Isopropyl Chloroformate: Past, Present, and the Road Ahead
A Journey Rooted in Chemical Curiosity
Winding back to the days before digital databases, chemists relied on painstaking experimentation in search of new reagents. Isopropyl chloroformate entered the scene during that period of brimming curiosity. Compared to the stalwarts like phosgene, its introduction brought a safer, more manageable way to carry out acylations, and it quickly found a place in peptide and pharmaceutical synthesis. People might overlook its historical value, brushing off these “middleman” reagents, but every reagent with a good story behind it deserves a closer look. Its emergence paralleled industrial demand for milder chlorinating agents, offering a tighter grip on selective reactions—a real turning point back in the day.
Defining the Substance
Isopropyl chloroformate doesn’t catch the spotlight. It's a transparent, colorless liquid that fumes in damp air, bearing a sharp odor that hints at its reactive nature. A whiff triggers instant respect for its potency. With a formula of C4H7ClO2, it mixes a chloroformate functional group with an isopropyl backbone, bringing out the best of both worlds in lab work. Boiling point lands around 90°C, and its density hovers near 1.1 grams per cubic centimeter. While those numbers don’t tell the whole story, they remind us that even familiar reagents demand attention to detail.
Why Technical Details Matter
Precision frames every step involving isopropyl chloroformate. Labels warn of moisture sensitivity and volatility, and every shipment must arrive in airtight containers. Small leaks don’t just mean cleanup; they signal a potential incident. It decomposes on contact with water, spitting out hydrochloric acid and carbon dioxide. That decomposition might seem manageable, but those who have worked in tight quarters know just how quickly things escalate. Manufacturers shift their labeling in line with safety specifications laid out by authorities, including GHS pictograms and hazard statements. It’s not bureaucracy for the sake of bureaucracy; these details save lives and prevent incidents.
Preparation in the Lab and Beyond
Chloroformates aren’t products of chance. Crafting isopropyl chloroformate calls for reacting isopropanol with phosgene, typically under controlled conditions with a base like pyridine. If that base sounds fancy, ask anyone who’s tried to handle phosgene in a hurry. No casual chemistry here; tight temperature controls and ventilation rigs protect against emissions. Runoff and waste get treated before disposal because even a “simple” batch can introduce persistent pollutants. Researchers learned the hard way that scaling up from bench to plant requires more than bigger flasks—process safety fundamentals take center stage every time.
Reactivity and Possibility
At its core, isopropyl chloroformate delivers value as an acylating agent. This means it turns amines into carbamates and carboxylic acids into anhydrides or esters, unlocking new possibilities for building complex molecules. Peptide chemistry benefits the most. Instead of juggling harsher reagents, chemists reach for this tool where selectivity matters. Tinkering with its structure leads to tweaks in solubility or reactivity, paving the way for exploring new derivatives. Not every tweak succeeds, but each one marks a bit of progress.
Names and Notoriety
Not many casual lab workers rattle off synonyms, but the lexicon matters. Isopropyl chloroformate goes by isopropyl carbonochloridate and carbonochloridic acid isopropyl ester, among other trade names. Misreading those labels means risking the wrong chemical in the wrong place, and everyone who’s spent time among crowded reagent shelves can recall stories where a mix-up led to trouble.
Safety: Respect the Dangers
Even a few drops can cause chemical burns, so every protocol turns cautious. Guidelines require splash-proof goggles, gloves rated for organic acids, and, ideally, fume hoods vented to the outside. Spills get handled with specific neutralizing agents; simple paper towels won’t cut it. Respiratory protection isn’t up for debate, especially where ventilation is spotty. Local exhaust, evacuation plans, and regular drills round out good practice. Regulations force companies to rethink storage and movement down to the last detail, and after seeing what one overlooked seal can cause, it’s tough not to advocate for extra vigilance.
Practical Applications
Isopropyl chloroformate works behind the scenes in pharmaceuticals and advanced polymers. In making life-saving drugs, its contribution comes in key steps—joining molecules to build protected intermediates, especially in peptide and urea synthesis. Agrochemicals and specialty coatings rely on its reactivity. Some research circles use it to introduce protective groups when crafting new antibiotics or fine-tuning potential cancer drugs. On the ground, it may not have household fame, but its role threads through products that people depend on without realizing.
Ongoing Research and New Directions
Chemists keep searching for milder conditions, hoping to cut costs and reduce hazards. Labs try swapping hazardous solvents for greener alternatives, automation for dull or dangerous steps, and smarter detectors for leaks. Efforts to cut down on byproducts matter because each gram of waste means more cleanup or higher disposal fees. Some groups take inspiration from biological systems, experimenting with enzyme catalysts to handle similar reactions more gently and selectively. While the jump to sustainable methods isn’t instant, every pilot study chips away at the old perception of chemicals as purely “hazardous necessities.”
Toxicity and Human Costs
Research shows that exposure above the recommended limit brings acute effects. Skin blisters, eye injuries, and respiratory distress all pop up in case studies. Long-term exposure hasn’t received as much attention, but animal tests point towards cumulative organ toxicity. Workers in production plants carry most of the risk, which puts occupational medicine front and center. Routine monitoring for symptoms beats responding too late, and medical professionals on these teams keep their senses sharp during every shift. Lessons from past accidents underline that real safety practice means consistent vigilance, not just a checklist.
Pushing Toward a Safer Future
The future of isopropyl chloroformate looks driven by two trends: increasing demand for complex molecules in pharmaceuticals and mounting regulatory scrutiny. Companies push for alternatives with fewer hazards and lower environmental footprints. Academic research teams hunt for new synthetic steps offering the same performance with less toxicity. Demand for monitoring technology grows, including real-time sensors that can halt a reaction before it gets out of hand. Anyone working with this reagent or its cousins has a stake in the push for cleaner, safer chemistry because progress on those fronts sets the stage for what comes next—in research, manufacturing, and daily safety for the people behind the process.
What is Isopropyl Chloroformate used for?
Nitty-Gritty Applications in Chemical Synthesis
When chemists talk about Isopropyl Chloroformate, they tend to focus on its strong role as a reagent. In practical terms, this stuff helps piece together molecules that would otherwise be tough to build. People in pharmaceutical labs often rely on it during the process of making certain drugs, especially where careful control over reactions can make or break the final product. It's a real backbone chemical for folks who work with peptides or compounds that need a so-called “protecting group.” These protecting groups help keep parts of a molecule safe while other sections get switched, built up, or cleaned up.
A Tool for Drug Makers and Material Scientists
Anyone who's spent time in a pharmaceutical manufacturing plant knows speed and accuracy matter. Isopropyl Chloroformate acts fast – it latches onto amine groups in molecules. This makes it an easy choice for making carbamates and urethanes, both of which show up in antiviral drugs, anesthetics, and even some cancer medicines. Years ago, I watched a chemist argue with her boss about swapping in this reagent for a slower one. The team saved hours on just one batch, proving that the right chemical can have a ripple effect through an entire production line.
These days, the compound pops up beyond drug-making. Material scientists use it to help craft specialty polymers. Some coatings used in electronics come from reactions built with this reagent. It’s the sort of thing that doesn’t catch headlines, but without it, the end products lose performance or cost more to make.
Safety Risks and Handling
People working with Isopropyl Chloroformate have learned not to underestimate its hazards. Breathing in fumes or spilling a small amount can lead to trouble – coughing, skin burns, or worse. There have been plenty of near misses in small research labs where a splash caught someone off guard. That’s why anyone using it keeps goggles and gloves close at hand. Fume hoods aren’t just a recommendation; they’re a necessity. Safety data sheets back this up, warning about the dangers of moisture and accidental mixing with incompatible chemicals. The chemical industry tracks accident rates involving this reagent, and evidence shows firms with routine safety training keep those numbers lower.
A Look Toward Safer Chemistry
People have been hunting for safer alternatives, but it’s not always simple. Some replacements fail to react as quickly or leave behind impurities. Others ramp up costs or cause new hazards. Green chemistry feels like the way forward. I’ve seen labs experiment with less toxic or recyclable reagents, although none have matched the speed and reliability that Isopropyl Chloroformate delivers just yet. Still, regulatory pushes in regions like the European Union force companies to rethink their stock chemicals. Some suppliers now offer improved containers, better labeling, and digital tracking to prevent mishandling.
Final Thoughts
Isopropyl Chloroformate sits right in the middle of modern chemical industry work. Its value comes not only from how well it performs, but from the blend of practicality, safety, and necessity. There’s a balance to strike — reap the benefits in research and industry while treating every bottle with respect and caution. Real progress lies in open communication about handling risks and continual searches for new options, so health and innovation go hand in hand.
What are the safety precautions when handling Isopropyl Chloroformate?
Why Bother with Safety?
Plenty of chemicals have a reputation, but isopropyl chloroformate doesn’t play around. This liquid’s uses span labs and industry, so I’ve seen folks get comfortable with it. That comfort can lead to slips, literally and figuratively, and that’s when things head south fast. Exposure can mean breathing problems, skin burns, or irritated eyes—not exactly a minor issue. The right safety steps go beyond common sense; they draw from lessons learned the hard way.
Keep It Off Your Skin and Out of Your Lungs
Picture an old college lab: no gloves, someone cracks a bottle open, and before anyone blinks, sharp, stinging vapors fill the air. Memories of chemical burns stick with me, and I’ve met a few people who ignored gloves “just for a second.” Not worth it. For every task, nitrile or neoprene gloves block direct contact. Goggles or full-face shields guard the eyes—splashes hurt, and permanent damage isn’t rare. Lab coats and long sleeves stop the stuff from touching skin.
Isopropyl chloroformate belongs nowhere near lungs. A reliable fume hood stands between safe handling and a trip to the clinic. It’s a straightforward move: work inside the hood, close the sash, and let ventilation do the heavy lifting. Ordinary rooms with basic fans might as well be ignored; they don’t keep vapors at bay.
Solid Storage Stops Trouble Before It Starts
This chemical reacts with water and makes toxic fumes, so it doesn’t sit well next to sinks or humid windows. Dry, cool, and well-ventilated spaces with clear hazard signs stop confusion and accidents. Dedicated cabinets for dangerous reagents cut down on the “wrong shelf” mix-ups. From years in busy workspaces, I know labels wear off or fade—fresh, legible ones matter more than folks admit.
Clean-Up and Disposal Must Be Spot-On
I remember spills handled with nothing but paper towels and a shrug. That shortcut brings more pain than it saves effort. Absorbent pads, proper spill kits, and immediate cleanup limit exposure. Waste goes into clearly marked containers, never poured down the drain or tossed in regular bins. Nobody wins when a janitor gets hit with surprise fumes.
Training Isn’t Optional
Companies and schools sometimes skip safety talks—they figure folks already know the basics. In reality, solid training marks the line between safe practices and risky shortcuts. Refresher sessions keep people awake to chemical hazards, and posted emergency numbers and first-aid steps act as extra guardrails. If your lab lacks regular reviews, it’s time to speak up.
Real Solutions, Not Red Tape
Safety routines work best with the right support. Supervisors should check stock regularly, keep protective gear in reach, and make sure equipment stays up to code. Regular audits sound dull, but small checks prevent big problems. If someone spots an issue—leaks, missing gear, old labels—address it today. Teams who look out for each other build stronger habits. In my own experience, nothing replaces simple respect for chemicals and the people working nearby.
Handling isopropyl chloroformate safely takes gear, training, and teamwork. People matter more than shortcuts, and habits built in the lab carry over anywhere hazardous work happens. The risk isn’t imaginary; putting safety first turns a stressful job into just another day at work.
How should Isopropyl Chloroformate be stored?
A Chemical That Commands Respect
Isopropyl chloroformate brings back memories of my early days in the lab, studying organic syntheses. That sharp, stinging odor meant one of my team was capping a flask or double-checking labels. This chemical isn’t just another bottle in a storeroom. It reacts easily with water, can irritate the lungs in seconds, and loves starting fires with the wrong neighbors. That’s enough reason to treat storage as a daily ritual, not just a regulatory checklist.
What Proper Storage Looks Like
Letting isopropyl chloroformate sit on a shelf with paint thinners and peroxide sounds like a shortcut to disaster. Flammable and unstable substances ask for careful isolation. Anyone storing this chemical should use a well-ventilated, cool room, far from any sources of heat or direct sunlight. Too much warmth makes the pressure build up. Even mild heat prompts breakdown into noxious fumes. Good ventilation stops vapors from spreading and being inhaled.
Labs often insist on airtight containers for a reason. Moisture slips in through loose seals, even in humid air, and prompts rapid decomposition. Using glass bottles with Teflon-lined caps or chemical-resistant seals cuts down on leaks. A secondary containment tray turns an accident into an inconvenience, rather than a crisis covering the whole shelf.
Keeping People and Chemicals Apart
Locking up dangerous reagents protects both chemicals and people. I remember a story from a research institute—one careless student grabbed the wrong bottle, thinking it was ethanol. Mild confusion ended in a sobering trip to the hospital. Labeling can’t cover every hazard. A separate storage cabinet, clearly marked and fire-resistant, spells out the risks. GHS-compliant hazard signs keep everyone on their toes.
This chemical doesn’t belong in crowded under-sink cupboards or catchall supply rooms. Having a dedicated chemical safety fridge helps, but only if that fridge is specifically designed for flammable liquids. Regular kitchen fridges aren’t built to contain leaks or stop a spark. A spill response kit nearby, with protective gloves and goggles, gives users a fighting chance if an accident happens.
Paying Attention to Shelf Life and Inventories
Over time, isopropyl chloroformate breaks down, producing gases that quietly raise the risk of an explosion. No one benefits by holding on to outdated bottles. Logging purchase and opening dates makes it easy to rotate supplies and get rid of expired stock before safety degrades. Trained staff should check cabinets often. Anything older than six months probably should go for disposal, according to the safety data sheets I’ve read.
Better Habits and National Safety Guidelines
The importance of proper training can’t be overstated. OSHA, NIOSH, and similar authorities around the world layout expectations for chemical handling and storage. Following their rules doesn’t just keep the workplace safe; it avoids expensive fines and protects an organization’s reputation. I’ve met frustrated managers who learned this the hard way, scrambling after an inspection.
If everyone—from the person who signs the order form to the technician who decants a bottle—understands both the risks and the storage requirements, accidents lose their chance to happen.
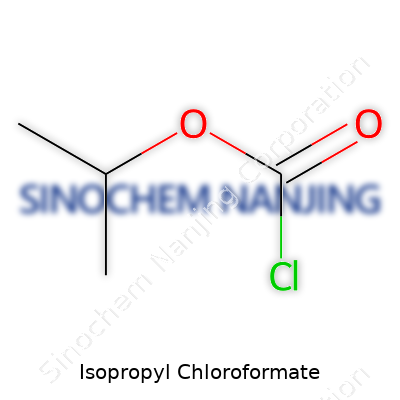
What is the chemical formula of Isopropyl Chloroformate?
The Formula Behind Isopropyl Chloroformate
The chemical formula for isopropyl chloroformate is C4H7ClO2. This formula tells you a lot. Four carbons, seven hydrogens, a single chlorine, and two oxygens—this cocktail creates a highly reactive compound. Chemists see this formula and immediately know it’s packing both a chloroformate group and an isopropyl group. To picture it, the isopropyl group hooks up with a carbonyl (C=O), which then links to chlorine and one extra oxygen.
From the Bench to the Bottle: Real Uses
In the lab, people reach for isopropyl chloroformate to introduce the carbamoyl group into molecules. It’s a key player in making carbamates and ureas, which turn up in everything from pharmaceuticals to agricultural chemicals. Drug makers depend on it in the step-by-step dance of assembling complicated molecules with specific effects on the human body. If you’ve been prescribed certain medicines or bought specific crop protection products, you’ve already crossed paths with what started as a bottle of this stuff.
Personal Experience from Lab Days
Recalling time in an organic lab, using isopropyl chloroformate brought a potent mix of respect and caution. A spill would clear the room—its pungency sticks in memory long after leaving the fume hood. Handling it gave me a practical lesson on just how crucial it is to understand a chemical’s structure: knowing the formula pointed to its reactivity, which explained its behavior. I learned quickly that the chlorine atom makes it pretty eager to react, especially with anything willing to swap out atoms, and the isopropyl group nudges it toward forming stable carbamates.
Risks and Responsibility
Isopropyl chloroformate does more than help make products. It also brings risk. The same chemical groups that make it useful also make it hazardous to both skin and lungs. Its vapors can choke or burn, it reacts with water to release gases no one wants to inhale, and it’s flammable. These dangers highlight the broader issue of chemical safety—for professionals but also for the people down the line who might encounter residues or byproducts.
How to Lower the Hazards
Safety practices save more than just the person wearing gloves. Labs and manufacturers must run tight ships: storage under cool, dry conditions, fume hoods, and spill kits close at hand. Regulations already demand special care for transport and disposal. Training matters even more than labels or alarms—my own mistakes as a student proved this point. Real understanding of a formula turns into thoughtful action, not rote procedures.
Respect for Chemistry and Accountability
The formula C4H7ClO2 is more than numbers and letters. It points to possibilities and pitfalls. Choosing to use this compound means holding responsibility—for the purity of medicines, the safety of coworkers, the integrity of the environment, and the well-being of communities. Open reporting on incidents and transparent environmental monitoring build public trust.
What are the potential hazards of Isopropyl Chloroformate?
The Real Dangers in the Lab
Every chemical brings baggage, and isopropyl chloroformate drags along quite a bit of trouble. Folks in research or manufacturing know this one for its use in making carbamates and other organic compounds. The risk goes way beyond ordinary skin irritation or needing to work under a good fume hood. Breathing its vapors is no light concern, with exposure inviting headaches, nausea, dizziness, and even trouble breathing. At high enough concentrations, lungs can feel like they’re burning, leading to coughing fits and chest pain. There’s no overstating the discomfort of even mild exposure, so proper precautions can’t ever drop off the checklist.
Chemical Burns and Eye Damage
Spilling isopropyl chloroformate on skin causes burning and blistering, not just mild irritation. Eyes react even more severely—splashing leads to intense pain, tearing, and can do real damage to vision. Many lab veterans have a story or two about someone not wearing eye protection and paying the price. Washing immediately helps, but the pain sticks around for days. Handling this chemical without gloves or goggles turns a short job into a visit to the occupational health nurse.
Explosive Reactions: More Than a Film Fantasy
Mix this substance with water, acids, bases, or even the wrong solvents, and it can release toxic gases like phosgene, hydrogen chloride, and carbon monoxide. These aren’t just theoretical threats. There have been incidents where a bit of spilled reactant and some stray moisture kicked off a nasty fume cloud in a matter of seconds. Those gases don’t only smell bad—they’re dangerous to lungs, and a large enough leak puts people at real risk. I remember a time when a container with a crusty lid released a sharp hiss after opening, reminding everyone to respect storage rules and double-check their PPE.
Flammability: Fire Hazards on the Bench
Isopropyl chloroformate doesn’t just sit quietly—its vapors catch fire well below the temperature of boiling water. Flames and hot surfaces in the lab bring big risks. Practically, this means storing it away from heating elements and making sure spill kits include materials that can contain both chemical and fire risks. People sometimes forget this point, putting the bottle too close to an active setup. A bit of spilled liquid, a spark from a static discharge, and suddenly the lab’s in chaos.
Long-Term Effects and Environmental Concerns
Short-term exposure problems grab the most attention, but repeated low-level contact brings lingering issues. Isopropyl chloroformate breaks down into compounds that stress the liver and kidneys. It also damages aquatic life, so pouring waste down the sink brings more than a legal issue—it hurts local ecosystems. Proper disposal can sound tedious—but skipping that step builds up risk for everyone downstream. Labs working with this chemical should set up clear collection and disposal routines that never rely on shortcuts.
Handling It Right
Calling out dangers only matters if solutions make a difference. Fume hoods, goggles, gloves, and lab coats form the baseline. Training goes a bit deeper—showing how to inspect for leaks, store it in tightly sealed containers, and plan work in a way that never puts people alone with the substance. Emergency eyewash stations and spill kits close by have saved more than one person from a rough day. Trust among colleagues grows when everyone pitches in to keep the place safe, and that sense of responsibility is what keeps accidents rare.
| Names | |
| Preferred IUPAC name | Propan-2-yl carbonochloridate |
| Other names |
Carbonochloridic acid, 1-methylethyl ester Chloroformic acid isopropyl ester Isopropyl chloro(carbonate) Isopropyl chlorocarbonate Isopropyl carbonochloridate |
| Pronunciation | /ˌaɪsəˈproʊpɪl klɔːˈrɒfərˌeɪt/ |
| Identifiers | |
| CAS Number | 3319-31-1 |
| 3D model (JSmol) | `Isopropyl Chloroformate JSmol string: CO(C(=O)Cl)C` |
| Beilstein Reference | 1071244 |
| ChEBI | CHEBI:132962 |
| ChEMBL | CHEMBL135887 |
| ChemSpider | 16001 |
| DrugBank | DB08729 |
| ECHA InfoCard | 03ee2f3c-d05c-4ad2-ab44-4e9750a8d6db |
| EC Number | 208-760-7 |
| Gmelin Reference | Gmelin 8336 |
| KEGG | C19261 |
| MeSH | D016437 |
| PubChem CID | 6589 |
| RTECS number | NT1225000 |
| UNII | 1Q7V6T0E80 |
| UN number | UN2406 |
| CompTox Dashboard (EPA) | DTXSID5020698 |
| Properties | |
| Chemical formula | C4H7ClO2 |
| Molar mass | 122.55 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Pungent |
| Density | 1.11 g/mL at 25 °C |
| Solubility in water | Decomposes in water |
| log P | 1.85 |
| Vapor pressure | 10 mmHg (20°C) |
| Acidity (pKa) | 1.3 |
| Basicity (pKb) | pKb = 7.07 |
| Magnetic susceptibility (χ) | -53.0e-6 cm³/mol |
| Refractive index (nD) | 1.403 |
| Viscosity | 2.47 mPa·s (20 °C) |
| Dipole moment | 2.00 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 350.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −348.9 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -1825 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS02,GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H302, H314, H331, H335 |
| Precautionary statements | P210, P261, P271, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P370+P378, P403+P233, P405, P501 |
| Flash point | 50 °F (10 °C) |
| Autoignition temperature | 451°F (233°C) |
| Explosive limits | 4.5–11% |
| Lethal dose or concentration | LD50 oral rat 1,100 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 946 mg/kg |
| NIOSH | RD3325000 |
| PEL (Permissible) | PEL: 0.1 ppm (0.5 mg/m3) as ceiling |
| REL (Recommended) | REL: 0.5 ppm (2 mg/m³) |
| IDLH (Immediate danger) | IDLH: 10 ppm |
| Related compounds | |
| Related compounds |
Methyl chloroformate Ethyl chloroformate n-Propyl chloroformate Phenyl chloroformate Benzyl chloroformate |

