Isopropenyl Acetate: A Practical Look at Its Past, Present, and Future
Historical Development
Isopropenyl acetate found its footing in the world of chemicals during the mid-20th century, as industries sought new ways to synthesize intermediates for pharmaceuticals, fragrances, and specialty chemicals. Scientists discovered that this compound, a colorless and distinctive-smelling liquid, opened new doors for organic synthesis. Early research focused on its use in forming acetic acid derivatives. By the 1960s, chemical companies pushed the boundaries, leveraging improved catalytic systems to boost yields and improve purity levels. Over time, production facilities across Europe, Asia, and North America incorporated the compound, taking advantage of increasingly efficient routes, such as the reaction between acetone and acetic anhydride under acidic conditions.
Product Overview
Coming off the production line, isopropenyl acetate often catches attention for its sharp, sweet odor and transparent appearance. Chemists turn to it as more than just a curiosity. Its composition—C5H8O2—gives it a unique edge in the laboratory. The molecule carries both acetate and alkene functionalities, making it a valuable intermediate. Many production plants sell it in bulk form, packaged tightly in sealed drums or stainless containers to guard against air and moisture exposure. The global market stays steady, with key consumers in the pharmaceutical, agrochemical, and flavoring sectors.
Physical & Chemical Properties
Take a closer look at the physical traits: isopropenyl acetate boils at around 96 degrees Celsius and melts well below freezing. The liquid form brings a low viscosity, so it pours quickly and is easy to handle in processing. It barely dissolves in water, but it mixes well with common organic solvents like alcohol, ether, and chloroform. Chemically, the acetate group serves as both a protective and reactive site for synthetic reactions, and the alkene group plays a role in addition reactions or polymerization. The molecule exhibits stability under normal storage, though it’ll degrade with strong acids or bases.
Technical Specifications & Labeling
Manufacturers must watch standards set by regulatory agencies. They test batches for purity, water content, and acidity, since high impurities compromise downstream applications. Labels list chemical identity, hazard pictograms, recommended storage temperatures, and the United Nations (UN) number for shipping. These details help suppliers and users reduce mishaps, and labels must communicate hazards like flammability and possible eye or skin irritation. Quality specifications commonly call for purity above 98% and restrict water down to less than 0.1%.
Preparation Method
The dominant production route starts with acetone and acetic anhydride, catalyzed by a strong acid like sulfuric acid. The trick involves careful control of temperature and the removal of water formed during the reaction, as any stray moisture can reverse the process or lower yield. Some plants explore gas-phase techniques to optimize throughput. Another approach leverages isopropenyl alcohol, reacting directly with acetic acid in the presence of acid catalysts—a method favored for its simplicity in laboratory settings. Each process requires robust separation steps, like distillation under reduced pressure, to keep the final product pure.
Chemical Reactions & Modifications
Organic chemists often use isopropenyl acetate to introduce acetate groups into other compounds or as a starting point for more complex molecules. It undergoes hydrolysis in the presence of acids to yield isopropenyl alcohol and acetic acid—useful in both synthetic and analytical chemistry. The molecule reacts well in addition reactions, picking up halogens or even participating in Diels-Alder synthesis when combined with suitable partners. Its dual-functionality makes it a candidate for polymer modification, especially when seeking to graft acetate chains onto backbones or tailor polymer properties for specific end-uses.
Synonyms & Product Names
Depending on the region, isopropenyl acetate goes by names like 1-methylethenyl acetate, 2-propen-1-yl acetate, and methylpropenyl acetate. Custom blends or technical grades sometimes bear trade names, though the chemical industry often defaults to the IUPAC designation for clarity. This makes communication clearer during cross-border shipping and regulatory filings.
Safety & Operational Standards
Handling this chemical calls for care. Vapors irritate the eyes and respiratory tract, and the liquid can cause dermatitis after repeated skin contact. Storage guidelines recommend cool, well-ventilated areas, away from open flames and sources of static electricity since isopropenyl acetate ignites easily. Most factories rely on explosion-proof pumps and sealed transfer systems to control spills. Facilities preparing or using this compound keep emergency shower stations and eye-wash kits nearby for quick response. Safety data sheets from trusted sources lay out recommended personal protective equipment—goggles, gloves, and flame-resistant lab coats. Regulatory standards reinforce these requirements, with OSHA and EU-REACH regularly updating permissible exposure limits and best-practice guidelines.
Application Area
Isopropenyl acetate finds work as an intermediary in the synthesis of pharmaceuticals—helping build molecules for pain relief, infection control, or even cancer therapy. The fragrances industry uses it to build complex aromatic compounds found in perfumes and flavorings. Chemical manufacturers take advantage of its reactive sites to craft agrochemicals, including certain herbicides and pesticides. Some labs reach for it to prepare specialty monomers for polymers, tuning properties for everything from adhesives to specialty coatings. Because it participates cleanly in Diels-Alder and Michael addition reactions, it helps organic chemists build intricate molecular architectures that support everything from material science to drug discovery.
Research & Development
Current research explores new catalytic systems that speed up production and limit waste—base and acid catalysis compete, and scientists regularly test heterogeneous catalysts that could allow continuous production and easier purification. There's also a hunt for greener processes, reducing reliance on fossil-derived acetone or minimizing acidic waste. Teams in academia and industry have teamed up to test bio-based routes, such as fermentative production of precursors. On the synthetic chemistry front, new reactions expand the versatility of isopropenyl acetate. The molecule has popped up in recent patent applications for specialty polymers, UV-curable coatings, and new drug scaffolds.
Toxicity Research
Toxicology studies show that acute exposure causes irritation to mucous membranes, with animal tests pointing to moderate toxicity at high concentrations. Chronic studies look for longer-term effects, especially inhalation in closed industrial settings. So far, research demonstrates low risk of carcinogenicity, but caution wins. Researchers continue examining metabolites and breakdown products to ensure environmental safety, especially as more of the molecule moves through global supply chains. Medical surveillance data keeps regulatory agencies alert to any occupational hazards, driving updates to industrial hygiene and first-aid protocols.
Future Prospects
Looking down the road, isopropenyl acetate stands to benefit from new catalysts and better process integration—pushing yields higher, trimming costs, and shrinking the environmental footprint. Researchers from both industry and universities chase ways to recycle waste streams and capture volatile emissions. The molecule’s unique chemistry acts as a springboard for new pharmaceutical agents and advanced polymer systems. With increased focus on sustainable practices, producers invest more into bio-derived raw materials and closed-loop production. The future likely brings smarter labeling, tighter exposure thresholds, and new milestones for green chemistry—making isopropenyl acetate not just a relic from industrial history but a tool for next-generation products and responsible manufacturing.
What is Isopropenyl Acetate used for?
What Stands Out About Isopropenyl Acetate?
Most folks don’t hear about isopropenyl acetate unless they have a foot in the lab or work with chemicals. This clear liquid packs plenty of punch, with a sharp, familiar smell you might catch a whiff of in certain solvents and glues. Chemists often notice how quickly it evaporates, leaving practically no trace behind—except for the action it’s already helped set in motion.
Paints, Inks, and Sticky Situations
The paint and coatings world taps isopropenyl acetate for its strong ability to break down and dissolve substances. Manufacturers bank on its quick-drying nature to speed up drying times in both paint and ink. This liquid helps spray paints set faster or inks lock onto surfaces without smearing. That brief window where a painted object feels tacky but soon dries smooth owes a bit to solvents like this one.
People using glues at home or in workshops may not know they’re handling isopropenyl acetate, yet its presence helps thicken or liquefy adhesives where needed. Whether you’re patching a leaky pipe or fixing the sole of a shoe, the chemistry traces back to this small but mighty ingredient.
Building Blocks in Chemical Manufacturing
Isopropenyl acetate often gets used not just for what it is, but for what it creates down the line. Chemists take this liquid and convert it into isopropenol (commonly called isopropyl alcohol) and acetic acid. This reaction, called hydrolysis, pops up throughout industrial processing. Isopropyl alcohol, its popular by-product, shows up everywhere from medicine cabinets to warehouses as a disinfectant, cleaner, or solvent. Acetic acid, on the other hand, lines the shelves both as a food additive and as a base material in making plastics.
Big companies look for ingredients that pull double-duty. If a chemical can act alone but also transform into something just as valuable, it becomes a reliable piece in their toolkit. This makes isopropenyl acetate especially useful when resources are tight or when factories aim for efficient supply chains.
Balancing Safety and Everyday Utility
Working with chemicals in factories brings risks. Isopropenyl acetate gives off vapors that can burn the nose or eyes, and overexposure makes workers uneasy or even sick. I spent a stretch of time in a facility that handled volatile solvents, and nothing replaced good ventilation and careful training. Relying only on personal protective equipment never truly covers all the bases. Down-to-earth safety practices, plus real investment in exhaust systems, keep people from dangerous exposures and long-term harm.
Rules in the United States, Europe, and East Asia set strict exposure limits for this and similar chemicals. Clear protocols for storage and disposal also keep spills from getting out of hand. Our experience on the factory floor underlines the need for periodic air quality checks—those readings let operators spot issues before they turn critical.
Where Next?
With industries shifting focus to safer, greener ingredients, pressure now lands on chemical makers to innovate. Companies explore recycling more volatile solvents or swapping them for alternatives with less impact on human health and the environment. Any switch needs to match performance without sacrificing safety, resistance to breakdown, or cost. Until then, isopropenyl acetate stays on center stage, handling the jobs that keep manufacturing, maintenance, and repairs running smoothly.
What are the safety precautions when handling Isopropenyl Acetate?
Introduction
People sometimes underestimate just how risky common lab chemicals can be. Isopropenyl acetate rarely comes up at backyard barbecues, but it shows up in all sorts of labs and industries—from coatings to fragrances. Setting safety aside, though, can lead to both personal injury and environmental headaches.
Personal Protection That Actually Works
Direct contact with isopropenyl acetate isn’t something to brush off. My own chemistry professor warned us to keep it off our skin, explaining that the liquid irritates eyes and can cause burns if you spill it. Whenever I’ve worked with this solvent, I put on heavy-duty nitrile gloves, not those thin plastic ones that rip at the drop of a hat. Safety goggles might feel clunky, but losing your eyesight is worse. Long sleeves, closed-toe shoes, and even a face shield come in handy when pouring larger quantities. Breathing in the vapors never sat right with me; using a well-fitted respirator rated for organic vapors is a choice I’ve made after one too many dizzy spells—even in a ventilated room.
Why Ventilation Matters
Even in the best labs, fumes always sneak around. Cracking open a window or turning on the exhaust fan cuts the smell fast, and more importantly, moves the vapors out before you get a headache or worse. I once underestimated the stuffiness on a windless day and regretted it for hours. Fume hoods really earn their keep with solvents like these. No one loves the noisy blowers, but they suck out dangerous air, keeping lungs clear and accident risks way lower.
Storing and Dispensing Without Risking Trouble
You’ll notice serious labels on any isopropenyl acetate container: flammable, irritant, keep away from sparks and open flames. That’s not paranoia. A surprising number of lab fires start with careless solvent handling. I keep it in a flammables cabinet, never near oxidizers. Metal containers with tight caps stop leaks and keep the vapor from building up. Pouring out a little bit at a time—and only what you need—limits exposure and makes cleanup much less of a hassle.
Clean Up and Disposal: Not Just Red Tape
Accidents happen—spills, splashes, or even a bottle tipping over. From bitter experience, I can say it’s faster to act immediately. Absorbent pads or spill kits, followed by sealing waste in a proper container, make a huge difference. Pouring used solvent down the drain not only damages pipes, but can create toxic gas if it mixes with other waste. As much as it’s tempting to just mop up and move on, calling in trained waste handlers keeps everyone out of legal and medical trouble.
Why It All Matters
No one needs to end up in the emergency room because of shortcuts. According to OSHA, exposure to isopropenyl acetate can cause respiratory irritation and skin issues. Underreporting of accidents just makes it harder for workers down the line. Sharing knowledge—real experience and clear rules—helps build a workplace where safety isn’t just a checklist, but a real habit. Those careful, sometimes annoying steps protect our health now and down the road, and let us get the job done without disaster.
What is the chemical formula and structure of Isopropenyl Acetate?
Understanding Isopropenyl Acetate
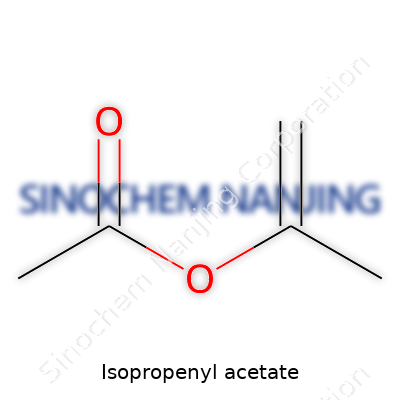
Isopropenyl acetate isn’t something most people talk about at the kitchen table, but it plays a big role in a range of industrial processes. This organic compound gets noticed in chemistry circles thanks to its unique structure and practical uses, especially as a building block for other chemicals. The formula is pretty straightforward: C5H8O2. That stacks up to five carbons, eight hydrogens, and two oxygens in every molecule.
Digging Into Its Chemical Structure
The backbone of isopropenyl acetate contains an isopropenyl group attached to an acetate group. Visualizing this, you have a double bond between two carbons in the isopropenyl piece. One of these carbons is also bound to a methyl group, while the other hooks into an acetate group, which comes from acetic acid. Chemists often write the structural formula as CH2=C(CH3)OCOCH3. Those double bonds in the molecule stand out, giving it certain reactivity in organic synthesis.
Why Structure Matters
Even outside of a laboratory, knowing how these atoms connect tells us a lot. For instance, the double bond means isopropenyl acetate is pretty reactive compared to saturated esters. In practice, that double bond lets it take part in polymerization reactions, which turn simple molecules into much bigger, useful plastics or specialty materials. The acetate group anchors the molecule’s smell—subtle and sweet. That leads perfumers and flavor chemists to try it out in creative blends, though it’s never as popular as its cousin, isoamyl acetate (banana oil).
Safety and Environmental Insights
I always pause when a compound includes double bonds and an acetate group. These features mean volatility can be high; a spill could evaporate quickly, sending vapors into the workspace. That’s why it’s essential to be careful about ventilation and exposure limits. While studies show moderate toxicity, prolonged contact can irritate the skin, nose, or lungs. Good safety standards and protective equipment cut down on risk for people who handle it daily.
Real-World Uses and Importance
Isopropenyl acetate brings value for chemists designing synthetic routes. It’s used to make isopropenyl alcohol and methyl isobutyl ketone, both of which end up in coatings and solvents. The double bond makes it a good candidate in specialty polymer manufacturing. Its use in scents or flavors might feel niche, but every ingredient counts in industrial recipes. These are the overlooked details that help manufacturers meet needs in coatings, adhesives, and some fine chemicals.
Tackling Challenges and Safer Chemistry
Making and using isopropenyl acetate raises questions about greener processes. Some researchers look to bio-based acetates or milder reaction conditions. Efficiency isn’t just a buzzword—it means fewer by-products and lower waste. There’s real value in pursuing routes that cut down on hazards and make recycling easier. I’ve seen improvements in handling methods, including smarter containers and improved air monitoring. Good stewardship keeps compounds like this useful while lowering risks.
Moving Forward
Curiosity about chemicals like isopropenyl acetate pushes science and industry forward. Each part of its structure offers opportunities to innovate—faster reactions, safer storage, even new end-products. Learning how and why certain atoms hold together gives us a roadmap for both safety and creativity in chemistry.
How should Isopropenyl Acetate be stored?
Recognizing the Risks
Isopropenyl acetate has found its way into labs, plants, and warehouses worldwide, used in making flavors, fragrances, and sometimes in specialty paints. Folks might not consider its hazards at first glance. Yet anyone who’s worked around chemical solvents knows what can go wrong when storage goes wrong—leaks, explosions, toxic fumes, the whole mess. One overlooked container or a shortcut for convenience leads to emergency room visits or environmental fines.
This chemical catches fire easily. Its vapors travel far, and all it takes is a spark. So, anyone working with it ought to know more than just the flash point listed in a data sheet. Years ago, a friend worked at a warehouse storing a range of chemicals, including isopropenyl acetate. One Friday, someone stacked drums too close to a heat vent. By Monday, fire officials had taped off the area. They were lucky—nobody got hurt. Others haven’t been so fortunate.
Storage Settings Make a Difference
It’s not rocket science—just common sense and caution. Store isopropenyl acetate in a cool, well-ventilated area where temperatures stay steady. Heat builds up in closed rooms or direct sun, and that triggers pressure inside the container or vapor leaks. I’ve seen well-designed facilities with concrete floors, metal racking, and ceiling fans humming—not because someone wanted to please an inspector, but to keep people safe from fumes and cuts from rusty drums.
Keep it in original, labeled containers—usually steel drums with tight-fitting lids. No plastic jugs or unknown canisters from the back shelf. Clear labels make a difference during inventory and especially in an emergency. I’ve sat through safety drills where someone grabbed the wrong fire extinguisher because the barrel next to it wasn’t marked. Small detail, big impact.
Protection Against Spills and Vapor
Even careful people spill on occasion. Don’t store these drums next to acids or oxidizers. Separating incompatible chemicals saves lives. Good spill containment means installing trays or dikes at ground level. Most pros have mopped up at least one leak, but a proper barrier keeps a drip from spreading to other stuff.
Ventilation is not just about comfort. Vapors build up, especially if a drum seeps around the bung or gasket. Efficient exhaust fans and open spaces keep concentrations down below explosive levels. Those with windows and doors know to keep them open. I once worked in a lab where we installed a simple exhaust system just to keep headaches and dry eyes in check—not fancy, but it made the job safer.
Practical Knowledge in the Real World
Supervisors and line workers should know the fire plan—where the sand, foam, and CO₂ extinguishers sit. Water won’t do the job with isopropenyl acetate. Personal protective equipment (PPE) brings that last line of defense—splash goggles, gloves, and sometimes respirators.
Regulations exist for a reason, and anyone who’s ever faced an Occupational Safety and Health Administration (OSHA) inspection can tell you, they’re there because stories of injury and loss drove them into policy. Training, routine checks, and a commitment to correct labeling all stack up to protect people and property. Real stories and common sense matter more than textbook guidelines. Learning from mistakes—our own and others’—makes the difference.
Is Isopropenyl Acetate hazardous to health or the environment?
Understanding the Risks of Isopropenyl Acetate
Growing up around a hardware store, familiar chemical smells always hung in the air. Solvents, cleaners, little bottles with labels that looked like tongue-twisters. I caught onto the hidden dangers pretty young, so learning about a chemical like isopropenyl acetate brings back that old curiosity—and concern.
Manufacturers often use isopropenyl acetate in the making of flavors, fragrances, and as a chemical intermediate. It doesn’t get headlines like benzene or asbestos, but ignoring it comes at a cost. Breathing in its vapors brings headaches, dizziness, and throat irritation. High enough concentrations could knock someone out. Not everyone reads technical data sheets, but the stuff can really pack a punch if handled carelessly.
On the skin, this chemical acts as a moderate irritant. Years ago, helping my uncle clean canned spray paint off his hands, we both ignored the warning about solvents. Red, itchy fingers reminded me fast: not everything that washes off easily goes away quietly. Lab studies point out isopropenyl acetate can harm eyes as well. Even a small splash stings and waters like crazy. The solution sounds simple—gloves, goggles, good ventilation. Still, workers, students, and home experimenters skip protection when in a rush.
Environmental Impact and Hidden Dangers
Spilled chemicals vanish quickly from smooth floors. We wipe, mop, and try to forget. With isopropenyl acetate, evaporation doesn’t signal safety. Once in the air, it breaks down fairly fast, thanks to sunlight and chemical reactions. This pace suggests a lower risk for persistent air pollution, but it doesn’t erase its toxicity. High vapor levels trip up indoor air quality fast. Outdoors, plants and animals are at lower risk, yet dumping or leaking in waterways pushes the problem downstream. Aquatic life can get hurt by relatively small amounts, and few folks notice until the damage shows up days or weeks later.
Putting Knowledge to Use
The truth is, no everyday user buys isopropenyl acetate straight off the shelf. Large-scale handlers see warning labels and Material Safety Data Sheets, but there’s a gap. Small workshops, schools, and even science hobbyists rarely invest in full training. That gap creates room for accidents—vapors collecting in small, closed spaces, improper storage near heat or flames, or mixing with other chemicals without a second thought.
Regulations limit emissions, require labeling, and set workplace air standards. But these rules don’t reach every corner. Real progress starts with better training and more accessible safety information. Anyone handling or storing volatile chemicals needs easy-to-read labels, clear advice on gloves and masks, and plain instructions about spills. Waste collection programs and community education events can shrink the odds of careless disposal.
Working with chemicals like isopropenyl acetate means trading convenience for risk. A fast-evaporating solvent handles tough jobs, but speed isn’t always an advantage. People learn best from real stories—a burned hand, a ruined project, a school evacuated for fumes—so sharing those lessons helps. Sometimes, all it takes is a memory of stinging eyes or an uncle’s red hands to remind us: short-term wins aren’t worth long-term headaches.
| Names | |
| Preferred IUPAC name | Propan-2-yl acetate |
| Other names |
2-Propen-1-one, 1-methoxy- Isopropenyl ethanoate 1-Methylethenyl acetate Prop-1-en-2-yl acetate Isopropenyl ester of acetic acid |
| Pronunciation | /ˌaɪ.səˈproʊ.pə.nɪl əˈsɛt.eɪt/ |
| Identifiers | |
| CAS Number | ['108-22-5'] |
| 3D model (JSmol) | `Isopropenyl Acetate` JSmol 3D model string: ``` CC(=C)OC(=O)C ``` |
| Beilstein Reference | **635873** |
| ChEBI | CHEBI:44668 |
| ChEMBL | CHEMBL372651 |
| ChemSpider | 54629 |
| DrugBank | DB13873 |
| ECHA InfoCard | 100.049.725 |
| EC Number | 205-565-2 |
| Gmelin Reference | 6177 |
| KEGG | C09706 |
| MeSH | D013497 |
| PubChem CID | 7905 |
| RTECS number | NI4370000 |
| UNII | 2BXY7U8BA8 |
| UN number | UN2344 |
| CompTox Dashboard (EPA) | DTXSID8067558 |
| Properties | |
| Chemical formula | C5H8O2 |
| Molar mass | 100.12 g/mol |
| Appearance | Colorless liquid |
| Odor | fruity |
| Density | 0.86 g/cm³ |
| Solubility in water | Insoluble |
| log P | 0.97 |
| Vapor pressure | 6.2 kPa (20 °C) |
| Acidity (pKa) | 13.3 |
| Basicity (pKb) | pKb: 12.62 |
| Magnetic susceptibility (χ) | -44.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.393 |
| Viscosity | 1.47 mPa·s (20 °C) |
| Dipole moment | 2.45 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 365.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -322.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | −2729 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Precautionary statements | P210, P280, P305+P351+P338, P370+P378, P403+P235 |
| NFPA 704 (fire diamond) | 2-3-2 |
| Flash point | 7 °C (closed cup) |
| Autoignition temperature | 455°C |
| Explosive limits | 1.4–8.5% |
| Lethal dose or concentration | Lethal dose or concentration (LD50, LC50) of Isopropenyl Acetate: "LD50 (oral, rat) 1,550 mg/kg; LC50 (inhalation, rat, 4h) 3,680 ppm |
| LD50 (median dose) | LD50 (median dose): 4.48 g/kg (rat, oral) |
| NIOSH | KIW145 |
| REL (Recommended) | 250 ppm |
| IDLH (Immediate danger) | IDLH: 400 ppm |
| Related compounds | |
| Related compounds |
Vinyl acetate Isopropenyl formate Isopropenyl propionate Isopropenyl butyrate |

