Digging Into Isopropyl Chloroacetate: Beyond the Basics
A Walk Through Its Story
Synthetic chemistry rarely gets a spotlight outside labs, but the story of isopropyl chloroacetate deserves a closer look. This compound didn’t just pop up overnight; over decades chemists have tinkered with its backbone to push forward industrial chemistry and modern manufacturing. The roots lie in early esterification studies, where finding the right alcohol and acid match brought out new flavors and reactivity profiles. Isopropyl chloroacetate combines the predictable volatility of an isopropyl ester with the reactivity boost that comes from a chlorine atom swinging off the acetic acid piece. Over time, this combo carved out a place in both synthetic chemistry and applied industrial fields. For folks working with carboxylic acid derivatives, it became the kind of “tool in the toolbox” you reach for when regular acetate esters don’t quite cut it.
What Sets It Apart
Pure isopropyl chloroacetate shows up as a colorless, sometimes faintly straw-tinted liquid, with a smell that chemists liken to a cross between fruits and solvents. Because it mixes a strong ester group with a reactive chloromethylene, you get a compound ready to play in everything from agrochemical intermediates to pharmaceutical building blocks. The boiling point sits in a comfortable midrange, allowing for controlled reactions and manageable storage. It burns readily, so chemists working at a bench have to give it the respect once reserved for more notorious flammables. Most labs store it in dark glass, away from open flames or oxidizers, and for good reason.
Physical & Chemical Details
On paper, the molecular details confirm its reputation: a low-to-moderate boiling point, moderate density, and fair solubility in typical organic solvents like ether, acetone, and benzene. Water doesn’t agree with it for long — hydrolysis can sneak in and turn it into isopropanol and chloroacetic acid, which flips its utility into a safety headache if mishandled. The chlorine atom, often overlooked, transforms the chemical’s personality. It pushes the molecule toward more ambitious reaction schemes, as that reactive spot gets targeted in transformations that plain esters can’t pull off.
Technical Specs and Label Talk
Bottles of isopropyl chloroacetate leave factories labeled with warnings that pull no punches: corrosive, flammable, keep off skin, avoid inhaling fumes. Even small splashes can irritate eyes or hands because the chlorine atom cranks up the chemical’s bite. Safety data doesn’t dance around: goggles, gloves, lab coats, and fume hoods matter every time. Industry standards, governed by global agencies and local workplace safety rules, call for careful tracking from delivery to disposal. Trained chemists learn early on that a shortcut with this ester invites equipment corrosion or worse, personal harm.
In Search of the Right Recipe
Making isopropyl chloroacetate usually means bringing together isopropanol with chloroacetyl chloride, often in the presence of a base like pyridine to mop up the acidic fumes. Every batch commands caution. Reaction temperatures stay tightly controlled, as the side effects can snowball quickly if heat or mixing gets out of hand. After the reaction, neutralization steps and careful washing strip out any lingering acid or byproducts, leaving a liquid pure enough for downstream synthesis. Years of lab fails and successes shaped this method, giving manufacturers procedures that work as reliably on a kilo scale as in a 100-gram flask for research.
The Reaction Playground
Most seasoned chemists know isopropyl chloroacetate as more than a static ingredient. The compound jumps into nucleophilic substitution reactions, sometimes serving as an acylating agent when other esters lag behind. It stands ready for modifications into acids, amides, or other esters, and the chlorine atom opens doors for repeated molecular tweaking. Researchers in medicinal chemistry find value in the way it hands over its group to build up complex molecules or fine-tune drug candidates. A handful of specialty syntheses depend on its reaction profile, giving it a niche value that doesn’t always make headlines but gets the job done in the background.
Alias and Name Soup
Like many industrial chemicals, isopropyl chloroacetate collected an armful of aliases: 2-chloroacetic acid isopropyl ester, isopropyl monochloroacetate, and more. The IUPAC conventions satisfy regulators and journals, but most folks in the lab know it by its simplest version—whatever rolls off the tongue or prints clearly on a label. Documentation and transport paperwork bounce between these names, but for someone running a reaction it’s the hazards and the purity that matter most.
Staying Safe, Staying Sharp
Lab safety drills don’t happen just for the optics. With isopropyl chloroacetate, serious protocols become second nature: ventilated storage, bonded containers, spill kits poised for action. Anyone handling this substance learns that the chlorine crowd brings increased toxicity and accident risk. Gloves become armor, and any sniff of its vapors calls for thicker barriers or a quick step to the fresh air. Proper disposal isn’t just best practice—it’s about keeping unwanted chlorinated residues out of drains or soil. A handful of stories about careless handling keep folks on their toes; nobody wants to clean up a spill with this one or explain an incident to a safety officer.
Where It Shows Up
The real value of isopropyl chloroacetate shows up downstream. In pharmaceuticals, it steps in as a building block for integrating functional groups onto bigger molecules. Agrochemical makers turn to it for its reactivity, pushing it into pesticides or herbicide precursors that help keep crops productive. Sometimes it enters the fragrance industry, where its ester backbone gives perfumers intermediate options for crafting new scents. Fine chemistry manufacturers appreciate the versatility, and the reliability in scale-up helps ensure that new molecules can go from grams to kilograms without missing deadlines or budgets.
On the Frontier: Research & Development
Recent years have delivered a growing bank of published reactions harnessing isopropyl chloroacetate. Efforts to squeeze more selectivity from classical reactions often start by swapping out less reactive esters for this chlorinated version, hoping for sharper yields or new products. Green chemistry advocates continue to experiment with alternative preparation methods or safer base additives, seeking to cut the footprint from both process waste and worker exposure. Analytical chemists look for faster, cleaner detection in finished products, always under pressure from tightening quality regulations.
Weighing Toxicity and Human Risk
Workplace exposure studies point to both acute and chronic toxicity risks. Fumes can irritate airways and eyes, while skin contact leads to burns more aggressively than milder esters. Ingestion or large exposures spell trouble, as the compound’s breakdown products pose risks beyond the parent molecule. Regulatory agencies keep expanding guidelines for air quality, storage limits, and permissible exposure levels at worksites. Toxicity assessments bring added scrutiny into disposal practices, and calls for alternative green reagents echo from industry watchdogs and university labs alike. Every new study pushes producers to refine safety messaging and adapt training protocols before permits get renewed.
Looking Beyond Today
Chemical manufacturing never stands still. As industries chase more sustainable and safer options, isopropyl chloroacetate will likely see tweaks in both how it gets made and the kinds of products it flows into. New research aims at reducing waste and hazards in its synthesis and finding biodegradable or less toxic replacements in sensitive applications. For now, its role in specialty synthesis and industrial projects looks secure, but long-term growth will ride on improvements in both worker safety and environmental impact. Researchers and factory managers keeping one eye on emerging regulations and the other on customer needs will be the first to adapt, staying ahead in a world where health, performance, and responsibility are knotted together tighter every year.
What are the main applications of Isopropyl Chloroacetate?
Chemistry in Action: Far Beyond the Lab Bench
Isopropyl chloroacetate rarely pops up in dinner table talk, but it quietly shapes a bunch of things we take for granted. This chemical carries a punch, both inside research labs and in products that end up all around us. Folks in industry handle it with care because it doesn’t just lie around; it reacts, it builds, and it unlocks possibilities for everything from cancer drugs to bug killers. I’ve watched research teams weigh it out in fume hoods, gloves on, eyes peeled—one slip-up, and the whole bench stinks like vinegar and cleaning fluid.
Pharmaceuticals: The Unsung Player in Drug Making
One of the largest jobs for isopropyl chloroacetate comes from pharmaceutical synthesis. It helps add a two-carbon group onto molecules, setting the stage for medicines with life-changing power. I know chemists who swear by this reagent for building up cancer drugs and antibiotics. They chase high purity, and that starts with raw materials like this—one hiccup in the supply chain can slow a whole drug project. According to industry reports, its role in producing intermediates for anti-cancer agents has made it a staple, despite health and handling risks.
Pesticides and Agrochemicals: Protecting Harvests
Pesticide makers use isopropyl chloroacetate to make active ingredients that keep bugs and weeds at bay. Growers end up relying on these chemicals to keep food on our plates. What makes this compound critical is its knack for efficiently connecting pieces in complex molecules. A few years back, a shortage dragged out deliveries of a common rice pesticide—the bottleneck came down to the availability of isopropyl chloroacetate, not the finished product. This showed how even a small step in the process can hit the whole food chain.
Solvents and Specialty Chemicals: Keeping Industry Moving
Some specialty applications see this chemical as a go-to for making solvents or additives that tune the behavior of paints and coatings. I’ve seen it in action when mixing up batches meant to treat specialty glass or metals. Surface treatments call for chemicals that evaporate cleanly or leave behind just the right film—here, isopropyl chloroacetate holds its own. Its volatility makes it useful, but demands proper ventilation and storage. It’s not something you want on your skin or in your lungs, so handling rules are strict.
Safety and Environmental Responsibility
Every company using isopropyl chloroacetate faces two key questions: Can we keep workers safe, and can we keep it out of the environment? This chemical can irritate eyes and lungs and doesn’t play nice with water—spills become an emergency fast. Federal safety data sheets highlight the need for goggles, gloves, and fume hoods. For the broader picture, strict rules on disposal help prevent it from showing up in rivers and soil. As countries place more weight on environmental and worker safety, best practices keep evolving. Companies now look for ways to reclaim, recycle, or substitute it rather than just ship off spent solutions for burning.
Towards Safer and Smarter Chemistry
Teaming up chemists with safety experts leads to smarter choices about how and where to use isopropyl chloroacetate. Swapping in greener alternatives for certain steps cuts risk for both workers and the world outside the plant gate. Whether it’s in a university lab or a chemical warehouse, the same truth holds: paying attention to how we handle chemicals like this means fewer emergencies and better-performing products.
What is the chemical formula and structure of Isopropyl Chloroacetate?
Chemical Basics of Isopropyl Chloroacetate
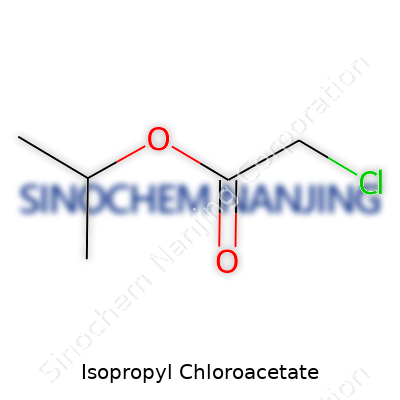
Isopropyl chloroacetate finds its importance in laboratories and specialty chemical industries. To break it down, its chemical formula is C5H9ClO2. This compound features an isopropyl group and the ester of chloroacetic acid. On the molecular level, it contains five carbons, nine hydrogens, one chlorine atom, and two oxygens. The structure centers around the acetate component, where a chlorine atom bonds to the alpha carbon, giving it both its “chloro” and “acetate” properties. The ester forms as the isopropyl alcohol attaches through an oxygen bridge to the acyl group of chloroacetic acid. The simplified skeletal structure shows a ClCH2COO– side linked to the isopropyl chain (–OCH(CH3)2).
Practical Chemistry and Where It Shows Up
My time working in an R&D lab taught me to respect chemicals capable of more than just sitting quietly on a shelf. Isopropyl chloroacetate has that reactive ester and a chlorine that acts as a powerful handle in synthesis. Small tweaks in its structure lead to a wide array of products ranging from pharmaceuticals to specialized agricultural chemicals. This ester sometimes serves as an intermediate—acting as a middle step people often overlook until the reaction stalls. It brings a certain reliability to organic syntheses. When chemists look for ways to add an acyl group or introduce an ester in the presence of a reactive halide, this compound fits the bill.
I remember a colleague who tried to replace it with a less reactive ester to cut costs, only to watch the yields drop and troubleshooting eat away more hours. The right chemical tool for the right step can save not just money, but morale, too. Overlooking the functional group flexibility and the stability of the isopropyl group leads to headaches further downstream.
Why Structural Awareness Matters
A part of working safely with substances like isopropyl chloroacetate means understanding more than its formula. The chlorine atom brings reactivity, but also a risk of irritation. Lab manuals don’t highlight enough how a splash can cause skin burns or respiratory distress. Practical experience says double-glove and work in a fume hood, even if the hazard label stays vague. The structural features can lead to hydrolysis if left open on the benchtop in a humid environment. It breaks down, releasing fumes and leaving messy residues in glassware. It pays to clean up right away.
Every chemist remembers the stumble when storing halogenated esters incorrectly. They don’t last long under sunlight or near heat. This isn’t just a shelf-life issue—improper storage risks pressure buildup or unexpected exposure. Smart storage uses amber bottles, secondary containment, and avoids overcrowded chemical cabinets. That's not optional if you share space with others or train lab assistants who learn more from your habits than a written protocol.
Pathways Toward Safer and Smarter Handling
Isopropyl chloroacetate shows up because it helps chemical reactions progress quickly and cleanly. To keep it useful and avoid trouble, handling instructions need to go beyond “store in a cool, dry place.” Labs and factories benefit from regular training—walk through spill drills as part of onboarding, keep safety data sheets visible, and inspect expired containers monthly. Encourage anyone mixing or storing the compound to describe out loud what happens if things go wrong. That kind of practical storytelling makes the properties real and keeps both new scientists and seasoned workers alert.
At the end of the day, the chemical formula points to a real, hands-on piece of a much bigger process. Recognizing its structure, hazards, and best practices ensures it truly serves its role in delivering reliable results and protecting the people working with it.
What are the storage and handling precautions for Isopropyl Chloroacetate?
Understanding Why It Demands Careful Attention
People who’ve spent time in a lab or warehouse know that not every bottle on the chemical shelf tells the full story. Isopropyl chloroacetate draws attention because of what it can do if left unchecked. Leaks, fumes, and reactions can change a safe workspace into a hazardous one pretty fast. I once discovered a bottle that had been set near a sunny window. The fumes hit me long before I got close, and it turned out some light heat had loosened the lid. That made me double down on safe storage, no matter how short the stop at this shelf.
Keep All Containers Tightly Closed
Make the tight seal part of the routine. Isopropyl chloroacetate does not hold back when it comes to vapors. Small gaps let fumes escape. Workers near the bottle start breathing in what they shouldn’t. In my years on research benches, cracked lids explained more unexplained symptoms than most newcomers might guess—especially for anything volatile. So each time a bottle leaves a bench, a twist to lock that cap makes a difference.
Pick a Cool, Dry, Well-Ventilated Spot
Heat messes with this chemical, turning liquid into trouble. That means shelving anything containing isopropyl chloroacetate well away from direct sunlight, heaters, or motors that might throw sparks. Humidity also poses a real risk, since moisture can lead to slow decomposition or boost unwanted reactions. In one instance, a colleague underestimated a humid storeroom; corrosion led to a slow leak that went unchecked. A dry room with fans running every shift offers a layer of protection. Ventilation also keeps fumes from building up, turning a regular space into a hazard zone.
Keep Away from Incompatibles
Any good safety manual highlights who shouldn’t play together on the shelf. Strong bases, amines, and oxidizers react fast—and dangerously—with organic chlorides. Setting isopropyl chloroacetate next to bleach, strong alkalis, or acids invites accidents. I’ve seen labels fade and bottles drift from their assigned spots, so regular checks and clear signage help everyone remember which shelves keep apart.
Don’t Forget the Right Containers
Glass or high-grade plastic works for holding isopropyl chloroacetate. Metal or regular plastic starts to fail over time, especially once chlorine gets involved. Corroded caps or softened bottle walls break or crack just as easily, so fresh containers and honest labeling matter. On my own benches, I swapped containers before they showed wear, dodging more leaks and questions from younger lab mates.
Fire Safety: A Priority
Flammability ranks at the top of risk lists with this compound. It catches fire from a spark or pilot light, so keep sources of ignition far from storage areas. Spill kits and fire extinguishers aren’t just for looks; they ought to be checked and within reach. Remind new employees at every training, too—memorization doesn’t always keep pace with stress.
Education and Oversight: The Real Solution
It’s not enough to follow printed rules. Safe chemical storage becomes a habit through repetition, open conversation, and strong examples set by long-time staff. I always encourage questions, no matter how basic, instead of letting someone pretend to know their way around. Supervisors and team leads should audit storage spaces often, replacing labels, reviewing playbooks, and talking through the logic behind every shelf and sign. That keeps everyone thinking before they reach, move, or pour.
Is Isopropyl Chloroacetate hazardous or toxic to health?
Why Chemical Safety Catches My Attention
Workplaces that handle chemicals teach hard lessons in respect. Everything from solvents to cleaners carries risks. A name like isopropyl chloroacetate might not pop up during a trip to the store, yet it underscores why reading labels and safety sheets is not pointless bureaucracy.
Health Risks That Matter
Isopropyl chloroacetate lands on lists that raise red flags for researchers and plant operators. Its chemical relatives have landed folks in the ER, and this one isn’t much friendlier. Exposure often means trouble for lungs, skin, and eyes. Liquid spills evaporate, creating fumes. Even short stints without carelessness can end with nose and throat irritation. Prolonged encounters ramp damage upward: coughing, headaches, dizziness, and chest discomfort. Absorption through skin can follow, which is not something anyone enjoys learning late.
I remember a colleague who trusted gloves “just for a moment” during a late cleanup—he wound up with redness and blisters running up his forearm. Turns out, isopropyl chloroacetate doesn’t always announce itself right away. Hours later, swelling and burning popped up, confirming the need for proper gear and thorough washing.
Long-Term Risks and Regulations
Science draws links between repeated contact with this chemical and potential nerve damage, especially if inhaled often in small doses. Lab tests flag liver and kidney strain over time. Ignoring proper protection or skipping ventilation stacks up risk, which over years becomes hard to reverse.
Regulatory agencies do not joke around once enough data comes in—OSHA and similar groups lay out boundaries and penalties for unsafe use. Material Safety Data Sheets spell things out: keep the stuff contained, wear barrier gloves and face shields, and install ventilation. Companies keep track of airborne levels because too much escapes fast, causing not just medical bills, but sometimes permanent damage.
The Role of Training and Equipment
From my years near the loading docks, I saw wise supervisors invest in hands-on seminars and mandatory walkthroughs. Refreshers once a season kept everyone alert to poor habits sneaking in: open containers, reused gloves, and shortcuts on goggles. Firms that take safety culture seriously invite fewer accidents and lawsuits.
Having the right equipment makes or breaks safety. Not every glove stands up to isopropyl chloroacetate. Real protection means picking gear designed for the specific chemical, checking seals, and tossing anything with pinholes or rips. Workers trade stories about gear failures—most of them end with stern warnings to the next shift.
Common-Sense Solutions
Trusted suppliers and labeled containers cut down on confusion. Eyewash stations and quick showers within reach make all the difference during emergencies. Supervisors who set clear steps before, during, and after each shift prevent a lot of regret later.
In the end, treating isopropyl chloroacetate with respect keeps people healthy. Knowledge beats luck every day in rooms where chemicals get stored, shipped, or mixed.
What is the shelf life and stability of Isopropyl Chloroacetate?
Understanding the Chemical’s Behavior
Isopropyl chloroacetate doesn’t show up in everyday conversations, but anyone working in a lab or handling chemicals in industrial settings recognizes the need for trustworthy information about shelf life and stability. If you’ve ever had to clean out an old chemical closet, you know that faded bottles of anything with “chloro” in the name should cause more than a little caution. The shelf life of isopropyl chloroacetate can make or break not just an experiment, but lab safety as a whole.
From experience, even unopened containers can become risky if left for years. Isopropyl chloroacetate is an ester, and like many esters, it undergoes slow decomposition, especially if exposed to moisture or heat. With a boiling point around 153°C and a reputation for hydrolysis, things get tricky when labs don’t keep bottles well-sealed or stored away from water.
The Impact of Storage Conditions
If you've worked in labs with spotty climate control, you know temperature swings accelerate breakdown. High humidity welcomes hydrolysis, breaking down the compound into chloroacetic acid and isopropanol, both of which bring their own risks. Glass bottles with tight, chemical-resistant stoppers avoid some problems. Polyethylene caps don’t always hold up against long-term storage of chlorinated compounds; slow leaching or vapor migration could start, affecting purity before anyone notices.
Direct sunlight produces heat, so even a clear glass bottle left out on a bench feels the effect in a few days. The more you keep the chemical away from light, the longer it holds its original form. In my own undergraduate lab, forgotten bottles sitting near a window taught students about color changes and pressure buildup in real time. Not a safe way to learn, but memorable.
Recognizing Signs of Decomposition
Most chemical suppliers recommend using isopropyl chloroacetate within two years of receipt if storage stays dry and cool. The sharp, slightly fruity smell turns acrid as decomposition creeps in. Yellowing of the clear liquid signals it’s past its prime. Even so, relying on labels alone can backfire. Old stock, or bottles without date stamps, pushes staff to guess—never a good plan in chemical handling.
Personal experience says don’t trust anything that smells “off.” If you lift a cap and pick up extra pungency, the safety sheet’s warnings about toxicity start to hit home. Decomposition products like HCl gas form slowly, corroding both closures and your confidence. Keep your inventory lean so nothing lingers long enough to cause problems.
Minimizing Risks, Maximizing Utility
Several steps keep isopropyl chloroacetate where it should be: inside well-labeled containers, away from water, and out of sunlight. Dry, dedicated chemical storage cabinets with desiccants work better than regular shelves. Always date bottles at receiving—permanently, with a marker. Inventory checks every few months let staff recognize which bottles need disposal. Don’t ignore changes in odor, color, or clarity.
A robust chemical hygiene plan can’t rely on memory or luck. Stick close to manufacturer guidelines but develop a policy that requires regular turnover. Trained staff stay ready to recognize unsafe chemicals and understand disposal procedures. Experience reminds everyone: erring on the side of caution beats cleaning up after a shelf-life failure.
| Names | |
| Preferred IUPAC name | 2-chloro-1-methylethyl acetate |
| Other names |
Chloroacetic acid isopropyl ester Isopropyl 2-chloroacetate 2-Chloroacetic acid isopropyl ester |
| Pronunciation | /ˌaɪ.səˈproʊ.pɪl ˌklɔːr.oʊ.əˈsiː.teɪt/ |
| Identifiers | |
| CAS Number | 543-27-1 |
| 3D model (JSmol) | `Isopropyl Chloroacetate` JSmol 3D model string: ``` isopropyl chloroacetate C(CCl=O)OC(C)C ``` This is the SMILES string representing the molecular structure which can be used to generate a 3D model in JSmol. |
| Beilstein Reference | '1718736' |
| ChEBI | CHEBI:77804 |
| ChEMBL | CHEMBL502697 |
| ChemSpider | 16193 |
| DrugBank | DB14347 |
| ECHA InfoCard | 100.010.948 |
| EC Number | 205-962-2 |
| Gmelin Reference | **Gmelin Reference:** 83667 |
| KEGG | C19806 |
| MeSH | D017355 |
| PubChem CID | 7443 |
| RTECS number | TX8575000 |
| UNII | V2QEH3W08K |
| UN number | UN1682 |
| CompTox Dashboard (EPA) | DTXSID1052327 |
| Properties | |
| Chemical formula | C5H9ClO2 |
| Molar mass | 136.58 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | Pungent |
| Density | 1.172 g/mL at 25 °C (lit.) |
| Solubility in water | Decomposes in water |
| log P | 1.38 |
| Vapor pressure | 3.6 mmHg (20°C) |
| Acidity (pKa) | pKa ≈ 2.8 |
| Basicity (pKb) | Isopropyl Chloroacetate" does not have a measurable pKb value because it is not a base; it is an ester. |
| Magnetic susceptibility (χ) | -50.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.414 |
| Viscosity | 1.523 cP (20°C) |
| Dipole moment | 2.10 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 383.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -423.65 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -669.1 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS02,GHS06 |
| Signal word | Danger |
| Precautionary statements | P261, P280, P301+P312, P305+P351+P338, P304+P340, P330, P332+P313, P337+P313, P362 |
| NFPA 704 (fire diamond) | 3-2-1-W |
| Flash point | Flash point: 54 °C |
| Autoignition temperature | 530°C |
| Explosive limits | Explosive limits: 2–11% |
| Lethal dose or concentration | LD50 oral rat 730 mg/kg |
| LD50 (median dose) | LD50 (median dose): 730 mg/kg (rat, oral) |
| NIOSH | NIOSH: IM1455000 |
| PEL (Permissible) | PEL: 1 ppm (5 mg/m³) |
| REL (Recommended) | 0.1 ppm |
| IDLH (Immediate danger) | IDLH: 10 ppm |
| Related compounds | |
| Related compounds |
Chloroacetic acid Isopropyl acetate Methyl chloroacetate Ethyl chloroacetate Chloroacetyl chloride |

