Isopropyl Isobutyrate: A Look at Its Journey, Uses, and Prospects
Historical Roots and Industrial Footing
The story of isopropyl isobutyrate traces back to the mid-20th century chemical renaissance, during a wave when industrial chemists branched out beyond the classic esters in search of compounds that could balance low toxicity and pleasant sensory profiles. Back in the day, industries needed solvents and flavor additives that could handle high temperatures and various synthetic routes. Isopropyl isobutyrate, landing amid the proliferation of esters for commercial use, settled in as a cost-effective option for both specialized production lines and consumer goods. Factories, especially in Europe and the U.S., expanded capacity throughout the 1960s and 1970s to match the rising demands in personal care and coatings, making the compound far less of a specialty product and more of an industrial staple. Since then, the regulatory scene has reshaped the landscape, emphasizing product traceability, clear labeling, and safer synthesis.
Getting Acquainted: What Sets Isopropyl Isobutyrate Apart
Not every ester can claim the versatility found in isopropyl isobutyrate. As a clear, colorless liquid with a sweet, slightly fruity odor, it finds a welcome spot both in professional laboratories and on consumer shelves. Anyone who has worked in personal care or flavor chemistry likely stumbled upon it as a skin-conditioning agent or carrier for fragrances. Product marketers often push its clean profile and mildness, especially as folks lean toward less reactive and less sensitizing chemical bases in cosmetics. Just ask someone in the coating or plastics industry: a solvent that evaporates evenly and leaves barely a trace tightly fits their long search for consistency. Engineers rely on its dependable boiling point and hydrophobic character to keep manufacturing lines ticking.
Physical and Chemical Traits Worth Knowing
The ester has a molecular weight hovering around 144.21 g/mol and a boiling range sliding near 157–158 °C. Its melting point sits well below zero, which makes it easy to pump or blend even during chilly warehouse winters. With a vapor pressure that sits squarely in the middle range for small esters and a flash point around 46 °C, people handling it daily rarely report surprise evaporation or dangerous vapor clouds—as long as ventilation and spill management keep up. Solubility data paints it as a near-perfect match for a wide roster of hydrocarbons, oils, and other esters, yet it barely mingles with water, so storage tanks stay dry and product separation happens without fuss. Chemically, the compound does not stay inert in the face of strong acids or bases, but it shrugs off casual moisture or slow oxidation in typical air, which makes for steady shelf-lives in finished goods.
Technical Details and Labeling in Practice
Chemical manufacturers adopt direct, no-nonsense labeling that flags its CAS number (97-85-8) and identifies its use classes—often fragrance, flavor, or solvent. Technical sheets emphasize assay values above 98% purity for most cosmetic or food contact applications. Production lines use GC (gas chromatography) profiles for quality checks, but finished drums get a simple hazard label based on GHS standards. Shipping and warehouse managers look for pictograms highlighting flammability and routine handling cautions. Anyone in charge of workplace safety tacks up SDS (safety data sheet) summaries pointing to the need for eye and skin protection during major spills or repeated daily exposure. European REACH compliance and US TSCA listing have kept the product in line with global trade rules.
Making It: How Synthesis Plays Out
Industrial plants turn out isopropyl isobutyrate through the tried-and-true reaction of isobutyric acid with isopropanol, using acid catalysts such as sulfuric acid or tosic acid. The process leans heavily on the Fischer–Speier esterification pathway: reactants bubble in a stirred, heated vessel and the product ester lifts off either by distillation or solvent extraction. Recovery cycles reclaim unreacted alcohol and acid, pushing up yields and keeping waste down. Most mid-sized producers opt for continuous reactors to tune output sizes for both custom batches and bulk, letting them tweak ratios or catalyst loads in real time. Those familiar with organic synthesis know this route strikes a practical balance—moderate energy input, few exotic chemicals or byproducts, and robust purification steps that suit GMP (good manufacturing practice) environments.
Chemical Reactions and Adaptations
Folks trained in synthesis or formulation understand that isopropyl isobutyrate fits snugly into a wide array of modifications. Its ester group takes the spotlight—hydrolysis snaps it back to isobutyric acid and isopropanol under alkaline or acidic conditions, which means recycling or breaking down spent batches becomes simple. For industrial chemists, the molecule can act as a precursor to more functionalized esters or amides, serving as an entry point for making derivatives with unique properties. In labs, it gets oxidized, reduced, or swapped into complex reaction sets when the target product needs a midweight hydrophobic anchor. Rarely, you’ll hear about transesterification with other alcohols, leading to designer esters in fragrance labs or pilot-scale pharmaceutical routes.
What’s In a Name? Synonyms and Market Identities
The world knows the compound by several names—Isopropyl isobutyrate matches up with 2-Propanol isobutyrate, Propan-2-yl 2-methylpropanoate, and even the handy abbreviation IPiB. Fragrance and flavor catalogues often drop the numbers for smoother branding, while chemical wholesalers stick to standardized labels for cross-border shipments. You might see older trade names in legacy product data sheets, but most digital platforms and regulatory filings converge on one or two core names. Consistent naming pays off, stopping confusion in busy procurement offices or fast-paced international shipping.
Safety and Handling in Real Workplaces
Any worker who has spent time in chemical storage or blending knows how crucial it is to treat liquid esters with respect. Isopropyl isobutyrate doesn’t trigger acute toxicity at typical workplace exposures, which brings peace of mind for users in labs or production areas. But experienced hands don’t skip gloves or eye shields—skin contact causes irritation for some, and high vapor concentrations need quick ventilation. Fire marshals note the moderate flash point, putting extra emphasis on grounded drums, bonded containers, and static controls. Most plants use down-to-earth spill kits with pads and granules, backed by regular staff training and drills. Environmental teams keep an eye on effluent streams, as residual esters can push up BOD (biological oxygen demand) if they reach waterways untreated. Audit reports reinforce the need for robust maintenance schedules, not just paperwork compliance.
Where Isopropyl Isobutyrate Gets Put to Work
Few chemicals bounce between so many industries with as much ease. If you check personal care labels, you’ll find it in skin creams, hair conditioners, and wash-off cosmetics, prized by formulators for its emollient touch and effortless blending of oils. Fragrance mixers credit its neutral scent carrier properties, letting delicate botanicals shine through without overpowering. It has earned a patch in synthetic flavor manufacturing, especially in confectionery or fruit flavor builds, since it amplifies wild berry or banana notes at trace concentration. Paint and coatings engineers value fast drying and low-residue performance, supporting dries-to-the-touch finishes on plastics, metal, or wood. Industrial workers handling adhesives and sealants bump into it frequently, where it thins viscous mixtures without major fire hazards or regulatory headaches.
Horizon of Research and Growing Knowledge
If you track patent registrations and academic journals, the past decade has seen a shift from pure solvent use to exploring isopropyl isobutyrate in eco-friendlier formulations. Materials scientists dig into new biocomposite resins, seeking to replace legacy plasticizers and softeners. Biomedical engineers pick up the compound for advances in tissue-compatible gels and microencapsulation techniques. Green chemistry groups study its breakdown in the presence of sunlight or common microbes, hoping to work up more sustainable waste routines. Research grants tip towards examining structure–activity relationships, dissecting why some esters excel in delivering topical drugs or stabilizing flavors. The scientific appetite isn’t just for “what works”—but “what’s safer, smarter, and longer-lasting.”
Digging Into Toxicity and Long-Term Exposure
Occupational health research shows isopropyl isobutyrate rates low for acute systemic toxicity in rodents and test tubes, with high exposure causing only transient mild symptoms in most cases. Workers in busy plants rely on this solid dataset, pushing for risk assessments that focus on preventing chronic skin or airway issues instead. Safety committees track sensitization responses, allergic signs, and signs of repeated exposure through annual medical reports and hydrocarbon monitoring. Environmental risk teams focus on aquatic toxicity, aiming for effluent treatments that break down esters before factory discharge points. Toxicology experts urge companies to invest in long-term studies, tracking subtle endpoints like developmental or reproductive health signals in valuable animal models. When regulations shift toward greater transparency, routine sharing of findings with the community and local authorities keeps trust strong.
Looking Ahead: Challenges and New Directions
Chemical producers and users face a shifting landscape. Consumer brands push for fewer “red-flag” ingredients, preferring those with short persistence and low ecological impact. Engineers and supply managers step up recycling, solvent capture, and plant upgrades that keep emissions in check. There’s growing pressure to swap out aromatic hydrocarbon solvents for esters like isopropyl isobutyrate, aiming to balance price, safety, and green credentials for new regulations. Lab teams get tasked with innovating—bio-based production methods, direct enzymatic syntheses, or feedstocks reclaimed from agri-industrial waste. Sector leaders invest hard in certification programs and co-creation with downstream users, aiming to pin down safer, more reliable supply chains. As next-gen markets ask for even milder and more sustainable chemical solutions, collaborative research and regulatory openness will decide who leads the charge in the years ahead.
What is Isopropyl Isobutyrate used for?
Finding Solutions in Chemistry
Growing up with a family that ran a small textile shop, I learned early how chemicals shape the products we use every day. Isopropyl isobutyrate doesn’t jump out as a name anyone recognizes in household conversations, but it’s one of those chemicals that keeps things running smoother beneath the surface.
Isopropyl Isobutyrate in Cosmetics and Personal Care
Cracking open a moisturizing lotion or a sunscreen, it's easy to appreciate the silky texture. That touchable softness often owes a lot to isopropyl isobutyrate. Chemists count on it for its light feel and ability to spread evenly on skin. Unlike heavier oils, it doesn’t block pores. This makes it especially useful for face creams and sprays aimed at those with sensitive skin. Personal care brands trust it to give lotions the smooth slip that people expect.
I’ve spoken with people who struggle with acne, and they always look for products claiming to be ‘non-comedogenic’—meaning they don’t clog pores. Isopropyl isobutyrate fits that niche well. Dermatologists recommend products using lighter emollients to avoid skin breakouts, and formulators keep going back to this ester because it keeps things light.
Industrial Uses and Performance Boosts
Beyond bathroom counters, isopropyl isobutyrate proves its worth in paint, ink, and coatings. Its solvent properties help dissolve colors and binders, so everything mixes well. I remember volunteering at a local art class and seeing how certain paints dried faster and kept colors brighter for longer. The instructor talked about additives improving performance in all sorts of climate conditions. Isopropyl isobutyrate is exactly the kind of substance behind those dependable results. It helps paints glide on smoother and dry with fewer visible streaks.
This chemical can even show up in cleaning products and metal polishes. For factories that build car parts or electronics, using reliable solvents isn’t a luxury—it prevents costly mistakes. Even small improvements in drying time or spread can translate into energy and time savings on the shop floor.
What Science and Safety Say
Safety matters more today than it ever did. Regulators keep an eye on which chemicals enter everyday products. Reliable research shows that isopropyl isobutyrate stays stable, doesn’t break down into harmful compounds quickly, and isn’t considered toxic at typical concentrations used in consumer goods. I’ve scanned safety data sheets out of curiosity when choosing paints for home projects, and it’s reassuring to see products contain ingredients with a low hazard ranking.
Sustainability does challenge the chemical industry. Using chemicals like isopropyl isobutyrate responsibly means promoting proper handling—and disposing of manufacturing waste the right way. Industry guidelines set limits on exposure, and workplaces train employees to follow those rules. Companies that stick to these practices earn a better reputation, and shoppers who read labels notice which brands take safety seriously.
Looking to the Future
People expect transparency and accountability from the companies behind their favorite lotions, paints, and sprays. Isopropyl isobutyrate isn’t the only ingredient that matters, but its use draws attention to the careful choices made in product development. Scientists innovate, but real progress means keeping health, safety, and the environment front-and-center. That’s how chemistry quietly shapes safer, higher-value experiences for everyone—from manufacturers to families shopping at the corner store.
Is Isopropyl Isobutyrate safe for skin?
Looking at the Science
A lot of skin care products list ingredients that sound more at home in a chemistry textbook. Isopropyl Isobutyrate pops up in lotions and makeup primers because it helps them smooth onto skin. It’s an ester, blending isopropyl alcohol and isobutyric acid. Most people only start paying attention to the label after a reaction or breakout. Isopropyl Isobutyrate might catch the eye as something unfamiliar and maybe worrying, but the facts tell a straightforward story.
Over the years, researchers put this ingredient through testing. According to reviews by the Cosmetic Ingredient Review (CIR) panel and studies cited by the European Commission, Isopropyl Isobutyrate has a strong track record for safety when used in the concentrations found in consumer products. It does not cause cancer, doesn’t break down into anything toxic, and isn’t flagged for organ toxicity.
My Experience and Common Concerns
After years of dealing with dry patches from eczema, I became pickier about every ingredient on my skin. I started paying extra attention to how products felt and whether they led to trouble spots. Out of curiosity, I tried a moisturizer with Isopropyl Isobutyrate fairly high on the ingredient list. The product did spread smoothly and didn’t leave behind that oily film I tried to avoid. After a month, there was no reaction—no redness, no clogged pores.
That matched reports from friends and feedback in online skincare forums. Most people using products with this ingredient do not experience irritation or allergic reactions. Still, a minority with very sensitive or compromised skin may report problems. Reactions like contact dermatitis don’t strike everyone equally and depend on unique immune responses. Contact allergy is rare but possible, usually in people who already react badly to esters or certain fragrances.
Why the Concern?
People get cautious about Isopropyl Isobutyrate for several reasons. Social media amplifies stories of adverse reactions, even though scientific evidence points to overall safety. Sometimes, isopropyl-based compounds get thrown into the same mental bucket as harsh alcohols that dry out skin. But in this case, the compound doesn’t evaporate in the way pure isopropyl alcohol does. It’s designed to soften and condition, not strip moisture.
Another concern comes from the broader movement toward all-natural or “clean” ingredients. That makes sense, if only because some people with chronic skin issues blame synthetic chemicals for flare-ups. Yet the origin of an ingredient doesn’t always predict irritation or safety. Poison ivy is natural but not good for anyone’s skin, for example.
Responsible Use and Next Steps
Having spoken with dermatologists and read the clinical literature, it’s clear the best answer comes down to knowing your own skin. Dermatologists recommend patch testing new skincare, especially for anyone with a history of allergic rashes or eczema. Companies including global brands keep using Isopropyl Isobutyrate because complaints are rare, and testing continues to support its safe record.
Manufacturers could help by listing concentrations and providing clarity on ingredient sources. More transparency would lower the anxiety for people interested in tracking what hits their skin. For those with unpredictable skin conditions, consulting a healthcare professional before trying new formulas brings peace of mind.
Ultimately, an ingredient like Isopropyl Isobutyrate gets green-lit in the cosmetic world because experts and regulatory panels have seen enough data to feel comfortable, and broad real-world use confirms that. The loudest stories often come from negative experiences, but numbers back up a calmer story: Isopropyl Isobutyrate is safe for the overwhelming majority of people and works well for silky skin without causing drama.
Is Isopropyl Isobutyrate an irritant or allergen?
Digging Beneath the Label
Spotting a name like isopropyl isobutyrate on a lotion bottle triggers my curiosity. The jumble of letters looks intimidating, but it’s much more common than you’d expect—showing up in moisturizers, sunscreens, and even makeup primers. I remember flipping over my favorite hand cream, searching for the culprit behind an odd burning feeling. There it was, wedged between other mysterious ingredients. So what is this stuff? And should we worry about slathering it onto our skin?
Understanding How People React
Most folks want simple answers. Either an ingredient is safe, or it’s trouble. Isopropyl isobutyrate doesn’t play by those rules. Chemically, it’s an ester, which means it offers that lightweight, non-greasy feel most of us love in cosmetics. Companies use it because it helps lotions spread evenly and sink in fast, which matters for anyone avoiding sticky residue.
Big dermatology journals and ingredient safety databases—like the Cosmetic Ingredient Review panel and European Scientific Committee on Consumer Safety—scrutinize ingredients for risk. Both these groups have given isopropyl isobutyrate a green light. They’re not rubber-stamping anything; they churn through lab studies and reports of real-world reactions. In their reviews, this ingredient rarely causes trouble. In over-the-counter use, it usually glides across skin with few complaints.
When Sensitive Skin Pushes Back
Skin doesn’t always read the memo, though. I’ve seen a handful of cases where people run into redness, stinging, or itchy patches after using products with isopropyl isobutyrate. Dermatologists classify this as irritation, not a full-blown allergy. The difference matters. Irritation can stem from a high concentration, repeated use, or rubbing product onto already damaged or dry skin.
Allergic reactions look different. With a classic allergen like nickel, your immune system gears up and spots the invader every time. For isopropyl isobutyrate, reports of allergy are almost nonexistent. Patch testing in clinics across the US and Europe rarely links isopropyl isobutyrate to immune-driven rashes. Its chemical structure doesn’t match that of common allergens, and researchers find it less likely to spark a true allergy.
Practical Approach to Skin Health
I’ve learned not to fear every unfamiliar name in the ingredient list. For nearly everyone, isopropyl isobutyrate stays on the safe side. If your skin flares up, it’s more about sensitivity than a universal problem. For anyone with eczema or damaged skin, products with minimal extras make more sense. A patch test on your inner forearm can sort out what your skin actually likes.
Public data from Poison Control centers and hospital dermatology clinics in the past decade backs up these findings. Out of thousands of calls and visits for skin irritation, this ingredient barely makes a blip compared to fragrance, certain preservatives, and harsh acids.
Looking Forwards: Consumer Awareness
Keeping track of new research still matters. Companies launch new product lines every year, sometimes pairing isopropyl isobutyrate with fragrance, dyes, or other known irritants. For those who trust but verify, websites like the Environmental Working Group or the US Food and Drug Administration offer up-to-date safety rundowns. If you’ve had a rough encounter with a cosmetic, save the label and check with a dermatologist before tossing everything labeled “chemical.”
Better transparency lets everyone decide what works. For now, isopropyl isobutyrate rarely causes problems, but every skin type has its own opinion.
Can Isopropyl Isobutyrate be used in cosmetics?
Understanding the Ingredient
Sometimes a new name on a cosmetic label throws people for a loop. Isopropyl isobutyrate, for instance, sounds more at home in a chemistry lab than a bathroom cabinet. But it pops up more often as brands hunt for ways to balance feel, function, and skin tolerance in their products.
Manufacturers and formulators aren’t just picking random chemicals. They weigh safety records, how a substance blends with others, and how it wears over the day. Isopropyl isobutyrate turns heads because it changes the way creams and lotions spread on the skin. For those who have applied something that felt sticky, tacky, or left behind a greasy finish, they know how a slick, silky finish makes a huge difference. This ester, made from isopropanol and isobutyric acid, aims to solve some of those texture problems.
Safety Takes the Front Seat
Every ingredient in a skincare line faces intense scrutiny. People read labels and want reassurance about both short-term and long-term effects, especially on the skin. Isopropyl isobutyrate has survived its share of safety reviews. The Cosmetic Ingredient Review panel checked available studies, and toxicology research covers everything from basic skin irritation to what happens if it makes contact with the eyes. Published research points to low-risk results for most skin types. Allergy rates haven’t spiked where this compound is common, and animal studies haven’t flagged anything alarming at cosmetic use concentrations.
This doesn’t mean everyone’s skin will welcome it. I’ve dealt with dozens of clients who develop redness or acne from a new cream, and sometimes even so-called gentler alternatives set things off. Anyone with a history of breakouts or sensitive skin benefits from patch testing unfamiliar ingredients, whether they come from natural or synthetic sources. Even water can cause trouble for a few people. So it makes sense to start slow, check how skin reacts, and not just trust a safe label.
Performance in Products
Companies don’t just throw in isopropyl isobutyrate for texture. It helps dissolve oil-based actives and delivers them more evenly. I’ve worked on projects where the texture of a product could make or break launch day. Products that went on heavy or left residue ended up sitting on store shelves. Lightweight emollients like this one help products sink in, leaving skin soft without that coated feeling.
Another practical point: It plays well with both water-based and oil-based ingredients, which gives chemists more room to design products that fit a range of skin types. For makeup, sunscreens, and light moisturizers, this ingredient contributes to that sought-after “barely there” finish.
Transparency and Consumer Confidence
People want more than unfamiliar names and long ingredient lists. If brands explain why an ingredient is there and outline the science, trust grows. Regulatory bodies in places like the United States, Europe, and Japan already review cosmetic ingredients for safety. Isopropyl isobutyrate passes current standards, but that doesn’t shut down curiosity. I see more brands publishing source information and science on their websites, which helps shoppers make choices with confidence.
Concerns around long-term effects keep people reading further, and folks with specific allergies look for alternatives. Conversations between cosmetic brands, scientists, and users steer the industry forward. With digital resources and open dialogue, clients have better odds of ending up with something that feels good—and stays safe—on their skin.
What are the benefits of using Isopropyl Isobutyrate in personal care products?
Smoother Skin, Lighter Feel
People care about how lotions and creams feel on their skin. Greasy formulas often get left behind in the medicine cabinet, no matter how well they work. Isopropyl isobutyrate steps in here. This clear, lightweight liquid helps finished creams and sunscreens glide on without that telltale heavy or sticky after-feel. It spreads quickly and sinks right in, leaving skin with a smooth touch. That sensory difference stands out when compared to some older emollients.
Better Absorption, Visible Results
Personal care isn’t just about what’s in a formula—it’s about how well it works. Isopropyl isobutyrate helps other ingredients reach the skin’s surface without pooling or evaporating too fast. That means more of the hydrating, nourishing, or protective ingredients reach where they’re supposed to go. In a hot climate, people want skin care to evaporate fast but not disappear before hydrating the skin. Isopropyl isobutyrate delivers on this balance, which matters for sunscreen and lotions alike.
Less Shine, More Comfort
Shine can be a major drawback, especially with moisturizing creams and SPF products. Some emollients leave a glossy sheen or even feel sticky, which makes users wash the product off instead of sticking to a daily routine. Isopropyl isobutyrate provides a mattifying finish. I have tried a few facial sunscreen sprays that use it, and I’m far less likely to skip my routine because of that light, dry touch.
Gentle Enough for Sensitive Faces
Sensitive skin types steer clear of oils that cause breakouts or pore clogs. Isopropyl isobutyrate scores low on comedogenicity charts, which means it doesn’t block pores or trigger more blemishes. Dermatologists often recommend it for acne-prone or oily skin. Younger people entering the skincare world benefit from this, since early choices often shape years of habits and skin health.
Works in Humid and Dry Places
My own experience living in different cities taught me how products behave in weather extremes. In Texas, heavy oils felt smothering and melted off my face. Out in Colorado, thin gels dried up too fast. Isopropyl isobutyrate adapts, working as a carrier in dry climates so creams don’t evaporate instantly, but also keeping that fresh feeling in humid air. This adaptability makes it a favorite of cosmetic chemists aiming for worldwide markets.
Scientific Backing and Safety
Research gives isopropyl isobutyrate a solid safety profile. The Cosmetic Ingredient Review (CIR) panel reviewed it and didn’t flag any major concerns at levels typically used in over-the-counter products. Manufacturers have included it in products for decades, and real-world use backs up those studies—complaints are rare, even among frequent users.
What Makes It Stand Out
Brands want products that feel appealing and keep their promises through daily use. Isopropyl isobutyrate helps formulas feel elegant, supports effective delivery of nutrients or UV filters, and keeps skin looking and feeling balanced. This ingredient isn’t about empty marketing; it solves long-standing issues with greasy skin care, irritation, and product wear-off during the day.
Ideas for the Future
Formulators keep looking for safer, lighter, and more sustainable choices. Continued research may focus on greener sourcing or new blends with natural oils to answer growing consumer demand for both performance and conscience. But as of now, isopropyl isobutyrate sets a high bar for those seeking both comfort and effectiveness without trade-offs.
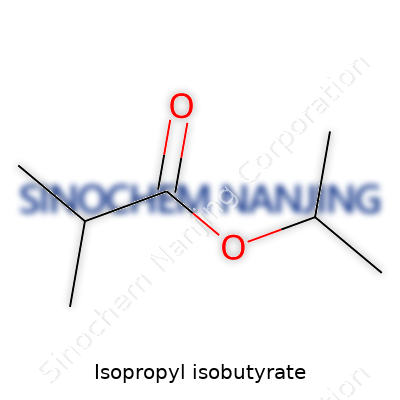
| Names | |
| Preferred IUPAC name | Propan-2-yl 2-methylpropanoate |
| Other names |
Isobutyric acid, isopropyl ester Isobutyric acid isopropyl ester Isopropyl 2-methylpropanoate |
| Pronunciation | /ˌaɪ.səˈproʊ.pɪl ˌaɪ.səˈbjuː.tɪ.reɪt/ |
| Identifiers | |
| CAS Number | 97-85-8 |
| 3D model (JSmol) | `Isopropyl Isobutyrate: CC(C)C(=O)OCC(C)C` |
| Beilstein Reference | 1421147 |
| ChEBI | CHEBI:89073 |
| ChEMBL | CHEMBL3187877 |
| ChemSpider | 115096 |
| DrugBank | DB16668 |
| ECHA InfoCard | 100.044.428 |
| EC Number | 246-360-3 |
| Gmelin Reference | 8587 |
| KEGG | C21155 |
| MeSH | D011805 |
| PubChem CID | 31249 |
| RTECS number | NI0175000 |
| UNII | G07GZ97H60 |
| UN number | UN2730 |
| CompTox Dashboard (EPA) | DTXSID5020659 |
| Properties | |
| Chemical formula | C7H14O2 |
| Molar mass | **144.21 g/mol** |
| Appearance | Clear, colorless liquid |
| Odor | Fruity |
| Density | 0.857 g/cm3 @ 25 °C |
| Solubility in water | Insoluble |
| log P | 2.8 |
| Vapor pressure | 0.30 mmHg (20°C) |
| Acidity (pKa) | pKa ≈ 25 |
| Magnetic susceptibility (χ) | -7.14 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.4080 |
| Viscosity | 5 mPa·s (25°C) |
| Dipole moment | 2.06 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 417.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -454.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4645 kJ/mol |
| Pharmacology | |
| ATC code | D02AE08 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | H319: Causes serious eye irritation. |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P280, P303+P361+P353, P370+P378 |
| Flash point | 77 °C |
| Autoignition temperature | 400 °C |
| Explosive limits | 1.0% - 7.0% |
| Lethal dose or concentration | LD50 (oral, rat): 13,400 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 13,400 mg/kg |
| NIOSH | WT2930000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Isopropyl Isobutyrate: Not established |
| REL (Recommended) | 19 mg/m³ |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Isopropyl acetate Isopropyl myristate Isobutyl isobutyrate Isopropyl palmitate Isobutyric acid |

