing to invest in alternative synthesis and lower-toxicity substitutes will have the chance to set the agenda. As markets lean toward greener and safer materials, expect more pressure to swap in renewable carbon sources and closed-cycle reactors that recapture any escapees. For now, the compound remains a critical niche intermediate, and future breakthroughs will depend on the ability of manufacturers, regulators, and scientists to work together. In this space, balance between progress and protection proves to be the most valuable product of all.
What is Isopropyl Isocyanate used for?
Why Isopropyl Isocyanate Matters in Industry
Isopropyl isocyanate is not the kind of chemical most people come across in daily life. Yet, in many industries, it plays a quiet but important role. Those working in the manufacturing of specialized coatings, polymers, or pesticides know that its presence is the difference between a lab experiment and a finished product. Factories rely on it to start reactions, especially for making compounds that need durability and resistance to heat and chemical exposure.
The Science and the Risks
Having spent time in a small research lab while studying chemistry, the first lesson drilled into all newcomers was safety with isocyanates. Even at low concentrations, exposure can bring trouble: respiratory irritation, skin reactions, and more. The safety data sheet is not just paperwork in these settings—it’s a shield. Workers suit up with goggles, gloves, and proper hoods out of respect, not just regulation.
Production plants use isopropyl isocyanate mostly as a reagent. Paints and coatings made to last in tough environments often depend on it. Car bumpers, dashboards, and industrial floors come out tougher, resistant to wear and tear. In agriculture, making certain pesticides and herbicides often involves this compound. Many of the advances that help crops survive blights or pests get their start in a flask where isopropyl isocyanate triggers the key chemical change.
Balancing Innovation with Health and the Environment
Those in charge of factories, labs, and shipping companies face a double challenge with chemicals like isopropyl isocyanate. Quality products are the goal, but there’s also the very real risk to people and the planet. Handling spills takes practice, strict planning, and gear. Once my team found faulty seals on a transport barrel, and the scramble to contain it and alert authorities cut through red tape because everyone understood the stakes. That day, plans saved lives—nobody breathed or touched what leaked.
Real Solutions—Not Just Rules
Mistakes can and do happen, so better detection and monitoring make a real difference. Modern facilities have alarms and sensors designed for isocyanate leaks. Training goes beyond the basics: drills now include response to unexpected chemical releases, based on lessons from past accidents. Government oversight helps, but nothing beats an organization taking responsibility for its people’s safety.
Green chemistry holds promise too. Researchers keep searching for alternatives that match isopropyl isocyanate’s performance, without its risks. Substitutes are being developed, though none have hit the right balance of cost and effectiveness. Until then, focusing on safe handling, real training, and accountability marks the smartest path forward. Transparent supply chains and responsible sourcing also matter; knowing where chemicals come from improves safety and trust throughout the process.
The Bottom Line
Isopropyl isocyanate is not a household name, but it makes possible many things relied on each day. From sturdy floors to better crop protection, its benefits stack up—but only so long as health, safety, and the environment are never afterthoughts. Respect for chemicals like this doesn't slow progress; it makes future innovation possible.
What are the safety precautions when handling Isopropyl Isocyanate?
What Makes Isopropyl Isocyanate So Risky?
Anyone who's spent time around chemical storage rooms or industrial labs develops a healthy respect for compounds like isopropyl isocyanate. This substance doesn’t forgive casual mistakes. It irritates skin, eyes, and airways badly, and breathing its vapors can land even experienced folks in plenty of trouble. If someone calls it volatile, they aren’t exaggerating. Isopropyl isocyanate reacts fiercely with water, acids, and plenty of other everyday chemicals, so once the jug is open, there’s no room for taking shortcuts.
Protective Gear Isn’t Optional
In my own years around industrial chemicals, I never witnessed a seasoned technician skip their goggles or gloves with something like isopropyl isocyanate. Direct skin contact brings burns or rashes. Splashing just a little on uncovered skin can mean days of irritation. A full-face shield and chemical-resistant gloves give good coverage. People who handle this chemical also put on long sleeves, closed shoes, and sometimes a disposable apron. Handling smaller batches might mean a lab coat, but moving drums or larger containers calls for a full protective suit and a respirator to avoid breathing any vapor.
Ventilation Keeps Workers Safe
One thing I always check before dealing with isocyanates: how well the fume hood or exhaust system works. Without steady airflow, vapors build up, and the risk for the whole crew rises. Sometimes, a job requires remote handling tools, minimizing time spent near open containers. If air monitoring equipment is available, folks use that for real-time exposure checks.
Preparation Means Fewer Accidents
Nobody should ever open a bottle or drum of isopropyl isocyanate without a plan for emergencies. The storage area needs a working eyewash station and an emergency shower steps away from the action. Safety data sheets belong somewhere visible, not tucked away in a drawer. A spill cleanup kit, built for harsh chemicals, should never stay sealed. Workers keep neutralizers and absorbents near the work site so they can stop a minor release from becoming a disaster.
Storage Conditions Matter
Isopropyl isocyanate stays stable only if it's kept cool, dry, and protected from any moisture. Anyone who’s seen the aftermath of stored drums mixing with water learns the lesson fast. The containers sit away from acids, alkalis, and anything else that might start a reaction. I’ve seen teams dedicate entire cabinets just for this chemical, lined with metal or chemical-resistant materials—not a single cardboard box in sight.
Training Makes a Difference
No tools or labels substitute for real, hands-on training. In places with a good safety culture, new hires practice scenarios, including spill response and proper disposal, right along experienced techs. Safety talks don’t happen once a year; reminders come up during every shift. Some organizations use color-coded labels or extra signage for high-risk chemicals, helping even the most distracted worker keep track of what’s on the bench.
Better Solutions Going Forward
Chemists and engineers keep on searching for ways to make dangerous chemicals safer to use. Substitution remains the gold standard: if a less hazardous substance does the job, the whole shop breathes easier. Until then, solid routines, working gear, and a culture of looking out for each other save real lives. Mistakes with isopropyl isocyanate carry heavy costs, so no one gets cavalier about handling it, no matter how many years of experience they have.
What are the potential health hazards of Isopropyl Isocyanate exposure?
The Real Risks in the Air
Isopropyl isocyanate isn't a household name, but it shows up across manufacturing and chemical plants. Workers might breathe it in or get it on their skin, and that's where things get uncomfortable—fast. I spent a part of my early career near chemical mixing tanks, and I've seen firsthand how quick a cough or stinging eyes can hit once these vapors aerosolize in the air.
Breathing in isopropyl isocyanate can kick off headaches, burning throats, and a raw feeling in the nose pretty quick. A little exposure might irritate your lungs. More of it, especially in an unventilated area, sends folks wheezing or reaching for rescue inhalers. Reports by the Centers for Disease Control and Prevention and NIOSH warn about asthma flares and longer-term breathing issues for workers stuck in these environments. You won't see it in clear air, but your body knows. The risk doesn't stay in the lungs, either; this chemical also irritates the eyes and skin. You'd be shocked how many workers lose days from rashes, swelling, and in some cases, blisters where they came in contact with the stuff.
Where Things Get Dangerous
The real trouble starts with prolonged or intense exposure. Once isopropyl isocyanate makes its way into the lungs over and over, some people end up with occupational asthma. This isn't just a fleeting cough—it's lifelong breathing problems. The European Agency for Safety and Health at Work tags isocyanates among the top causes of workplace asthma across the globe. I remember a plant foreman who loved his job, sticking with it for decades. He had to leave because his breathing never bounced back after a heavy leak in the warehouse.
The American Conference of Governmental Industrial Hygienists recommends strict limits for isocyanate-related compounds in the air workers breathe. These thresholds aren't just numbers to hit; they draw the line before hospital trips start happening. That's the everyday reality behind these scientific guidelines. OSHA's advice is rooted in real cases of sickness, missed paychecks, and bills that follow.
Tackling the Problem
It's not enough to hand out dust masks and call it safety. Where isocyanates show up, proper ventilation makes a difference. Scrubbers and exhaust fans need to run well—regular checks and real investment go a long way, both for a company's bottom line and workers' long-term health. Chemical-resistant gloves, tight-fitting goggles, and full-face respirators turn near-misses into forgotten close calls instead of ER visits. Even the toughest workers can't just power through a chemical burn or asthma attack.
Training plays its part, too. Many workers don't realize how quietly exposure builds up over time. Regular reminders and updates on proper handling cut back on careless mistakes. Instead of treating symptoms later, investing in safety upfront prevents most problems entirely. Plenty of places use sensors and alarms to catch VOC spikes—those tools matter, especially on night shifts or during cleaning cycles.
Those in charge—plant managers, shift leaders, OSHA reps—have the responsibility to look out for the crew. Doctors who work with these workers keep an eye out for unusual coughs or rashes. Sharing what they find in these circles helps others spot problems earlier. Industry and government working together share lessons learned and keep pushing for safer workspaces. If fewer people get sick, that's proof the work matters.
How should Isopropyl Isocyanate be stored and disposed of?
No Room for Carelessness
Working with chemicals such as isopropyl isocyanate feels a bit like moving around a pit bull—one wrong move triggers problems you never wanted. I once walked into a lab with a faint, sweet, sharp smell—a sign something wasn’t quite right. We traced it to a cracked bottle of isocyanate, which shows that storage and disposal aren’t chores to brush off. This compound doesn't forgive mistakes. It reacts with water, spits out toxic fumes, and attacks your lungs in a heartbeat if leaks or spills pop up. Real-life stories remind us this isn’t a textbook hazard.
Practical Storage Lessons
Every time you pop open a bottle of isopropyl isocyanate, you face risk. Keeping it in an ordinary cabinet turns that risk into a ticking time bomb. Instead, secure it in a tightly sealed, chemical-resistant container—something I picked up early as a rookie in a research lab. Use strong secondary containment. A good rule: handle it in a well-ventilated space and keep that bottle under an inert atmosphere like nitrogen. Storing it below room temperature helps slow down any unwanted reactions or pressure build-up. I once saw someone ignore that advice; their storage closet ended up with warped shelving and nasty chemical stains.
Don’t just rely on technology either. Place flammable signs, keep fire extinguishers nearby, and don’t mix it anywhere close to acids, water, or bases. I learned this after seeing a cleanup crew scramble when a neighbor stored acids beside isocyanates—the spill nearly triggered an evacuation.
Disposal: Responsibility Has No Shortcuts
Disposal isn’t about pouring leftovers down the drain. Isopropyl isocyanate breaks down into nasty stuff—cyanides, noxious gases. Mixing it with water or sending it into sewer lines almost guarantees a call from emergency services. I remember a technical director at a plant who thought water dilution would render it harmless. The local community found out with a shelter-in-place alert that still makes headlines at safety conferences.
Partner with certified hazardous waste handlers. Experts come armed with the proper gear and chemical know-how. They often neutralize small quantities using solutions that deactivate the compound, but these procedures involve chemical fume hoods and careful stepwise addition of deactivators like dilute acids. And for large-scale work, incineration at a licensed facility is the right call. Every step of this chain matters: skips in paperwork or mislabeling attract steep fines and endanger people down the line.
Solutions Worth Investing In
Training pays off more than fancy labels or thick manuals. Staff who actually understand chemical risks—those who’ve seen a malfunctioning fume hood or cleaned up a spill—don’t get sloppy. Clear protocols, posted emergency contacts, and regular safety drills set up workers for success. I’ve also seen smart storage software help track shelf life, flag expired chemicals, and prevent stockpile surprises.
In the end, chemicals don’t forgive shortcuts. Storage and disposal standards protect workers, neighbors, and the planet. If anyone doubts that, they haven’t spent a night scrubbing up after someone who skipped proper procedures. Raising awareness, insisting on accountability, and investing in safety gear keep everyone out of trouble—your lungs and reputation both depend on it.
What is the CAS number and chemical formula of Isopropyl Isocyanate?
The Basics: Identification and Chemistry
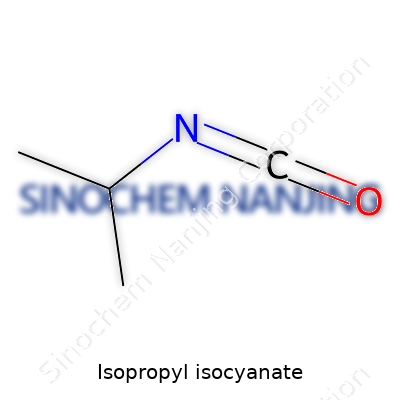
Every chemical needs to be clearly identified, both for science and safety. The CAS number gives us that common language. For isopropyl isocyanate, the CAS number is 409-71-0. Its chemical formula reads C4H7NO. In labs, every small bottle carries those numbers—no matter the brand—making accidents less likely and tracing issues faster.
Real-World Experience: Handling Hazards and Risks
Anyone who has worked with isocyanates knows the sharp, pungent scent and the irritation it brings on contact. My first memory of isopropyl isocyanate comes from a chemical synthesis lab—tight gloves, hood fans running at full blast, and frequent reminders to double-check every label before placing a bottle on the workspace. These rules aren’t just for show. Isopropyl isocyanate reacts quickly with water, even the moisture on your skin. Direct exposure leads to burns and breathing problems. The CAS number isn’t just bookkeeping—it signals to experienced chemists and health professionals what to expect, which gear to use, and how to treat exposure.
Industrial and Research Importance
In manufacturing, isopropyl isocyanate plays a role as a building block for pesticides, pharmaceuticals, and specialty chemicals. The smallest contamination in the process can ruin weeks of work, which is why strict labeling and inventory controls matter. Mislabeling or confusion about a chemical’s identity can halt work, trigger safety drills, or in the worst cases, endanger lives.
I once witnessed a colleague mistake a flask for another reagent—caught only because the CAS number on the inventory register didn’t match. That small label forced a stop, checked every step, and restored order. It’s easy to see chemical formulas as abstract, but in the daily grind of research and production, accuracy saves money, time, and health.
Sourcing, Regulation, and Security
Isopropyl isocyanate doesn’t sit on the general market shelf. Regulations control who buys it and how it can be shipped. The CAS number functions as its fingerprint in global trade systems and customs. Anyone shipping hazardous materials knows that missing or incorrect CAS numbers block shipments at borders or lead to hefty fines. For chemical suppliers and buyers, daily workflow depends on keeping these details straight.
Addressing Problems: Communication and Safety Training
Incidents happen most often from miscommunication. The solution comes back to training and clear processes. All new lab staff get drilled on label checking, personal protective equipment, and emergency steps. People who have spent years in the field keep their eyes on every chemical number, not just the product name. Auditing those habits, supporting them with checklists, and keeping documentation straight actually matters more than flashy safety posters. It’s about embedding these checks into habit and culture.
Isopropyl isocyanate may not headline news, but in research and industry, it highlights how identification, knowledge, and routine vigilance build safer outcomes and better science. Learning to keep track of a single CAS number sometimes means the difference between routine and disaster.
| Names | |
| Preferred IUPAC name | propan-2-yl isocyanate |
| Other names |
2-Isocyanatopropane 2-Propanamine, N=C=O Isocyanic acid, isopropyl ester Propan-2-yl isocyanate |
| Pronunciation | /ˌaɪsəˈproʊpɪl ˌaɪsoʊsaɪˈæneɪt/ |
| Identifiers | |
| CAS Number | 4083-64-1 |
| Beilstein Reference | 1362052 |
| ChEBI | CHEBI:148612 |
| ChEMBL | CHEMBL278592 |
| ChemSpider | 16853 |
| DrugBank | DB14440 |
| ECHA InfoCard | 100.005.728 |
| EC Number | 209-851-3 |
| Gmelin Reference | 8226 |
| KEGG | C19338 |
| MeSH | D015432 |
| PubChem CID | 13745 |
| RTECS number | NN1575000 |
| UNII | YSD6L4PXI8 |
| UN number | 2480 |
| Properties | |
| Chemical formula | C4H7NO |
| Molar mass | 101.12 g/mol |
| Appearance | Colorless liquid |
| Odor | Sharp, pungent |
| Density | 0.87 g/cm3 |
| Solubility in water | Reacts with water |
| log P | 0.86 |
| Vapor pressure | 36.6 kPa (20°C) |
| Acidity (pKa) | 15.43 |
| Magnetic susceptibility (χ) | -58.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.382 |
| Viscosity | 0.685 cP (20 °C) |
| Dipole moment | 3.96 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 325.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | “-134.6 kJ/mol” |
| Std enthalpy of combustion (ΔcH⦵298) | -2006 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS06 |
| Pictograms | GHS02,GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H225, H301, H311, H331, H314, H317, H334 |
| Precautionary statements | P210, P261, P273, P280, P285, P301+P310, P304+P340, P305+P351+P338, P310, P320, P330, P362+P364, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-3-2-W |
| Flash point | 6 °C (43 °F; 279 K) - closed cup |
| Autoignition temperature | 456 °C (853 °F; 729 K) |
| Explosive limits | Explosive limits: 2.4–15% |
| Lethal dose or concentration | LD50 oral rat 57 mg/kg |
| LD50 (median dose) | LD50 (median dose): 180 mg/kg (rat, oral) |
| NIOSH | NIOSH: NI4375000 |
| PEL (Permissible) | PEL: 0.005 ppm (as an 8-hour TWA) |
| REL (Recommended) | 0.005 ppm |
| IDLH (Immediate danger) | IDLH: 50 ppm |
| Related compounds | |
| Related compounds |
Methyl isocyanate Ethyl isocyanate n-Propyl isocyanate Phenyl isocyanate |

